Abstract
Background
Pharmaceutical waste includes drugs that have expired, are unused, spilt, and/or contaminated. 1 This waste represents a significant cost burden to pharmacy operations and management. For instance, a study conducted at a major health care institute in the United States found that 2 out of 3 dispensed medications were unused, with national projected costs ranging from $2.4B to $5.4B. 2 In another US study, the estimated total costs associated with drug wastage in lung cancer patients were $16.6M over a 2-year period. 3 Others have reported that cancer drugs alone account for $1.8B in medication waste annually. 4 Unused opioids have contributed to the opioid epidemic that continues to ravage the US. For example, the 2023 National Survey on Drug Use and Health in the US estimated that 39% of people who misused opioid pain relievers received their most recent pain relievers from a friend or relative with unused pills. 5 It is thought that developing safe disposal methods for unused prescription opioids is a key prevention strategy in combating the opioid epidemic. 6 Furthermore, improperly discarded pharmaceutical waste appears in the environment, including within surface waters, groundwater, drinking water, and tissues of freshwater/terrestrial wildlife. 7 Ultimately, humans can be exposed to improperly disposed pharmaceuticals. 8 Therefore, proficient and cost-effective drug disposal methods are urgently required.
In the past decade, numerous drug disposal systems/technologies have been developed and, in some cases, marketed to the public. The mechanisms for treating/inactivating pharmaceutical waste vary greatly among the systems. 9 In general, the unused/expired medication is placed into a waste receptacle (eg, bottle or container) where the medication can interact with chemical agents already present in the receptacle. One common mechanism among marketed products is adsorption, in which chemicals such as charcoal (carbon) or clay will “soak up” the drug. Unfortunately, this mechanism is not always irreversible, raising concerns of its widespread utility. 9 An alternative method is treating drug waste with a combination of hydrogen peroxide and iron that makes up the Fenton reaction, a powerful oxidative process that can chemically degrade pharmaceuticals and facilitate safe disposal. 10
Propofol (Diprivan) is an intravenous agent widely used as an anesthetic during surgical procedures. It is thought to produce its sedative/anesthetic effects via action at the ligand-gated gamma-aminobutyric acid (GABA)-A receptor in the central nervous system. 11 Propofol is recognized as a commonly wasted drug in health care settings, whereby significant amounts of left-over infusion solutions are typically discarded rather than administered to patients.12,13 Since propofol is highly lipid soluble and poorly degradable under normal circumstances, there are serious concerns about bioaccumulation and environmental toxicity to aquatic/terrestrial life if not properly disposed. 12
Due to poor water solubility, propofol injection is formulated as an emulsion containing, in part, soybean oil, egg lecithin, and glycerol. 11 Propofol has been associated with the occurrence of health care–related infections, which are thought to be a result of microbial contamination of its lipophilic formulation, a medium that strongly supports extrinsic bacterial growth such as Escherichia coli (E. coli) at room temperature as well as the fungus Candida albicans (C. albicans). 14 Anecdotal reports suggest mold-like contamination occurs when propofol solutions are left open to air and not properly disposed of. Hence, there is immense potential for microbial contamination of propofol waste, and if insufficient disposal occurs in the surgical, clinical, or pharmacy setting, serious health and environmental repercussions may arise. Henceforth, the primary purpose of this study was to compare decontamination of propofol solutions inoculated with medical pathogens among 2 drug disposal systems with different treatment mechanisms and secondarily assess chemical degradation of propofol after exposure to Fenton reagents.
Methods
Chemical and Reagents
Authentic propofol (European Pharmacopoeia reference standard) was purchased from Millipore Sigma (Burlington, Burlington, Massachusetts). Propofol injectable emulsion 200 mg/20 mL (Hospira Inc; Lake Forest, Illinois) was acquired by our institute’s clinic. HiPerSolv Chromanoform high-performance liquid chromatography (HPLC)-grade methanol and water were purchased from VWR International, LLC (Radnor, Pennsylvania). Lauri Bertani broth (LB) and YPD (Yeast, Peptone, and Dextrose) were both purchased from Fisher Scientific (Waltham, Massachusetts). All other chemicals and reagents were of the highest purity available and purchased from commercial sources.
Pharmaceutical Disposal Devices
Two medication disposal devices were evaluated in this study. A prototype instrument, described in more detail in a separate publication, 10 and a marketed product that was acquired from an online retailer. The commercial product, according to the package label, is a mixed solution consisting of activated carbon, water, and unspecified ingredients.
Kirby-Bauer Diffusion Disk Assay
The disk diffusion method of Kirby and Bauer is a standardized procedure to assess microbial survival in the presence of medications. 15 Twenty-five (25) mL of Mueller-Hinton agar in the presence or absence of 20 g/L of dextrose was poured into 100 mm sterile petri dishes and allowed to solidify in a laminar flow hood. Thereafter, 2 × 108 CFUs/mL of E. coli or C. albicans was spread on the Mueller-Hinton agar plates using a sterile swab. The plates were allowed to dry, and sterile paper disks (BD BBL) impregnated with undiluted propofol (10 mg/mL) or 25-, 50-, 100-fold diluted propofol solutions (0.4 mg/mL, 0.2 mg/mL, and 0.1 mg/mL, respectively) were placed onto the agar using sterile forceps. Thereafter, the plates were incubated at 37°C overnight. Control experiments included sterile paper disks impregnated with sodium hypochlorite (bleach). Upon completion of the incubation period, the zone of inhibition was determined. Experiments were repeated 3 times (triplicate).
Assessment of Microbial Survival After Fenton Treatment
Nine-hundred (900) mL each of 25-, 50-, and 100-fold dilutions of propofol were inoculated with 100 mL of 3.10 × 105 CFUs/mL of E. coli and C. albicans and subjected to decontamination by the prototype instrument for 16 minutes. Thereafter, the chemically treated effluent was collected under aseptic conditions and analyzed for microbial growth. The experiment was repeated in triplicate for each dilution.
Growth Assays
To determine if propofol solutions support the growth of microorganisms, 180 µL of 25-, 50-, and 100-fold dilutions of propofol (0.4 mg/mL, 0.2 mg/mL, and 0.1 mg/mL, respectively) were inoculated with 20 µL of 3.10 × 105 CFUs/mL of E. coli and C. albicans. Thereafter, solutions were incubated at 20°C and 37°C and 10 µL aliquots of samples were removed at 24-, 48-, and 72-hour intervals, diluted, and plated on media containing LB broth and YPD. Plates were subsequently incubated at 37°C for 24 hours. The number of CFUs (Colony Forming Units), a unit of measurement used to estimate the number of viable microbes in a sample, was determined after incubation. The experiment was repeated 3 times.
A growth assay was also conducted with the commercial drug disposal device. Aliquots of 25-, 50-, and 100-fold diluted propofol solutions were poured into a solution already residing in the container of the commercial product. One-hundred eighty (180) µL of the resultant mixture was inoculated with 20 µL of 3.10 × 105 CFUs/mL of 3.10 × 105 E. coli and C. albicans and incubated for 24, 48, and 72 hours at 20°C and 37°C. A total of 10 mL aliquots of the solutions were removed at 24- and 48-hour intervals, diluted, and plated on LB and YPD media; then subsequently incubated at 37°C for 24 hours. The number of CFUs was determined after incubation. The experiment was repeated 3 times.
Propofol Analytical Assay
A reversed-phase HPLC assay with ultraviolet detection was developed according to our previous procedure. 10 The separations module consisted of a Waters e2695 Alliance system (Milford, Massachusetts), which was coupled to a Waters 2998 photodiode array (PDA) detector. The system was managed by Empower 3 software (Waters Corp). A GL Sciences (Tokyo, Japan) Inertsil C8 analytical column (4.6 × 50 mm; 3 μm) was used for separation. The column was protected by an Avantor ACE C8 guard cartridge (Radnor, Pennsylvania) and maintained at 25°C in a column oven. The mobile phase consisted of a mixture of methanol and water in a ratio of 75:25. Flow rate was set at 0.5 mL/min, and ultraviolet (UV) absorbance was monitored at 272 nm. Injection volume was 25 μL.
A stock solution of propofol (400 μg/mL) was prepared in a mixture of methanol and water (75:25). Seven calibrator samples (0.1-6.4 μg/mL) were prepared by diluting stock solution with appropriate volumes of methanol and water (75:25). The general procedures for determining assay analytics (eg, stability-indicating, intra-day and inter-day precision and accuracy, and lower limit of quantification [LLOQ]) were performed according to our previous publication. 10
Chemical Destruction Experiments
Chemical processing in the prototype instrument utilized Fenton’s reagent (a mixture of ferrous sulfate and hydrogen peroxide in water) as previously described. 10 In brief, 20 mL of propofol emulsion was added to 980 mL of sterile water and stirred to yield a 200 μg/mL solution. A small aliquot (100 µL) was withdrawn (this served as the “before treatment” sample). The remaining solution was poured into the instrument (via an inlet valve) and underwent 2 pulsatile treatment cycles (6 minutes apart) with Fenton’s reagent, each cycle consisting of pumping 5 mL each (via different tubes) of 30% hydrogen peroxide and 1M solution of ferrous sulfate into the solutions, with mixing occurring with each cycle. Final eluent was pumped out of the instrument’s drain line into a large glass beaker and was brownish-yellow in color. An aliquot of eluent was filtered through a 13-mm JT Baker (Phillipsburg, New Jersey) PTFE syringe filter (0.20 μm porosity) to yield a clearer solution (this served as the “after” treatment sample). The presence (concentration) of propofol in filtrate was determined by the propofol analytical assay.
Results
Kirby-Bauer Diffusion Disk Assay
No zones of inhibition were observed for any diluted or undiluted samples of propofol. For the control (8.25% sodium hypochlorite), 10- and 14-mm zones of inhibition were observed for E. coli and C. albicans, respectively.
Survival Assay
No viable microbes were observed in any diluted propofol solutions after treatment in the prototype instrument, indicating chemical treatment with Fenton reagents inhibits microbial survival.
Growth Assay
For diluted solutions of propofol, an average of 3.13 to 3.33 × 105 CFUs/mL was observed after 24, 48 and 72 hours of growth at 20°C (Table 1). For samples incubated at 37°C, the average range of CFUs/mL was 5.20 to 5.53 × 105 (Table 1). These data collectively indicates propofol solutions serve as a medium for microbial growth.
Growth of Microorganisms Following a 24-, 48-, and 72-Hour Incubation at 20°C and 37°C. a

Initial inoculum was 3.10 × 105 CFUs/mL of each microorganism (E. coli and C. albicans).
Data are shown for experiments conducted with the 25-fold diluted propofol solutions.
For the commercial product, an average of 4.2 to 4.4 × 105 CFUs were observed in samples incubated at 20°C for 24, 48, and 72 hours (Table 1), and in incubations kept at 37°C, there was an average range from 5.20 to 5.4 × 105 CFUs/mL. These findings demonstrate that treatment with the commercial product lacks antimicrobial properties that were observed with the prototype instrument.
Propofol Assay Performance
Under the analytical conditions described, propofol eluted at approximately 4.5 minutes (Figure 1). The assay was found linear over 7 calibration points ranging from 0.10 μg/mL to 6.4 μg/mL (R 2 = 0.999). The equation of the regression line was y = 20 425x − 1455. Furthermore, the assay was shown to be stability-indicating and the intra-sample precision of the assay, expressed as percentage relative standard deviation (%RSD), was ≤11%. The intra-day and inter-day coefficients of variations were ≤8%. Regression analysis of the calibration curve calculated an LLOQ of 0.13 μg/mL (130 ng/mL).
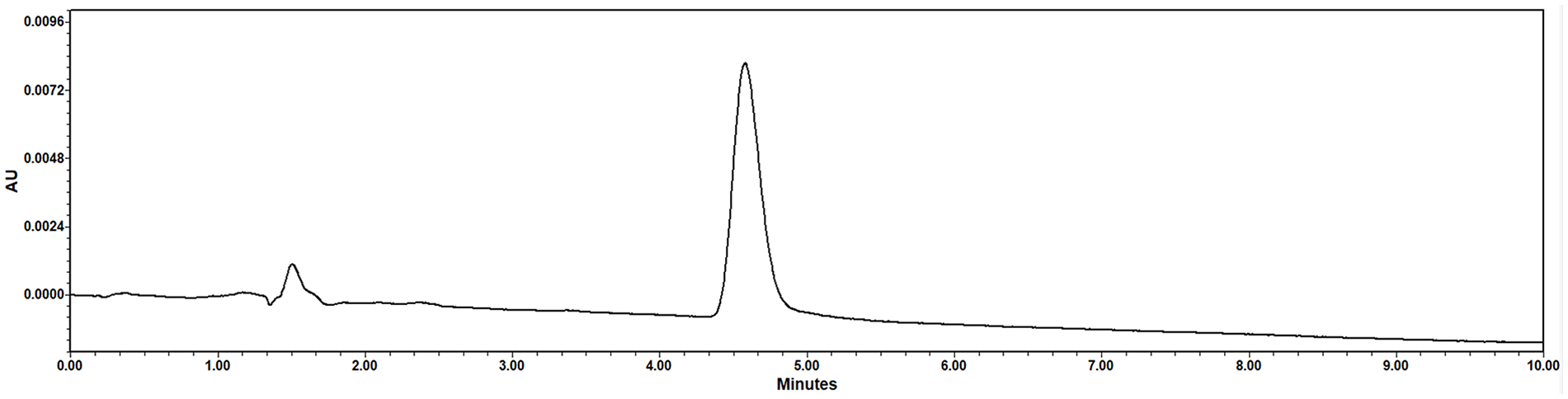
Representative HPLC-UV chromatogram following injection of 0.25 μg/mL of propofol prepared in methanol and water co-mixture. Propofol eluted at approximately 4.5 minutes.
Chemical Destruction of Propofol Infusion Solutions
After treatment of propofol infusion solution in the prototype instrument, we observed no propofol peak when a sample of the eluent was analyzed by HPLC (Figure 2). Therefore, the total extent of degradation was limited to the HPLC assay sensitivity. This suggests that in chemically treated samples of propofol solutions, there was less than 0.13 μg/mL (0.13 ppm) of drug remaining.
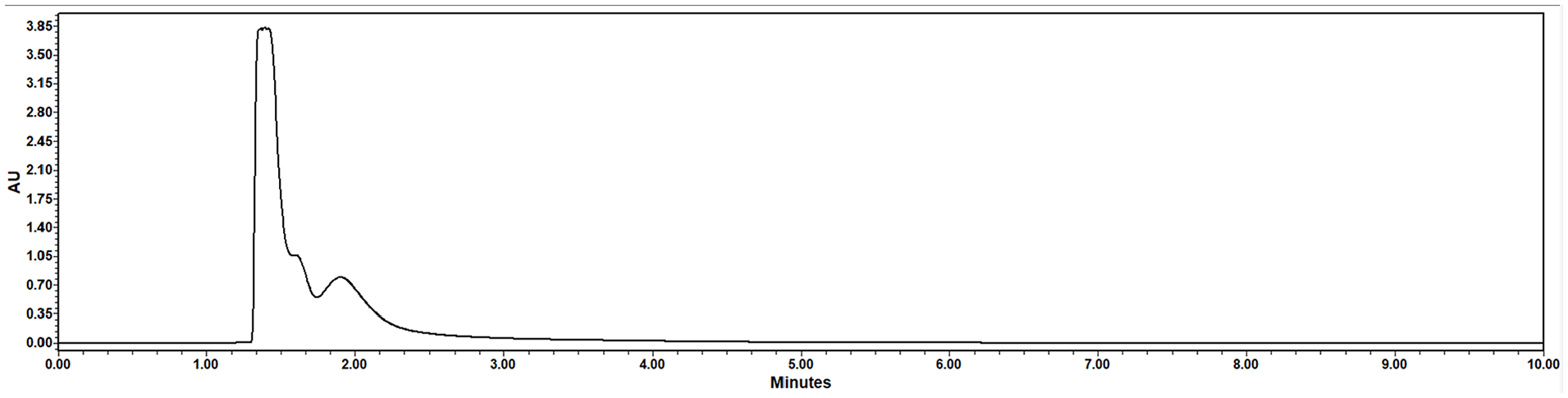
HPLC-UV chromatogram following chemical processing of propofol emulsion in the prototype instrument. No propofol peak was detected, indicating significant chemical degradation.
Discussion
The first finding of this study was that propofol injectable emulsion (undiluted) failed to demonstrate antimicrobial properties, as demonstrated on the disk diffusion assay. This is intriguing since the package insert states the emulsion contains benzyl alcohol that inhibits microbial growth for up to 12 hours. 11 However, we observed no zones of inhibition against E. coli and C. albicans under the experimental conditions described herein. Our findings also demonstrate that propofol emulsion and propofol-diluted solutions were suitable mediums for growth of E. coli and C. albicans at 20°C and 37°C. These data along with those found in the literature 14 suggest improperly disposed propofol waste (eg, remaining open to the air) can grow medical pathogens, predisposing to local contamination and possible spread of dangerous microbes into the surrounding environment.
We further demonstrated that chemical treatment of propofol emulsion solutions (inoculated with E. coli and C. albicans) with Fenton reagents effectively kills these medical pathogens. Contrastingly, the marketed product studied herein does support the growth of these microorganisms, suggesting drug disposal devices that rely on a sequestration (adsorption) mechanism would be problematic in disposing of propofol waste in the pharmacy and medical setting.
The final objective of this study was to determine if treatment of propofol solutions with Fenton reagents results in degradation of the propofol molecule. In fact, the prototype instrument was capable of degrading solutions to undetectable quantities of propofol (<0.13 ppm) as measured by the HPLC assay before and after treatment with Fenton reagents. These data complement previous findings 10 in which Fenton reagents were shown to degrade an assorted array of drug molecules with differing chemical structures and properties.
This study has several limitations. The evaluation of the devices was conducted in a controlled laboratory environment, which may not fully reflect the variability of usage in a real-world pharmacy setting. Factors such as environmental conditions, user knowledge, and behavior could influence the performance of the devices. Furthermore, the prototype device was tested in its developmental stage. As such, performance and functionality may differ in future versions that are optimized for practical use. Finally, we used E. coli and C. albicans to demonstrate the potential growth of these microorganisms in propofol solutions and to evaluate device capabilities. However, other pathogens associated with nosocomial infections, such as Staphylococcus aureus, Enterococcus faecalis, and Pseudomonas aeruginosa, could be used to further test the efficacy of these devices. Despite these limitations, this study provides valuable insights into the decontamination and disposal of propofol solutions.
Conclusion
In conclusion, medical pathogens inoculated into propofol emulsion and diluted propofol solutions are effectively killed after exposure to Fenton reagents. Finely tuned, this method of treatment holds promise in developing novel, compact drug disposal devices for real-time use in pharmacy and health care facilities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a grant from Clear River Enviro, Inc.
