Abstract
Keywords
Introduction
Obesity presents a worldwide health issue with a limited number of pharmacotherapies available. The 2023 Adult Obesity Prevalence Maps from the Centers for Disease Control and Prevention (CDC) revealed that all US states and territories had an obesity prevalence exceeding 20%. 1 In 2019, medical costs for obesity-related health care in the US alone exceeded $170 billion USD.2 -4
Multiple pharmaceutical agents are approved for weight loss, including glucagon-like peptide-1 receptor agonists (GLP-1 RA). The first GLP-1 RA on the US market was exenatide (Byetta), which was approved by the US Food and Drug Administration (FDA) for the treatment of type II diabetes on April 28, 2005. 5 The mechanism of action of this class of medications is to mimic the effects of endogenous glucagon-like peptide-1 (GLP-1), a hormone involved in glucose homeostasis. 6 In response to glucose-containing food intake, GLP-1 elicits insulin secretion from pancreatic beta cells, modulating blood glucose levels. Its efficacy in weight loss includes inducing sensations of satiety in the hypothalamus, reducing the desire for food and caloric intake, slowing gastric emptying leading to a sense of fullness and reduced meal frequencies. Common adverse effects include nausea, diarrhea, vomiting, abdominal pain, headache, fatigue, and dizziness. 7 These adverse effects must be weighed against the potential benefits of weight loss. Randomized controlled trials have shown Wegovy, a GLP-1 RA approved in the United States for weight loss in June 2021, reduces body weight by up to 15% over a 68-week treatment period. 8
In 2023, Rocky Mountain Poison & Drug Safety and Denver Health reported an increasing number of GLP-1 RA exposures since 2014. 9 Sixty-nine percent of these exposures resulted from user error, specifically injecting more often or higher doses than prescribed. 10 The risk for consequences from unintended exposures increased with the use of GLP-1 RA for weight loss.
The increased exposures to GLP-1 RA may reflect the expansion of approved uses. Although GLP-1 RA were initially developed for type II diabetes, 11 the FDA approved Saxenda (liraglutide) for chronic weight management on December 23, 2014, which generated US-based revenues of $256.2 million in 2017 and $469.7 million in 2023—a modest sum compared with the $4.2 billion generated by Wegovy (semaglutide) in 2023.12,13 Amid the growing popularity of GLP-1 RA such as Ozempic (semaglutide), these antidiabetes medications have been increasingly prescribed off-label for weight loss. The approval of Wegovy by the FDA on June 4, 2021, for chronic weight management has further increased demand.14,15 However, the limited time in which GLP-1 RA have been approved for weight loss means they lack the safety data for GLP-1 RA used in type II diabetes. The rapidly increasing use of GLP-1 RA in the nondiabetic population creates a greater risk for misuse and adverse events. Clinical trials only reviewed potential risks at the prescribed doses of GLP-1 RA, and among those whose body mass index are more than 30 or 27 with one or more weight-related comorbidities.7,16,17 The incidence of hypoglycemia in nondiabetic patients was not captured in these clinical trials. 7
In this study, we analyzed changes in exposures reported to a statewide poison control system serving approximately 10% of the US population related to reported GLP-1 RA exposures before and after FDA’s approval of Wegovy for chronic weight management. In addition, the study sought to characterize the clinical effects and outcomes reported from exposures, including the reason for the exposure, reported indication for use, product type, concomitant use or concurrent exposure to other antidiabetic agents (if reported), and hospitalization. The goal was to describe trends or differences in GLP-1 RA exposures following the approval of Wegovy.
Methods
This study was a retrospective medical record review assessing changes in GLP-1 RA exposures reported to the California Poison Control System (CPCS) from December 1, 2017, to December 31, 2023, comparing data from before and after the FDA approval of Wegovy as a weight loss medication on June 4, 2021. 18
Poison control data in the United States have been identified as a critical resource for nearly real-time information on exposures, making it possible to assess novel drug exposures that can affect public health.19,20 Consistent with published guidelines in the research literature, we addressed the inherent limitations of poison control data (which are applicable to multiple administrative datasets) through (1) hypothesis testing (the expectation of increased exposures after FDA approval of Wegovy for weight loss), (2) generation of a detailed template for data collection, (3) assessment of linked hospital records in records review, and (4) discussion of possible discrepancies between reported and nonreported cases. 21
The data used in this study were human cases coded as GLP-1 RA exposures and drawn from the CPCS database. These records were coded by CPCS toxicologists in real time during poison hotline calls, adhering to America’s Poison Centers’ National Poison Data System (NPDS)-defined codes. The NPDS-defined codes for exposure type are unintentional (general; therapeutic error; misuse; unknown), intentional (suspected suicide; misuse; abuse; unknown), other (contamination/tampering; malicious), adverse reaction to the drug, and unknown reason. The NPDS-defined codes for medical outcome are: no effect, minor effect (minimally bothersome symptoms), moderate effect (non-life-threatening, but pronounced, prolonged symptoms or systemic symptoms), major effect (life-threatening symptoms resulting in significant residual disability or disfigurement), death, not followed (judged as nontoxic or minimal clinical effects possible), unable to follow but judged as potentially toxic exposure, unrelated effect (exposure probably not responsible) and confirmed non-exposure.
There were 1074 cases extracted; personal identifying information was redacted. After removing duplicates, 1070 cases were coded using REDCap (Research Electronic Data Capture), a secure, web-based software platform designed to support data capture for research studies.22,23 The REDCap instrument was created by 2 authors with more than 5 years of cumulative experience coding CPCS records for data analysis, using the measures described below.
The following variables were extracted from each CPCS case: age, gender, location, nature of the call (exposure or informational), single or multiple substance exposure, GLP-1 RA medication, concomitant antidiabetic medications for single GLP-1 RA exposures and co-ingested antidiabetic medications for polysubstance GLP-1 RA exposures, indication for GLP-1 RA medication, symptoms, presence and characteristics of hypoglycemia, reason for exposure, exposure management site, and medical outcome of the exposure. Hypoglycemia was defined as a blood glucose of <70 mg/dL.
The research team consisted of 8 coders: 5 CPCS toxicologists and 3 PharmD candidates. Interrater reliability was assessed by drawing a sample of 150 cases and assigning each case to 2 different coders. The result was a Gwet’s AC1 coefficient of 0.71 to 0.72, indicating consistent coding. The 3 PharmD candidates performed final coding into REDCap. Cases that were unclear to coders were reviewed for accuracy by the 5 CPCS toxicologists as a team until a consensus was reached.
Inclusion criteria for cases analyzed in this study were confirmed GLP-1 RA human exposure with an exposure date between December 1, 2017 and December 31, 2023. Cases were considered non-exposures if there was insufficient information to confirm exposure or if they were only informational requests. Special circumstances were treated as follows:
Cases with patients not followed or lost to follow-up were included;
Cases with unrelated clinical effects were included to determine the characteristics and reason for the exposure, but excluded from the clinical effects and medical outcome analysis;
Cases involving compounded GLP-1 RA products co-formulated with cyanocobalamin and exposures with 2 GLP-1 RAs reported were coded as sole GLP-1 RA exposures if no other substances were involved;
Blood glucose reported as a range (eg, “50s”) was coded as the midpoint of the upper and lower limit of the reported range (eg, “55”);
No additional information was assumed in cases with missing or obscure data fields, and any obscure information was treated as missing;
Conflicts between measures coded into the CPCS system via NPDS codes and information reported by a healthcare facility were treated as a failure to update the record, and information from the healthcare facility documented in the case narrative took precedence.
Changes in the number of exposures and hospital utilization from exposures were assessed using an interrupted time series analysis (ITSA); the intervention was FDA approval of Wegovy for chronic weight management in June 2021. Regression analyses and t-tests of pre-approval versus post-approval comparisons of means were conducted. Clinical effects and medical outcomes were compared using Fisher’s exact tests. Relative risk (RR) ratios and 95% confidence intervals (CI) were calculated. All statistical analyses were completed using Stata v17 and OpenEpi (version 3.01). The University of California, San Francisco Institutional Review Board approved this study, #23-38977, as exempt on September 18, 2023.
Results
A total of 1070 cases were reviewed and 1047 cases met inclusion criteria. The median reported age in the sample was 54 years (interquartile range: 41-64) and 74.2% of the sample was female. We identified 233 (22.3%) reported exposures before FDA approval of Wegovy for weight loss in June 2021, and 814 (77.7%) after approval.
Prior to FDA approval of Wegovy, reported GLP-1 RA exposures did not exceed 20 cases per month. By 2023, reported exposures ranged from 40 to 60 cases per month. The ITSA results (see Figure 1a) estimated a starting level of 2.219 reported GLP-1 RA exposures per month and reported exposures increased significantly every month prior to FDA approval by 0.163 per month (CI = 0.093, 0.234, P < 0.001). One month after FDA approval, there was a significant increase in reported monthly GLP-1 RA exposures of 1.16 per month (CI = 0.570, 1.802; P < 0.001).
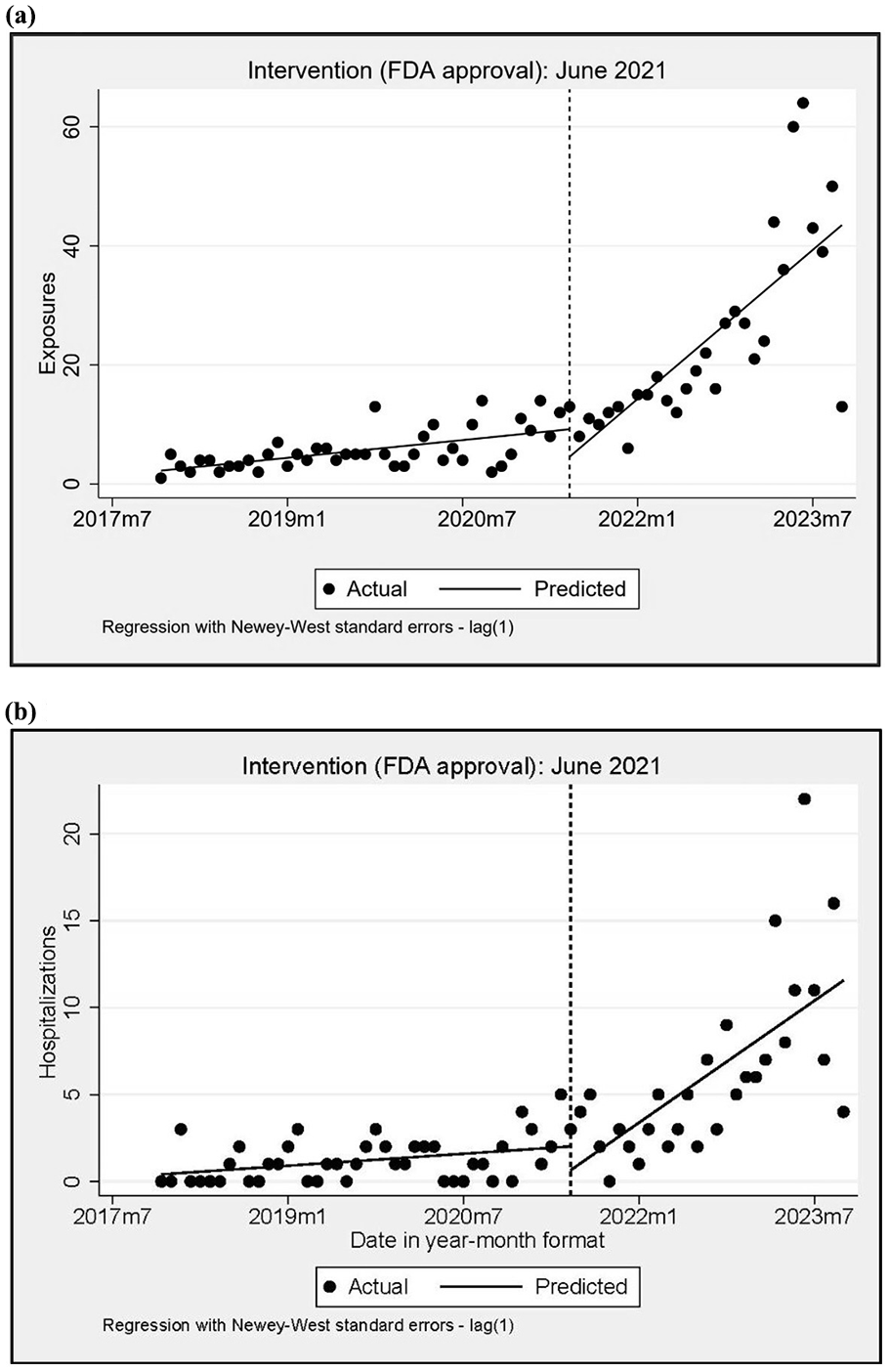
Interrupted time series analysis of GLP-1 receptor agonist exposures (a) and hospital utilization (b) in California after the FDA-approved Wegovy for weight management in June 2021. (a) Exposures. (b) Hospital utilization.
We examined the rate of hospital utilization related to GLP-1 RA exposures before and after the FDA approval of Wegovy for weight loss in June 2021 using ITSA (see Figure 1b). Exposures involving hospitalization in 2017 were estimated at 0.406 per month and appeared to increase in the months prior to approval by 0.038 per month (CI = 0.003, 0.073; P = 0.032). One month after approval and through 2023, reported exposures involving hospital utilization increased significantly by 0.351 per month (CI = 0.159, 0.544; P = 0.001).
Of the 1047 cases, 996 (95.1%) involved sole GLP-1 RA exposures, and 51 (4.9%) were GLP-1 RA & polysubstance exposures. Semaglutide was the most frequently reported exposure (64.6%), followed by liraglutide (17.8%), and dulaglutide (15.7%). We identified 36 compounded GLP-1 RA exposures, which all involved semaglutide and were sole GLP-1 RA exposures (see Supplement, Table S1).
Unintentional therapeutic error was the most common reason for exposure (n = 838, 80.0%), followed by adverse reaction (n = 95, 9.1%), and intentional misuse (n = 40, 3.8%). Glucagon-like peptide-1 receptor agonist indication was reported in 338 (32.3%) cases; of these, 54.4% reported use for weight loss and 45.6% reported use for diabetes mellitus. The most frequent unintentional therapeutic error scenarios were GLP-1 RA inadvertently administered twice (n = 313, 29.9%) (eg, administered a double dose); other dosing errors (n = 276, 32.9%) (eg, misreading the syringe or pen during administration); followed by dose received too close together (n = 76, 7.3%) (eg, administered daily instead of weekly).
The most reported clinical effects (see Table 1) were nausea (n = 295, 28%), vomiting (n = 267, 25.5%), dizziness (n = 63, 6.0%), other gastrointestinal symptoms that included constipation, diarrhea, and bloating (n = 60, 5.7%), and abdominal pain (n = 54, 5.1%). Sole GLP-1 RA exposures had a greater risk for nausea (RR=2.47; 95% CI = 1.16-5.26) and vomiting (RR=6.78; 95% CI = 1.74-26.49), whereas GLP-1 RA and polysubstance exposures had a greater risk for central nervous system (CNS) effects (RR=0.07; 95% CI = 0.03-0.14) and hypoglycemia (RR=0.06; 95% CI = 0.04-0.11).
Clinical effects and medical outcomes.
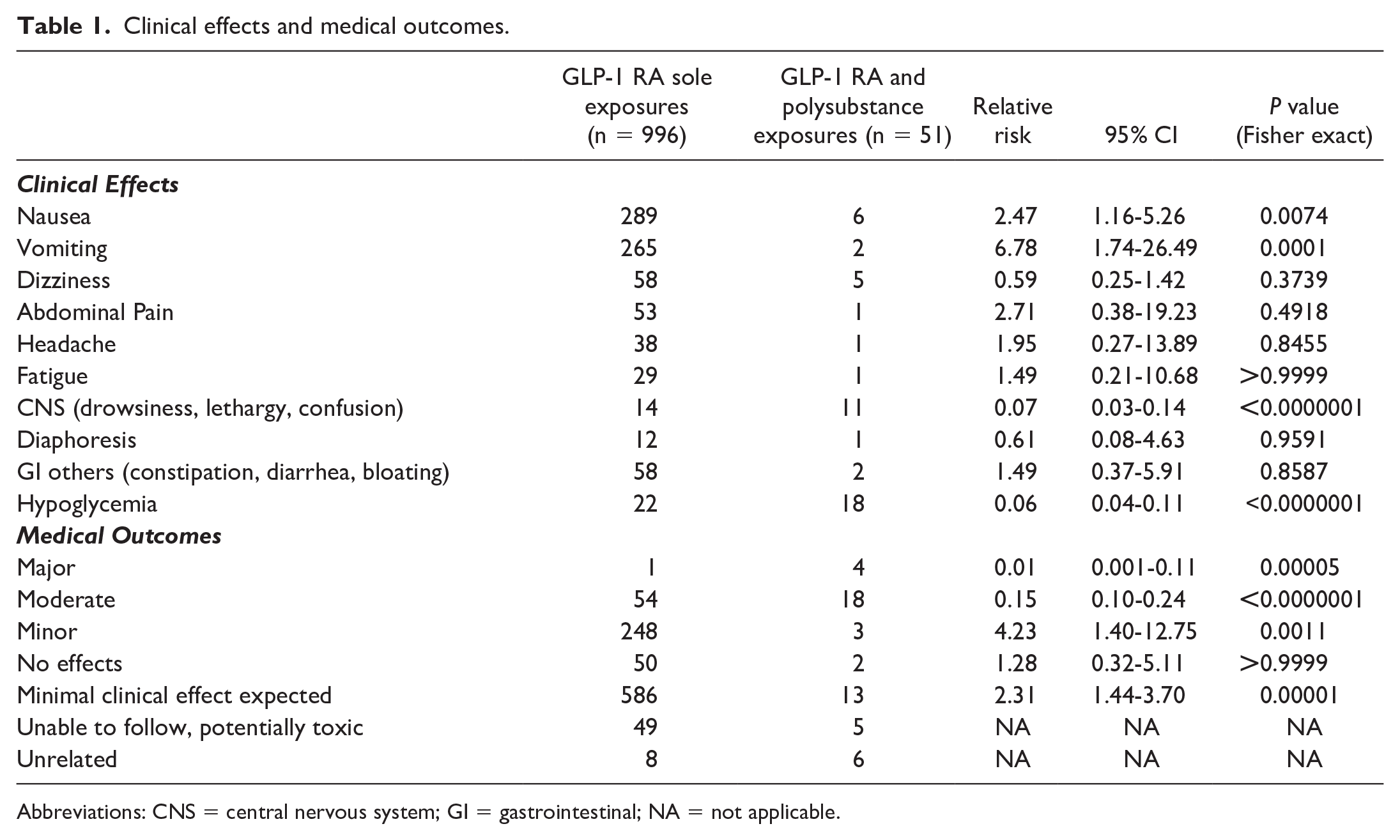
Abbreviations: CNS = central nervous system; GI = gastrointestinal; NA = not applicable.
No deaths were reported in our study and most patients experienced mild outcomes. The coded medical outcomes were as follows: 57.2% (n = 599) minimal effects expected, 24.0% (n = 251) minor effects, and 5.0% (n = 52) no effects (see Table 1). Moderate and major (case narratives in Supplement, Table S2) outcomes were 6.9% (n = 72) and 0.5% (n = 5), respectively, and there was no significant difference in the risk for these outcomes before (n = 20) and after (n = 57) Wegovy approval (RR 1.23; 95% CI = 0.75-1.99, P = 0.4933). However, GLP-1 RA and polysubstance exposures had a greater risk for major (RR=0.01; 95% CI = 0.001-0.11) and moderate (RR=0.15; 95% CI = 0.10-0.24) medical outcomes compared with GLP-1 RA sole exposures.
Hypoglycemia related to GLP-1 RA exposures occurred in 40 (3.8%) cases, and the lowest reported blood glucose level was 20 mg/dL. Within this cohort, 22 (55.0%) cases were GLP-1 RA sole exposures, and 19 (47.5%) cases had self-reported indications for GLP-1 RA use (diabetes n = 16, weight loss n = 3). Among the sole GLP-1 RA exposure, 7 (31.8%) cases reported concomitant use of other antidiabetic agents (insulin, metformin, and empagliflozin); unintentional therapeutic errors occurred in 11 (50.0%) cases and 2 reported the use of a compounded GLP-1 RA product. All 18 (46.2%) cases with GLP-1 RA & polysubstance exposures involved an antidiabetic agent; these included insulins, sulfonylurea, metformin, glitazones, and empagliflozin. Thirty-one (77.5%) of the cases in this subgroup involved medical outcomes classified as moderate and 16 (41%) were admitted to noncritical care units (see Table 2). The was no difference in the risk for hypoglycemia before (n = 13) and after (n = 27) Wegovy approval (RR = 1.68, 95% CI = 0.88-3.21, P = 0.1705).
Characterization of GLP-1 RA Exposure Cases With Hypoglycemia.
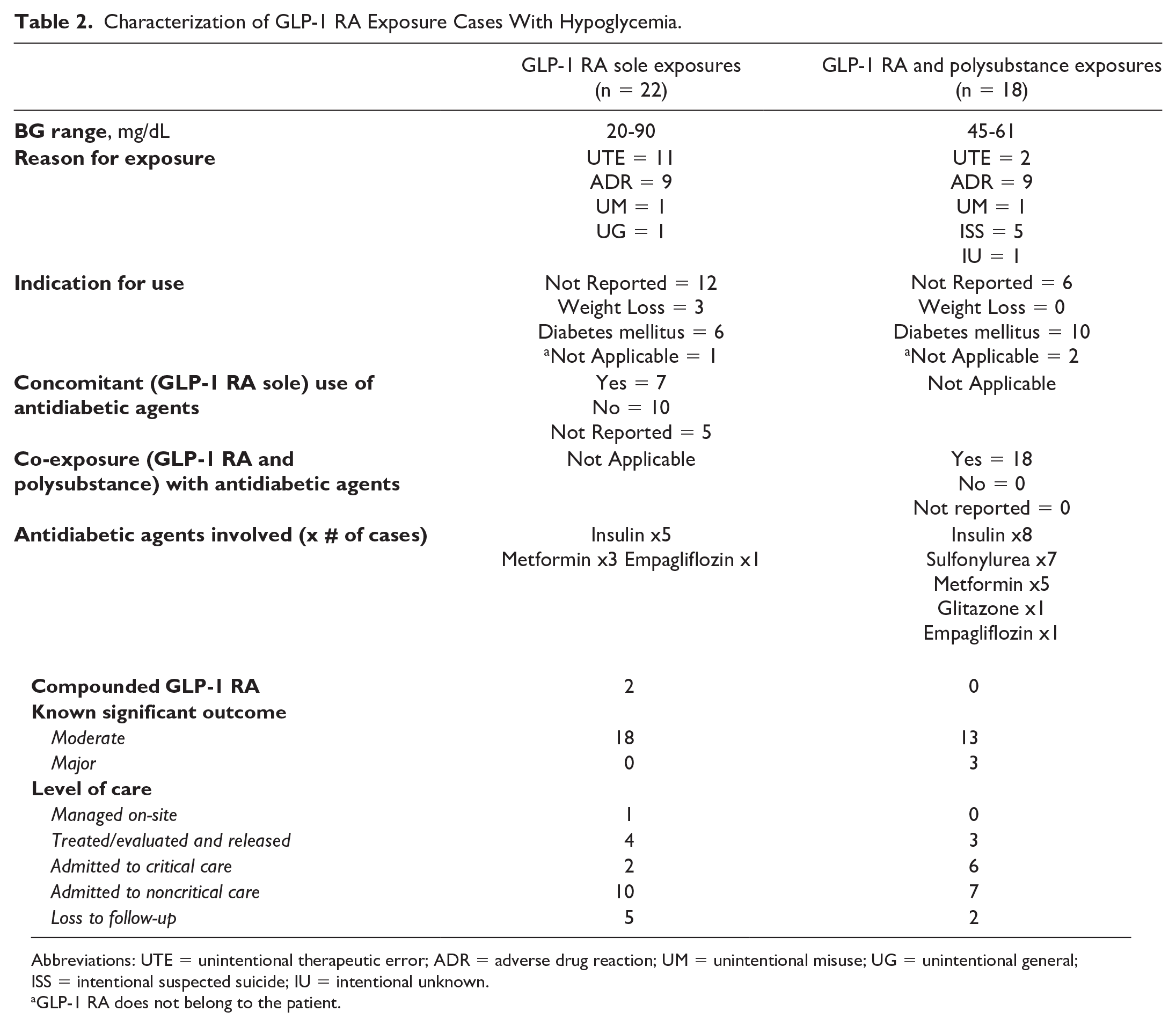
Abbreviations: UTE = unintentional therapeutic error; ADR = adverse drug reaction; UM = unintentional misuse; UG = unintentional general; ISS = intentional suspected suicide; IU = intentional unknown.
GLP-1 RA does not belong to the patient.
Most of the reported GLP-1 RA exposures (n = 696, 66.5%) were managed on-site; however, 25.4% (n = 266) resulted in hospital utilization. Among the hospitalized, 82.7% (n = 220) were released from the emergency department, 17.3% (n = 46) were admitted, and of those admitted, 1.3% (n = 14) were admitted to a critical care unit (Table 3).
GLP-1 RA Exposure Management Sites.

Thirty-six sole GLP-1 RA exposures involved compounded GLP-1 RA products; none were GLP-1 RA and polysubstance exposures. Among these exposures, 35 (97.2%) cases occurred after Wegovy approval. Analyzing this subgroup, we identified 33 (91.7%) cases involving unintentional therapeutic error, and the most frequent scenarios were other-incorrect-dose (n = 14, 42.4%) and 10-fold dosing error (n = 11, 33.3%), followed by confusion regarding units of measure (n = 5, 15.2%). These compounded GLP-1 RA products have various concentrations and injection volumes reported as opposed to the pre-filled single-dose branded pen injectors. Thirteen of 36 cases (36%) reported the use of compounded GLP-1 RA for weight loss, 2 cases (5.6%) for diabetes mellitus, and 21 cases (58.3%) did not report an indication. Most of the compounded GLP-1 RA exposures (n = 27, 75.0%) reported outcomes classified as mild (minor, no effect, or minimal clinical effect expected); only 2 (5.5%) cases reported moderate outcomes (see case narrative in Supplement, Table S3). Fourteen (38.9%) cases required hospital utilization related to the exposure; 13 (36.0%) cases were evaluated and released from the emergency department, and 1 (2.8%) was admitted to a noncritical care unit for observation.
Pediatric exposures account for 2.9% (n = 30) of the total cases in the study and most involved sole GLP-1 RA (96.7%, n = 29). Fourteen cases involved ages less than 5 years old, 9 cases were 6 to 12 years old, and 6 cases were 13 to 19 years old. Unintentional general (eg, pediatric exploratory exposures) account for 46.7% (n = 14) of cases, followed by unintentional therapeutic error (30.0%, n = 9). Semaglutide was the most frequent GLP-1 RA reported (70.0%, n = 21) with the tablet formulation, Rybelsus accounting for 33.0% (n = 7). Most of the medical outcomes were classified as mild (n = 26, 86.7%) except for one case of moderate outcome.
Discussion
Our study confirmed an increase in GLP-1 RA exposures and hospital utilization from exposures after semaglutide, rebranded as Wegovy, was approved for weight management. Unintentional therapeutic errors persist as the primary reason for exposure with mild medical outcomes, particularly gastrointestinal symptoms, being common in sole GLP-1 RA exposures; these findings are consistent with previous GLP-1 RA exposure studies that examined national trends and data reported by a single poison center.24-26 Serious outcomes (moderate and major) increased significantly with combined GLP-1 RA and polysubstance exposures; this trend persisted before and after Wegovy approval. We hypothesized that alterations in the indication for GLP-1 RA would exert minimal impact on the clinical outcomes observed in cases of overdose. Recent meta-analyses have suggested a risk of pancreatitis, and gallbladder and biliary disease associated with the prolonged use of GLP-1 RA.27,28 However, our data, which specifically examined the acute effects following exposure from overdose, did not identify these adverse outcomes. Although ileus and gastroparesis are serious post-market adverse effects of GLP-1 RA, only one such case was reported in our study.8,29
There was no difference in the overall low incidence of hypoglycemia before or after Wegovy approval; however, hypoglycemia occurred in a small number of sole GLP-1 RA exposures with or without concomitant antidiabetic agent use. Seventeen of the 22 sole GLP-1 RA exposures had documented blood glucose levels of <70 mg/dL; 8 cases reported no concomitant antidiabetic agent use. The infrequent occurrence of hypoglycemia in cases without concomitant use of antidiabetic agents parallels the low frequency of hypoglycemia reported in clinical trials of semaglutide monotherapy; 8 these observations warrant caution with GLP-1 RA use for nondiabetic indications. The underlying mechanism for this hypoglycemia remains undetermined. Our cases frequently included patients experiencing significant nausea and vomiting, accompanied by minimal or absent food intake for prolonged periods. Further research is required to assess the impact of excessive GLP-1 RA exposures on glucose homeostasis during extended fasting or starvation conditions. No hypoglycemia-related seizures were reported, and hypoglycemia resolved with food, dextrose, or both. In contrast, GLP-1 RA and polysubstance exposures involving any antidiabetic agent significantly increase hypoglycemia risk, likely due to synergistic effects (increasing insulin endogenously or exogenously) and impacting glucose homeostasis (inhibiting gluconeogenesis by metformin or decreasing renal glucose reabsorption by empagliflozin).
Most compounded GLP-1 RA exposures occurred after Wegovy approval. There were similar reasons for exposure and medical outcomes compared with the branded GLP-1 RA. A notable distinction was observed in the administration scenario despite the relatively small number of identified cases. We hypothesize that this difference is due to confusion related to the variable concentrations and injection volumes of compounded products or inadequate patient education, as these are often obtained through online sources. 30 As access to these compounded products rises, the implication of such variability remains uncertain.
Pediatric exposures were infrequent, likely attributable to storage, administration, and accessibility of GLP-1 RA (eg, refrigeration and injectable preparation). There was one moderate outcome case involving a 2-year-old male child who self-injected a dulaglutide autoinjector, with glucose nadir to 69 mg/dL, 5 hours later in the emergency department. Also observed, exploratory pediatric exposures were more common in children younger than 6 years old with Rybelsus (semaglutide oral tablet formulation) due to ease of access compared with injectables, warranting further study of its impact in this age group.
Study Limitations
Like all administrative datasets, poison control records have limitations. Some of them can be addressed methodologically; others are discussed below. A retrospective study can identify associations but not causality. Poison control data are known to underestimate exposures due to reliance on self-reporting; 31 self-reported data may also omit key information if callers fail to accurately recognize subjects of concern. Glucagon-like peptide-1 receptor agonist indication and concomitant antidiabetic agent use were unreported in 67.7% and 47.8% of cases, respectively. Moderate and severe outcomes may be underestimated as 5.2% of potentially toxic exposures did not follow poison control recommendations for medical attention. Accurate coding by poison control clinicians was another limitation, which we addressed through a qualitative review of the clinician notes to adjust for discrepancies and misinterpretations in NPDS codes. Laboratory sample collection times were unavailable and missing initial blood glucose readings in some hospitalized cases may underestimate hypoglycemia incidence. An FDA warning on counterfeit Ozempic products containing insulin glargine was issued in late 2023, near our study’s end period.30,32,33 It is unclear whether counterfeit products contributed to our cases, particularly those with hypoglycemia, as callers may not have been aware or disclosed how the GLP-1 RA were obtained.
Conclusion
Glucagon-like peptide-1 receptor agonist exposures and hospital utilization from exposures increased significantly after Wegovy approval by 1.16 and 0.351 exposures per month, respectively. Unintentional therapeutic errors were the dominant reason for exposures with mild outcomes common for sole GLP-1 RA exposures. Hypoglycemia risk was higher with co-exposures involving antidiabetic agents but also observed, though not significant, in sole GLP-1 RA exposures. Our findings highlight the need for stricter regulations of compounded GLP-1 products, better patient education on compounded versus branded products, and increased healthcare provider awareness to educate patients on proper use and monitoring, especially hypoglycemia in nondiabetic patients. Further studies are needed to evaluate the population health impacts of compounded GLP-1 RA.
Supplemental Material
sj-docx-1-pmt-10.1177_87551225251332212 – Supplemental material for Changes in Glucagon-Like Peptide-1 Receptor Agonist (GLP-1 RA) Exposures Following Recent Demand for Weight Management: A Retrospective Review of California Poison Control System Data
Supplemental material, sj-docx-1-pmt-10.1177_87551225251332212 for Changes in Glucagon-Like Peptide-1 Receptor Agonist (GLP-1 RA) Exposures Following Recent Demand for Weight Management: A Retrospective Review of California Poison Control System Data by Raymond Y. Ho, Hsiaoting Regelman, Anita Ma, Shu Yi Lee, Saveena Sandhu, Sarah Shapiro, Justin Lewis, Ben Tsutaoka and Dorie E. Apollonio in Journal of Pharmacy Technology
Footnotes
Acknowledgements
The authors acknowledge Terry Carlson of CPCS for assistance in extracting medical records for review.
Data Availability
The data were previously presented as a poster at the 2024 Annual Meeting of the North American Congress of Clinical Toxicology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
