Abstract
Objective
This retrospective study was performed to assess the clinical and radiological variables associated with proximal type IA endoleak (EL) in patients treated with elective endovascular repair for abdominal aortic aneurysms.
Methods
The chi-square test, t-test, and logistic regression analysis were performed as appropriate. A P value of <0.05 was considered statistically significant.
Results
The data of 79 patients were analyzed. No mortality occurred. During follow-up (median, 28.5 months; interquartile range, 12.8–43.0 months), 10 patients developed type IA EL. In the logistic regression analysis, undersizing of the endograft diameter by <10% significantly affected the occurrence of type IA EL. When the diameter was used for measurements, less oversizing was significantly associated with a higher risk of type IA EL. When the area was used for measurements, oversizing of >20% significantly affected the occurrence of type IA EL.
Conclusion
When sizing endografts, a discrepancy was noted between the measurements of the diameter and area of the proximal neck. The area might represent a more accurate measurement than the axial diameter to optimize the proximal sealing and lower the risk of developing type IA EL.
Keywords
Introduction
Since its introduction to clinical practice about 30 years ago, endovascular abdominal aortic repair (EVAR) has become beneficial in terms of early survival compared with open repair in anatomically suitable patients. 1 Nevertheless, aortic complications requiring reinterventions are more likely to develop in the mid-term after EVAR than after open repair. 2 Among these complications, the occurrence of proximal type I endoleak (EL) and endograft migration are the most frequent causes of reinterventions, 3 and both require prompt correction because of the persistent risk of aneurysm rupture.
Although achievement of a good seal between the endograft and the proximal aortic neck seems to be a key point, 4 the factors that predispose to proximal type I EL remain incompletely understood.
The present study was performed to assess the clinical and radiological variables that are significantly associated with the occurrence of proximal type I EL in patients treated with elective EVAR for abdominal aortic aneurysms (AAAs).
Materials and methods
Ethical approval was obtained from the Ethics Committee of San Raffaele Hospital (167/int/2020) for this retrospective, single-center study. Explicit patient consent was not required.
Clinical and radiological data of patients who underwent elective EVAR for AAAs from January 2011 to December 2018 were retrospectively collected and analyzed. Patients were included in the study if they had complete computed tomography angiography (CTA) data available for both the preoperative analysis and a ≥1-year follow-up. Patients who had undergone CTA scans with a slice thickness of >1.5 mm per layer were excluded.
All patients underwent preoperative CTA of the abdomen to evaluate the feasibility of endovascular treatment and planning of the procedure. The surgery was performed in a dedicated operating room equipped with a mobile X-ray machine. The EVAR procedure was performed using a standard technique 5 according to the instructions for use (IFU) reported for each endograft. Patients were excluded from standard EVAR if they had inadequate iliac-femoral access, if the aortic neck diameter was >32 mm, if the aortic neck length was <15 mm, and if the proximal aortic neck angulation was ≥60°. The patients were followed up three times during the first year postoperatively (at 1, 6, and 12 months) using a duplex scan. A CTA scan was also performed at 12 months. After the first year, the patients underwent an annual duplex scan. CTA was performed if complications were detected or the duplex scan was nondiagnostic. In addition, thoracoabdominal CTA was performed every 3 years to evaluate possible progression of the aneurysmal pathology in the thoracoabdominal or thoracic district.
The following clinical data were extracted from the medical records: sex, age, presence of cardiovascular risk factors, and history of oral anticoagulant drug use. Cardiovascular risk factors included the presence of diabetes mellitus (defined as a fasting glucose concentration of >100 mg/dL or the intake of at least one drug to control the serum glucose concentration), hypertension (defined as systolic blood pressure of >140 mmHg and/or diastolic blood pressure of >90 mmHg, or as the intake of at least one drug to control the blood pressure), smoking habit (defined as current or past smoker versus no smoker), coronary artery disease, chronic renal failure (defined as an estimated glomerular filtration rate of <60 mL/min/1.73 m2), and chronic obstructive pulmonary disease.
The radiological data were collected by viewing the CTA scans using the software Carestream Health® (Algotech India Pvt. Ltd., New Delhi, India) on a transverse plane. The centerline with curved planar reformation and multiplanar reconstruction modes were used to obtain the images for analysis in the coronal and sagittal axes.
For each CTA study, the following variables were measured on the transverse plane perpendicular to the aorta:
Anteroposterior (AP) (inner-to-inner) diameter of the proximal aortic neck at the level of the emergence of the lower renal artery (level 0) and at 5 and 10 mm lower Laterolateral (LL) (inner-to-inner) diameter of the proximal aortic neck at level 0 and at 5 and 10 mm lower Area of the proximal aortic neck at level 0 and at 5 and 10 mm lower, including the thrombus and parietal calcifications, and internally to the positioned endograft (in the case of postoperative CTA)
The maximum sac diameter and the length and angulation of the proximal neck relative to the suprarenal aortic axis were also recorded.
Two independent operators (GMaz and GZ) performed the measurements using the same work tools and the same rules.
The following parameters were also calculated from the preoperative and postoperative recorded data:
average AP diameter (recorded at level 0 and at 5 and 10 mm lower) average LL diameter (recorded at level 0 and at 5 and 10 mm lower) average of these last two values [using Lachat’s formula: (AP diameter + LL diameter) / 2] average of calculated areas (recorded at level 0 and at 5 and 10 mm lower)
The following parameters were also calculated:
difference between preoperative and postoperative diameters of the proximal neck using Lachat’s formula difference between preoperative and postoperative areas of the proximal neck
Considering the dimensions of the implanted endograft, the area of the neck of the endograft was calculated from the diameter (d) of the main body, assuming a circular section [area = π × (d/2)2].
Finally, the amount of oversizing of the endograft relative to both the diameter and area of the preoperative native aortic neck was calculated.
The primary outcomes were the occurrence of death and type IA EL during follow-up, defined as the presence of iodinated contrast medium on CTA within the aneurysmal sac and outside the prosthesis lumen, coming from the proximal neck.
All collected data were inserted into a database and analyzed as appropriate using the software Stata/IC (StataCorp, College Station, TX, USA). Continuous variables are reported as median and interquartile range (IQR), and categorical variables are presented as number (percentage). Kaplan–Meier analysis with the log-rank test was used to estimate the rate of freedom from EL over time. The chi-square test and t-test were used as appropriate to compare variables. Logistic regression analysis was performed to assess any variable that could be associated with the occurrence of EL during follow-up. A P value of <0.05 was considered statistically significant.
Results
During the examined period, 402 patients underwent elective EVAR for AAAs. However, complete data from the preoperative and follow-up analyses were available for only 79 patients. Of these patients, 11 were female (13.9%). The patients’ median age was 74.5 years (IQR, 66.2–78.6 years). As shown in Table 1, most patients were affected by hypertension (84.8%), and 40.4% of them had a previous or current smoking habit.
Patients’ demographic and clinical characteristics.

Data are presented as median (interquartile range) or n (%).
COPD, chronic obstructive pulmonary disease; CAD, coronary artery disease.
All patients were treated according to the IFU of the chosen endograft (Table 2). The proximal neck diameter was >30 mm (33.6 mm) in only one patient.
Preoperative and postoperative features of aortic aneurysms on computed tomography angiography.

Data are presented as median (interquartile range).
Statistically significant P values are indicated by boldface text.
CIA, common iliac artery; AAA, abdominal aortic aneurysm.
An endograft with suprarenal fixation of the proximal neck was placed in most patients (49 patients, 62%): Cook Zenith (Cook Medical LLC, Bloomington, IN, USA) in 37 patients, Incraft (Cordis, Santa Clara, CA, USA) in 10 patients, and Ovation (Endologix, Inc., Irvine, CA, USA) in 2 patients. A Gore Excluder (W.L. Gore and Associates, Flagstaff, AZ, USA) was used in the remaining 30 patients.
Technical success was achieved in all patients; no endoleaks or graft thrombosis was detected in the perioperative period. Moreover, no immediate conversion was required and no mortality occurred.
A good correlation was observed between the measurements performed by the two operators (K ≥ 0.8 in all cases).
During follow-up (median, 28.5 months; IQR, 12.8–43.0 months), type IA EL was identified in 10 patients, all of whom underwent endovascular correction with a proximal cuff. None of the ELs were associated distal migration of the graft. According to the Kaplan–Meier analysis, the mean rate of freedom from ELs was 97.2% ± 1.9% at 1 year and 77.3% ± 7.3% at 5 years (Figure 1).

Kaplan–Meier estimate of freedom from EL during follow-up, with life table.
During follow-up, modest dilatation of the proximal neck was noted in terms of diameter (average, 1.11 mm; P = 0.04) and area (average, 19 mm2; not statistically significant) (Table 2).
The mean oversizing of the diameter that was applied between the endoprosthesis and the preoperative native aortic neck was 30%. However, the mean circular section of the main body of the endograft was about 70% greater than the preoperative area of the proximal aortic neck that was measured on CTA (P < 0.00001).
The logistic regression analysis showed no statistically significant correlation between the clinical factors analyzed and the occurrence of type IA EL (Table 3a). However, the analysis of radiological data showed some significant correlations (Table 3b). In particular, incorrect measurement of the endoprosthesis with undersizing of the graft diameter by <10% was significantly related to the occurrence of type IA EL (P = 0.01). In particular, when the diameter was used for measurements, less oversizing was significantly associated with a higher risk of type IA EL (P = 0.003; correlation coefficient, −2.98). Conversely, when the area was used for measurements, oversizing by >20% was strongly correlated with the occurrence of type IA EL (P = 0.001; correlation coefficient, 3.23). Moreover, the theoretical area of the proximal aortic neck calculated from the mean diameter, assuming a circular shape of the aortic neck, was significantly larger than the area that was directly measured (366.1 ± 199.2 vs. 232.4 ± 128.1 mm2, respectively; P < 00001).
Regression analysis of (a) clinical and (b) radiological factors affecting the occurrence of type IA endoleak during follow-up.
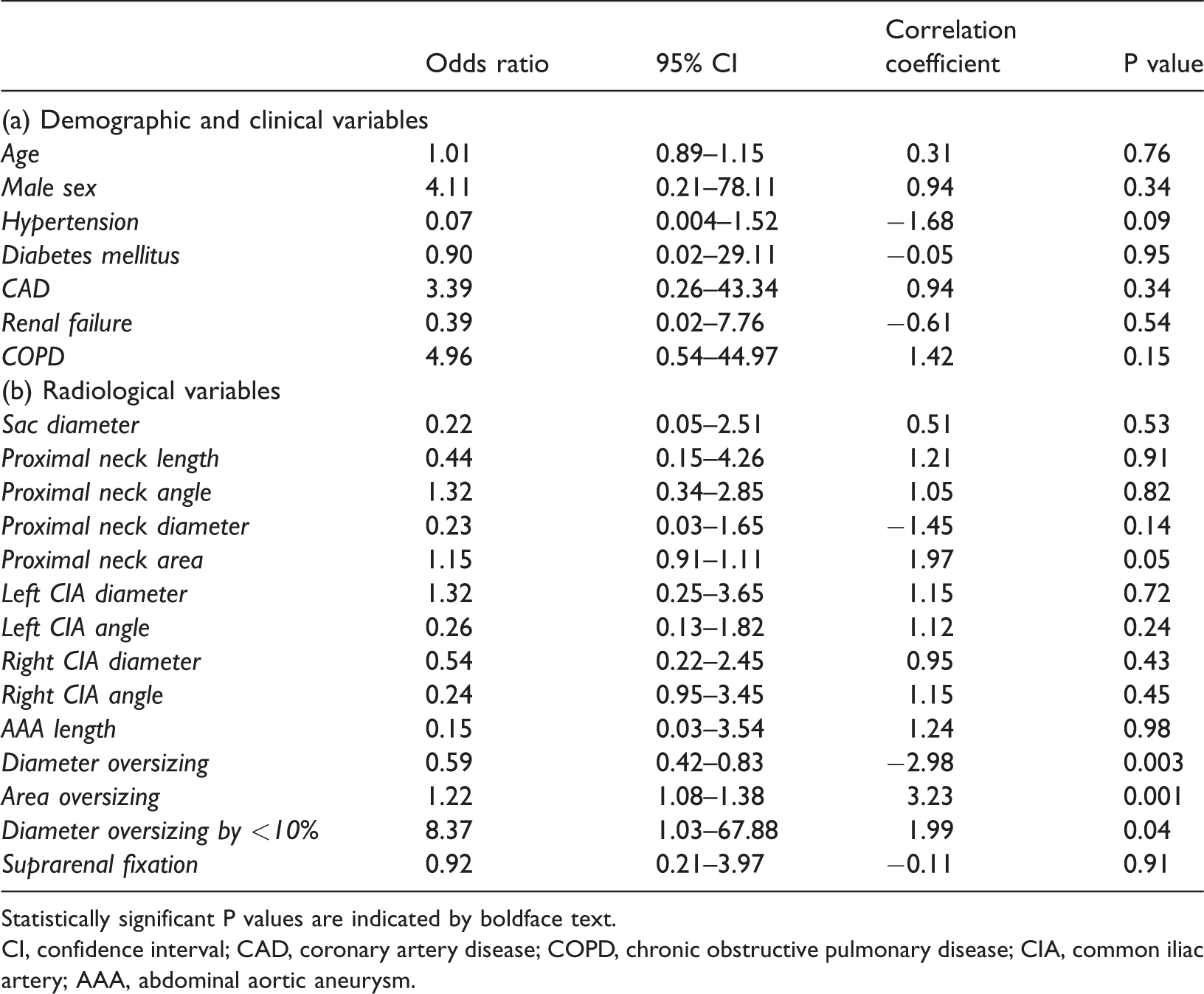
Statistically significant P values are indicated by boldface text.
CI, confidence interval; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CIA, common iliac artery; AAA, abdominal aortic aneurysm.
The use of endografts with suprarenal fixation did not affect the occurrence of type IA EL.
Discussion
Since their introduction into clinical practice, endovascular techniques have allowed the treatment of aortic pathologies in patients not suitable for traditional open repair because of their lower invasiveness with reduced postoperative mortality and morbidity rates. 6
Nevertheless, the advantages of EVAR have been lost over time because of a higher incidence of complications necessitating reinterventions. 7 Strict postoperative surveillance is therefore needed to detect complications that can lead to reintervention, especially aortic rupture. Among these complications, proximal (type IA) EL is the most frequent cause of reintervention and aortic rupture after EVAR. 3
Many studies have been performed to identify the most important factors predisposing to the occurrence of type IA EL. In particular, loss of the proximal seal of the endograft is the most important cause of type IA EL. The proximal seal can be lost over time because of changes in the proximal aortic neck, progression of the native aneurysmal disease, or, according to some authors, geometrical remodeling of the proximal neck due to the radial force of the endograft acting on the aortic wall, especially in cases of excessive graft oversizing. 8 In the present study, slight dilation of the proximal aortic neck was recorded over time in terms of both the aortic diameter and area. However, the dilation was not statistically significant and was not related to the occurrence of type IA EL.
A second important and well-recognized issue is anatomic hostility of the proximal aortic neck, which prevents adherence to the IFUs of the employed device. A hostile aortic neck is characterized by marked angulation or shortness of the proximal neck, which can result in difficulty achieving a proper seal and an increased occurrence of type IA EL.9,10 Moreover, patients with a proximal neck of >29 mm in diameter are at higher risk of developing a type IA EL after EVAR. 11 Fenestrated or branched EVAR should be considered in such patients if they are unfit for open repair.
As a third issue, appropriate sizing of the endograft has been advocated as important to achieve a proper proximal seal, which can reduce the risk of developing type I EL and distal migration. 12 In particular, the guidelines of the European Society of Vascular Surgery recommend 10% to 20% oversizing of the endograft diameter on the aortic neck 13 to optimize the seal. In their review, Van Prehn et al. 14 found that oversizing by up to 25% may reduce the risk of type IA EL, while the risk of graft migration with >30% oversizing remains unclear.
Similarly, in our case series, diameter oversizing by <10% was associated with a higher risk of type IA EL. In particular, less oversizing was associated with a higher risk of developing type IA EL. In our series, the five cases in which the endograft was undersized by <10% based on diameter occurred in the first part of our experience and were not standard practice; they were instead likely due to technical measurement errors. In contrast, when the measurements were performed using the area of the proximal neck instead of the diameter, oversizing by >20% was significantly related to the occurrence of type IA EL.
These findings lead us to question how endograft sizing is carried out using CTA images. In fact, the optimal method for endograft sizing has not been adequately validated. In most cases, the sizing is performed on a static CTA image. In some centers, however, electrocardiographically gated (dynamic) CTA can be executed to minimize cardiac-induced aortic wall motion and thus obtain more precise definition of the aortic neck. Iezzi et al. 15 demonstrated significant variation of the proximal aortic neck diameter during the cardiac cycle, which can lead to inadequate endograft sizing in up to 18% of evaluated cases. Finally, some authors have argued that the diameter may not be the most appropriate tool for endograft sizing.
Buijs et al. 16 assessed the degree of discrepancy between direct measurements of the aortic neck diameter and those obtained by a circumference-based method and then examined the association of this discrepancy with the development of type IA EL. They compared 12 patients with postoperative EL to 48 matched controls without EL and found that the diameters that were directly measured were smaller than those derived by the circumference, although the difference was not statistically significant and was not correlated with the occurrence of type IA EL. Therefore, the authors concluded that the proper method for endograft sizing is still a matter of debate. Tielliu et al. 17 argued that the circumference-based method is better than the traditional method for endograft sizing, especially when the proximal neck does not have a circular shape or is irregular because of the presence of thrombi and calcifications.
Similarly, in our study, the area of the proximal neck that was directly measured was smaller than that obtained geometrically from the mean diameter (assuming a circular shape of the aortic neck), as if the measurement of the diameter somehow overestimated the real size of the aortic neck. These findings may justify the data regarding endograft oversizing with respect to the area of the proximal neck, which was significantly higher than the oversizing obtained using the diameter. From a geometrical point of view, this difference cannot be overlooked. Additionally, from a clinical point of view, this difference seems to translate into an increased risk of proximal EL. These findings underline how the proximal aortic neck is not “perfect” and that a methodological error is likely to occur if we consider its cross section to be a circle.
Excluding cases in which an obvious sizing error has occurred (i.e., cases in which the diameter oversizing was <10%), we could argue that measurement of the diameter is probably not the best tool for endograft sizing based on the findings of our study. The area might be an effective alternative or adjunctive tool for calculation of graft oversizing, especially when the proximal neck has an irregular shape. In light of these assumptions, manufacturers of endoprostheses should give the measures of their graft not only according to the diameter of the proximal aortic neck, but also with reference to the area of the main body that best suits the area of the proximal neck.
Some centers employ endografts with suprarenal fixation to achieve a proper seal, with the idea of minimizing the risk of graft migration. Nevertheless, according to some authors, suprarenal fixation of the graft may be associated with more rapid dilatation of the proximal aortic neck with an increased risk of developing type IA EL.18 In our experience, however, suprarenal fixation did not seem to affect the occurrence of type IA EL during follow-up.
Our study has some limitations, the most important being its retrospective nature and small sample size. Moreover, the measurements were performed manually by two operators on static CTA images, which might have been impaired by the aortic wall motions during the cardiac cycle.
Conclusion
We noted a discrepancy between the measures of the diameter and area of the proximal neck when sizing the endograft, suggesting that the area might represent a more accurate measurement than the axial diameter to optimize proximal sealing and lower the risk of developing type IA EL. Undersizing of the endograft diameter by <10% was significantly related to the occurrence of type IA EL. When the area was used for measurements, oversizing by >20% was strongly correlated with the occurrence of type IA EL.
