Abstract
Aim:
This study aimed to investigate the impact of different types of baby wipes on the barrier recovery of compromised skin after repeated wiping.
Methods:
The forearm skin of adult females was compromised through repeated tape-stripping. Baby wipes with different compositions and water and cloth were used to repeatedly wipe the compromised forearm skin. The health of the skin barrier was evaluated by transepidermal water loss and erythema over the course of 5 days.
Results:
Skin wiped with pH-buffered baby wipes containing emollients had better barrier health in comparison to skin wiped with baby wipes lacking these features. Additionally, one of the pH-buffered wipes with emollients was gentler on compromised skin than the other baby wipes, as well as water and cloth and performed similarly to unwiped skin.
Conclusion:
Baby wipes composition can affect the barrier health of compromised skin after wiping.
Introduction
Infant skin, like adult skin, is comprised of 3 main layers that include the epidermis, dermis, and hypodermis. 1 The epidermis is the outermost skin layer, and it is divided into multiple sub-layers with different structures and functions. The outermost layer of the epidermis, the stratum corneum, is of particular interest as it acts as a barrier to the external environment. This layer is the first line of defense against skin irritants and it is characterized by its slightly acidic pH and by its diverse microbiome. The stratum corneum is responsible for regulating heat, ion balance, and protecting from mechanical stress.1 -5
Although a full-term infant has a competent skin barrier at birth, its maturation continues throughout their first year of life. Thus, protecting the skin barrier from mechanical damage and irritation is of utmost importance to support healthy skin and overall health. This is especially true for premature infants, as their underdeveloped skin barriers are thinner and more fragile than the skin barriers of full-term infants, making them more susceptible to irritation.6 -10 The skin in the diapered area is prone to irritation due to prolonged exposure to excess moisture and to digestive enzymes from feces that can breakdown the skin barrier, leading to the onset of inflammation.11,12 Therefore, gentle and effective cleaning of diapered skin is key to caring for the delicate skin of infants, especially for premature newborns.
Supporting the natural acidic pH of the skin barrier is another important factor that must be considered to help maintain healthy skin. This slightly acidic pH known as the acid mantle promotes skin barrier maturation, helps establish a healthy skin microbiome and helps protect from fecal enzyme-induced skin irritation.4,7,12,13 Although the skin of a healthy full-term infant acidifies quickly within the first few days of birth, this process can take longer for premature infants, putting them at risk for irritation and infection.14,15 Therefore, when cleaning diapered skin, it is important to use products that do not disrupt the natural acidity of the skin barrier. Although water alone is commonly used to clean infant diapered skin, water is generally neutral to basic in pH 16 and does not match the acidic nature of healthy infant skin, which could affect the acid mantle and compromise protection against the effects of fecal enzymes.
The Association of Women’s Health, Obstetric and Neonatal Nurses recommends the use of a properly formulated baby wipes as a safe and gentle way to clean infant diapered skin.17,18 Furthermore, multiple studies have demonstrated the safety and efficacy of using baby wipes on fragile premature skin.9,19 To increase gentleness when cleaning diapered skin, some baby wipes contain ingredients commonly referred to as emollients to help reduce the friction against the skin during wiping. 20 Studies have shown the positive impact that emollients may have on skin health, which is largely attributed to the hydrophobic nature of these materials which serves to replenish the protection provided by natural skin lipids.21 -24 Emollients have been demonstrated to improve symptoms related to inflammatory skin disorders such as psoriasis and atopic dermatitis and increase barrier health and hydration of infant skin.18,24 -30 Despite the known benefits of emollients, usage in baby wipes remains low due to their hydrophobic nature, making emollients difficult to stabilize in a largely aqueous formulation. Therefore, emollient rich baby wipes are uncommon and there are few, if any, studies that demonstrate the benefits of using such wipes on infant diapered skin.
The purpose of this study was to understand how different baby wipes impacted skin barrier recovery of compromised skin after it was wiped. These baby wipes were compared to cotton washcloths wetted with water per guidelines for diapering infants. 31 This study aims to provide care-givers with information on how wipes with different compositions may impact compromised skin.
Material and Methods
Subjects
The protocol used in this study was reviewed and approved by Sterling Institutional Review Board (ID#5995-001). All subjects provided written informed consent prior to enrollment in the study. For this study, 40 generally healthy women between the ages of 18 to 55 with a Fitzpatrick skin type of I-IV were recruited. Participation in the study was voluntary and participants were compensated for their time and travel costs to/from the clinical site. To ensure compliance with the study protocol, participants agreed to avoid tanning, and not use excluded medications and skin preparations during the study. Continued compliance to the study requirements was confirmed at each visit. Participants with known or documented allergies to adhesives or products containing perfumes, a skin cancer diagnosis within the previous 12 months, or any condition on the inner forearm that might prevent a clear assessment of the skin were not enrolled in the study. In addition, participants met a baseline trans-epidermal water loss (TEWL) measurement of <10 g/m2/h at all skin testing sites prior to continuation with the testing procedures.
Study Design
The study design was controlled, single-blind, randomized complete-block where each subject tested up to 5 treatments simultaneously over 5 days. One additional randomized forearm test site remained untreated. Serial tape stripping was performed at each test site except the untreated control using D-Squame tape (Cuderm, Dallas, TX) and pressing the tape with the tape strip applicator ensuring even pressure. Tapes were removed with forceps in a slow, backwards motion. Periodic TEWL checks during the tape stripping process were completed until the target TEWL level of 40 to 50 g/m2/h was achieved. This TEWL value simulates the compromised skin barrier found in premature infants at approximately 26 to 28 weeks of gestational age.32,33 Forearm test sites were wiped with baby wipes or water and cloth using an exaggerated wiping protocol previously described. 33 For this, each forearm site was wiped 60 times per session and 4 sessions per day for a total of 4 days by a single trained wiper to limit variability. A fresh wipe or wetted cloth was used every 10 wipes to ensure consistent treatment to the skin. One tape stripped site was left unwiped and referred to as the unwiped control. One test site was not tape stripped or wiped and was referred to as the untreated control site. Subjects returned to the testing location the following testing day to have skin barrier and erythema measurements collected prior to continuing the next set of wiping sessions, except for test day 5 where only measurements were collected.
Skin Barrier and Erythema Measurements
Skin barrier as measured by transepidermal water loss (TEWL) using the Cortex DermaLab® TEWL probe was collected at each forearm skin test site averaging 30 one second readings after a 30 second stabilization period. Measurements were collected on test day 1 prior to serial tape stripping (Baseline), again after target TEWL levels were obtained and at the start of each subsequent testing day for total of 5 days. Erythema was measured by tri-stimulus colorimetry using the Courage & Khazaka Skin Colorimeter and CIE L * a * b * color space at each forearm skin test site. An average of 3 readings at the test site were reported. Measurements were collected on test day 1 after target TEWL levels were met and at the start of each subsequent testing day for total of 5 days.
Baby Wipes and Water and Cloth
For this study, 3 commercially available baby wipes and cotton cloth wetted with water were compared. Baby wipes A (Huggies® Natural Care® Extra Sensitive wipe by Kimberly-Clark®) contained emollients per manufactures ingredient list (Table 1) and was buffered to a slightly acidic pH. Baby wipes B (a sensitive wipe produced by Procter and Gamble®) contained an ingredient(s) classified as emollients or skin conditioners per the manufacturer’s ingredient list (see Table 1) and was buffered to a slightly acidic pH. Baby wipes C (a water-based wipe with grapefruit seed extract produced by WaterWipes®) did not contain an emollient ingredient, was not buffered, and was at a basic pH (Table 1). Cotton cloth wetted with sterile distilled water was also tested because this method has been recommended to clean diapered skin. 31 For this, 100% cotton washcloths were saturated in sterile distilled water and wrung out to reduce dripping before wiping forearms. The pH values of the products provided in Table 1 were obtained from hand compressing the baby wipes and measuring the pH of the expressed solution that was collected.
Ingredient List of Baby Wipes Tested.

Avg., average; st. dev., standard deviation.
Data Analysis
TEWL and erythema were standardized across subjects by subtracting untreated site (control) measurements from test measurements. All analyses were conducted on the change from untreated response. Each time point was analyzed separately using ANOVA. Tests for statistical significance were conducted comparing baby wipes treated sites to the tape strip only (unwiped) site using a Dunnett’s test at the 95% confidence level.
Ethical Approval and Informed Consent
The methodology used in this work received ethical approval through an expedited review process by Sterling Institutional Review Board (ID#5995-001). All subjects participating in this work provided voluntary consent utilizing an in-person Informed Consent process.
Results
Emollient Rich, pH-Buffered Baby Wipes Improved Skin Barrier Health Compared to Other Wipes
The first aim of the study was to determine the barrier recovery of skin after it was wiped with baby wipes or cloth and water. For this, forearms were serially tape stripped to model compromised skin and transepidermal water loss (TEWL) was measured over the course of 5 days after the skin was wiped with baby wipes a total of 240 times each day. Results showed that skin wiped with baby wipe A had TEWL values that were no different than the unwiped skin on days 4 and 5 (Figure 1 and Table 2). Skin sites wiped with baby wipe C, baby wipe B, and water and cloth showed higher TEWL values compared to unwiped skin (Table 2). These data indicate that barrier recovery of skin wiped with baby wipe B and C was delayed compared to baby wipe A. Moreover, the barrier health of skin wiped with wipe A was equivalent to the unwiped control site by days 4 and 5.

TEWL values of skin wiped with emollient rich, pH-buffered baby wipes were similar to unwiped skin. The graph shows the change in TEWL values from untreated control skin. Error bars denote mean ± standard error.
Difference in Average Transepidermal Water Loss Values Versus Untreated Site at Day 4 and 5.

Avg., average; SG, significance; Y, yes; N, no.
Furthermore, TEWL values of skin wiped with water and cloth or baby wipe C (no emollient, not pH buffered) showed the highest TEWL values over the course of testing (Figure 1) indicating barrier recovery was the slowest with water and water-like baby wipe formulations. Taken together these data indicate that baby wipe A supported skin barrier health better than the other baby wipes tested.
Erythema on Skin Wiped with Emollient Rich, pH Buffered Baby Wipe was Equal to Unwiped Skin
To determine how baby wipes and water and cloth affected skin erythema, skin redness (a*) was measured over the course of the study after the skin was wiped with the baby wipes or cloth and water. Similar to TEWL results, redness of skin wiped with baby wipes A was no different than unwiped skin on days 4 and 5 (Figure 2 and Table 3). Baby wipes B, C, and cloth and water showed statistically higher erythema measurements on days 4 and 5 as compared to the unwiped control skin (Table 3). These data indicated that baby wipes A were less irritating on compromised skin than the other wipes that were tested.
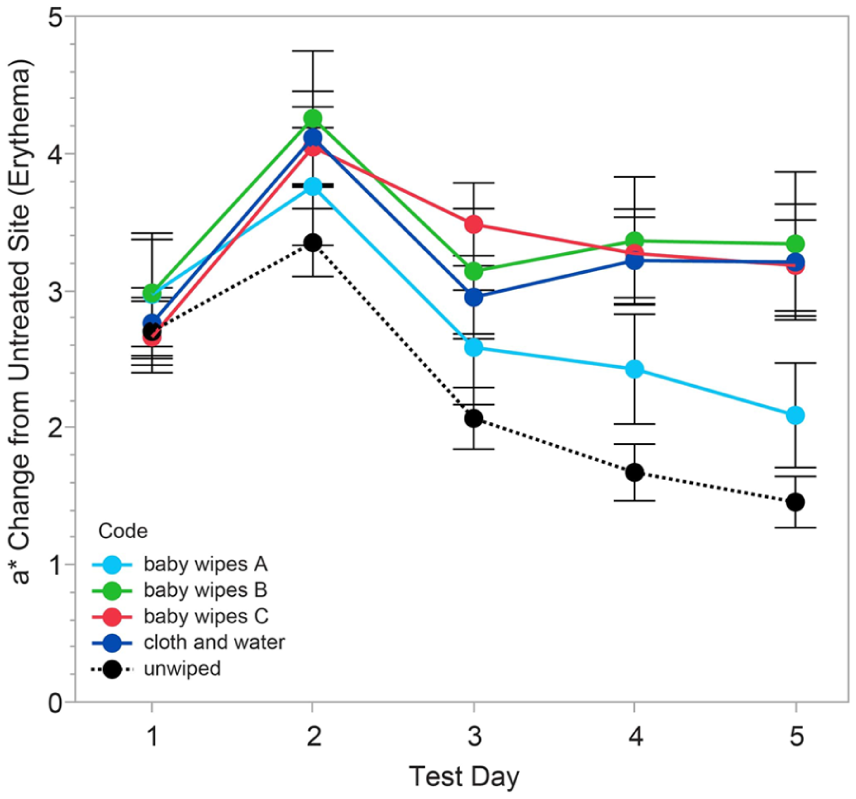
Skin redness (a*) of sites wiped with baby wipes A was no different than unwiped skin on days 4 and 5 that was tape stripped. The graph shows the difference in skin redness values from untreated control skin. Error bars denote mean ± standard error.
Change in Skin Redness (a*) from Untreated Site on Days 4 and 5.

Avg., average; SG, significance; Y, yes; N, no.
Discussion
Using products that gently and effectively clean infant diapered is important to prevent irritation and maintain healthy skin, especially for the underdeveloped and fragile skin of premature infants.7,8 The use of properly formulated baby wipes has been demonstrated to be a gentle and effective way to clean diapered infant skin and this approach is recommended by the Association of Women’s Health, Obstetric and Neonatal Nurses.9,18,34 -37 In this study, an adult forearm compromised skin model was used to evaluate the gentleness of different baby wipes or cloth and water when wiping compromised skin.
Skin wiped with baby wipes A experienced a faster barrier recovery than skin wiped with baby wipes B or C. By day 4, the barrier health and erythema of skin wiped with baby wipes A had recovered to levels comparable to unwiped skin, despite repetitive wiping. On day 5, the average TEWL value of skin wiped with baby wipes A was 9.25 g/m2/h and the erythema score was 2.09. These values were consistent with and not statistically different from the unwiped skin sites, which showed values of 8.82 and 1.46 g/m2/h, respectively. This suggests that baby wipes A enabled a faster recovery of the compromised skin barrier than the other tested baby wipes. Additionally, skin sites wiped with baby wipes B, C, and water and cloth exhibited greater redness and irritation compared to those wiped with baby wipes A. Consequently, these results indicate that baby wipes A are gentler on compromised skin than baby wipes B, C, and cloth and water, and comparable to unwiped skin on days 4 and 5.
Baby wipes A and B contained emollient ingredients and were pH-buffered. Emollients are known to help reduce the coefficient of friction between a material and the skin, helping the material glide across the skin. 20 Furthermore, they also help in protecting the skin barrier due to their hydrophobic properties.21 -24 In addition, baby wipes A and B were pH buffered to support the natural acidic pH of healthy skin, which is an essential attribute for barrier maturation and health of the skin microbiome.1,13 Wipes that contained emollients and were pH-buffered minimized damage to the compromised skin upon repetitive wiping compared to wipes that lacked these features, thereby enabling faster recovery of the skin barrier. These findings indicate that emollients and pH-buffering are key attributes for gentle and effective cleaning of compromised skin. Although both baby wipes A and B were pH-buffered and contained an emollient, with demonstrated gentleness over wipes without these ingredients, results indicate that baby wipes A was gentler than all other baby wipes tested, suggesting that other components or characteristics of these baby wipes may have contributed to their superior gentleness.
Baby wipes C did not contain an emollient to reduce friction of the wipe on skin and was not pH buffered to support skin’s acid mantle. Baby wipes C performed similarly to cloth and water in this study, leading to increased TEWL and redness over the course of the study, compared to the unwiped control site. These data suggest that baby wipes without emollients and pH-buffering were harsher on skin and slowed down barrier recovery as compared to wipes that contained emollients and were pH-buffered.
When choosing baby wipes to clean and care for the delicate skin of infants, especially for preterm newborns, it is important to select products designed with skin health in mind and considering the unique needs of infant skin. Premature infant skin would benefit from baby wipes formulated to have a pH that complements the slightly acidic pH of healthy skin and are rich in emollients to help minimize friction against the skin. Care must be taken when selecting water-like baby wipes that are not pH buffered or when cleansing with washcloths wetted with water as, in this work, both were shown to delay barrier recovery of compromised skin.
Conclusions
This study demonstrated that that the composition of baby wipes can impact the recovery of compromised skin barrier after repeated wiping. Baby wipes containing emollients and pH buffer were more effective in supporting the health of compromised skin. Additionally, baby wipes A were gentler on compromised skin compared to the other baby wipes tested and to cloth and water.
Footnotes
Ethical Considerations
The methodology used in this work received ethical approval through an expedited review process by Sterling Institutional Review Board (ID#5995-001). All subjects participating in this work provided voluntary consent utilizing an in-person Informed Consent process.
Consent to Participate
Each subject provided written consent. Participation in the study was voluntary and participants were compensated for their time and travel costs to/from the clinical site.
Consent for Publication
No pictures or photographs of people are in the manuscript.
Author Contributions
RV contributed to study design, data analysis and interpretation, and led drafting of the article. AB contributed to study design and led data analysis. DN contributed to study design and data analysis and interpretation. LS contributed to study design and collected data. SM, KR, CK contributed to study design and data interpretation. All authors contributed to either drafting and/or critically reviewing the article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from Kimberly-Clark Corporation.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are scientists employed at Kimberly-Clark Corporation. The study was funded by Kimberly Clark Corporation.
