Abstract
Keywords
Background
Apixaban is an oral anticoagulant used in the primary and secondary prevention of embolic events, such as cerebrovascular accidents as well as venous thromboembolisms.1 -3 Although apixaban is used for these indications, its abrupt discontinuation has also been associated with thromboembolic events, making medication access and adherence a major concern for patients. 1 Apixaban is most commonly prescribed to patients with nonvalvular atrial fibrillation and other risk factors in an effort to prevent an acute ischemic stroke (AIS).1,2 In an analysis conducted on prescribing data in Medicare Part D beneficiaries, the use of apixaban was noted to have increased from 75 948 active patients in 2013 to 7 741 247 patients in 2018. 4
Apixaban exerts its mechanism of action through the inhibition of Factor Xa, one of the rate-limiting steps in the production of thrombin. 5 The drug has a relatively high plasma protein binding of about 87% with an associated volume of distribution (Vd) of 21 L and undergoes biotransformation primarily through oxidative metabolism via cytochrome P450 enzymes CYP3A4/5.5,6 To a lesser extent, apixaban is metabolized by CYP1A2, CYP2C8, CYP2C9, and CYP2C19.5,7,8 Apixaban has a half-life of approximately 12 hours in healthy individuals, which can be prolonged in patients with renal impairment. 5 Renal excretion is responsible for about 27% of the total clearance of apixaban, whereas the majority (73%) is eliminated through other mechanisms, including biliary and intestinal routes.5,7 -9
Plasma exchange is a either a centrifugation or filtration-based treatment modality used to clear endogenous or exogenous molecules from blood plasma and has demonstrated therapeutic use in a wide variety of settings, including the removal of autoimmune-mediated antibodies, cytokine storms, and select toxins. 10 With regard to medication clearance, the ability of plasma exchange to filter a drug is directly related to its physiochemical properties. 11 A positive correlation exists regarding the plasma protein binding capacity of a drug and its ability to be filtered through plasma exchange. Similarly, medications with a low Vd, indicating their propensity to remain in the plasma as opposed to extrapolating out of the vasculature and into tissues, are more likely to be removed via plasma exchange. 11
Given apixaban’s drug properties, it is plausible to assume that it may be readily filtered out of systemic circulation by plasma exchange. The following patient case describes the implications of plasma exchange on the efficacy of apixaban.
Case Report
Written patient consent was obtained with regard to the following case. We describe a 50-year-old African American male patient (162 cm, 105 kg) who presented to our facility with new onset aphasia and right-sided weakness. According to the patient’s spouse, the patient experienced a fall in the restroom and was found to have a left gaze deviation, right-sided weakness, and was generally unresponsive. The patient arrived at our facility via emergency medical service transport and was documented to have a National Institute of Health Stroke Severity (NIHSS) score of 23, indicating a severe ischemic stroke. The patient was intubated for airway protection in the emergency department and sent for emergent imaging. The initial computed topography scan of the head was negative for hemorrhagic changes, although further imaging of the head and neck demonstrated a M1 middle cerebral artery occlusion. As the patient was taking apixaban for his atrial fibrillation for the past 4 years, he was not a candidate for thrombolysis with alteplase; however, he did undergo an emergent thrombectomy, which was successful in restoring perfusion as evidenced by a post-operative Thrombolysis in Cerebral Infarction (TICI) score of 3.
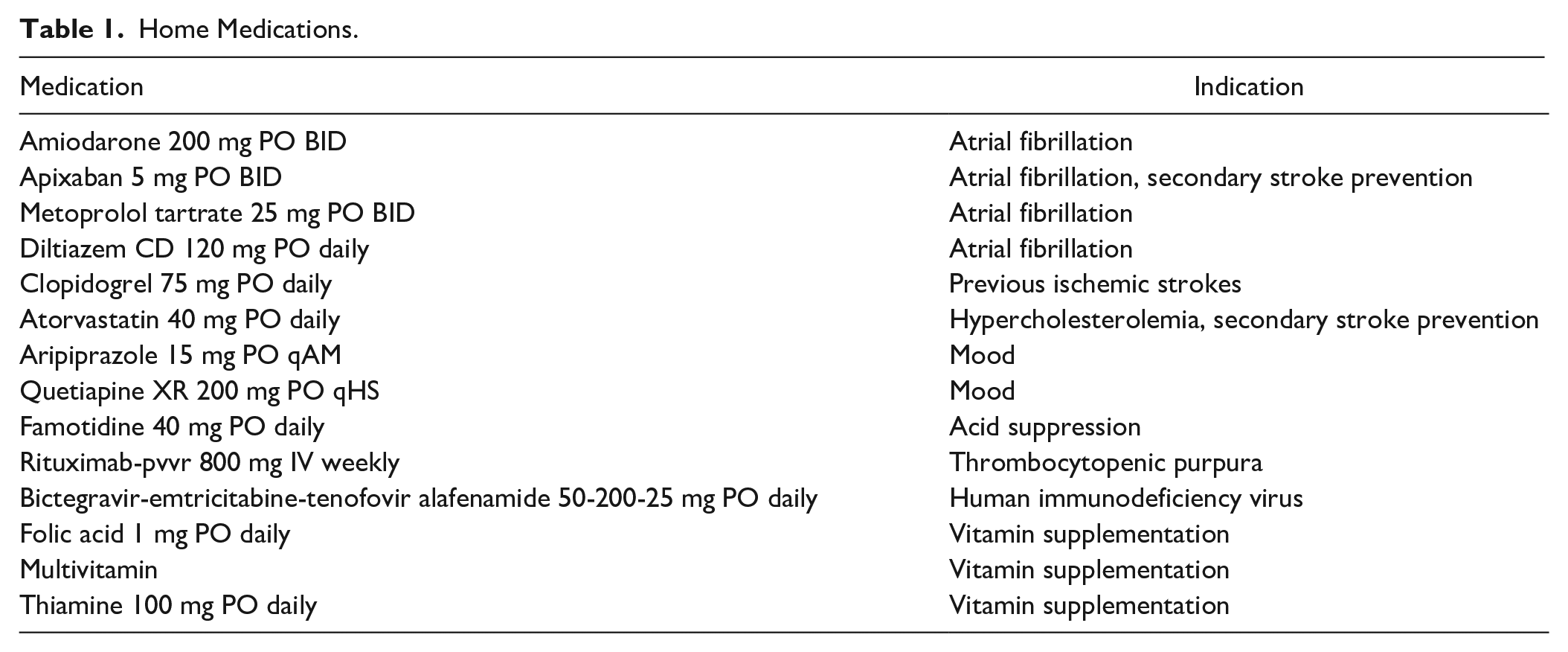
The patient has a significant pertinent past medical history including multiple previous ischemic strokes due to which he suffers from chronic right hand numbness and difficulty with left hand dexterity and required assistance with activities of daily living prior to this admission. In addition, the patient has a history of alcohol and substance abuse, mood disturbances following his previous strokes, hypertension, atrial fibrillation (CHA2DS2-VASc = 3), chronic kidney disease (estimated glomerular filtration rate [eGFR] = 41 mL/min/1.73 m2), obstructive sleep apnea, thrombotic thrombocytopenic purpura (TTP) for which he receives twice weekly plasma exchange in addition to immunomodulatory medication therapy, and is human immunodeficiency virus positive. A complete list of medications can be found in Table 1.
Home Medications.
A myriad of imaging and laboratory tests were obtained on the patient’s presentation to the emergency department, which surprisingly led to the discovery that the patient’s international normalized ratio (INR) was normal despite the patient being on therapeutic anticoagulation with apixaban. Although anti-factor Xa levels were not ordered and are only available at our facility via send out, and the fact that apixaban may only marginally increase a patient’s INR and prothrombin time (PT) levels, concern was raised over the lack of anticoagulation markers in the patient. A conversation with the patient’s wife revealed that the patient had undergone a 4-hour plasma exchange session within the previous 48 hours and that she occasionally forgets to administer her husband’s evening dose of apixaban. It was learned that the patient underwent plasma exchange via a Spectra Optia Apheresis System and was anticoagulated with citrate dextrose solution. The patient received approximately 4200 mL of plasma per exchange. In reviewing records from the outpatient plasma exchanges, it appears as though the intention was to continue with plasma exchanges until the patient became stable on his rituximab therapy. Pertinent baseline laboratory results are included in Table 2.
Admission Laboratory Results.
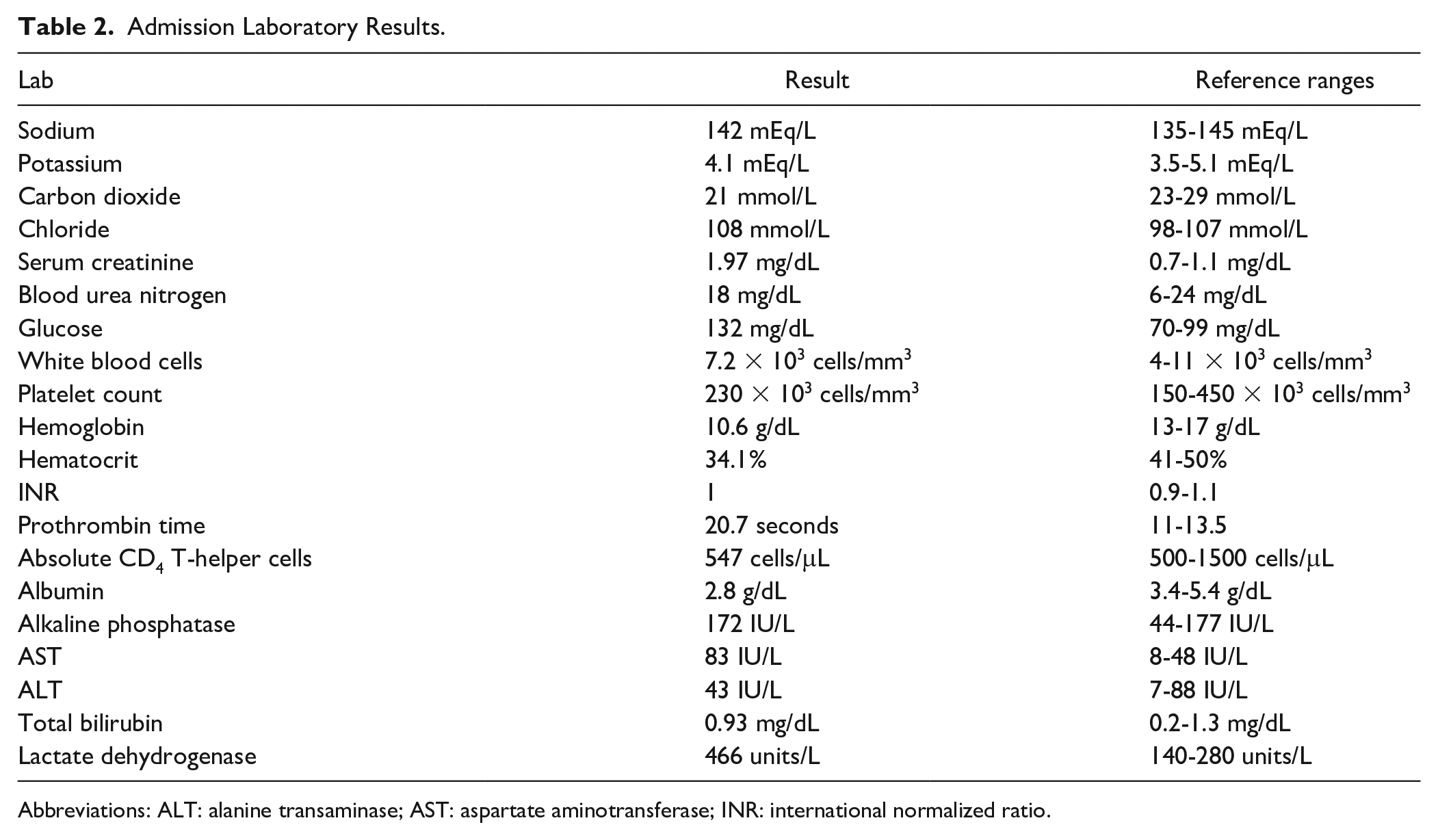
Abbreviations: ALT: alanine transaminase; AST: aspartate aminotransferase; INR: international normalized ratio.
The patient experienced a complicated 2-week hospital course marked by an 80% reduction in his baseline absolute CD4 T-helper cell count, concerns for pneumonia and a gastroenteric infection, drug-induced liver injury associated with anidulafungin, the placement of a percutaneous endoscopic gastrostomy tube, and poor ventilator compliance. Ultimately, the patient’s family made the decision to transfer the patient to another facility in hopes of receiving a higher level of care.
Discussion
The patient’s presentation of AIS despite appropriate dosing of therapeutic oral anticoagulation with apixaban arises the concern of clearance by plasma exchange. With a history of TTP, the patient underwent scheduled therapeutic plasma exchange twice weekly for 4 hours and received approximately 4200 mL of plasma with each exchange. The patient received total plasma exchange (TPE) approximately 24 hours prior to admission. The Naranjo Adverse Drug Reaction Probability score for this case was assessed as 3, suggesting that it is “possible” that the adverse event was attributable to the removal of apixaban via plasma exchange. 12
There is a multitude of evidence demonstrating the risks of thrombosis with abrupt cessation of anticoagulation, which may occur with drug clearance by TPE.13,14A systematic review conducted by the fact that the AIS occurred shortly after TPE, the high NIHSS score, and confirmatory imaging all contribute to the possibility of elimination of apixaban by TPE. Factors that could also influence risk of AIS that were unable to be assessed include patient adherence to medications and timing of oral anticoagulation in regard to plasma exchange. There were no prior admission medication interactions to our knowledge that decreased serum concentrations of apixaban. Of note, the patient’s history of TTP is prothrombotic in nature and is associated with an increased risk of AIS. 15
A critical review by Cheng and colleagues describes medication-related properties that increase risk of drug removal by plasma exchange: high protein binding and low Vd. 16 Both characteristics increase the amount of time that drug will remain in the plasma, thereby increasing risk of drug removal during TPE. Other medication-related properties include half-life and endogenous clearance rates. Of the amount of apixaban absorbed into circulation, approximately 87% is bound by plasma proteins. 13 In addition, the Vd is only approximately 21 L. 13 Both characteristics suggest that apixaban is vulnerable to drug removal during TPE. Limited data exist describing the effect of plasma exchange on drug removal.
Only one other case report exists for the removal of apixaban during TPE. The case report, written by Lam and colleagues, describes the use of TPE for apixaban reversal due to hemorrhagic tamponade requiring emergent intervention. 17 They measured an anti-Xa (aXa) level, calibrated for low-molecular weight heparins (LMWHs), 23 hours from the last dose of apixaban but prior to TPE, which was elevated at 0.84 IU/mL. The patient underwent 1 plasma volume exchange over the course of 2 hours, receiving 3 L of fresh frozen plasma with calcium replacement. Directly after TPE, and 31 hours post-apixaban dose, another aXa level was taken and found to be 0.35 IU/mL. An aXa level was also taken of the plasma waste which was found to be 0.82 IU/mL. Unlike the Lam et al report, there are no aXa data in our patient case to support the possibility of drug removal from TPE; however, the patient in their case report only received 2 hours of apheresis.
Despite multiple factors playing a role in the development of AIS in this patient, subtherapeutic levels of apixaban due to clearance by routine plasma exchange is a plausible differential. Given the pharmacokinetic parameters of apixaban, mechanism of plasma exchange and available literature, it is likely that apixaban is removed by plasma exchange but to what degree is uncertain. Another factor to consider is the patient’s morbid obesity with a body mass index (BMI) of 40 kg/m2. Regarding direct oral anticoagulants (DOACs) in obese patients, there are concerns of reduced peak concentrations and decreased drug exposure. Despite current literature suggesting DOACs are efficacious in patients with high BMI and weight, there is significantly less evidence with apixaban compared with rivaroxaban.18,19 Although not routinely recommended, patients receiving plasma exchange may benefit from aXa-apixaban level monitoring to ensure drug is detectable at therapeutic level. It is important to note, however, that with aXa-apixaban levels, there are no standard laboratory therapeutic ranges, and thus no recommendations for dose adjustments. 20 In the patient case, aXa-unfractionated heparin (UFH) and aXa-LMWH were not obtained which could have potentially been used to detect the presence of apixaban but would not be useful in quantifying the therapeutic amount of apixaban in circulation. Other clot-based coagulation parameters including activated partial thromboplastin time (aPTT) and PT/INR are influenced depending on the type of oral anticoagulant, timing of dose, and reagents used. 21 Elevation of PT/INR and aPTT has been seen in patients with apixaban levels as low as 200 ng/mL and during peak onset of 3 to 4 hours. 22
A cofounding variable that warrants discussion are the multiple interacting medications that the patient was receiving at the time of the AIS. Although not all may be clinically relevant, one of potentially clinical significance is the interaction between diltiazem and clopidogrel.23,24 A review of the literature reveals that the efficacy of clopidogrel may be diminished when administered with concomitant calcium channel blockers, although only a small percentage of patients were receiving diltiazem.23,24 The anticipated cause of this interaction stems from known substrate and inhibitor activity of many calcium channel blockers for CYP3A4, a minor metabolic activation pathway for clopidogrel.23,24 Although the clinical significance of this interaction has not been unequivocally quantified, it remains possible that it could have played a small part in this patient case.
This case describes a patient with a history of atrial fibrillation on apixaban, prior ischemic strokes, and TTP requiring therapeutic plasma exchange presenting with an AIS. Considering the onset of symptoms and timing of plasma exchange, it is possible that apixaban extraction by plasma exchange contributed to the AIS. Despite the correlation between events discussed in this case, many limitations exist that deserve attention. The absence of available laboratory values that could otherwise support or refute our interpretation of data present the largest detraction, especially the lack of aXa levels. Platelet levels were normal during admission, suggesting no relapse of TTP, although no ADAMTS13 was obtained to confirm. Given the patient’s PT/INR was not prolonged, we cannot deduce or exclude the presence of apixaban in our patient. The inability to confirm patient adherence to apixaban therapy as an outpatient also presents a significant detraction. Finally, the only other case report published on apixaban removal by TPE described a patient who received a 2-hour apheresis session, as opposed to our patient who received 4 hours. These highlight the need for further investigation of drug clearance, specifically DOACs, in plasma exchange.
Conclusions
A paucity of data exists regarding the centrifugation and filtration of medications via plasma exchange, with even more uncertainly surrounding the clinical ramifications on drug efficacy and safety. It is imperative that clinicians correlate known medication pharmacokinetic and pharmacodynamic properties with the duration and frequency of plasma exchanges to make informed inferences as to the anticipated drug clearance. In the case of apixaban, it may be advisable to consider pre-exchange and post-exchange therapeutic drug monitoring; however, the applicability of this theorem to clinical practice has not yet been elucidated in the literature.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
