Abstract
Background
Post-COVID-19 fatigue is the most prevalent symptom among people who have overcome the disease, whether they were hospitalized or not. The aim was to analyze fatigue in subjects who had experienced COVID-19, with or without hospitalization, compared to their pre-infection status. We also sought to examine the relationship between descriptive characteristics of subjects and various holistic health indicators with the presence or absence of fatigue, as well as the fatigue severity.
Methods
A cross-sectional observational study was carried out following the Strengthening the Reporting of Observational Studies in Epidemiology guideline. Subjects were classified into 2 groups: hospitalized and non-hospitalized. Both groups were divided into 2 subgroups based on the absence or presence of fatigue: non-fatigue and fatigue. Descriptive characteristics, fatigue before versus after COVID-19, and holistic health indicators were measured. Correlation and regression analyses were conducted.
Results
A total of 262 subjects were divided into hospitalized (n = 70) and non-hospitalized (n = 192) groups and non-fatigue and fatigue subgroups. Results showed increased fatigue after COVID-19 in both groups, with hospitalized subjects experiencing more fatigue (p < .001). Before COVID-19, neither group exceeds the cut-off point for fatigue, but after the disease, the hospitalized group surpassed it. Hospitalization and body mass index (BMI) emerged as key predictors of the presence of post-COVID-19 fatigue. In addition, post-COVID-19 fatigue was associated with poorer quality of life, health status, functionality, and physical activity, as well as greater frailty, anxiety, depression, and dysphagia. Functionality and frailty were the most relevant predictors of post-COVID-19 fatigue.
Conclusions
These findings underscore the need for personalized rehabilitation strategies that address both descriptive characteristics and holistic health indicators. However, further studies with larger sample sizes are recommended to validate and expand upon these results.
Post-COVID-19 fatigue is the most prevalent symptom in subjects who have recovered from the infection, regardless of hospitalization. In addition, functionality, physical activity, frailty, anxiety, depression, and swallowing difficulties may also be affected. Together, these factors contribute to a marked decline in quality of life. Fatigue levels increased significantly after COVID-19 in both hospitalized and non-hospitalized subjects, with greater severity in the hospitalized group. Hospitalization and body mass index (BMI) emerged as key predictors of post-COVID-19 fatigue. The severity of post-COVID-19 fatigue was linked to poorer quality of life, health status, functionality, and physical activity, as well as to greater frailty, anxiety, depression, and dysphagia. Among these factors, functionality and frailty were the most relevant predictors.Quick Look
Current knowledge
What this paper contributes to our knowledge
Introduction
COVID-19 is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). 1 Symptoms severity varies: some individuals are asymptomatic, 2 while symptomatic cases range from mild (40%) and moderate (40%)—not requiring hospitalization—to severe (15%) and critical (5%), which involve hospitalization.3,4 The most common COVID-19 symptoms are fatigue, cough, fever, ageusia, anosmia, and dyspnea.5,6 Symptoms persistence varies with disease severity, 7 occurring in 30% of non-hospitalized and 80% of hospitalized patients. 8 Post-COVID-19 fatigue is the most prevalent persistent symptom, especially in hospitalized subjects. 5
Post-COVID-19 fatigue is defined as a decrease in mental and/or physical performance caused by central, psychological, and/or peripheral factors related to COVID-19. 9 Central factors may involve central nervous system invasion by SARS-CoV-2, 10 affecting neurotransmitters, inflammation, demyelination, and neuronal excitability.9,11 Psychological factors can also contribute to the appearance or exacerbation of post-COVID-19 fatigue. 12 Peripheral factors arise from COVID-19 infecting multiple tissues, either directly via angiotensin 2–rich cells or indirectly through cytokine release and homeostatic disruption.12,13 Fatigue, whether central, psychological, or peripheral, is the most prevalent post-COVID-19 symptom. 14 Other affected areas include functionality, physical activity, frailty, anxiety, depression, and dysphagia, reducing quality of life.15–18
Therefore, an integrated approach is needed to better understand post-COVID-19 fatigue. This study addresses the gap in research by comparing pre- and post-infection fatigue and examining the combined influence of subject characteristics and holistic health indicators on fatigue in both hospitalized and non-hospitalized subjects. The aim was to analyze fatigue in subjects who had experienced COVID-19, with or without hospitalization, compared to their pre-infection status. We also sought to examine the relationship between subjects’ characteristics and various holistic health indicators with the presence or absence of fatigue, as well as the fatigue severity.
Methods
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline was used to conduct this cross-sectional observational study. The study protocol was registered on ClinicalTrials.gov (NCT05731817) and approved by the Biomedical Research Ethics Committee of Andalusia, Spain. It was conducted in accordance with the 1975 Helsinki Declaration, and all subjects provided informed consent.
From February 2022 to June 2023, subjects were identified, recruited, and evaluated virtually by the University of Granada research team. Participants were divided into hospitalized and non-hospitalized groups. Hospitalized subjects were recruited via the Pneumology Department of Hospital Universitario Virgen de las Nieves; pulmonologists informed patients before discharge, and interested individuals gave consent to share their contact with the research team. Non-hospitalized subjects were recruited using snowball sampling, starting with participants from the researchers’ personal and professional circles. Inclusion criteria: adults ≥18 years, access to a compatible device and stable internet, basic Google Meet skills, willingness to participate, informed consent, confirmed COVID-19 diagnosis, and recovery from acute infection (or hospital discharge for hospitalized subjects). Exclusion criteria: serious comorbidities, mental/physical/organic problems posing risk, or prior participation in rehabilitation programs between COVID-19 diagnosis and study evaluation, in order to assess the natural course of fatigue without the potential confounding effects of prior therapeutic interventions. The sample size was based on participant availability, without a prospective calculation for the primary endpoint. 19
Recruited subjects received an initial telephone call explaining the study, discussing inclusion/exclusion criteria, and answering questions. Those who agreed gave initial consent and scheduled the assessment. A specific date was arranged, and a Google Meet link was provided with instructions. Each evaluation lasted 1–1.5 h. Data were stored in a secure Microsoft Excel database, accessible only to authorized researchers. Assessment timing depended on clinical status: hospitalized subjects were assessed after discharge and at least 1 week after a negative COVID-19 test, while non-hospitalized subjects were assessed at least 1 week after a negative test.
The variables related to the descriptive characteristics of subjects were as follows and were measured using the specified instruments. Age, sex, height, weight, BMI, and days between COVID-19 diagnosis and assessment date were collected using an online form. Comorbidities were assessed with the Charlson Comorbidity Index, which predicts short-term mortality risk. 20 Classification: 0–1, no comorbidity; 2, low; ≥3, high. Annual mortality (<3 years): 0 (12%), 1–2 (26%), 3–4 (52%), and ≥5 (85%). 21
Fatigue, the primary outcome, was assessed using the Modified Borg Scale and the Fatigue Severity Scale (FSS). The Modified Borg Scale measured perceived fatigue on a 0 (no fatigue) to 10 (extreme fatigue) scale. 22 Subjects were asked to rate their fatigue in different contexts: at rest and during activities of varying intensities. Light activities included walking on level surfaces, slow strolling, or walking at approximately 2.7 km/h; moderate activities comprised Pilates, yoga, or aquatic exercises; and intense activities involved jogging, running in place, or rope skipping. Ratings were based on their usual experiences.23–26 The FSS is a 9-item, 7-point Likert questionnaire, with fatigue threshold at ≥36 or mean ≥4. All subjects rated fatigue before and after COVID-19, with pre-COVID levels obtained by recall-based self-assessment. 27
Secondary outcomes included a range of holistic health indicators: quality of life, functionality, frailty, anxiety and depression, dysphagia, and physical activity. Quality of life was measured with the European Quality of Life-5-Dimensional Questionnaire (EuroQoL-5D), divided into 2 sections. The first assesses mobility, self-care, daily activities, pain/discomfort, and anxiety/depression, with severity scored 1 (no problems), 2 (some), and 3 (many). The second is a Visual Analogue Scale from 0 (worst health) to 100 (best health). 28 Functionality was assessed with the Functional Independence Measure (FIM), the Post-COVID-19 Functional Status Scale (PCFS), and the Patient-Specific Functional Scale (PSFS). The FIM has 18 items, scored 18–126 (higher = more independence). 29 The PCFS grades post-COVID functional limitations 0–4 (none to severe). 30 The PSFS asks subjects to rate 3 difficult activities from 0 (disability) to 10 (full ability), with the final score as the average. 31 Frailty was measured with the FRAIL frailty scale (FRAIL), which includes 5 components: fatigue, resistance, walking, illness, and weight loss. Scores range 0 (best) to 5 (worst), classifying subjects as robust (0), prefrail (1–2), or frail (3–5). 32 Anxiety and depression were assessed with the Hospital Anxiety and Depression Scale (HADS), which has 14 items divided into anxiety and depression. Total scores range 0–42, with higher scores indicating greater anxiety or depression. A score of ≥13 indicates the presence of anxiety or depression. 33 Dysphagia was evaluated using the Eating Assessment Tool-10 (EAT-10), a 10-item questionnaire rated 0 (no problem) to 4 (severe problem). Higher scores indicate greater swallowing difficulty, and a total score of ≥3 suggests potential issues with safe and effective swallowing. 34 Finally, physical activity was measured with the International Physical Activity Questionnaire (IPAQ). Subjects were classified as low/inactive (category 1), moderate (category 2), or high (category 3). 35
Statistical analyses were performed with SPSS 28.0. Normality was checked visually (histograms, Q-Q plots) and analytically (Kolmogorov-Smirnov test, n ≥ 30). Continuous data were expressed as mean (SD)–normal or median (interquartile range (IQR))–non-normal. Depending on normality, parametric or non-parametric tests were used. Although some variables did not show a normal distribution, according to the Central Limit Theorem,36,37 with a large enough sample size (n ≥ 60 in each group), the sampling distribution will be approximately normal. Therefore, the normality was assumed and the Welch test was applied. For the study of fatigue before versus after COVID-19 within groups, Student’s t-test for paired samples or Wilcoxon signed-rank were used as appropriate. Levene’s test checked variance equality. Categorical data were reported as n (%), analyzed with chi-square test. Significance: p < .050. No missing data were present.
For holistic health indicators, groups were divided by post-COVID-19 fatigue into fatigue (FSS ≥ 36) and non-fatigue subgroups. 27 Correlation analysis assessed relationships with FSS scores. Spearman’s rho was used for ordinal variables and/or non-normally distributed variables; Pearson’s r was used for approximately normally distributed continuous variables. Pearson’s r interpretation: 0–0.19 none/slight, 0.20–0.39 low, 0.40–0.69 moderate, 0.70–0.89 high, and 0.90–1 very high.38,39 Multiple linear regression identified subject characteristics and health indicators influencing post-COVID fatigue (β, 95% CI, p-values, R2). Separate models were made for hospitalized and non-hospitalized subjects. Multiple logistic regression identified predictors of fatigue (OR, 95% CI, p-values, R2). Assumptions of linearity, normality, independence, and homoscedasticity were verified. Multicollinearity was assessed via Variance Inflation Factor (VIF) and tolerance.
Results
A total of 346 subjects were screened, with 84 excluded. The final sample included 262 subjects: hospitalized (n = 70) and non-hospitalized (n = 192). Each group was divided into non-fatigue and fatigue subgroups: hospitalized—non-fatigue (n = 25) and fatigue (n = 45); non-hospitalized—non-fatigue (n = 128) and fatigue (n = 64). The flow diagram (Figure S1) is presented in Supplemental File A1.
The descriptive characteristics of hospitalized and non-hospitalized subjects.

Notes. *p < .001. aWelch test p-value; bchi-square test p-value; cStudent’s t-test p-value.
Abbreviations. BMI = body mass index; SD = standard deviation.
Fatigue before versus after COVID-19 in hospitalized and non-hospitalized subjects.
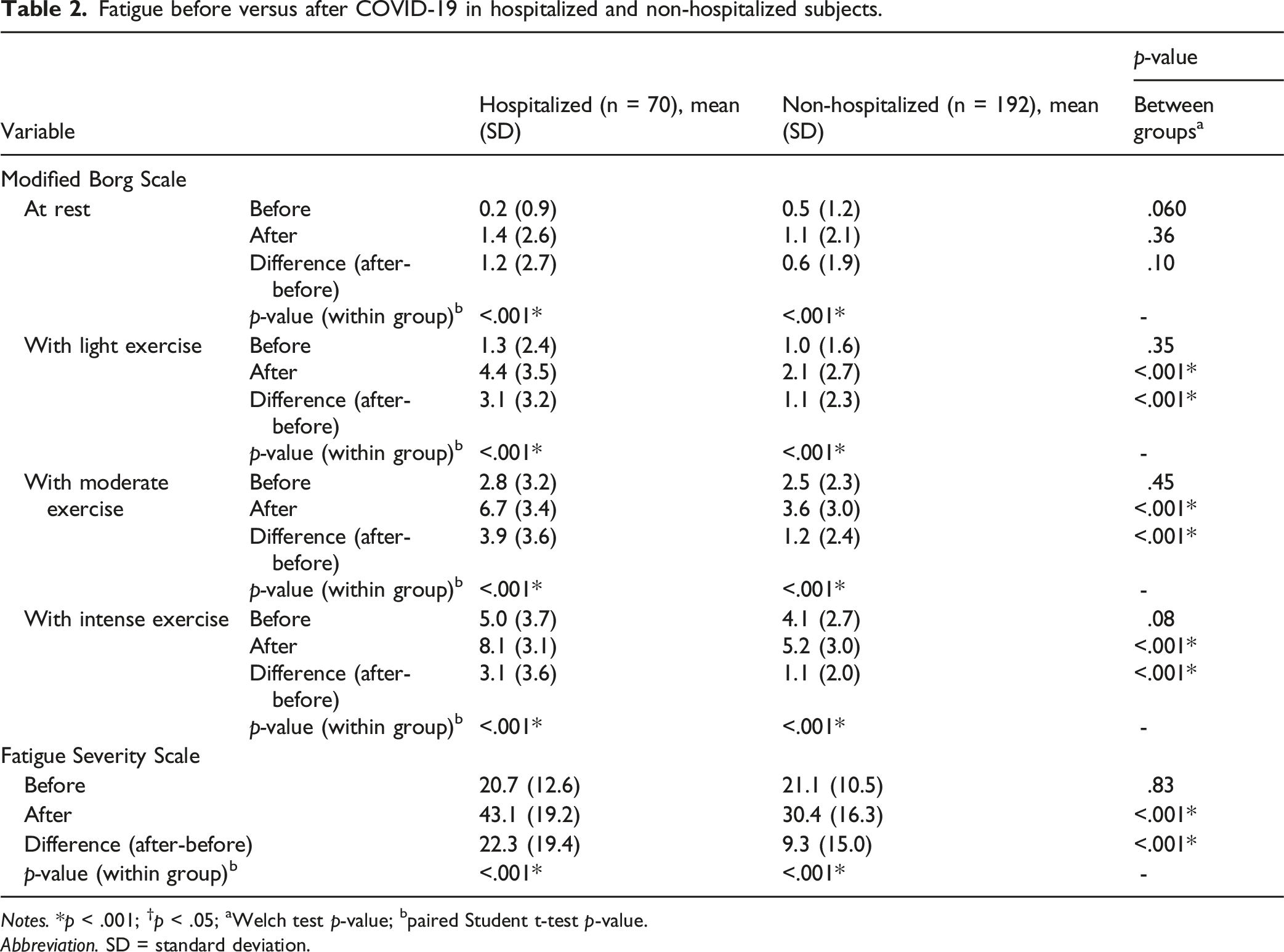
Notes. *p < .001; †p < .05; aWelch test p-value; bpaired Student t-test p-value.
Abbreviation. SD = standard deviation.
Holistic health indicators in hospitalized and non-hospitalized subjects, with or without fatigue.
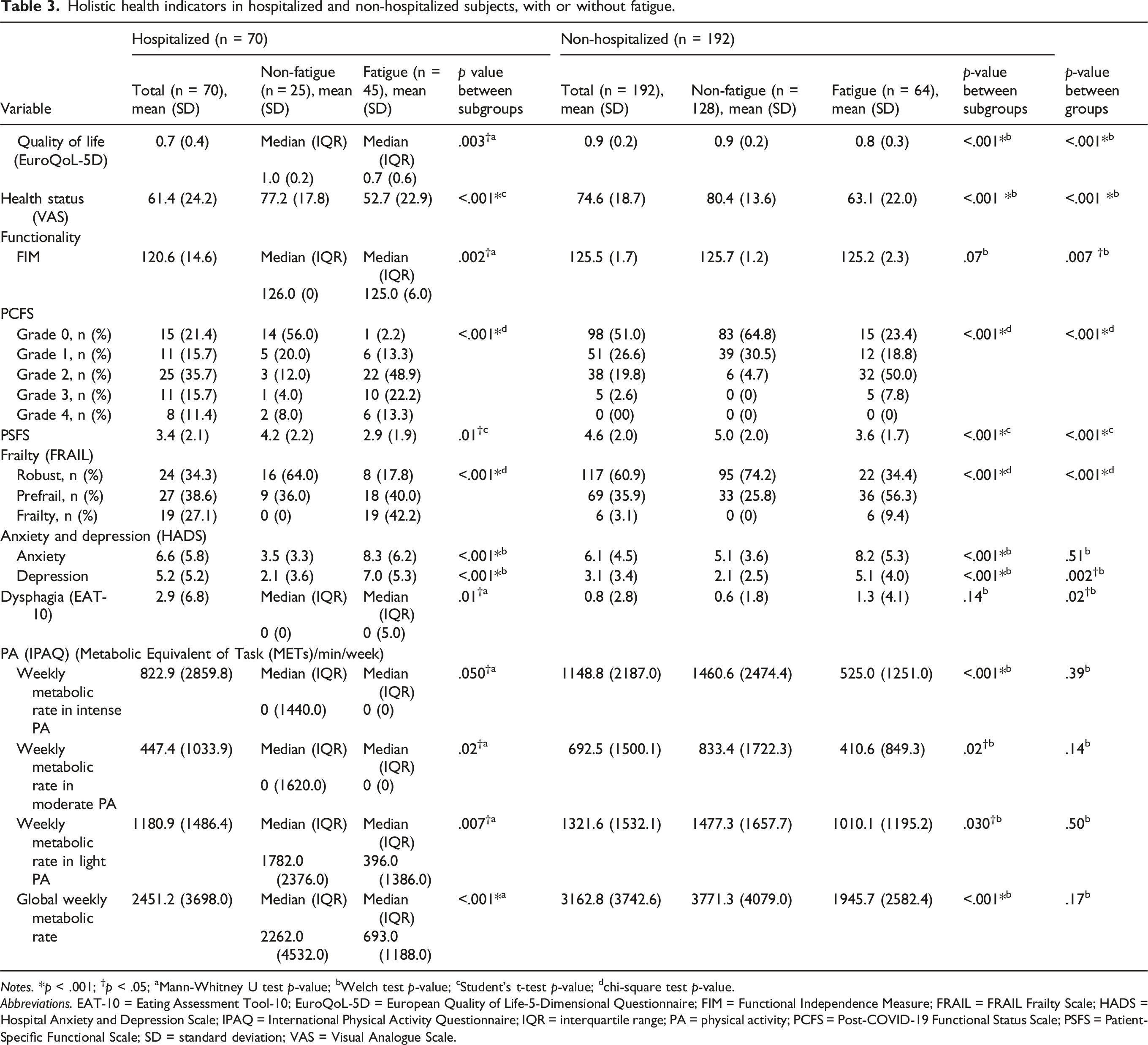
Notes. *p < .001; †p < .05; aMann-Whitney U test p-value; bWelch test p-value; cStudent’s t-test p-value; dchi-square test p-value.
Abbreviations. EAT-10 = Eating Assessment Tool-10; EuroQoL-5D = European Quality of Life-5-Dimensional Questionnaire; FIM = Functional Independence Measure; FRAIL = FRAIL Frailty Scale; HADS = Hospital Anxiety and Depression Scale; IPAQ = International Physical Activity Questionnaire; IQR = interquartile range; PA = physical activity; PCFS = Post-COVID-19 Functional Status Scale; PSFS = Patient-Specific Functional Scale; SD = standard deviation; VAS = Visual Analogue Scale.
The FIM found that subgroups with fatigue had a greater dependency than subgroups without fatigue, in both groups. Differences were significant in the hospitalized group (p = .002). Nevertheless, the non-hospitalized showed a greater independence than the hospitalized group (p = .007). The PCFS revealed that the hospitalized group had a higher percentage of subjects in grades 2, 3, and 4 (greater functional limitation). By contrast, the non-hospitalized had a higher percentage of subjects in grades 0 and 1 (the functional limitation did not exist or was insignificant). Subgroups with fatigue in both groups had a higher percentage of subjects in grades 2, 3, and 4, and subgroups without fatigue had a higher percentage of subjects in grades 0 and 1 (p < .001 between subgroups). Subjects who had been hospitalized and those experiencing fatigue exhibited greater functional limitations. The PSFS showed a greater disability in subgroups with fatigue compared to subgroups without fatigue in both groups. Differences between subgroups were significant (p = .01 and p < .001, respectively). The disability was greater in hospitalized compared to non-hospitalized subjects (p < .001). Regarding frailty, significant differences were observed between groups (p < .001). In the hospitalized group, 27.1% of subjects were categorized as frailty. In contrast, within the non-hospitalized group, this percentage was significantly lower, at 3.1%. Similarly, the percentage of robust subjects differed considerably between groups (34.3% vs 60.9%). The HADS revealed that, in both groups, subgroups with fatigue showed higher levels of anxiety and depression than subgroups without fatigue. Significant differences were observed between subgroups for anxiety and depression (p < .001). The hospitalized group showed higher anxiety and depression levels than the non-hospitalized. Significant differences between groups were only showed for depression (p = .002).
The EAT-10 showed greater difficulty swallowing in subgroups with fatigue compared to those without fatigue, both in hospitalized and non-hospitalized. Differences between the fatigue and non-fatigue subgroups were significant in the hospitalized group (p = .01). However, only subjects who had been hospitalized with COVID-19 and who presented fatigue exceeded the cut-off point for dysphagia. Consequently, it can be concluded that subjects who had been hospitalized and experienced fatigue had the greatest difficulty swallowing.
Finally, subgroups with fatigue in both groups reported lower physical activity compared to subgroups without fatigue. Differences between subgroups were significant (p < .050) in hospitalized and non-hospitalized. The non-hospitalized group showed higher levels of physical activity compared to the hospitalized. This emphasizes the impact of both fatigue and hospitalization on physical activity levels. However, non-significant differences were observed between hospitalized and non-hospitalized.
The correlation analysis between post-COVID-19 fatigue severity (FSS scores) and the study variables are presented in Supplemental File A2, Table S1. Regarding subjects’ characteristics, correlation analysis indicated a significant correlation between fatigue and age (rho = .22, p < .001), indicating that higher levels of fatigue were correlated with higher age. Similarly, a significant correlation was observed between fatigue and BMI (rho = .31, p < .001), between fatigue and hospitalization (rho = .29, p < .001), and between fatigue and comorbidities (rho = .17, p = .005). Higher levels of fatigue were correlated with higher BMI and with more comorbidities. Hospitalized subjects tended to report higher levels of fatigue than non-hospitalized subjects did. However, there was no correlation between fatigue and sex (rho = −.01, p = .92).
In hospitalized subjects, fatigue was significantly correlated with BMI (rho = .41, p < .001), indicating that higher levels of fatigue were correlated with higher BMI. In addition, fatigue and sex were significantly correlated (r = .26, p = .032). Women tended to report higher levels of fatigue than men did. However, there was no correlation between fatigue and age (r = .08, p = .50). The correlation between fatigue and comorbidities was slight and non-significant (rho = .15, p = .23). In non-hospitalized subjects, fatigue is positively correlated with BMI (r = .19, p = .01). However, there was no correlation between fatigue and comorbidities (rho = −.03, p = .71) or between fatigue and sex (r = .02, p = .76). The correlation between fatigue and age was slight and non-significant (r = .12, p = .10).
Considering the holistic health indicators, there was a significant correlation between fatigue and all of them. On the one hand, fatigue was negatively correlated with quality of life (rho = −.54, p < .001), health status (rho = −.60, p < .001), functionality (FIM: rho = −.36, p < .001; PSFS: rho = −.36, p < .001), and physical activity (rho = −.39, p < .001); indicating that higher levels of fatigue were correlated with lower quality of life, health status, functionality, and physical activity. On the other hand, fatigue was positively correlated with functionality (PCFS: rho = .71, p < .001), frailty (rho = .53, p < .001), anxiety (rho = .39, p < .001), depression (rho = .56, p < .001), and dysphagia (rho = .26, p < .001); indicating that higher levels of fatigue were correlated with higher functional limitation, frailty, anxiety, depression, and dysphagia.
In hospitalized subjects, there was a moderate significant correlation between fatigue and all holistic health indicators. On the one hand, fatigue was negatively correlated with quality of life (rho = −.47, p < .001), health status (rho = −.62, p < .001), functionality (FIM; rho = −.54, p < .001; PSFS: rho = −.40, p < .001), and physical activity (rho = −.48, p < .001); indicating that higher levels of fatigue were correlated with lower quality of life, health status, functionality, and physical activity. On the other hand, fatigue was positively correlated with functionality (PCFS: rho = .62, p < .001), frailty (rho = .66, p < .001), anxiety (rho = .49, p < .001), depression (rho = .62, p < .001), and dysphagia (rho = .47, p < .001); indicating that higher levels of fatigue were correlated with higher functional limitation, frailty, anxiety, depression, and dysphagia. In non-hospitalized subjects, there was a significant correlation between fatigue and holistic health indicators, except with dysphagia. The correlation between fatigue and dysphagia was slight and non-significant (rho = .12, p = .12). On the one hand, fatigue was negatively correlated with quality of life (rho = −.49, p < .001), health status (r = −.57, p < .001), functionality (FIM: rho = −.18, p = .02; PSFS: r = −.31, p < .001), and physical activity (rho = −.29, p < .001); indicating that higher levels of fatigue were correlated with lower quality of life, health status, functionality, and physical activity. On the other hand, fatigue was positively correlated with functionality (PCFS: rho = .65, p < .001), frailty (rho = .41, p < .001), anxiety (r = .41, p < .001), and depression (r = .50, p < .001); indicating that higher levels of fatigue were correlated with higher functional limitation, frailty, anxiety, and depression.
The multiple linear regression analysis of post-COVID-19 fatigue severity is summarized in Supplemental File A3, Table S2. The multiple linear regression model with subjects’ characteristics was statistically significant (p < .001). The model explained 15% of the variance of fatigue severity, indicating that the variance explained on the dependent variable was limited. Accordingly, only BMI (β = .2; p < .001) and hospitalization (β = .2; p = .008) were found to have a significant effect on post-COVID-19 fatigue severity. The multiple linear regression model with the holistic health indicators was statistically significant (p < .001). The model explained 61.3% of the variance of fatigue severity. Accordingly, health status (β = −.1; p = .02), functionality (FIM: β = .2; p < .001; PCFS: β = .5; p < .001), frailty (β = .2; p = .006), anxiety (β = .1; p = .032), and physical activity (β = −.1; p = .01) were found to have a significant effect on post-COVID-19 fatigue severity. The linearity, normality, homoscedasticity, and multicollinearity assumptions were verified. We confirmed the absence of multicollinearity in multiple linear regression model with subjects’ characteristics (tolerance was above 0.4 and all VIFs were below 3) and with holistic health indicators (tolerance was above 0.3 and all VIFs were below 3).
The multiple linear regression analysis of post-COVID-19 fatigue severity in hospitalized subjects is reported in Supplemental File A4, Table S3. The multiple linear regression model with subjects’ characteristics was statistically significant (p = .008). The model explained 16% of the variance of fatigue severity, indicating that the variance explained on the dependent variable was limited. Accordingly, only BMI was found to have a significant effect on post-COVID-19 fatigue severity (β = .3; p = .003). The multiple linear regression model with the holistic health indicators was statistically significant (p < .001). The model explained 58.8% of the variance of fatigue severity. Accordingly, only functionality (PCFS: β = .4; p = .01) and frailty (β = .3; p = .030) were found to have a significant effect on post-COVID-19 fatigue. The linearity, normality, homoscedasticity, and multicollinearity assumptions were verified. We confirmed the absence of multicollinearity in multiple linear regression model with subjects’ characteristics (tolerance was above 0.9 and all VIFs were below 2) and with holistic health indicators (tolerance was above 0.3 and all VIFs were below 4).
The multiple linear regression analysis of post-COVID-19 fatigue severity in non-hospitalized subjects is displayed in Supplemental File A5, Table S4. The multiple linear regression model with subjects’ characteristics was statistically significant (p = .02). The model explained 3.9% of the variance of fatigue severity, indicating that the variance explained on the dependent variable was very limited. Accordingly, only BMI was found to have a significant effect on post-COVID-19 fatigue severity (β = .2; p = .031). The multiple linear regression model with the holistic health indicators was statistically significant (p < .001). The model explained 58.9% of the variance of fatigue severity. Accordingly, health status (β = −.2; p = .007), functionality (FIM: β = .1; p = .045; PCFS: β = .5; p < .001), and physical activity (β = −.1; p = .01) were found to have a significant effect on post-COVID-19 fatigue. The linearity, normality, homoscedasticity, and multicollinearity assumptions were verified. We confirmed the absence of multicollinearity in multiple linear regression model with subjects’ characteristics (tolerance was above 0.9 and all VIFs were below 2) and with holistic health indicators (tolerance was above 0.4 and all VIFs were below 3).
The multiple logistic regression analysis is presented in Supplemental File A6, Table S5. A logistic regression analysis investigated the association between the presence of post-COVID-19 fatigue (cut-off of the FSS) as the dependent variable and subject characteristics or holistic health indicators as independent variables. The model for subjects’ characteristics was statistically significant (p < .001). The model explained 14.4% (Nagelkerke R2) of the variance in fatigue and correctly classified 66% of cases. BMI was a dominant predictor (OR = 1.08, 95% CI: 1.01–1.14, p = .02), indicating a substantial increase in the odds of fatigue. Hospitalization was also significant (OR = 2.76, 95% CI: 1.29–5.93, p = .009), indicating that for individuals who were hospitalized, the odds of fatigue are higher than for those who were not hospitalized. Age, sex, and comorbidities did not reach statistical significance, indicating the limited predictive power for post-COVID-19 fatigue in this model.
The model for holistic health indicators was statistically significant (p < .001). The model explained 56.8% (Nagelkerke R2) of the variance in fatigue and correctly classified 80.2% of cases. Functionality, assessed with FIM (OR = 1.07, 95% CI: 1.02–1.12, p = .005), PCFS (OR = 2.87, 95% CI: 1.76–4.67, p < .001), and PSFS (OR = 0.83, 95% CI: 0.68–1.00, p = .046), was a dominant predictor. On the one hand, a worse functional status (measured with PCFS) was associated with higher odds of fatigue, whereas higher functionality (measured with PSFS) was associated with lower odds of fatigue. Frailty was also significant (OR = 1.98, 95% CI: 1.20–3.25, p = .007), indicating a substantial increase in the odds of fatigue. The results also indicated that an increase of 100 METs/min/week decreased the odds of fatigue (OR = 0.99, 95% CI: 0.98–1.00, p = .047). However, the statistical significance is marginal. The rest of the outcomes did not reach statistical significance, indicating the limited predictive power for post-COVID-19 fatigue in this model.
Discussion
The results of this study revealed a significant increase in fatigue after COVID-19 in both hospitalized and non-hospitalized groups, with the hospitalized group reporting significantly higher fatigue levels. While neither group exceeded the clinical cut-off for fatigue before infection, the hospitalized group surpassed this threshold after COVID-19, showing a marked difference compared to the non-hospitalized group. Hospitalization emerged as a significant factor influencing fatigue severity. Hospitalized individuals had higher odds of experiencing post-COVID-19 fatigue, and within this group, women reported greater fatigue than men. Among the descriptive variables, age, BMI, and comorbidities were correlated with fatigue severity, but only BMI showed a significant and consistent effect across both groups. Moreover, BMI was a dominant predictor of the presence of post-COVID-19 fatigue. Holistic health indicators were also significantly correlated with post-COVID-19 fatigue. Subjects with fatigue—regardless of hospitalization—demonstrated significantly lower quality of life, health status, functionality, and physical activity, as well as greater frailty, anxiety, depression, and swallowing difficulties compared to those without fatigue. Functionality and frailty were the most dominant predictors of fatigue severity. In hospitalized subjects, fatigue severity was significantly associated with functionality and frailty, while in non-hospitalized individuals, functionality, health status, and physical activity were the most influential factors. Dysphagia showed a significant association only in hospitalized subjects.
Regarding the descriptive characteristic of the participants, the sample size of the hospitalized (27%) and non-hospitalized (73%) groups was uneven. This is because the proportion of subjects who had had COVID-19 without hospitalization (mild or moderate form) was higher than that of those who required hospital admission (serious or critical form): 80 versus 20% according to the Ministry of Health. 40 Hospitalized subjects were older than non-hospitalized ones, aligning with previous findings by Pérez-González et al 41 and Logue et al 42 Moreover, fatigue showed a significant positive correlation with age overall, indicating that higher levels of fatigue were correlated with higher age. Among hospitalized subjects, no correlation was found between fatigue and age. However, in the non-hospitalized group, the correlation was slight and not statistically significant. Although the literature does not usually distinguish between hospitalized and non-hospitalized subjects, older age has been reported as a risk factor for post-COVID-19 fatigue (OR = 0.04; 95% CI: 0.01–0.07). 43 In terms of body composition, hospitalized individuals had higher weight and BMI than their non-hospitalized counterparts. These results are consistent with Cottini et al 44 and Cai et al, 45 who found that excess weight and obesity increase the risk of developing severe COVID-19. BMI was significantly correlated with post-COVID-19 fatigue in both groups, showing a moderate correlation in hospitalized subjects. Moreover, BMI was found to have a significant effect on post-COVID-19 fatigue severity, in both hospitalized and non-hospitalized subjects. Finally, in both cases, BMI emerged as a dominant predictor, substantially increasing the odds of post-COVID-19 fatigue. Although the specific relationship between BMI and post-COVID-19 fatigue has not been previously studied, it has been observed that higher body weight is associated with more persistent symptoms after COVID-19 infection in hospitalized subjects. 46 Regarding comorbidities, while post-COVID-19 fatigue severity showed a low but significant correlation with comorbidities in the overall sample, this association was slight and non-significant in hospitalized individuals and absent in the non-hospitalized group. Comorbidities did not remain significant in the regression analysis and were not predictive of fatigue. However, hospitalized subjects had a higher burden of pre-existing conditions and a greater short-term mortality risk than non-hospitalized subjects. Nonetheless, our findings are consistent with Tian et al 47 who demonstrated that the most serious forms of COVID-19 occurred in subjects with previous comorbidities such as carcinoma, diabetes, or chronic lung diseases. Furthermore, Pott et al 48 stated that pre-existing comorbidities, such as chronic pathologies and less functional reserve, were key determinants of short-term survival in COVID-19.
Concerning the primary outcome, we found that no groups reported fatigue before COVID-19, while, after the disease, both hospitalized and non-hospitalized subjects had greater fatigue severity compared to their previous state. Nevertheless, after COVID-19 only the hospitalized group exceeded the cut-off point for fatigue. These results agreed with previous existing literature. 6 In the study by Goërtz et al, 49 87% of subjects who had had COVID-19, whether hospitalized or not, reported post-COVID-19 fatigue. However, fatigue levels after COVID-19 were higher in subjects who had been hospitalized during the illness. In fact, 30% of non-hospitalized subjects presented post-COVID-19 fatigue, while this percentage rose to 39% in hospitalized subjects. 5 Our findings are also supported by those of Hu et al 50 The authors reported that, although fatigue was the most prevalent persistent symptom of COVID-19 in both hospitalized and non-hospitalized subjects, it was closely related to the clinical severity of the infection. In our study, fatigue severity was significantly correlated with hospitalization. Hospitalized individuals not only reported higher fatigue levels than non-hospitalized subjects but also had increased odds of developing post-COVID-19 fatigue. Hospitalization thus emerged as a key factor influencing fatigue severity.
To our knowledge, no previous studies have directly examined the relationship between post-COVID-19 fatigue and a comprehensive set of holistic health indicators—including quality of life, functionality, frailty, anxiety, depression, dysphagia, and physical activity—in both hospitalized and non-hospitalized subjects. However, prior research has reported that subjects with post-COVID-19 fatigue tend to increased difficulty breathing or anxiety and depression, among other persistent symptoms. 16 In our study, anxiety and depression were positively correlated with post-COVID-19 fatigue severity in both hospitalized and non-hospitalized groups, indicating that greater fatigue was associated with higher levels of anxiety and depression. These results are supported by Meca-García et al, 51 who reported a significant relationship between fatigue and depression at 2, 12, and 24 months following severe COVID-19. Fatigue was also correlated with poorer perceived health status and reduced quality of life in both hospitalized and non-hospitalized individuals. Notably, health status significantly influenced fatigue severity, although this effect was observed only in the non-hospitalized group. These results align with the finding of de Arruda et al, 52 who described that post-COVID-19 fatigue was associated with a decline in both functional capacity and quality of life. Functionality was assessed using 3 validated instruments: FIM, PCFS, and PSFS. Greater post-COVID-19 fatigue severity was significantly associated with reduced functional ability. Furthermore, regression analysis identified functionality as a significant predictor of post-COVID-19 fatigue. This finding is consistent with the study by Candermir et al, 53 who have shown a close relationship between fatigue and functionality in this population. They found a moderate positive correlation between functionality measured by PCFS and fatigue measured by FSS (r = .49; p < .010), indicating that subjects with greater fatigue tended to have worse functional status. Furthermore, it was observed that for each increase in fatigue severity, the odds of having a greater functional limitation increased significantly (OR = 0.88; 95% CI: 1.68—2.10). These data support our findings and underline the importance of considering functionality as a key factor in the assessment and management of post-COVID-19 fatigue.
This study has some limitations that should be acknowledged. Although all questionnaires were validated for online use and administered by the same evaluator, assessments were virtual, requiring internet literacy, technology access, and stable connection, which may have biased the sample. Recall bias may have affected pre-COVID fatigue reports. As an observational, cross-sectional study, longitudinal research is recommended to track changes over time. The sample size was not formally calculated, limiting precision and generalizability; future studies should use larger, calculated samples and robust statistics. Finally, correlations were mostly low to moderate, and the variance in post-COVID fatigue explained by subject characteristics was limited.
The findings of this study provide relevant information for future research and clinical practice regarding factors influencing post-COVID-19 fatigue. Fatigue severity was associated with descriptive characteristics of subjects such as older age, higher BMI, comorbidities, and hospitalization, with BMI and hospitalization emerging as key predictors. These characteristics allow for early identification of subjects at higher risk of severe fatigue, which could directly impact the planning of rehabilitation programs by guiding the intensity, frequency, and progression of interventions. Furthermore, considering that swallowing disorders are clinically relevant in subjects with respiratory conditions, the potential presence of dysphagia should be assessed during the clinical evaluation of hospitalized post-COVID-19 patients. Incorporating this assessment may contribute to a more comprehensive and safer patient care, as well as support the design of individualized rehabilitation strategies. Finally, correlation and regression analyses showed consistent relationships with holistic health indicators, with functionality and frailty emerging as the main predictors of fatigue. This suggests that fatigue reflects not only basic clinical characteristics but also the subject’s overall functional capacity, and can be used as a useful clinical marker to individualize rehabilitation. In practice, subjects with higher fatigue, poorer functionality, and greater frailty require carefully dosed rehabilitation strategies with more individualized progressions, while those with lower fatigue and preserved functionality may benefit from more intensive approaches. Therefore, systematic assessment of fatigue, together with evaluation of functionality, frailty, and descriptive characteristics, should be integrated into clinical practice to guide patient stratification, tailor rehabilitation programs, and optimize recovery and quality of life.
Conclusions
This study revealed a significant increase in fatigue levels after COVID-19 in both hospitalized and non-hospitalized subjects, compared to before COVID-19, with greater severity in the hospitalized group, who exceeded the clinical cut-off. Hospitalization and BMI emerged as key predictors of post-COVID-19 fatigue. Post-COVID-19 fatigue severity was associated with poorer quality of life, health status, functionality, and physical activity, as well as greater frailty, anxiety, depression, and dysphagia. Functionality and frailty were the most relevant predictors of post-COVID-19 fatigue. These findings underscore the need for personalized rehabilitation strategies that address both descriptive characteristic and holistic health indicators. However, further studies with larger sample sizes are recommended to validate and expand upon these results.
Supplemental Material
Supplemental Material - Fatigue in hospitalized versus non-hospitalized individuals following COVID-19
Supplemental Material for Fatigue in hospitalized versus non-hospitalized individuals following COVID-19 by Laura Pérez-Gisbert, Beatriz Brea-Gómeza, Concepción Morales-García, José A Sánchez-Martínez, María V González-Gutiérrez, Marie C Valenza, Irene Torres-Sánchez in Respiratory Care Reports.
Footnotes
Acknowledgements
This article is part of the doctoral thesis written by Laura Pérez-Gisbert in the context of the “Clinical Medicine and Public Health Program” of the University of Granada, Spain.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Association of Physical Therapists of Andalusia (Ilustre Colegio Profesional de Fisioterapeutas de Andalucía), grant numbers: AI-05/2022 and AI-10/2022; the Spanish Society for Pneumology and Thoracic Surgery (Sociedad Española de Neumología y Cirugía Torácica, SEPAR), grant number: Proyecto: 1216; and The Spanish Ministry of Universities for the training of university teachers with a grant to Laura Pérez-Gisbert, number: FPU: 22/00430.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Supplemental material
Supplemental material for this article is available online.
