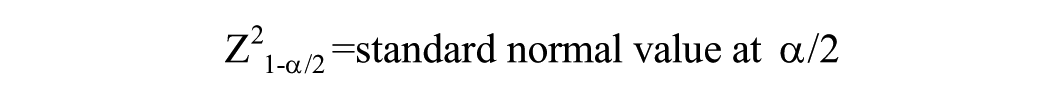Abstract
Objectives:
This study aimed to assess clinical outcomes and associated factors among children with meningitis at Ras Desta Damtew Memorial Hospital.
Method:
A retrospective cohort study was conducted from April 2022 to May 2024. Multivariable logistic regression analyses were performed after adjusting for potential confounders and P < .05 considered significant.
Results:
The magnitude of good clinical outcome was 90.9% (95% CI: 87.3, 94.4). Factors significantly associated were: age >2 months (AOR: 12.1; 95% CI: 3.39, 42.89; P < .001), duration of illness <72 hours prior to hospital admission (AOR: 4.75; 95% CI: 1.36, 16.56; P = .014), completed immunization status (AOR: 3.72; 95% CI: 1.19, 11.63; P = .024), and absence of severe acute malnutrition (AOR: 5.18; 95% CI: 1.18, 22.71; P = .029).
Conclusions:
Public health strategies should prioritize addressing severe acute malnutrition, ensuring complete immunization, and promoting early healthcare-seeking within 72 hours of symptom onset.
Introduction
Meningitis, an infection of the meningeal layers surrounding the brain and spinal cord, remains a significant public health challenge globally, particularly in the “meningitis belt” of sub-Saharan Africa, which extends from Senegal to Ethiopia.1,2 The disease accounts for approximately 2.5 million cases and over 250 000 deaths annually, with the highest burden in low- and middle-income countries where case fatality rates can reach 30%. 3 In these settings, the disease contributes disproportionately to childhood mortality, accounting for an estimated 5% to 10% of all under-five deaths.3,4 It is the most common central nervous system infection in neonates and infants, with case fatality rates of 2% to 30% and long-term complications including mental retardation, paralysis, and hearing loss occurring in 10% to 20% of survivors.5,6
In Ethiopia, despite the implementation of the Expanded Program on Immunization since 1980, which includes vaccines against major meningitis pathogens such as Haemophilus influenzae type b (Hib) and Streptococcus pneumoniae, meningitis continues to contribute substantially to childhood morbidity and mortality. 7 The 2024 to 2025 Ethiopia Mini Demographic and Health Survey and Report reported under-five mortality at 51 per 1000 live births, with infectious diseases including meningitis as major contributors. 8
Meningitis diagnosis is typically established through clinical presentation (eg, fever, neck rigidity, altered mental status, seizures, or bulging fontanel in infants) and cerebrospinal fluid (CSF) analysis showing pleocytosis, elevated protein, and reduced glucose, with or without microbiological confirmation.9,10 Clinical outcomes are categorized at discharge as good (resolution of symptoms without acute complications) or poor (death, complications, referral to higher facility, or ICU admission).11,12
While previous multicenter studies in Ethiopia have reported on pediatric meningitis outcomes across regional settings,6,13,14 these investigations have predominantly been conducted in tertiary referral hospitals located outside the capital, often with limited diagnostic infrastructure and variable access to pediatric subspecialty care. Notably, the multicenter study by Siraj et al (2025) included sites in Northwest Ethiopia but did not encompass any facility in Addis Ababa, leaving a critical gap in understanding whether outcomes in the capital where diagnostic tools such as cerebrospinal fluid analysis are more readily available, where specialist pediatric care is concentrated, and where vaccination coverage may differ; align with or diverge from regional findings. 14
Furthermore, existing studies have primarily focused on bacterial meningitis confirmed by microbiological methods, potentially excluding the substantial proportion of clinically diagnosed meningitis cases that represent the reality of frontline practice in Ethiopian hospitals.6,13 This study addresses these gaps by providing contemporary, facility-specific data from an urban hospital in Addis Ababa, capturing the full spectrum of clinically diagnosed meningitis cases. Its contribution lies not in claiming geographic uniqueness, but in offering a critical comparative benchmark against which to assess whether outcomes in a better-resourced urban setting differ from regional averages, and to identify whether previously established risk factors hold similar predictive value in this distinct healthcare environment. By doing so, this research generates locally actionable evidence to inform context-specific interventions and health policy tailored to Ethiopia’s diverse healthcare landscape. Therefore, this study aimed to assess the clinical outcomes and identify factors associated with outcomes among children admitted with meningitis at Ras Desta Damtew Memorial Hospital, Addis Ababa, Ethiopia.
Methods
Study Design and Setting
A retrospective cohort study was conducted at Ras Desta Damtew Memorial Hospital, a general hospital located in Addis Ababa, Ethiopia. Data were collected from medical records of children admitted between April 2022 and May 2024, with clinical outcomes assessed at the time of discharge. The hospital serves a catchment population of approximately 300 000 and admits an average of 761 pediatric cases annually, including 181 meningitis cases per year.
Inclusion and Exclusion Criteria
The study included charts of children aged 1 month to 14 years admitted with a clinical diagnosis of meningitis to the pediatric ward during the study period from April 2022 to May 2024. Charts of patients with a final outcome documented at discharge were included, while those whose initial diagnosis was subsequently changed to another condition or with unknown clinical outcomes were excluded.
Sample Size and Sampling Procedure
The source population comprised all children aged 1 month to 14 years admitted with a clinical diagnosis of meningitis to the pediatric ward at Ras Desta Damtew Memorial Hospital between April 2022 and May 2024. A total of 362 eligible meningitis admissions were recorded during this period. The sample size was calculated using the single-population proportion formula, assuming a 18.9% prevalence of poor clinical outcome from a previous study, 13 a 5% margin of error, and a 95% confidence level. After adding 10% to account for incomplete records, the final sample size was 260.
Eligible patient records were identified from the hospital’s electronic medical recording system and assigned unique numeric identifiers. Simple random sampling was performed using a computer-generated random number sequence to select 260 records. Following data extraction, 8 records were excluded due to incomplete documentation of the primary outcome or key independent variables, resulting in a final analytic sample of 252 records. Records with missing data for variables included in the multivariable model were excluded listwise, and no variables with >10% missingness were retained for analysis.
The formula used for sample size determination is
Where
Thus, for a confidence interval level of 95%, α is .05 and the critical value is 1.96 from standard normal table
p = expected proportion in population based on previous research which is 18.9% (0.19)
d = Absolute error (margin of error) which is 5% (0.05)
Then using correction formula 10% of n for incomplete charts = 24
Thus, our final sample size = 260
Data Collection
Data were collected using a structured, pretested checklist adapted from previous similar studies. The checklist captured: sociodemographic characteristics (age, sex, maternal occupation, season of admission), clinical presentation (symptoms, vital signs, physical examination findings), medical history (comorbidities, immunization status, nutritional status), laboratory findings (complete blood count, cerebrospinal fluid analysis), treatment details (antibiotics, corticosteroids), and clinical outcomes. Maternal occupation and season of admission were used in previous studies as an independent variable. Additionally meningitis is seasonal in Ethiopia according to some studies,15,16 and socio economic status of the families will also have an impact on the clinical outcome of meningitis (care-seeking delays, nutritional status, and immunization completion).17,18
Three data collectors (2 Bachelor of Science nurses and 1 general practitioner) were trained on the study objectives and data collection procedures. The checklist was pretested on 5% of the sample at a similar facility, and necessary modifications were made before actual data collection, which occurred from July 31 to August 15, 2024.
Variables and Operational Definitions
Treatment Protocol
At Ras Desta Damtew Memorial Hospital, the standard empirical antibiotic regimen for pediatric meningitis is ceftriaxone (100 mg/kg/day) plus vancomycin (60 mg/kg/day). However, adherence to this protocol is limited by drug stockouts, formulary restrictions, and empirical prescribing practices based on clinician preference. Consequently, alternative regimens including ampicillin plus gentamycin or ceftriaxone alone are frequently used, particularly in infants under 2 months or when vancomycin is unavailable.
Data Analysis
Data were checked for completeness and consistency, entered into Epi Data version 3.0.2, and exported to SPSS version 26 for analysis. Descriptive statistics, including frequencies and percentages, were computed for all sociodemographic, clinical, laboratory, and treatment variables. The primary outcome variable was clinical outcome at discharge, dichotomized as good versus poor. Bivariate logistic regression was first performed to assess the association between each independent variable and clinical outcome. Variables with a P-value <.25 in bivariate analysis were considered candidates for inclusion in the multivariable logistic regression model. To adjust for potential confounding, all candidate variables were entered simultaneously into a multivariable logistic regression model using the enter method to identify independent predictors of good clinical outcome. Statistical significance was set at P < .05, and adjusted odds ratios (AOR) with 95% confidence intervals (CI) were reported. All variables with missing data below 10% (duration of illness, immunization status, and SAM) were handled using listwise deletion, meaning cases with missing values for any of these variables were excluded from the multivariable regression analysis. Variables with more than 10% missingness (CSF analysis result and causative organism) were excluded from the multivariable model to avoid bias and instability. The final analytic sample consisted of 252 records with complete data for all variables included in the multivariable model. Prior to fitting the multivariable model, multicollinearity among independent variables was assessed using variance inflation factor (VIF) and tolerance. Variables with VIF >5 or tolerance <0.2 were considered indicative of problematic collinearity; no variables met this threshold, and all candidate variables were retained. Goodness-of-fit of the final multivariable model was assessed using the Hosmer-Lemeshow test, with P > .05 indicating acceptable model fit (χ2 = 8.24, df = 8, P = .41). Model discrimination was evaluated using the area under the receiver operating characteristic curve (AUC). An AUC value of 0.85 (95% CI: 0.78, 0.92) was obtained, indicating excellent discriminatory ability of the model to distinguish between good and poor clinical outcomes.
Results
Sociodemographic Characteristics
A total of 260 patient charts were initially selected using simple random sampling. After exclusions, 252 charts were included in the final analysis. The majority of participants (219, 86.9%) were aged 2 months and above, and 188 (74.6%) were female. Regarding maternal occupation, 116 (46.0%) were merchants, and 76 (30.2%) were private employees. Most admissions occurred during winter (135, 53.6%) and spring (75, 29.8%; Table 1).
Socio-Demographic Characteristics of Children Admitted with Meningitis (N = 252).

Clinical Characteristics
Comorbidities were present in 80 (31.7%) participants, including pneumonia (4.4%), sepsis (6.7%), severe acute malnutrition (15.5%), malaria (2.0%), and typhoid fever (2.4%). Presenting symptoms included poor feeding (74.2%), neck rigidity (90.5%), seizures (14.3%), irritability (81.0%), and bulged fontanel (0.8%). Positive Brudzinski and Kernig signs were observed in 75 (29.8%) patients. Fever (>37.5°C) was present in 246 (97.7%) of cases (Table 2).
Medical and Clinical Characteristics of Children Admitted with Meningitis (N = 252).

Laboratory Findings
Complete blood count revealed leukocytosis (>10 000 cells/mm3) in 168 (66.7%) participants. Cerebrospinal fluid (CSF) analysis was performed in only 102 (40.5%) cases. Among those with CSF analysis, 82 (80.4%) had turbid CSF, 49 (48.0%) had elevated protein (>50 mg/dl), and 65 (63.7%) had low glucose (<70 mg/dl; Table 3).
Laboratory Findings of Children Admitted with Meningitis (N = 252).

Treatment Characteristics
Most participants (213, 84.5%) had completed their immunization schedule. Corticosteroids were administered to 195 (77.4%) children, and oxygen therapy to 178 (70.6%). The most frequently used antibiotics were ampicillin (27.0%), gentamycin (25.0%), ceftriaxone (21.8%), and vancomycin (22.2%; Table 4).
Treatment Characteristics of Children Admitted with Meningitis (N = 252).

Clinical Outcomes
The magnitude of good clinical outcome among children admitted with meningitis was 90.9% (95% CI: 87.3, 94.4), with 229 children experiencing favorable outcomes and 23 (9.1%) experiencing poor outcomes.
Factors Associated With Clinical Outcome
In bivariate analysis, age, sex, duration of illness, immunization status, presence of comorbidity, corticosteroid use, oxygen administration, and severe acute malnutrition were associated with clinical outcome (P < .25) and were included in multivariable analysis.
In multivariable logistic regression, 4 factors remained independently associated with good clinical outcome: Children aged 2 months and above had 12 times higher odds of good clinical outcome compared to those younger than 2 months (AOR: 12.1; 95% CI: 3.39, 42.89). Children presenting within 72 hours of illness onset had 4.75 times higher odds of good outcome compared to those with longer duration (AOR: 4.75; 95% CI: 1.36, 16.56). Children who completed their immunization schedule had 3.72 times higher odds of good outcome compared to incompletely immunized children (AOR: 3.72; 95% CI: 1.19, 11.63). Children without severe acute malnutrition had 5.18 times higher odds of good outcome compared to malnourished children (AOR: 5.18; 95% CI: 1.18, 22.71; Table 5).
Factors Associated with Good Clinical Outcome Among Children with Meningitis (N = 252).
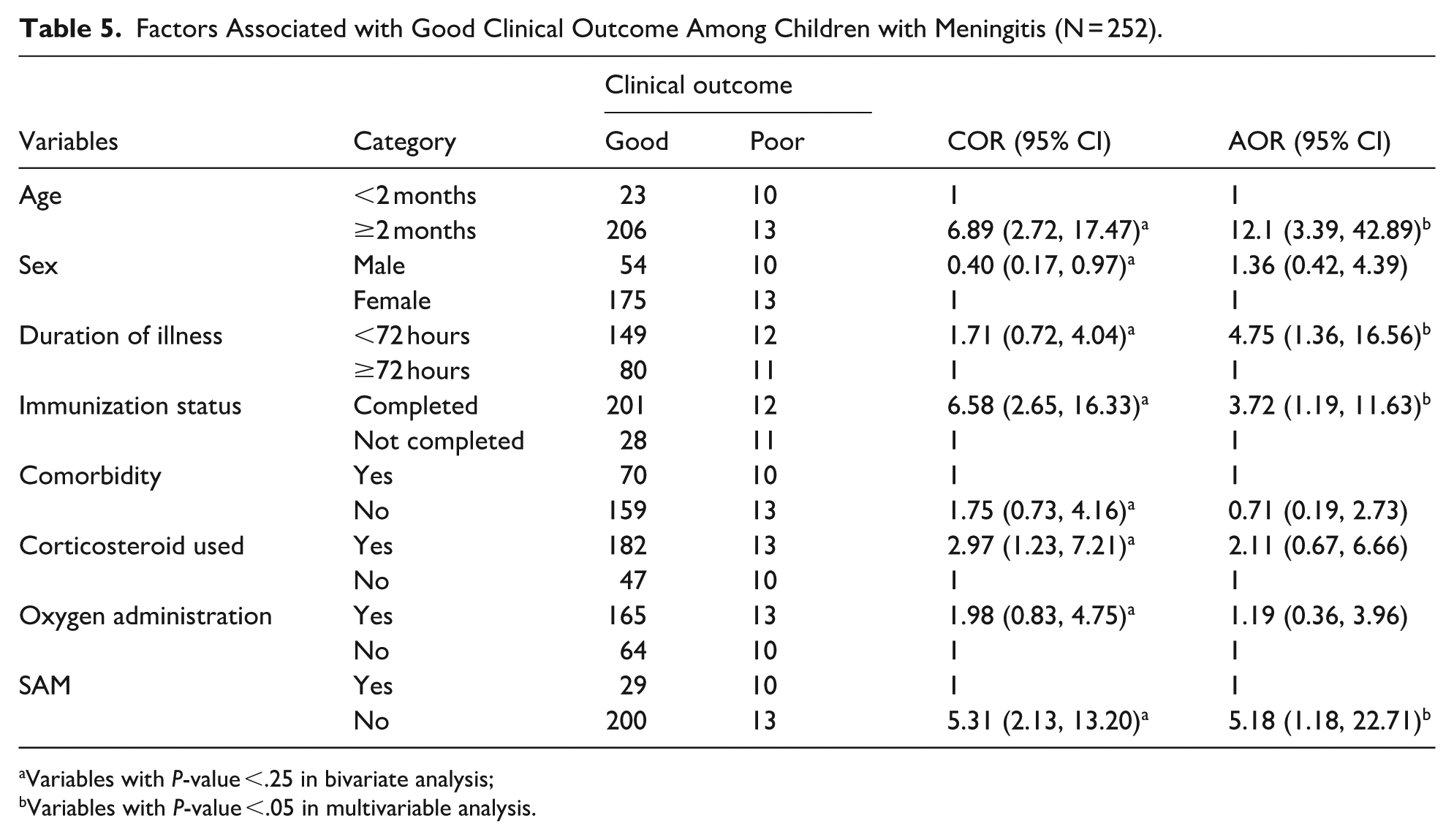
Variables with P-value <.25 in bivariate analysis;
Variables with P-value <.05 in multivariable analysis.
Discussion
The finding of 90.9% good clinical outcomes represents a higher rate than previously reported in Ethiopian studies. At Hiwot Fana Specialized University Hospital, Eastern Ethiopia, good outcomes were observed in 77% of cases, 20 while Felege Hiwot Referral Hospital and Jimma University Specialized Hospital reported 85% and 71.7% good outcomes, respectively.6,28 Several factors may explain this variation. While Addis Ababa has relatively better access to specialized care, diagnostic tools, and antimicrobial agents, differences in disease severity at presentation or referral patterns—whereby children with more severe illness may be referred to tertiary centers outside the capital—could also contribute to the observed outcome differences. Additionally, the higher proportion of clinically diagnosed meningitis cases (59.5% without CSF analysis) in this study may have included milder cases compared to studies requiring microbiological confirmation. Therefore, direct comparisons should be interpreted with caution.
The use of standard first-line therapy (ceftriaxone plus vancomycin) was observed in only 21.8% and 22.2% of cases, respectively, despite being the hospital’s protocol. This likely reflects the high proportion of clinically diagnosed meningitis (59.5% without CSF analysis), where clinicians may favor narrower-spectrum regimens pending diagnostic confirmation. Additionally, empirical use of ampicillin and gentamicin remains common in neonates, and occasional antibiotic stockouts in public hospitals may influence prescribing patterns.
The strong association between age and clinical outcome, with infants under 2 months having significantly poorer outcomes, aligns with findings from studies in the USA, Britain, and Nigeria.29 -31 Newborns and young infants have immature immune systems, making them more susceptible to severe infections and complications. 30 Their limited ability to mount an effective inflammatory response, combined with nonspecific clinical presentations that delay diagnosis, increases the risk of adverse outcomes including neurological damage and death. 31 This underscores the need for heightened clinical suspicion and aggressive management in this vulnerable age group.
The association between shorter illness duration (<72 hours) and better outcomes is consistent with previous Ethiopian research.6,28 Duration was measured from symptom onset to hospital arrival, indicating that early care-seeking by caregivers rather than in-hospital diagnostic delay was the key determinant of improved outcomes. Early medical intervention allows prompt administration of appropriate antibiotics, reducing bacterial proliferation and the associated inflammatory cascade that causes neurological damage. 20 Delayed treatment beyond 72 hours permits progression to more advanced disease stages, with significant inflammation and potential irreversible brain injury.20,28 These findings emphasize the critical importance of healthcare-seeking behavior and early diagnosis in improving meningitis outcomes.
The protective effect of complete immunization, with fully vaccinated children having nearly fourfold higher odds of good outcome, corroborates findings from studies in the USA and Ethiopia.6,29 Vaccination against H. influenzae type b, S. pneumoniae, and Neisseria meningitidis provides direct protection against major bacterial meningitis pathogens. 6 Additionally, immunized children may have more frequent healthcare interactions, enabling earlier detection and intervention if infection occurs.6,29 This finding reinforces the importance of maintaining high vaccination coverage as a cornerstone of meningitis prevention and control. The association between complete immunization and good outcome should be interpreted with caution, as vaccination status may serve as a proxy for socioeconomic advantage. Vaccinated children in this setting are more likely to reside in urban areas with better healthcare access and have caregivers with higher health literacy, factors independently associated with favorable outcomes.17,18 Residual confounding by these unmeasured variables cannot be excluded.
Children without severe acute malnutrition had fivefold higher odds of good outcome compared to malnourished children, consistent with studies from the USA and Eastern Ethiopia.20,29 Malnutrition impairs immune function, reducing the body’s capacity to fight infections and respond to treatment. 29 Essential nutrients required for immune competence and tissue repair are deficient in malnourished children, prolonging illness and increasing complication risk.29,30 Conversely, well-nourished children mount more effective immune responses and recover faster, highlighting the critical role of nutritional support in managing serious infections.
The absence of microbiological data limits interpretation of outcomes by causative organism. In this study, CSF analysis was performed in only 40.5% of cases, and causative pathogens were identified in a minority. This is a notable gap, as bacterial etiology strongly influences prognosis: Streptococcus pneumoniae typically carries higher mortality and neurological sequelae than Neisseria meningitidis or Haemophilus influenzae type b.11,29 The relatively high proportion of good outcomes in this study may partly reflect a case mix with fewer pneumococcal cases, though this cannot be confirmed without microbiological data.
The role of comorbidities in meningitis outcomes warrants consideration. In this study, 31.7% of children had at least 1 comorbid condition, with severe acute malnutrition (15.5%) being most common. The absence of systematic screening for tuberculosis and other chronic illnesses in this retrospective dataset limits our ability to assess their true impact. Future prospective studies should incorporate routine screening for underlying infections to better understand their contribution to meningitis prognosis
Limitations
This study has limitations that should be considered when interpreting the findings. First, the single-center design may limit generalizability to other settings. Second, retrospective data and incomplete medical records could introduce bias. Third, CSF analysis was performed in only 40.5% of cases, limiting etiological characterization and precluding subgroup analysis by microbiological confirmation. Fourth, as a cross-sectional study, causation cannot be established—only associations. Fifth, listwise deletion of missing data may have introduced bias if missingness was not completely at random. Finally, the small number of poor outcomes (n = 23) limited statistical power, and wide confidence intervals should be interpreted as indicating direction and magnitude rather than precise estimates. Future multicenter prospective studies with larger samples are needed.
Conclusion and Recommendations
In this study, the proportion of children with good clinical outcomes was higher than the 71.7% to 85% rates reported in previous Ethiopian studies conducted in regional settings. Age greater than 2 months, illness duration of less than 72 hours before presentation, complete immunization status, and absence of severe acute malnutrition were associated with favorable outcomes. Within the context of this single-center study, these findings suggest potential clinical and public health implications: clinicians should maintain a high index of suspicion for meningitis in young infants and prioritize early treatment, as delayed presentation was associated with worse prognosis. Strengthening routine immunization programs and integrating nutritional assessment and rehabilitation into pediatric meningitis care protocols may be critical interventions. From a public health perspective, promoting early healthcare-seeking behavior among caregivers, enhancing vaccination coverage, and addressing underlying malnutrition could contribute to reducing meningitis-related morbidity and mortality in similar urban healthcare settings. However, given the single-center design, these findings should be interpreted with caution and validated through larger, multicenter studies before broader generalization to the Ethiopian population.
Footnotes
Ethical Considerations
An ethical review clearance was received from Yanet Health Science College (Ref.No: YHSC/0223/07/2021) ethical review committee. Permission was obtained from was obtained from the Addis Ababa Health Bureau and Ras Desta Damtew Hospital Department of Pediatrics. Data were used solely for research purposes and were destroyed after 2 years.
Consent to Participate
Written informed consent on the participant’s behalf was obtained from the caregivers after explaining the purpose of the study. Patient confidentiality was maintained by omitting names and using medical registration numbers as identifiers.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be disclosed on a reasonable request from the corresponding author.*