Abstract
Background:
Arginine can stimulate growth hormone production. However, a synthesis of the evidence on the effect in children’s height is lacking.
Objectives:
To systematically evaluate the efficacy of oral arginine in improving height in prepubertal children with idiopathic short stature.
Design:
Systematic review and meta-analysis.
Data sources and methods:
We included clinical trials and cohort studies. We searched in 7 databases until May 31, 2025. Effects were pooled using standardized mean differences (SMDs) and re-expressed as mean differences (MDs).
Results:
We only found low-quality studies. Oral arginine administered for 6 or 12 months increased growth velocity (SMD 0.66, 95% CI 0.21-1.12), but the benefit was clinically small (re-expressed MD 0.59 cm/year [95% CI 0.19-1.00]), and the effect was not statistically significant when administered for exclusively 12 months.
Conclusion:
Oral arginine administered for 12 months may not affect growth. Well-designed RCTs are required.
Registration:
PROSPERO (CRD42024503778).
Introduction
Short stature is characterized by a height < –2 standard deviation score (third percentile) for the reference population; thus, by definition, 3% of all children are abnormally short.1,2 There are many conditions that can result in short stature, and in some cases short stature can be classified as idiopathic. 2 Management of short stature relies on the treatment of the underlying disorder inducing growth failure, or, in some cases, involves the use of drugs that directly act on the growth hormone/insulin-like growth factor 1 axis. 1
Arginine has the capacity to stimulate growth hormone production and because of this it is used for growth hormone stimulation testing. 3 Furthermore, there have been attempts to assess the effect of oral arginine to improve height in prepubertal children with idiopathic short stature (ISS).4 -6 However, a quality assessment and synthesis of the evidence on the effect of oral arginine in children with ISS has not been published.
The objective of our study was to systematically assess the efficacy of oral arginine as treatment to improve height in prepubertal children with ISS.
Methods
This study is a systematic review and meta-analysis, and its protocol was registered in PROSPERO (CRD42024503778). We followed the PRISMA 2020 guidelines for the reporting of systematic reviews. 7
Study Searches
From the inception until May 31, 2025, we searched the following databases: Scopus, PubMed, Web of Science, Embase, Cochrane CENTRAL Clinical Trials (Through OVID), International Clinical Trials Registry Platform (ICTRP) and ClinicalTrials.gov. If only a group of study subjects in an article were eligible, and there was specific extractable information about that group, these were included. Our search was limited to articles in English, Spanish or Italian. We also conducted hand searches of lists of citations for all the articles that were included and for relevant review articles to find studies that could be eligible. Search strategies of all engines are available in Supplemental Material.
Outcomes
Our protocol primary outcomes were change in height (ΔHt), change in height for age standard deviation score (ΔHt-SDS), change in growth velocity (ΔGV), and change in growth velocity for age standard deviation score (ΔGV-SDS). A SDS adjusts a measurement by age and sex. Thus, we prioritized data expressed as SDS such as Ht-SDS and GV-SDS. We did not include secondary outcomes in our protocol.
Study Selection
We included randomized controlled trials (RCTs), non-randomized trials (ie, quasi-experimental studies), and retrospective and prospective cohort studies assessing the effect of arginine, in any of its pharmaceutical formulations, on outcomes in prepubertal children with ISS. If, in any eligible study, some patients began puberty during the study period, we removed data from those subjects. If there was insufficient information to remove such data, we considered it reasonable to include studies in which no more than 10% of subjects began puberty during the study period, given the limited literature on our research question. We excluded other observational and animal studies. The current definition of short stature is a height for age < −2 SD. However, we were aware that old studies might use other definitions such as a height for age < 10th percentile. We did not exclude studies with other definitions of short stature since the available literature about our research question is scarce. All articles were downloaded to EndNote 20 and we deleted duplicates. We used Rayyan (https://rayyan.qcri.org/) to upload unique papers and begin the selection of eligible articles. Two authors independently screened titles and abstracts to identify relevant studies. The full texts of the selected articles were assessed in an independent manner also by 2 review authors, and the exclusion reasons were recorded. We conducted a hand search of reference lists of all included articles and relevant review articles to identify potentially eligible studies. Disagreements on the selection processes were resolved by consensus.
Data Extraction
The data from each chosen study was extracted separately by 2 reviewers using a standardized form in an Excel spreadsheet, which was tested prior to use. We extracted the following information: name of the first author, country, year of publication, research design, population and sample size, gender, age, comorbidities, group of comparison, treatments, adverse events and outcomes. Disagreements were resolved through consensus.
Risk of Bias Assessment
In each quasi-experimental study, 2 reviewers (M.V. and R.C.) evaluated the risk of bias independently using the Joanna Briggs Institute’s (JBI) Tool for risk of bias assessment for quasi-experimental studies. 8 Disagreements were resolved by consensus. Relevant domains of bias assessed by this tool are allocation and selection bias; temporal procedure bias; administration of treatment/exposure bias; confounding; participant retention bias; and outcome assessment, detection and measurement bias. 8 Final critical evaluation is reported in a narrative manner, since there are not thresholds given by the tool to classify bias according to its severity and leaves it to the reviewers in deciding these thresholds. 8
In each RCT, we assessed the risk of bias using the Cochrane Risk-of-Bias 2.0 tool for RCTs (RoB2). 9 Two authors (M.V. and R.C.) independently performed a risk of bias assessment. Any disagreements were resolved by discussion. Domains assessed by this tool were 5: risk of bias arising from the randomization process, risk of bias due to deviations from the intended interventions, risk of bias due to missing outcome data, risk of bias in measurement of the outcome and risk of bias in selection of the reported result. 9 Each of the domains is given a category of risk of bias: low, some concerns or high. 9 Overall risk of bias is equivalent to the higher risk of bias among the domains assessed. 9
Assessment of Certainty of Evidence
The certainty of the evidence per outcome was evaluated using the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach. 10 The GRADE methodology assesses 5 items: inconsistency, risk of bias, indirectness, reporting bias and imprecision. 10 The certainty of evidence assessment began as high and was decreased based on issues on 1 or more aspects described above. 10 We used the GRADEpro software (McMaster University and Evidence Prime, 2021; www.gradepro.org/) to create the Summary of Findings (SoF) table.
Statistical Analyses
We used inverse-variance random-effects models for meta-analyses. Effects of oral arginine on outcomes were described with mean differences (MD) and their 95% CI if units and measuring tools were the same across studies, or with standardized mean difference (SMD) when units or measuring tools were different across studies. Standardized mean differences (SMD) were estimated by Cohen’s d with their 95% confidence interval (CI). We employed the I2 statistic to assess statistical heterogeneity in the following manner: <30% mild, 30% to 60% moderate, and >60% high heterogeneity. We used the meta packed of R statistical software version 4.3.3 (www.r-project.org) for meta-analyses. A statistically significant result was defined as having a P < .05 for a two-tailed test.
For a better interpretation of the SMDs, the pooled GV SMDs of our meta-analyses were re-expressed choosing 0.9 as the reference GV SD, which is the mean GV SD for prepuberal boys and girls between 2 and 12 years according to Rikken and Wit. 11 We chose the age range of 2 to 12 years because that is the age range used by the studies included in our meta-analyses. Cochrane Handbook describes how to re-express SMDs with a reference SD from a representative study: SMD × reference SD = MD). 12
Ethical Approval and Informed Consent
Since this systematic review did not involve human subjects, either directly or indirectly, ethical approval and informed consent were not obtained.
Results
After electronic searches, we obtained 2786 references and assessed inclusion in 1954 references after removing 832 duplicates. We selected 6 papers,4 -6,13 -15 from which our hand search of reference lists yielded 4 more papers.16 -19 Thus, we selected 10 papers to perform full-text screening.4 -6,13 -19 We finally included 6 articles in our study4 -6,13,16,17 (Figure 1).

Study selection flow diagram.
The excluded a letter to the editor that did not report any anthropometric data, 14 a retrospective cohort that did not include subjects who met our inclusion criteria, 18 a retrospective cohort without any of our primary outcomes and growth velocity at the end of the study as the only anthropometric data, 15 and another retrospective cohort without our primary outcomes and no basal and follow-up anthropometric data to calculate any change in growth. 19
Selected studies were published from 1972 to 1993.4 -6,13,16,17 Four of them were performed in Italy5,6,13,17, one in France, 16 and the other was conducted in Spain. 4 All selected studies were experimental studies evaluating the effect of oral arginine for improving growth velocity in prepubertal children with short stature with characteristics consistent with ISS during 6 to 24 months of follow-up.4 -6,13,16,17 The study by Cicognani et al was an RCT controlled with placebo 13 and the other 5 studies had a quasi-experimental design.4 -6,16,17 We provided detailed general information and inclusion criteria of selected studies in Table 1.
Characteristics of Selected Studies.
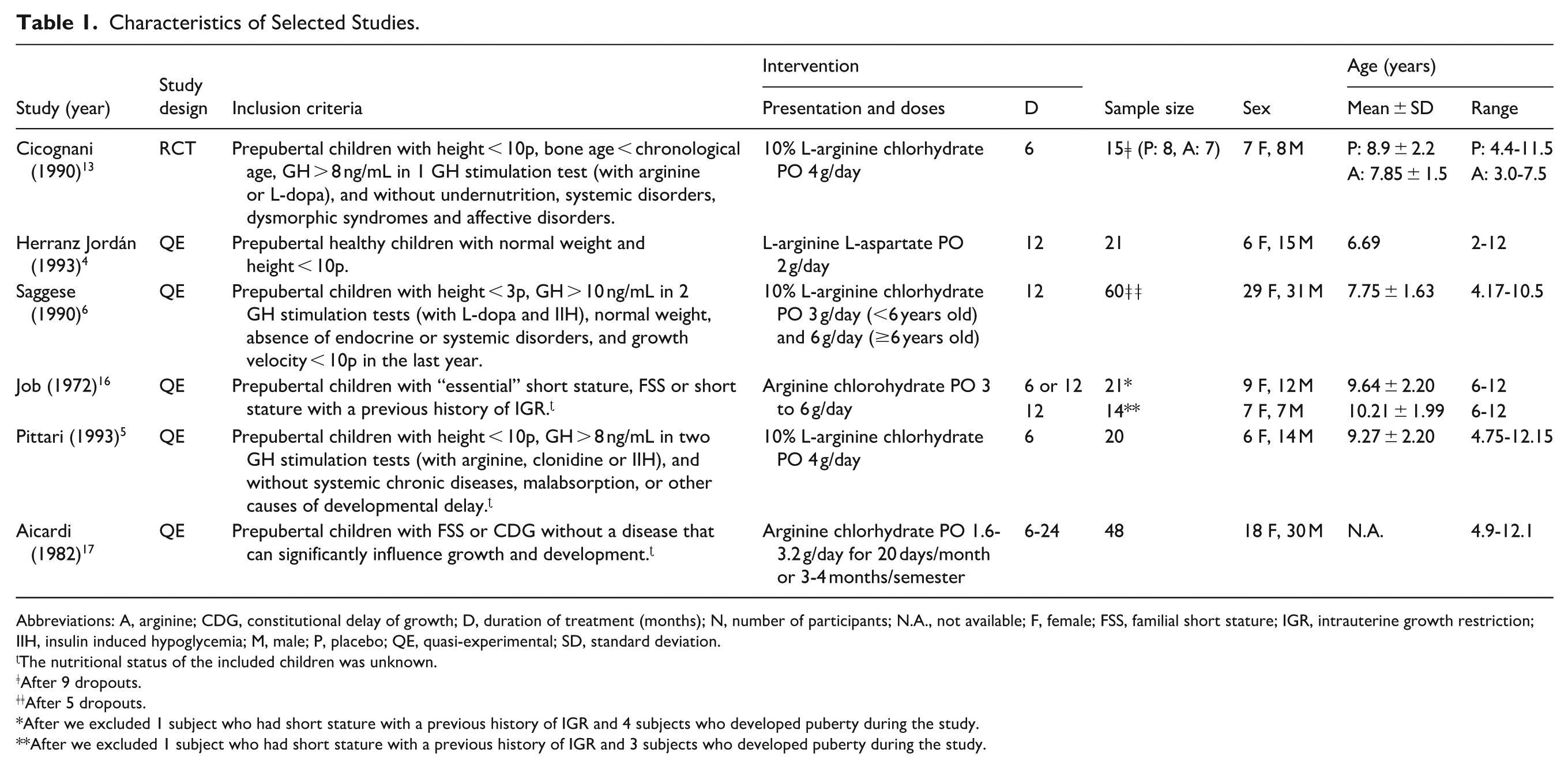
Abbreviations: A, arginine; CDG, constitutional delay of growth; D, duration of treatment (months); N, number of participants; N.A., not available; F, female; FSS, familial short stature; IGR, intrauterine growth restriction; IIH, insulin induced hypoglycemia; M, male; P, placebo; QE, quasi-experimental; SD, standard deviation.
The nutritional status of the included children was unknown.
After 9 dropouts.
After 5 dropouts.
After we excluded 1 subject who had short stature with a previous history of IGR and 4 subjects who developed puberty during the study.
After we excluded 1 subject who had short stature with a previous history of IGR and 3 subjects who developed puberty during the study.
In the study by Herranz Jordán et al, 4 2 children who received oral arginine started puberty during the study period. Since we considered that 2 subjects with puberty would not significantly change the results of our analyses and individual data of the subjects from the study by Herranz Jordán et al were lacking, 4 we included the results from the study by Herranz Jordán et al in our research.
In the study by Job, 16 1 child had a previous history of intrauterine growth restriction (IGR) and 4 children developed puberty during the study period. Since the author provided detailed individual data, 16 we excluded the information of the participant with history of IGR and the subjects who developed puberty during the study.
In the studies by Cicognani et al, 13 Saggese et al, 6 and Aicardi et al, 17 subjects who entered puberty during the study period were excluded. Pittari et al, 5 stated that none of the “older subjects” included at the beginning of their study entered puberty during the study period.
All studies measured GV in cm/year and/or GV-SDS before and after treatment.4 -6,13,16,17 The studies by Herranz Jordán et al 4 and Aicardi et al 17 only reported the mean ± SD of GV. The study by Pittari et al 5 only reported the mean ± SD of GV-SDS. The studies by Saggese et al 6 and Cicognani et al 13 reported the mean ± SD of GV and GV-SDS, and the article by Cicognani et al 13 also provided individual data that allowed us to calculate mean differences and confidence intervals by using the t-test (Table 2). The study by Job 16 showed individual data including age, sex and GV at 0, 6 and 12 months of treatment (some patients were followed only 6 months). Thus, we were able to calculate the mean ± SD of GV and GV-SDS before and after treatment using the data from the article by Job 16 and reference charts by Tanner et al. 20 Then, we were also able to calculate mean differences and confidence intervals by using the t-test (Table 2).
Outcomes of the Selected Studies.

Arginine, Placebo, Pre and Post values are expressed as
Abbreviations: CI, confidence interval; MD, mean difference; N.A., not available; N.S., no statistically significant difference; SD, standard deviation;
P-value reported in the article (it is unclear which test was used).
P-values reported in the articles (t-tests).
The RCT by Cicognani et al 13 showed no statistically significant difference in the ΔGV or ΔGV-SDS between the administration of oral arginine or placebo, and 2 quasi-experimental studies4,16 also showed no statistically significant effect (Table 2). Three quasi-experimental studies5,6,17 showed a statistically significant positive effect of the administration of oral arginine (Table 2).
Herranz Jordán et al 4 reported no statistically significant benefit of oral arginine to increase Ht-SDS in prepubertal children with ISS. They also reported GV before and after treatment. 4 However, they did not report the results of a statistical test to compare those results. 4
Job 16 did not report statistical tests to compare means. However, since complete individual data was available in his paper 16 we were able to calculate MDs with 95% CIs in those patients with 6 or 12 months of treatment, or in those with exclusively 12 months of treatment (Table 2).
Two quasi-experimental studies reported Ht-SDS before and after 1 year of treatment, showing no statistically significant effect4,6 (Table 2). However, since one of them reported the means of Ht-SDS measurements without standard deviations, 4 we could not perform a meta-analysis using ΔHt-SDS as an outcome. As detailed Ht (cm) data before and after intervention was reported by only 1 quasi-experimental study, 6 we could not perform a meta-analysis using ΔHt as an outcome.
After risk of bias assessment of the RCT by Cicognani et al, 13 we concluded that ΔGV-SDS and ΔGV had a high risk of bias due to deviations from the intended interventions (effect of adhering to intervention; Table 3).
Risk of Bias Assessment of Randomized Controlled Trials Using the Cochrane RoB2 Tool.

After risk of bias assessment of the quasi-experimental studies by Herranz Jordán et al, 4 Saggese et al, 6 Pittari et al 5 and Job, 16 we concluded that ΔGV-SDS and ΔGV had a low risk of bias related to participant selection and allocation (Table 4). Further, we concluded that ΔGV in the study by Aicardi et al 17 had a very high risk of bias related to participant selection and allocation; assessment, detection and measurement of the outcome; and invalid statistical analysis (Table 4). Thus, the study by Aicardi et al 17 was not included in our meta-analyses (Table 4).
Results of Critical Appraisal Using the Revised JBI Critical Appraisal Tool for Quasi-Experimental Studies.

It was unclear if there was a risk of bias related to the administration of the intervention when we assessed ΔGV-SDS and ΔGV in all the quasi-experimental studies since it is unknown if participants were not allowed to receive other interventions simultaneously4 -6,16,17 (Table 4). In the article by Herranz Jordán et al, 4 it was unclear which test was used to analyze ΔHt-SDS and it was unknown if a statistical test was used to analyze ΔGV (Table 4).
The study by Cicognani et al 13 could not be used to perform a meta-analysis since it was a RCT and the other studies were quasi-experimental studies.4 -6,16,17
We performed meta-analyses to assess the effect of oral arginine including the studies by Herranz Jordán et al, 4 Saggese et al, 6 Pittari et al 5 and Job. 16 Our meta-analyses prioritized the use of ΔGV-SDS as the main outcome because GV-SDS adjusts GV by age (and sex since reference charts provide different values according to age and sex). The studies by Herranz Jordán et al and Pittari et al only provided information about GV, and information needed to calculate GV-SDS was lacking. Thus, we used SMDs to include ΔGV from those studies in our meta-analyses.
The meta-analysis of the 4 mentioned quasi-experimental studies4 -6,16 showed that the effect of oral arginine administered for 6 or 12 months on GV was positive (SMD 0.66 [95% CI 0.21-1.12]; re-expressed MD 0.59 [95% CI 0.19-1.00]; Figure 2). However, the meta-analysis that included only studies and patients that received oral arginine for 12 months4,6,16 showed no statistically significant effect (SMD 0.58 [95% CI 0.10-1.27]; re-expressed MD 0.52 [95% CI 0.09-1.14]; Figure 3).

Effect of oral arginine on growth velocity after 6 or 12 months of treatment.

Effect of oral arginine on growth velocity after 12 months of treatment.
According to GRADE approach, in the RCT by Cicognani et al, 13 ΔGV-SDS and ΔGV provided very low certainty evidence due to very serious imprecision and strong suspicion of publication bias (Table 5). Similarly, in the quasi-experimental studies included in our meta-analyses,4 -6,16 change in growth velocity after 6 or 12, or only 12 months of treatment (measured as ΔGV-SDS in the studies by Job, 16 Saggese et al 6 and Pittari et al, 5 or ΔGV in the study by Herranz Jordán et al 4 ) provided very low certainty evidence, which was related to risk of bias, inconsistency, imprecision and strong suspicion of publication bias (Table 5).
Summary of Findings Table of the Quality of Evidence Per Outcome.

Abbreviations: CI, confidence interval; N.E., not estimable; SD, standard deviations; SMD, standardized mean difference; Ar., arginine group; Co., control group.
Very small sample size in the included study (placebo: 8 subjects, arginine: 7 subjects).
Evidence limited to one very small study.
Lack of allocation concealment and lack of blinding in all the studies.
I2 = 66.9%, P = .0286.
Small sample sizes in all included studies (15-60 patients).
Evidence limited to a few small studies.
I2 = 80.0%, P = .0067.
In the study by Herranz Jordán et al we analyzed GV, and in the studies by Job, Saggese et al and Pittari et al we analyzed GV-SDS.
Four studies reported no complications,5,6,16,17 another described an increase in appetite and sleepiness in 7 and 2 patients, respectively, 4 and the other 1 mentioned that 1 patient stopped arginine treatment due to nausea. 13
Discussion
There is not enough high-quality evidence to conclude an effect of oral arginine on growth. Our first meta-analysis (6 or 12 months of treatment) suggested that oral arginine increased growth, and the outcomes analyzed had a low risk of bias and very low certainty of evidence. Conversely, our second meta-analysis (only 12 months of treatment) suggested that oral arginine had no effect on growth, but the outcomes had the same risk of bias and certainty profile. Moreover, the outcomes from the only RCT we found also suggested that oral arginine had no effect on growth but had a high risk of bias and a very low certainty of evidence.
Amino acids are very popular because of their real or perceived properties, and they are widely consumed as dietary supplements. 21 Arginine is probably the most popular and widely consumed. 21 Arginine, a nonessential amino acid, is acquired from diet, protein turnover and produced endogenously; and is crucial for protein synthesis. It serves as a precursor to various molecules including urea, nitric oxide, polyamines, proline and agmatine. 22 In recent years, arginine metabolism has been studied due to its role in cellular activation and growth, and also inflammation, which are processes involved in tumor development. 22 Cancer cells are directly affected by the availability of arginine. 22 Prostate and breast cancer cells experience mitochondrial dysfunction, depletion of mitochondrial metabolites, changes in mitochondrial morphology and production of mitochondrial reactive oxygen species due to arginine deprivation. 22 The need of cancer cells for certain amino acids because of their inability to produce them has been the basis for treatment strategies that involve depriving the cells of these specific. 22 Arginine, for example, is necessary for the development of cancer cells, but it is not required for normal cells. 22
In human subjects, intravenous arginine stimulates GH production in GH stimulation tests.3,23 In short children, after performing different studies to diagnose secondary causes of short stature, growth hormone stimulation tests are usually needed to diagnose growth hormone deficiency. 24 When assessing the possibility of growth hormone deficiency, 2 GH stimulation tests with different stimuli should be done. 25 Insulin-induced hypoglycemia was the standard stimulation test. 23 However, since the publication of Root et al in 1969 and other studies showing the effectiveness of arginine on GH release,3,23 nowadays intravenous arginine is one of the most used drugs for GH stimulation testing, together with clonidine and also glucagon.3,23 The use of drugs different from insulin became a better choice since there were concerns regarding adverse effects related to insulin-induced hypoglycemia. 23
Since there has been a wide use of arginine as a GH stimulant for diagnostic purposes and there is evidence of the effect of arginine in cell growth promotion, it is reasonable to propose that it may have a potential utility as a treatment for short stature in children. However, once L-arginine is orally ingested, it is eliminated through extensive pre-systemic and systemic processes (ie, by gut bacteria and intestinal and hepatic arginases, respectively). 26 So, oral administration of L-arginine probably is not an efficient way to deliver this molecule to the target tissues.
Moreover, experimental evidence that assesses the effect of arginine as a treatment for ISS in prepubertal children is limited to 5 quasi-experimental studies4 -6,16,17 and 1 RCT 13 with small sample sizes.
The study by Aicardi et al 17 was a quasi-experimental study that showed a positive effect of oral arginine to improve growth velocity in prepubertal children with ISS: +1.37 ± 1.06 cm/year (mean ± SD), P < .001. Unfortunately, their methodology would invalidate their results. 17 Aicardi et al 17 gave arginine chlorhydrate PO 1.6 to 3.2 g/day for 20 days/month or 3 to 4 months/semester to prepubertal children. Total treatment duration was 6 to 24 months. 17 The authors did not provide outcome data stratified by each of the multiple possible treatment regimens administered. 17 Instead, they only provided outcome summary measures of all subjects. 17 This introduces bias due to the wide heterogeneity of the interventions given to each child.
On the other hand, Aicardi et al 17 showed the GV (before treatment and at follow-up) for all the study subjects, for those with familial short stature and those with constitutional delay of growth and puberty. However, they stated in the heading of their results table that the follow-up measurements are “growth velocity during treatment” but inside the table they mentioned that those measurements are “growth velocity during or after treatment.” 17 The authors did not provide other descriptions of the way follow-up measurements were taken. 17 If GV was measured “during treatment” without any clear criteria about when to perform the measurements it means that any GV detected after beginning treatment and before treatment stopped was used in the study; on the other hand, if GV was measured “during or after treatment” it means that any of the 2 measurement options was used in the study. 17 Both situations suggest that authors could choose the follow-up GV measurement of each subject according to their own subjective criteria, which is a biased and unethical behavior that would invalidate the results of their article.
Saggese et al 6 showed that oral arginine had a positive effect on growth in children with ISS and low GV. However, a low GV (<10th percentile 6 ) at the beginning of the study suggests these children possibly had undetected comorbidities such as an underlying nutritional deficiency, which could explain a positive response to treatment.
Pittari et al 5 found that oral arginine also had a positive effect. However, this study had a SMD confidence interval very close to the null value in our first meta-analysis (Figure 2) and a short duration of treatment (6 months). 5 Further, baseline GV in some participants was <1st percentile. 5 Thus, similarly to the hypothesis we proposed for the study by Saggese et al, 6 some participants possibly had undetected nutritional deficiencies that could explain the positive effect.
Regarding complications associated with oral arginine, the studies included in our review did not report severe adverse effects. However, there have been several pediatric case reports describing esophagitis or esophageal ulcers induced by L-arginine given orally as a treatment of short stature.27 -30 There has been also 1 report about a esophageal ulcer in a 22-year-old adult who started oral L-arginine 1 g/day to increase his cycling performance. 31
The meta-analysis that assessed the effect of oral arginine administered for 6 or 12 months showed a statistically significant positive effect (Figure 2). However, when the pooled GV SMD was re-expressed as an MD, the benefit was clinically small (0.59 cm/year), and the confidence interval included values close to zero (the lower bound was 0.19 cm/year). Furthermore, the effect became non-statistically significant when we analyzed the effect of oral arginine administered for exclusively 12 months (Figure 3). GV is measured in cm/year. Thus, GV or GV-SDS after 6 months of treatment can be calculated in 2 ways: (a) performing the measurement over a period that includes the 6 months prior to the start of treatment and the 6 months following the start of treatment, or (b) duplicating the observed 6-month growth to estimate a 1-year growth. Both calculation methods can induce bias. Therefore, the meta-analysis that assessed the effect of oral arginine administered for exclusively 12 months is probably more reliable than the one that assessed the effect of oral arginine administered for 6 or 12 months. Moreover, studies with longer treatment times are generally more reliable to assess growth outcomes, which increases the importance of the results of the meta-analysis that assessed the effect of exclusively 12 months of treatment.
Our study has several limitations: the outcome of the only RCT 13 included in our review had a high risk of bias (Table 3) and the other studies that accomplished our inclusion criteria were quasi-experimental4 -6,16,17 (Table 4); and the outcomes used in our meta-analyses had a very low certainty (Table 5). Further, since the literature found is not recent, some patients may have been misdiagnosed with idiopathic short stature due to the limitations of medicine at that time. If a patient has an undiagnosed disease inducing growth failure, no growth improvement is expected without etiological treatment.
In conclusion, our study suggests that oral arginine administered for 12 months may not have a significant effect on growth. However, we only found low-quality studies to perform our review. Therefore, contemporary, well-designed randomized controlled trials with standardized outcomes and adequate follow-up, blinding, and allocation concealment are required to address current evidence gaps.
Supplemental Material
sj-docx-1-gph-10.1177_30502225261433767 – Supplemental material for Efficacy of Oral Arginine as Treatment to Improve Height in Prepubertal Children With Idiopathic Short Stature: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-gph-10.1177_30502225261433767 for Efficacy of Oral Arginine as Treatment to Improve Height in Prepubertal Children With Idiopathic Short Stature: A Systematic Review and Meta-Analysis by Manuel André Virú-Loza, Ruth Elizabeth Chávez-Nomberto and Adrian V. Hernandez in Sage Open Pediatrics
Footnotes
Ethical Considerations
Since this systematic review did not involve human subjects, either directly or indirectly, ethical approval was not obtained.
Author Contributions
MAV-L: contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring itegrity and accuracy. REC-N: contributed to conception, contributed to acquisition and analysis, drafted manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring itegrity and accuracy. AVH: contributed to design, contributed to interpretation, drafted manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Universidad San Ignacio de Loyola funded the publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data used in our study is publicly available.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
