Abstract
Background
High-flow nasal cannula (HFNC) is the most frequently used noninvasive respiratory support (NRS) modality for pediatric critical asthma and is often administered with continuous short-acting β-agonists. However, aerosol delivery is inversely related to flow, whereas higher HFNC flows can reduce work of breathing—complicating simultaneous optimization of drug delivery and support. Although infants may benefit from 1.5 to 2.0 L/kg/min, older children may benefit from lower flows (0.5–1.0 L/kg/min). In addition to flow, cannula and circuit caliber may influence delivery. This study quantified continuous aerosol delivery during HFNC across four cannula–circuit configurations at flows of 0.5–1.0 L/kg/min in a simulated pediatric model.
Methods
A pediatric manikin was connected to a lung simulator (VT 150 mL, rate 28/min, I:E 1:2.4, PIF 22 L/min) via a collecting filter. Albuterol (15 mg/h) was nebulized via a vibrating mesh nebulizer placed upstream of a heated humidifier (37°C). Four cannula–circuit configurations were tested at 11 and 22 L/min (0.5–1.0 L/kg/min). Continuous nebulization ran 3 h per condition with hourly collections (three repeated measurements per condition). Albuterol was quantified by spectrophotometry. One-way ANOVA) with Holm–Sidak tests, one-way RM ANOVA, and multiple linear regression (flow, circuit, cannula, flow × circuit) were used.
Results
Inhaled dose ranged 0.7–5.4% of nominal. Increasing flow from 0.5 to 1.0 L/kg/min reduced delivery by ∼50–60% across configurations. Adult circuits delivered 2–3 times higher inhaled dose than junior circuits at both flows (e.g., 5.2–5.4% vs 1.6–1.9% at 0.5 L/kg/min; 2.3–2.6% vs 0.7–0.9% at 1.0 L/kg/min, P < .001). Regression identified circuit type as the strongest independent predictor with a significant flow × circuit interaction; cannula-related differences were modest by comparison.
Conclusions
In this simulated pediatric model, inhaled dose during HFNC was driven primarily by the interaction between gas flow and circuit caliber, with cannula design contributing more modestly. At flows of 0.5 and 1.0 L/kg/min, adult-caliber circuits delivered approximately two- to threefold higher inhaled dose than junior circuits. Pairing an appropriately sized cannula with an adult-caliber circuit may result in substantially higher relative inhaled dose during HFNC therapy.
Keywords
Introduction
High-flow nasal cannula (HFNC) has emerged as the most frequently used noninvasive respiratory support (NRS) modality for pediatric critical asthma, with regional and national registry data showing its use has increased dramatically over the past decade, reflecting both its ease of application and tolerability compared to mask-based interfaces.1–3 This growth has occurred despite limited evidence supporting its comparative effectiveness. 4 In this same patient population, frequent intermittent or continuous nebulized short-acting β-agonists (SABAs) remain a first-line therapy. 5 Recent AARC and PALISI Clinical Practice Guidelines conditionally recommend continuous over intermittent administration with low certainty of evidence 4 ; nonetheless, continuous administration is widely used in practice because it provides uninterrupted bronchodilator exposure, reduces respiratory therapist workload, and may accelerate early symptom improvement. 4 However, delivering continuous SABAs concurrent with HFNC therapy presents a dilemma: aerosol delivery declines significantly at flows >0.5 L/kg/min,6–8 yet higher flows are associated with a reduction in work of breathing. 9 This trade-off complicates efforts to optimize both bronchodilator delivery and respiratory support.
While flows of 1.5–2.0 L/kg/min have been shown to be optimal in reducing work of breathing in infants with bronchiolitis, there is a lack of data to support optimal flow rates in older children with asthma.9,10 In a recent narrative review of NRS for pediatric critical asthma, 11 Miller and Rotta summarized available evidence on HFNC flow settings in pediatric asthma. They noted that prior studies have used a range of approaches: in randomized trials conducted in the emergency department, flows were initiated at 1 L/kg/min and titrated to 2 L/kg/min to maintain target oxygen saturation, 12 or set at 2 L/kg/min for the first 10 kg with 0.5 L/kg/min for each kilogram thereafter. 13 Retrospective pediatric ICU data compared outcomes at flows ≤0.5 L/kg/min versus >0.5 L/kg/min and found no significant differences. 14 To add physiologic context, Weiler et al demonstrated in children under 3 years that effort of breathing improved in a dose-dependent manner with increasing flows, with the greatest benefit at 1.5–2.0 L/kg/min in those ≤8 kg. In contrast, among children >8 kg, higher flows did not significantly reduce work of breathing. 15 Consistent with this, Li et al. highlighted that inspiratory flow demand per kilogram declines with age, suggesting that flows effective in infants with bronchiolitis may be excessive for older children with asthma, where 0.5–1.0 L/kg/min is likely sufficient. 9 Synthesizing these data, Miller and Rotta concluded that an initial flow of 0.5–1.0 L/kg/min is reasonable in older children.
What remains uncertain is whether, at these flows, HFNC delivers a therapeutically meaningful inhaled dose. Prior in vitro reports of continuous delivery during HFNC have ranged from 14.8% to 8.58% to 1.16%, but those values were obtained at 0.13, 0.25, and 1.7 L/kg/min, respectively—outside the 0.5–1.0 L/kg/min range of interest here and under differing model/configuration conditions.7,16 Further complicating practice is the variety of cannula designs and circuit calibers available for this population, which may affect aerosol delivery.
7
Accordingly, the aim of this study was to quantify continuous aerosol delivery via HFNC at 0.5 and 1.0 L/kg/min across four cannula–circuit configurations and to determine whether the effect of increasing flow on inhaled dose differs by circuit caliber in a simulated pediatric airway model. High-flow nasal cannula (HFNC) is frequently used for pediatric critical asthma, and continuous short-acting β-agonist is often co-administered. For older children, flows of roughly 0.5–1.0 L/kg/min are likely sufficient. However, aerosol delivery via HFNC declines significantly as flows approach 0.5 L/kg/min. In addition to flow, cannula and circuit caliber may influence delivery. This bench study demonstrates that aerosol delivery during concurrent HFNC therapy is strongly influenced by both flow and circuit caliber. Across commonly used flows (0.5–1.0 L/kg/min), adult-caliber circuits delivered approximately two- to threefold higher inhaled dose than junior circuits. These findings indicate that pairing appropriately sized cannulas with adult-caliber circuits may substantially increase relative inhaled dose when co-administered with HFNC.Quick look
Current knowledge
What this paper contributes to our knowledge
Methods
A pediatric manikin (TruCorp AirSim Child Combo X, N. Ireland), with an anatomically correct airway based on CT DICOM data of a 6-year-old, was connected to a lung simulator (ASL 5000, IngMar Medical, Pittsburgh, PA) via corrugated tubing. The manikin was positioned upright to simulate a Fowler’s position with an open-mouth configuration as older children often breathe with their mouth open during respiratory distress, providing a clinically relevant model for aerosol delivery. Collecting filters (RespirGard-II 303EU, Vyaire Medical, Mettawa, IL) were placed in-line distal to the trachea and oriented at an angle ≥90° using a section of corrugated tubing to minimize possible condensate accumulation on the filter (Figure 1). Simulated pediatric model. Manikin connected to ASL-5000 via corrugated tubing; positioned upright (Fowler) with mouth open to mimic distressed breathing typical of older children during HFNC. An in-line collecting filter was placed distal to the trachea and oriented ≥90° using a 6-inch tubing segment to limit condensate, enabling tracheal (inhaled) dose measurement.
The lung model was configured using previously published parameters 7 (VT 150 mL, rate 28/min, I:E 1:2.4) to simulate a spontaneously breathing 5- to 7-year-old child (∼20–25 kg). Using a sinusoidal waveform, settings were adjusted to achieve a peak inspiratory flow (PIF) of 22 L/min, selected as a representative inspiratory demand for an older pediatric patient at HFNC flows of 0.5–1.0 L/kg/min, based on prior studies demonstrating lower per-kilogram inspiratory demand with increasing age.9,15 Accordingly, HFNC flows were set at 11 and 22 L/min to reflect this clinically relevant range, such that delivered flow matched or remained below the model PIF.
Three Optiflow cannulas (Figure 2) representing four cannula–circuit configurations (Fisher & Paykel Healthcare, Auckland, New Zealand) were evaluated: (a) XL Jr 2 (OJR418) w/Optiflow Jr circuit (RT330), (b) XXL Jr 2+ (OJR520) w/Optiflow Jr circuit (RT330), (c) XXL Jr 2+ (OJR520) w/Optiflow adult circuit (RT232), and (d) small adult (OPT942) w/adult single-limb circuit (RT114) (Figure 3). The pressure manifold supplied with the Optiflow Jr circuit (RT330) was used on the dry side of the humidifier for all conditions, with only the inspiratory limb exchanged between circuit configurations. The XL Jr and XXL Jr bifurcate into two flexible tubes (∼5 mm and ∼10 mm) that supply gas flow to each prong independently; the small adult uses a single ∼13-mm tube to supply both prongs simultaneously. Circuit internal diameters were ∼12 mm (Jr) and 19–20 mm (adult). All three cannulas incorporate secure connect adapters, allowing the XL and XXL Jr cannulas to attach to either Jr or adult circuits; the small adult connects to the adult Optiflow and includes a 15-mm OD adapter for single-limb ventilator circuits (Figure 4). Percent nare occlusion was quantified using both diameter-based and area-based approaches. Diameter occlusion was calculated as the ratio of prong outer diameter to nare diameter, reflecting common clinical visual sizing. Area occlusion was calculated as the ratio of prong cross-sectional area to nare cross-sectional area, representing a physical measure of lumen obstruction. Using the manikin nare of 9 mm, the XL Jr cannula (prong OD ≈ 4 mm) resulted in approximately 44% diameter occlusion and 20% area occlusion per nare, whereas the XXL Jr and small-adult cannulas (prong OD ≈ 5 mm) resulted in approximately 56% diameter occlusion and 31% area occlusion per nare. Cannulas were positioned with prongs fully inserted into each nare without exerting excessive pressure on the philtrum or intentionally sealing the nares, consistent with standard clinical practice. All cannula–circuit configurations were appropriate for the simulated model based on cannula size and manufacturer-specified flow capabilities and may represent combinations encountered in clinical practice for a 5- to 7-year-old child. HFNC cannulas tested: (a) Optiflow XL Jr 2, (b) Optiflow XXL Jr 2, and (c) Optiflow small adult. Cannula–circuit configurations tested: (a) Optiflow XL Jr 2 with Optiflow Jr circuit, (b) Optiflow XXL Jr 2 with Optiflow Jr circuit, (c) Optiflow XXL Jr 2 with Optiflow adult circuit, and (d) Optiflow small adult with adult single-limb circuit. Occlusion of manikin’s nares. (a) Optiflow XL Jr 2, (b) Optiflow XXL Jr 2, and (c) Optiflow small adult.


An oxygen flow meter attached to an air/oxygen blender (Precision Medical, Northampton, PA) was used to deliver the desired HFNC gas flow and
Inhaled dose was reported as both the absolute inhaled mass (mg) and as a percentage of the nominal dose (%), expressed as mean ± standard deviation. One-way analysis of variance (ANOVA) with Holm–Sidak post hoc testing was performed on mean inhaled mass (mg) per configuration to detect significant differences between cannula–circuit configurations at each flow setting. To assess potential time-dependent effects during sustained nebulization, inhaled dose was compared across hourly collections (hours 1–3) using one-way repeated-measures ANOVA. Multiple linear regression was performed on inhaled dose to evaluate the independent contributions of flow (0.5 vs 1.0 L/kg/min), cannula type (XL, XXL, and small adult), and circuit type (junior vs adult). An interaction term (flow × circuit) was included to test whether increasing HFNC flow affected inhaled dose differently in junior versus adult circuits. Assumptions of normality were assessed using the Shapiro–Wilk test. A P value <.05 was considered statistically significant. Statistical analyses were performed using SigmaPlot v15.0 (Grafiti LLC, Palo Alto, CA).
Results
Inhaled dose for all cannula–circuit configurations.

Data are presented as mean ± standard deviation.
Flow and circuit caliber together had the greatest impact on inhaled dose. Across all configurations, increasing flow from 0.5 to 1.0 L/kg/min reduced inhaled dose by approximately 50–60% (Figure 5). In junior circuits, inhaled dose fell from 1.6–1.9% to 0.7–0.9% (P < .001). In adult circuits, inhaled dose decreased from 5.2–5.4% to 2.3–2.6%, respectively (P < .001). Although the absolute change in inhaled dose (percentage points) was larger in adult circuits, the relative reduction was similar (∼53–56%). At 0.5 L/kg/min, adult circuits delivered approximately 3.6 percentage points more aerosol than junior circuits (mean inhaled dose 5.30% vs 1.75%, ∼200% relative increase), whereas at 1.0 L/kg/min this difference narrowed to 1.7 points (mean inhaled dose: 2.45% vs 0.80%) with a comparable relative increase. Mean inhaled dose at different flow rates (0.5 vs 1.0 L.kg/min) and cannula–circuit configurations. Inhaled dose decreased by approximately 50–60% as flow increased from 0.5 to 1.0 L/kg/min regardless of cannula–circuit configuration, *P < .05.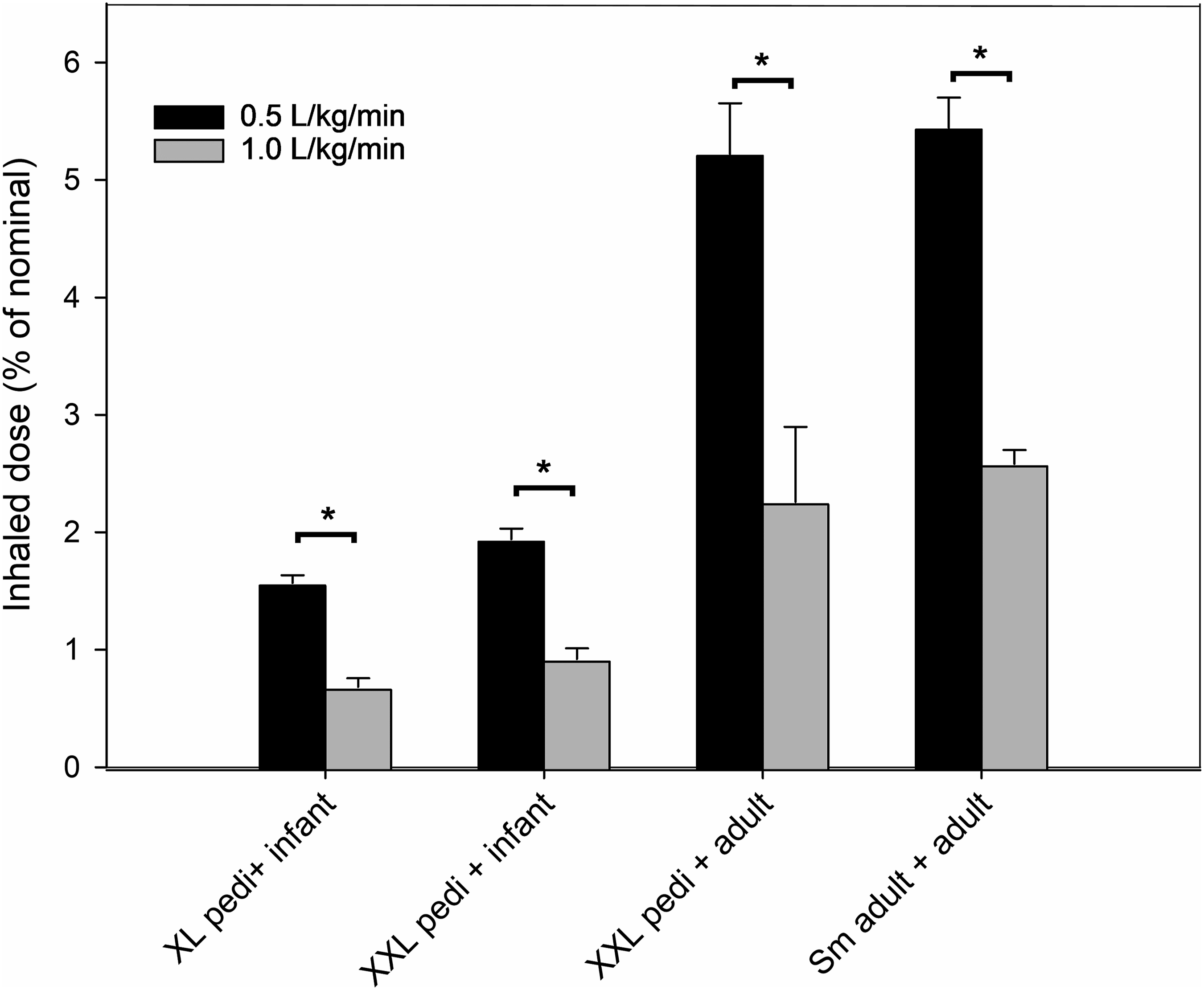
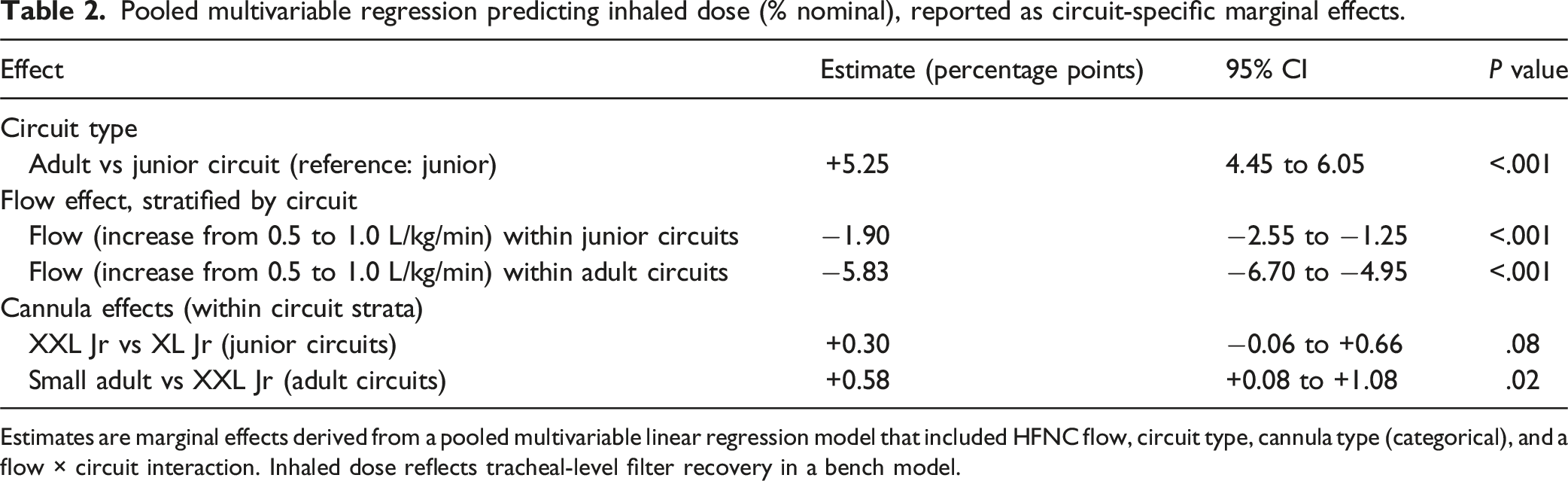
Regression analysis supported the descriptive findings, with circuit type/caliber emerging as the strongest independent predictor of inhaled dose. Adult circuits were associated with a 5.25-percentage point higher inhaled dose compared with junior circuits (95% confidence interval (CI), 4.45–6.05; P < .001). When expressed as marginal effects, increasing flow was associated with reduced inhaled dose in both circuit types, but the magnitude of this reduction differed substantially by circuit. Within junior circuits, increasing flow from 0.5 to 1.0 L/kg/min resulted in a 1.90-percentage point reduction in inhaled dose (95% CI, −2.55 to −1.25; P < .001). In contrast, within adult circuits, the same increase in flow corresponded to a larger 5.8-percentage point reduction in inhaled dose (95% CI, −6.70 to −4.95; P < .001) (Figure 6). Because a significant flow × circuit interaction was present (β = −3.9; P < .001), regression results are reported as circuit-specific marginal effects derived from the pooled model rather than as a single pooled slope (Table 2). Marginal effect of increasing HFNC flow from 0.5 to 1.0 L/kg/min on inhaled dose, stratified by circuit type. Bars represent absolute change in inhaled dose expressed in percentage points (not relative percent change) associated with this flow increase, derived from the pooled regression model; error bars indicate 95% confidence intervals. Pooled multivariable regression predicting inhaled dose (% nominal), reported as circuit-specific marginal effects. Estimates are marginal effects derived from a pooled multivariable linear regression model that included HFNC flow, circuit type, cannula type (categorical), and a flow × circuit interaction. Inhaled dose reflects tracheal-level filter recovery in a bench model.
The fitted multivariable regression model explained 98.0% of the variance in inhaled dose (R2 = 0.98), reflecting the controlled bench design and the inclusion of flow, circuit type, cannula configuration, and the flow × circuit interaction, which together accounted for most of the observed variability across test conditions. The regression equation was: Inhaled dose (% nominal) = 2.55 − 1.90 × Flow + 0.31 × Cannula_XXL + 0.58 × Cannula_Adult + 5.25 × Circuit − 3.92 × (Flow × Circuit), where flow was modeled as a continuous variable in L/kg/min, circuit was coded as a binary variable (0 = junior circuit, 1 = adult circuit), and cannula type was treated as a categorical predictor using dummy variables, with the XL Jr cannula as the reference category. Predicted inhaled mass (Minh) can be calculated as Minh (mg/h) = (D/100) × R, where D is the predicted inhaled dose (% nominal) and R is the nominal infusion rate (mg/h).
Cannula size/design had a modest influence on inhaled dose relative to flow and circuit caliber (Table 2). When modeled as a categorical predictor, the XXL Jr cannula was associated with a small, non-significant increase in inhaled dose compared with the XL Jr cannula (∼0.3 percentage points). The small-adult cannula was associated with a slightly larger increase relative to the XL Jr cannula (∼0.6 percentage points), which reached statistical significance but remained small in magnitude. These cannula-related differences were substantially smaller than the effects of circuit caliber and the flow × circuit interaction.
During continuous aerosol nebulization, condensation (rainout) was observed to varying degrees within the HFNC circuit and nasal cannula across all conditions. Rainout was minimal in most cannula–circuit configurations, typically limited to mild sputtering at the cannula prongs without requiring circuit drainage between filter changes. In contrast, the XXL Jr cannula exhibited substantial fluid accumulation within the bifurcated flexible tubing regardless of circuit type or flow rate (Figure 7). When paired with the junior circuit, pooling within the circuit produced visible circuit bounce and necessitated drainage between runs; this was not observed when the adult circuit was used. Fluid accumulation (“rain out”) observed with the XXL Jr cannula, resulting in pooling within the bifurcated flexible tubing.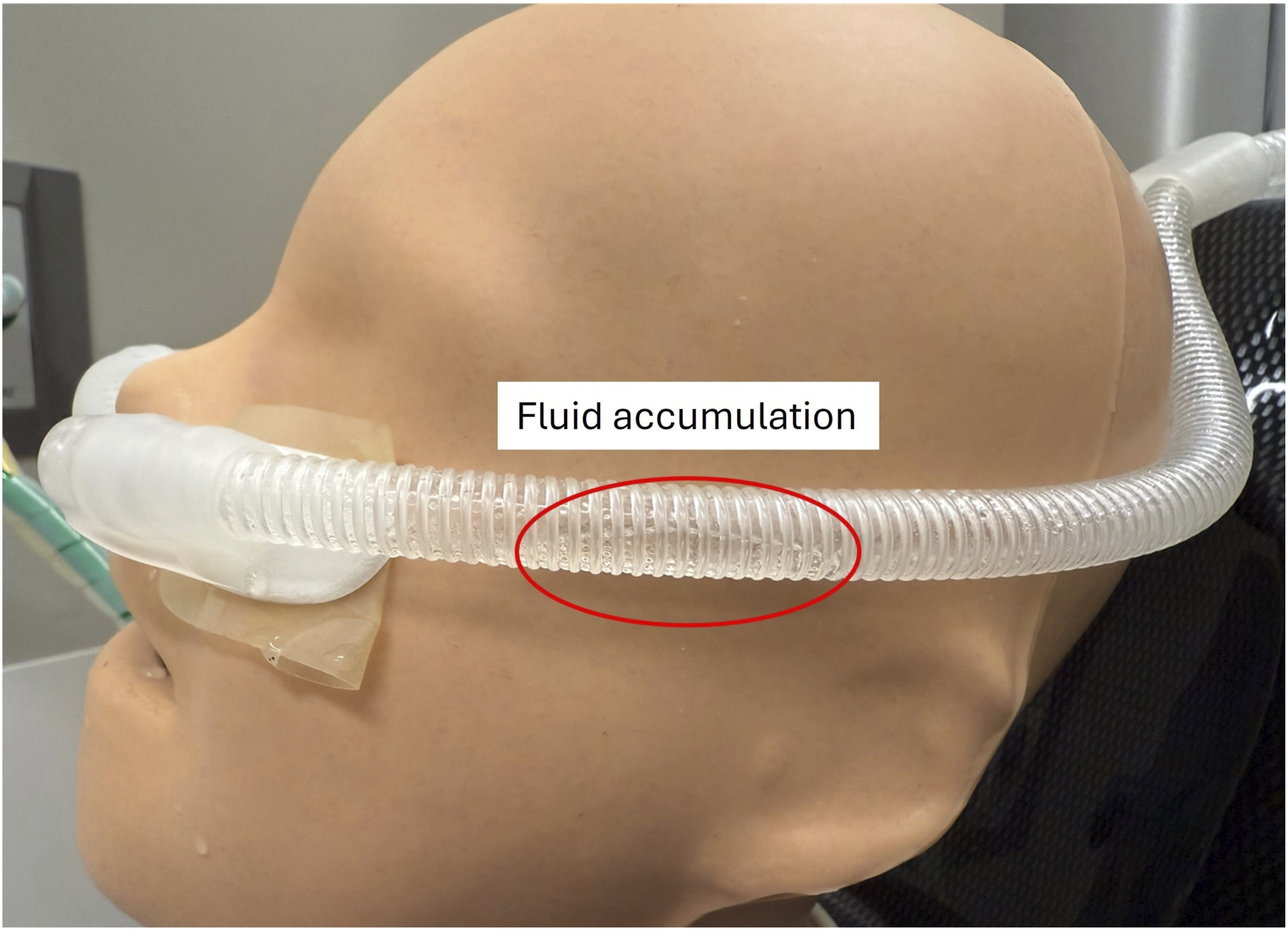
To assess whether condensate (rainout) accumulation was associated with time-dependent reductions in aerosol delivery, inhaled dose was evaluated across hourly filter collections during the 3-h continuous runs. When all cannula–circuit configurations were considered together, no statistically significant difference in inhaled dose was observed between hours 1, 2, and 3 (one-way repeated-measures ANOVA, P = .57). Within individual XXL Jr cannula configurations at 1.0 L/kg/min, where visible condensate accumulation was greatest, modest hour-to-hour declines in mean inhaled dose were observed (Figure 8); however, these trends varied across circuit pairings and did not demonstrate a consistent or statistically significant time-dependent reduction in delivery. Hourly inhaled dose (% nominal) measured during 3-h continuous aerosol delivery for XXL cannula configurations at 1.0 L/kg/min. Points represent mean inhaled dose for each hourly collection. Although modest hour-to-hour declines were observed, no statistically significant difference in inhaled dose was detected across hours 1–3 (one-way repeated-measures ANOVA, P = .57).
Discussion
This simulated model was designed to mirror common clinical configurations for a typical 5- to 7-year-old, the age range that comprises most pediatric critical asthma admissions,1,2,17,18 using manufacturer-compatible cannula–circuit pairings and HFNC flows of 0.5 or 1.0 L/kg/min. Under these conditions, continuous aerosol delivery fell by ∼50–60% as flow increased, and inhaled dose ranged from 0.7% to 5.4%. Adult circuits delivered approximately two- to threefold more aerosol than junior circuits, whereas differences attributable to cannula design were small in magnitude relative to flow and circuit effects.
The finding that inhaled dose declined as flow increased is consistent with prior unit-dose studies,19–24 and at least two in vitro studies evaluating continuous aerosol delivery via HFNC.7,16 Using the same simulated pediatric model, Moody and Ari 7 reported an inhaled dose of 14.8% during continuous aerosol delivery via HFNC using the same small-adult cannula–circuit configuration as in this current study; however, this was achieved at only 3 L/min (0.13 L/kg/min), below clinically relevant HFNC flows. While continuous aerosol was not evaluated at higher flows, unit-dose delivery was evaluated at 0.53 L/kg/min (12 L/min) using the same XL Jr cannula with junior circuit and the small-adult cannula with adult circuit, yielding inhaled doses of 2.3% and 5.7%, respectively. These values are comparable to the present study’s continuous-delivery results at 0.5 L/kg/min (1.6–1.9% with junior circuits and 5.2–5.4% with adult circuits). In a complementary 15 kg toddler model, Li et al. compared unit-dose and continuous aerosol delivery via HFNC. 16 Using a similar sized earlier XL Jr cannula paired with the same junior circuit as in the current study, they tested flows of 3.75 L/min (0.25 L/kg/min) and 25 L/min (1.7 L/kg/min). For continuous delivery, the inhaled dose reported at each flow represented the average of two tested concentration conditions delivering the same nominal dose (5 mg/mL at 4 mL/h and 1 mg/mL at 20 mL/h). On this basis, the averaged continuous-delivery inhaled doses were 8.58% at 0.25 L/kg/min and 1.16% at 1.7 L/kg/min.
Although these flows differ from those evaluated here, the directionality is consistent: inhaled dose declines as flow increases. This parallels the present findings, in which delivery fell from 1.6% to 1.9% at 0.5 L/kg/min to 0.7–0.9% at 1.0 L/kg/min with junior circuits, and from 5.2–5.4% to 2.3–2.6% with adult circuits. Compared with the current junior circuit results at 1.0 L/kg/min (0.7–0.9%), Li et al’s findings of 1.16% at higher flows (1.7 L/kg/min) are ∼30–65% higher. Part of the disparity likely reflects differences in simulated models and testing conditions: Li et al reported higher inhaled dose under closed-mouth breathing at higher flows, which was the condition used during continuous infusion administration in their model. In contrast, open-mouth breathing was associated with lower inhaled dose at elevated flows. They attributed this effect to high gas flow through the nasal prongs washing out the nasopharyngeal reservoir, resulting in loss of aerosol-laden gas through the oral opening during both inhalation and exhalation. In the present study, the open-mouth configuration likely promoted similar washout, contributing to increased aerosol dilution and fugitive loss at higher flows. Consequently, the inhaled dose values reported here likely represent a conservative, lower-bound estimate for patients breathing with an open mouth. However, sampling approach remains a plausible contributor to differences between studies. Li et al reported continuous delivery from 15-minute runs with the infusion pump set to 20 mg/h, normalizing to the nominal 5 mg expected in that interval. Because condensate (rainout) may accumulate within the circuit and cannula during prolonged nebulization and contribute to upstream aerosol losses, extrapolating a 15-minute sample or inferring hourly delivery from unit-dose measurements may not fully reflect sustained hourly delivery during extended administration.
Consistent with this, condensate was observed to varying degrees within the HFNC circuit and nasal cannula across all tested conditions, with the most pronounced rainout occurring in the XXL Jr cannula. This likely reflects its design: heating extends only to the cannula connection, leaving the longer, larger-bore dual limbs (∼10 mm) unheated. Compared with the XL Jr (∼5 mm dual limbs) and the small-adult cannula (single ∼13 mm limb), this greater unheated volume and surface area promote heat loss, condensation, and pooling (Figure 2). The circuit bounce observed with the junior circuit likely reflects its smaller diameter, where a given condensate volume more strongly alters compliance and flow.
Although a direct comparison between short-duration and sustained-hour sampling was not performed in this study, inhaled dose did not change significantly across hours 1–3 when all configurations were considered, indicating that any time-dependent reduction in delivery was small relative to the dominant effects of flow and circuit caliber. Descriptively, the greatest visible condensate accumulation and modest hour-to-hour decreases occurred in XXL Jr cannula configurations at 1.0 L/kg/min. Within these XXL Jr configurations, the observed hour-to-hour changes were inconsistent and do not indicate a uniform or progressive decline in aerosol delivery over time; however, hourly measurements for the XXL Jr cannula configurations were obtained with visible condensate actively drained between runs, which may have limited progressive upstream aerosol losses during testing despite visible rainout.
In clinical practice, where condensate may not be routinely cleared during prolonged administration, greater upstream losses could occur, underscoring the importance of attention to rainout accumulation and circuit maintenance during extended HFNC aerosol delivery. Importantly, this study was powered to detect configuration- and flow-related differences in inhaled dose rather than subtle time-dependent decay; thus, the absence of a statistically significant time effect should be interpreted cautiously and does not exclude clinically relevant delivery drift during prolonged therapy. Because condensate was not quantified or modeled as an independent variable, observed patterns suggest that condensate-related losses represent a configuration-dependent mechanism through which flow and circuit geometry influence inhaled dose, rather than an independent driver of delivery.
Although prior studies have identified flow as the principal determinant of inhaled dose during HFNC,7,8,19–26 the present study showed that flow and circuit type together had the greatest impact, with circuit type/caliber emerging as the strongest independent predictor. This reflects the large baseline difference observed between junior and adult circuits, with the latter delivering approximately two- to threefold higher inhaled dose across both flow rates. Flow remained the dominant driver of dose reduction, but circuit configuration modified that effect. The significant flow × circuit interaction observed here extends prior evidence by demonstrating that the magnitude of flow-related loss depends on circuit type/caliber. Larger-bore circuits lower gas velocity and turbulence at a given volumetric flow:27,28 this reduces losses at the cannula interface; when flow transitions from a larger caliber circuit into smaller-bore tubing/prongs at a lower velocity, inertial impaction decreases. Consequently, a greater fraction of aerosol remains suspended and reaches the airway compared with smaller-bore infant circuits.
Previous reports of the effects of cannula size/design on inhaled dose vary. Bhashyam et al 26 evaluated infant, pediatric, and adult cannulas (dual small-bore tubing) and found that inhaled dose did not differ significantly across cannulas (18.6–26.9%) at 3 L/min using an inhalation-only breathing pattern. In contrast, Perry et al21 examined infant, pediatric, and adult cannulas (also dual small-bore) over 3–40 L/min and reported higher inhaled dose with larger cannulas; however, for most flow–cannula combinations, delivered dose was low (0.0–2.5%) and unlikely to be clinically meaningful. The low recovery in that study was likely related to the HFNC system (Vapotherm), which required placing the nebulizer proximal to the interface via an adapter that retained 62–80% of the nominal dose. Even though both studies paired cannulas with the same circuit type/size, they varied flows and breathing parameters across cannulas and used non-anatomical airway models, confounding attribution of differences to cannula design alone.
Bennett et al 23 evaluated the effect of nasal cannula design on emitted dose using two adult HFNC cannulas (Comfort Flo and Comfort Flo Plus; analogous to the XL Jr and small-adult cannula used here) at 10, 25, and 40 L/min. The larger-bore design (Comfort Flo Plus) emitted a higher percentage of the nominal dose at each flow: 55.7%, 49.9%, and 40.4% at 10, 25, and 40 L/min, respectively, compared with 6.6%, 2.6%, and 0.8% for the smaller-bore cannula. The same adult circuit was used in both conditions, isolating the cannula effect. However, because no anatomical model or simulated breathing was used and measurements were limited to the cannula outlet, clinical applicability is limited. More recently, Wang et al 25 examined aerosol delivery with three Optiflow Jr cannulas (OPT314, OPT316, and OPT318) across multiple flows using the same circuit and observed differences in tracheal dose by cannula size; however, results were not subjected to statistical analysis, limiting the strength of inference.
In a prior bench study, using the same simulated pediatric model, Moody and Ari 7 paired the XL Jr cannula with the junior circuit and the small-adult cannula with the adult circuit at 3, 6, and 12 L/min (0.13, 0.27, 0.53 L/kg/min). Inhaled unit-dose was consistently higher with the small-adult pairing (12.6% vs 10.1% at 3 L/min; 10.5% vs 6.5% at 6 L/min; 5.7% vs 2.3% at 12 L/min), and the difference was attributed to cannula size; however, because cannula and circuit were coupled, that design could not isolate cannula from circuit effects. Hence, the higher delivery observed previously with the small-adult configuration likely reflected the larger-diameter adult circuit rather than cannula size/design alone.
In the current work, cannula comparisons were performed within matched circuit calibers (XL Jr vs XXL Jr on the junior circuit; XXL Jr vs small adult on the adult circuit), with the XXL Jr evaluated on both circuits, providing a partial bridge across circuit types. Under these conditions, cannula design had a modest influence on inhaled dose once flow and circuit were accounted for. When modeled categorically, the XXL Jr cannula demonstrated a small, non-significant increase in inhaled dose relative to the XL Jr cannula, whereas the small-adult cannula was associated with a slightly larger, statistically significant increase. However, these differences were small in magnitude compared with the dominant effects of flow and circuit caliber. Together, these findings suggest that discrepancies across prior studies are more likely driven by circuit caliber, measurement location, and test conditions rather than intrinsic cannula size or design alone.
Building on these findings, the fitted regression model can be used as a tool to illustrate how flow, circuit caliber, and cannula design interact to influence tracheal-level inhaled dose under controlled conditions. For example, for a 22-kg child (11 vs 22 L/min) receiving continuous albuterol at 15 mg/h, the model predicts an inhaled dose of ∼1.60% (0.24 mg/h) at 0.5 L/kg/min with a junior circuit and XL Jr cannula, decreasing to ∼0.65% (0.10 mg/h) at 1.0 L/kg/min. Using an adult circuit, predicted inhaled dose is higher at 0.5 L/kg/min (∼5.2%; 0.78 mg/h) and declines at 1.0 L/kg/min (∼2.56%; 0.38 mg/h).
For patients not receiving NRS, large-volume jet nebulizers (LVNs) with an aerosol mask are commonly used to delivery continuous SABAs. In the same simulated pediatric model used here, Moody and Ari 7 previously reported an LVN-mask inhaled dose of 3.2%, similar to unit-dose small-volume jet nebulizer efficiency (2.99–5.76%).7,16,29,30 In this study, junior-circuit configurations yielded ≤1.9% at 0.5 L/kg/min and ≤0.9% at 1.0 L/kg/min—values consistently lower than the ∼3% LVN–mask reference. In contrast, adult-circuit configurations demonstrated higher inhaled dose (2.3–5.4%), reflecting greater relative delivery efficiency; at 0.5 L/kg/min, measured values were higher than the LVN–mask reference (XXL Jr 5.2%, small adult 5.4%), whereas at 1.0 L/kg/min delivery efficiencies were closer to, but remained below, that reference (2.6%, 2.3%). Although the optimal loading or nominal dose for continuous SABAs is unknown 4 and no accepted therapeutic target exists for continuous SABA delivery via HFNC, the LVN–mask reference provides a useful bench-derived comparator for contextualizing relative aerosol delivery efficiency; however, the clinical implications of these bench-observed differences cannot be determined from bench data alone and require confirmation in human studies that account for patient response, disease severity, and clinical outcomes.
Limitations
This study has many limitations. An in vitro model of a single age/size with a fixed breathing pattern (fixed PIF), fixed humidification settings, and open-mouth breathing may not generalize to other ages, breathing patterns, or leak states. Only two flows (0.5 and 1.0 L/kg/min) were tested, so findings should not be extrapolated to higher or weaning flows; accordingly, estimated flow effects reflect a two-point comparison within the tested range rather than a continuous or linear relationship. Although the airway geometry was realistic, the manikin used in this study lacks mucosa, cilia, secretions, and heat/water exchange, which can alter delivery. A single HFNC platform was used; other brands, connectors, and circuit geometries may perform differently. The cannula/circuit orientation during testing likely impeded some gravitational condensate drainage, potentially affecting dose.
Cannula–circuit pairings were not fully factorial: the XL Jr was tested only with the junior circuit, the small-adult cannula only with the adult circuit, and the XXL Jr was the only interface spanning both circuits. As a result, cannula and circuit effects cannot be considered fully independent, and cannula-related findings are limited to comparisons within matched circuit calibers. Therefore, these results should not be generalized across platforms, circuit sizes, or manufacturers, and cannula effects were inferred though multivariable modeling rather than direct fully crossed experimental contrasts. Prong–nare fit was standardized, so real-world variability in nare size and prong depth was not modeled.
Each cannula–circuit–flow configuration was evaluated using three repeated hourly measurements during continuous nebulization runs. This level of replication is consistent with prior in vitro aerosol delivery studies7,16,22,31 and was sufficient to demonstrate repeatability and to detect large configuration and flow-related differences, which were the primary outcomes of interest. The study was not designed to detect subtle time-dependent decay in delivery within individual configurations. Given the factorial design, increasing replication would have required substantially longer testing duration and greater drug consumption. In addition, circuits were not routinely drained between hourly collections unless rainout was visually significant; alternative condensate management practices could influence time-dependent delivery.
Finally, measured “inhaled dose” at the trachea is a proxy for aerosol available to the lower airways, not a direct measure of distal deposition, and particle-size characterization was not performed. Future clinical studies should report device-level details (cannula–circuit pairing), index flow to inspiratory demand, and incorporate particle-size characterization in bench/simulation work while linking device choices to clinical outcomes.
Conclusion
In this simulated pediatric model, inhaled dose during HFNC was driven primarily by the interaction between gas flow and circuit caliber, with cannula design contributing more modestly. At flows of 0.5 and 1.0 L/kg/min, adult-caliber circuits delivered approximately two- to threefold higher inhaled dose than junior circuits, reflecting reduced flow-related aerosol losses within larger-bore systems. Pairing an appropriately sized cannula with an adult-caliber circuit may result in substantially higher relative inhaled dose during HFNC therapy at clinically relevant flows.
Footnotes
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosures
Author has no relationships to disclose.
A version of this paper was presented as an abstract by Mr Moody at AARC Congress 2025 held in Phoenix, Arizona, December 6–9, 2025.
