Abstract
Background/Objective:
Pediatric sepsis remains a leading cause of child mortality worldwide. Early risk stratification using organ dysfunction scores may improve management. Bangladesh bears a high burden of pediatric infectious disease and sepsis, particularly in children under 5. This study compared the predictive performance of admission PELOD-2, pSOFA, and Phoenix Sepsis Scores for in-hospital mortality among pediatric sepsis patients.
Methods:
We conducted a secondary analysis of an observational study at a tertiary hospital in Dhaka, Bangladesh (February-December 2022). Of 100 enrolled children aged 2 months to 18 years with sepsis, 96 had sufficient data for score calculation.
Results:
Median age was 8 months (IQR 5-18), and in-hospital mortality was 23%. Admission PELOD-2 >3 and pSOFA >6 were associated with increased mortality. AUCs for mortality were 0.83 (pSOFA), 0.82 (PELOD-2), and 0.79 (Phoenix).
Conclusions:
All 3 scores showed good mortality discrimination. pSOFA and Phoenix may be more feasible in low-resource settings.
Introduction
Pediatric sepsis is the leading cause of child mortality worldwide.1,2 Low- and middle-income countries (LMICs), particularly in South Asia and Africa have the highest burdens of pediatric sepsis incidence and mortality. 3 There have been calls globally to develop tools to more quickly and accurately identify and risk-stratify children with sepsis. 4
Sepsis risk stratification tools, such as clinical prognostication models, should ideally be easy to use, low-cost, minimally intrusive, and applicable in diverse settings. 5 Several pediatric organ dysfunction scoring systems have been proposed for aiding in clinical prognostication in pediatric sepsis, such as the Pediatric Logistic Organ Dysfunction (PELOD-2) score and the Pediatric Sequential Organ Failure Assessment (pSOFA) scores. The PELOD-2 was developed in 2013, as an update of the original PEdiatric Logistic Organ Dysfunction score. Later, in 2017, the pSOFA score was developed as an adaptation of the SOFA score recommended in Sepsis-3 which focused only on adult populations. 6 Both scoring assessments were developed and initially validated in high-income countries, while to date, few external validations among LMIC populations have been performed.7 -9 In addition, the primary datasets were not representative of the global pediatric population of sepsis cases, which has a higher proportion of patients <5 years of age.3,6,10
In January 2024, The Society of Critical Care Medicine (SCCM) published the Phoenix Sepsis Score, a new organ dysfunction score to specifically identify children with sepsis and septic shock that was both derived and validated specifically for sepsis diagnosis in children. 11 The Phoenix consensus score consists of 4 organ system variables: respiratory, cardiovascular, coagulation, and neurological. Mortality was higher in children who had organ dysfunction in at least 1 of 4 of these organ systems that were not the primary site of infection. 11
Bangladesh, an LMIC in South Asia, carries a significant burden of communicable pediatric diseases and concomitant sepsis, especially in the pediatric population under 5 years of age. At the International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b), mortality from pediatric sepsis in high-risk children approached 60% in 2010. 12 These high rates are due to a multitude of reasons including late identification of sepsis severity.1,13 Early recognition of sepsis severity in these patients is crucial to allow for aggressive goal-directed treatment and management. 2 In addition, there is utility in comparing these scores to one another to understand both their validity and feasibility. 10
Objectives
The original purpose of this study was to externally validate the pSOFA and PELOD-2 scores against the outcome of in-hospital mortality for pediatric patients in Bangladesh with sepsis. Since the recent publication of the Phoenix Sepsis Score, a secondary objective of this study was to perform the first comparison in an LMIC of these scores with the new Phoenix Sepsis Score outside of the original publication. 9
Methods
Study Design
This study was a secondary analysis of the Remote Electronic Monitoring and Decision-support for Improved Sepsis care (REMEDIES, Dhaka, Bangladesh, 2022) study, a prospective observational study of pediatric ICU patients admitted with sepsis to the ICU of Dhaka Hospital of icddr,b.14,15 Briefly, the aim of the REMEDIES study was to develop a machine learning-based mobile health tool for sepsis risk prediction using continuous physiologic data (heart rate, respiratory rate, temperature, positioning) obtained remotely from a wireless, wearable device. All participants in this secondary analysis were enrolled by the primary REMEDIES study.
Population and Setting
The REMEDIES study took place at Dhaka Hospital, a specialized care hospital operated by a non-profit organization (icddr,b). The hospital treats over 200 000 patients each year and serves a catchment population of 17 million people in Dhaka, Bangladesh, and surrounding areas. Patients between 2 months (prematurity corrected) and 18 years of age admitted to the ICU with signs of sepsis were eligible for enrollment. Sepsis was defined as meeting at least 2 systemic inflammatory response syndrome (SIRS) criteria with a suspected source of infection, according to the standard pediatric sepsis definition at the time of REMEDIES study enrollment in 2022. These 4 SIRS criteria include at least one of either temperature >38.5 °C or <36 °C or abnormal leukocyte count, age-dependent tachycardia or bradycardia (if <1 year), and tachypnea or mechanical ventilation. 16 In addition, prior to enrollment, all patients required a preliminarily diagnosis of sepsis with an infectious source by the clinical team physician. Exclusion criteria included prior enrollment in the study, known history of skin sensitivity to adhesives, prematurity-corrected age of <2 months, congenital cyanotic heart disease, and recent chest wall trauma or open wound. All participant recruitment activities took place in the intensive care unit (ICU) of Dhaka Hospital. While icddr,b was one of the sties for the original Phoenix Score study, there was no overlap of study patients used in the development of the Phoenix Sepsis Score and the REMEDIES study as icddr,b was one of the sites for the original Phoenix Score study.
Patient Enrollment, Consent, and Involvement
Dedicated research staff were hired and trained specifically to screen and enroll participants in the REMEDIES study, obtain informed consent, and collect and record data 24 hours/day in person. REMEDIES recruitment was continuous during the data collection phase from February 2022 to December 2022 with a maximum of 3 concurrent participants enrolled at a single time (due to the number of wearable devices available). Research nurses initially screened all potential patients for inclusion and exclusion criteria on admission to the Dhaka Hospital ICU and confirmed eligibility with the on-duty research medical officer. 15 There was no patient or public involvement in this secondary data analysis.
Funding Statement
This secondary research analysis received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. The parent REMEDIES study was funded by the National Institutes of Health Fogarty International Center (R33TW012211).
Data Acquisition
Data was collected by staff for each participant such as demographics, immunization history, manually collected vital signs, therapeutic interventions utilized during patient course and patient outcomes, laboratory tests including blood gases, and duration of hospitalization, and ICU and in-hospital mortality rate. Data was collected on paper case report forms and entered into a secure REDCap electronic database daily by study staff. 17 We chose to utilize “Next Observation Carried Backwards” for any missing data necessary to compute the participants’ PELOD-2, pSOFA, and Phoenix Sepsis Scores at admission when possible. 18
Data Analysis
The pSOFA score is an assessment of the severity of pulmonary, hepatic, cardiovascular, neurologic, and renal system dysfunction, and failure. 6 Each organ system is allocated a score of 0 to 4, with a maximum possible score of 24. 6 The pSOFA score includes age-related cardiovascular and renal sub-scores, as well as a respiratory score that used SpO2:FiO2 ratios rather than the PaO2:FiO2 originally used in the adult SOFA score. 4 This alternative ratio has been validated in children and allows for the use of the scoring system in settings and populations where arterial blood gas measurements cannot be easily obtained. 6 Cutoffs of 8 or 10 have been suggested to discriminate the risk of mortality in the pSOFA score.6,19
The PELOD-2 score is an assessment of the severity of Neurologic, Cardiovascular, Renal, Respiratory, and Hematologic system dysfunction and failure. 10 The PELOD-2 score includes 10 variables involving the above 5 organ dysfunctions. 10 Each of the above organ systems was allocated a maximum score between 2 and 10, for a maximum PELOD-2 score of 33.10,20 Cutoffs of 8, 9, 10, and 13 have been suggested as optimal to discriminate mortality in the PELOD-2.20 -22
The participant’s Phoenix Sepsis Score was calculated based on recently published data. 23 The Phoenix Sepsis Score is an assessment of the severity of respiratory, cardiovascular, hematologic, and neurological system dysfunction and failure. 23 Each of the above organ systems was allocated a maximum score of 0 to 6 (respiratory—3, cardiovascular—6, hematologic—2, neurologic—2), with the maximum possible Phoenix Sepsis Score of 13. 23 Patients with suspected infection and Phoenix Sepsis Score ≥2 are defined to have sepsis and patients with sepsis and a cardiovascular score ≥1 are defined to have septic shock. The Phoenix Sepsis Score was not intended to have a discriminatory score cutoff for mortality. More importantly, with this score, children with sepsis who had elevated scores in remote organ systems outside of their primary infectious source were found to have higher mortality. 23 Though all patients included in our analysis were missing d-dimer and fibrinogen levels, as this testing was not available at the study site, they could still achieve the maximum score the hematologic category (2) with the available components, platelets and INR. 23 Importantly, the authors who developed the Phoenix Sepsis Score stated that in its conception and validation, the Phoenix Sepsis Score was designed to accurately identify children with sepsis, even in settings where not all variables are available. In fact, “the score functions when not all variables are available because of its redundancy.” 23
Regression Analysis
Area under the receiver operating curve (AUC) was performed to assess the scores’ ability to discriminate the primary outcome of mortality, defined as an in-hospital death of a study participant or if the patient was a non-survivor at the time of study completion. Additionally, analyses controlled for potential confounders including sex, past medical history, age, severe acute malnutrition (SAM) status, and maternal education level as these factors have been previously found to have associations with increased mortality in similar populations, although they are not directly involved in the calculation of these scores.24 -29 Family income level was not included as a potential confounder as sensitivity analyses revealed that all study participants met criteria below the international extreme poverty line of USD 2.15/day. 30
Sex was defined as binary if the patient was the biological sex male or female. Age was divided into 3 categories—<24, ≥24 and <60, and ≥60 months. Maternal education has an established association with increased child mortality.27 -29 Severe malnourishment status was defined as a mid-upper arm circumference (MUAC) <11.5 cm or edema (suggestive of kwashiorkor) for children <60 months, or a BMI less than the age-adjusted −3 SD for those ≥60 months. 31
Model discrimination was assessed using the AUC analysis with 95% confidence intervals (CIs) to compare the performance of the various pediatric sepsis scores. 32 A sensitivity analysis calculating the AUC with 95% CIs including only participants without missing data (n = 91) was conducted to ensure the appropriateness of the “Next Observation Carried Backwards” method. Odds analysis was conducted to compare the sensitivity and specificity of various score cutoffs for mortality discrimination of both PELOD-2 and pSOFA for this sample population. Cut-offs were chosen, using Youden’s index, to maximize both the sensitivity and specificity of odds of mortality, thus maximizing the identification of true-positive cases, while also minimizing the identification of false-positive patients who are not at a higher risk of inpatient mortality.
All analyses were conducted in STATA (version number 17; StataCorp, College Station, TX, USA).
Ethical Approval and Informed Consent
The REMEDIES study was conducted in accordance with the Declaration of Helsinki and received approval from the Institutional Review Boards of icddr,b (PR-21062, approval date: June 10, 2021), and Rhode Island Hospital (CMTT/PROJ: 008121, approval date: June 9, 2021). Parents or legal guardians of eligible patients under the age of 18 years provided written consent in Bangla before REMEDIES study enrollment. For patients aged 8 to 17 years, written assent was also obtained from the child. Unaccompanied minors without a parent or guardian in attendance were not eligible to be enrolled in the study.
Results
A total of 463 patients were screened for enrollment from February to December 2022 for the REMEDIES study. Of the 141 patients who were eligible for enrollment, a total of 100 patients consented and were enrolled. Of those who did not enroll, 12 declined participation, 2 patients died before enrollment, and 27 patients were not enrolled as the maximum concurrent participant capacity (3) had already been reached (Figure 1).
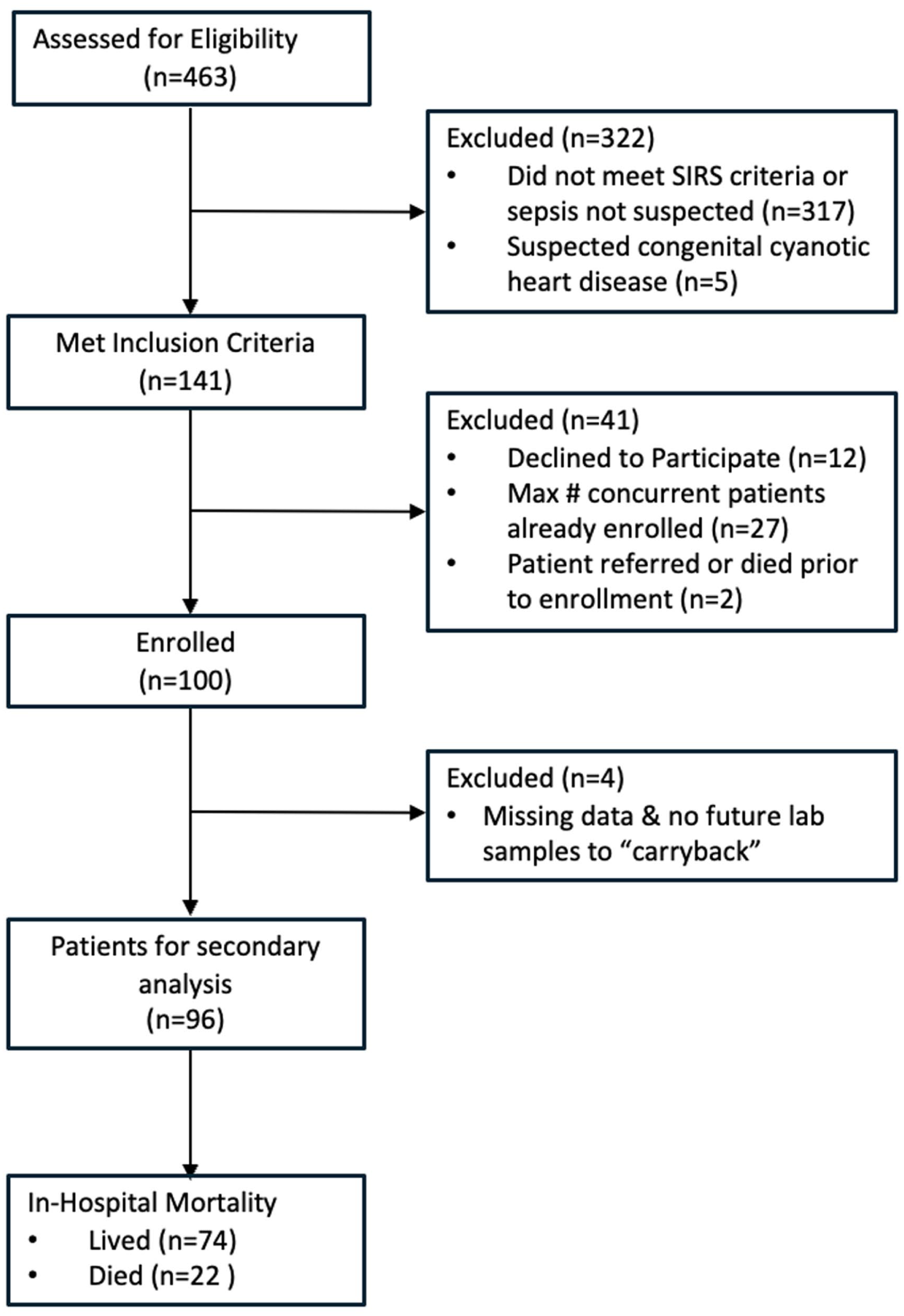
Study flow chart.
Of the 100 children enrolled in the primary study, 9 were missing laboratory data (bilirubin and/or platelets), due to hemolysis or inability to acquire adequate blood samples, and 4 were removed from further analysis as they did not have any further data to carry backward, leaving 96 for this secondary analysis. Of the included 96 children, 39 (41%) were female; the median age was 8 months (IQR 5-18 m), and 46 (48%) were defined to have SAM/underweight status. Total in-hospital mortality was 23%. Descriptive statistics of the cohort are shown in Table 1.
Description of Pediatric Sepsis Patients by In-Hospital Mortality, REMEDIES, Dhaka, Bangladesh, 2022 (N = 96).

Calculated using Wilcoxon rank sum test.
Participants were documented to have chronic illnesses as follows: epilepsy (1%), heart disease (2%), prematurity at birth (2%), and cerebral palsy (5%). There were no documented cases of asthma, diabetes, cancer, or chronic lung disease. Most children in this study had multiple sources of infection documented by the study clinical physicians. Of the final 96 patients analyzed, 94 (98%) of participants had a diagnosis of gastroenteritis, acute watery diarrhea, invasive diarrhea, or persistent diarrhea, 59 (61%) had a diagnosis of pneumonia, 4 (4%) had a diagnosis meningitis, and 2 (2%) had a diagnosis of urinary tract infection.
The median admission scores for PELOD-2 was 3 (IQR 2-5), for pSOFA was 5 (IQR 3-7), and for Phoenix Sepsis Scores was 2 (IQR 2-3). Based on criteria defined by the Phoenix Sepsis Score, 74 (77%) study participants had sepsis, and 23 (24%) had septic shock. Of these 23 who met criteria for septic shock by the Phoenix Sepsis Score at admission, mortality was 43% (10).
The 3 scores were found to have good discriminatory power for predicting mortality in this population by assessing AUC (Figure 2), with the AUC for PELOD-2 was 0.8163 (95% CI 0.69-0.94), for pSOFA was 0.8335 (95% CI 0.72-0.94), and for Phoenix was 0.7908 (95% CI 0.67-0.91). Sensitivity analysis revealed similar findings (Supplement eFigure 1).

Assessing mortality risk prediction: ROC curve analysis for PELOD-2, pSOFA, and Phoenix Sepsis Scores, REMEDIES.
Using Youden’s Index, a cutoff of 6 (rounded down from 6.5) maximized sensitivity and specificity for mortality discrimination in the pSOFA score in this sample population (sensitivity of 72.73% (95% CI 64%-82%) and specificity of 78.38% (95% CI 70%-87%; Supplement eTable 1). Similarly, for the PELOD-2 score, a cutoff of 3 (rounded down from 3.5) maximized sensitivity and specificity for mortality discrimination (sensitivity of 77.27% (95% CI 69%-86%) and a specificity of 75.68% (95% CI 67%-84%; Supplement eTable 2).
Among pediatric patients admitted for sepsis in the ICU at Dhaka Hospital included in the REMEDIES study, those with a PELOD-2 Score >3 (35 patients) had 12.47 times the adjusted odds (95% CI 3.65-42.46) of mortality (17 patients, 48.6%) compared to participants with PELOD-2 score ≤3 (5 patients, 8.2%), after controlling for sex, age, severe malnourishment, and maternal education (Table 2). Furthermore, those with a pSOFA score >6 (32 patients) had 10.51 times the adjusted odds (95% CI 3.15-35.08) of mortality (16 patients, 50.0%) compared to participants with a pSOFA score ≤6 (6 patients, 9.4%), after controlling for sex, age, severe malnourishment, and maternal education (Table 3). Lastly, those meeting the criteria for septic shock as per the new Phoenix criteria (23 patients) had 3.91 times the odds (95% CI 1.39-10.96) of mortality (10 patients, 43.5%) compared to participants without the Phoenix scoring criteria for septic shock (12 patients, 16.4%; Table 4).
Odds Ratios of Mortality Among Pediatric Patients Admitted for Sepsis When Assessing PELOD-2, REMEDIES.

Adjusted for categories including admission PELOD-2 score, sex, age, past medical history, malnourishment status, maternal education.
Odds Ratios of Mortality Among Pediatric Patients Admitted for Sepsis When Assessing pSOFA, REMEDIES.
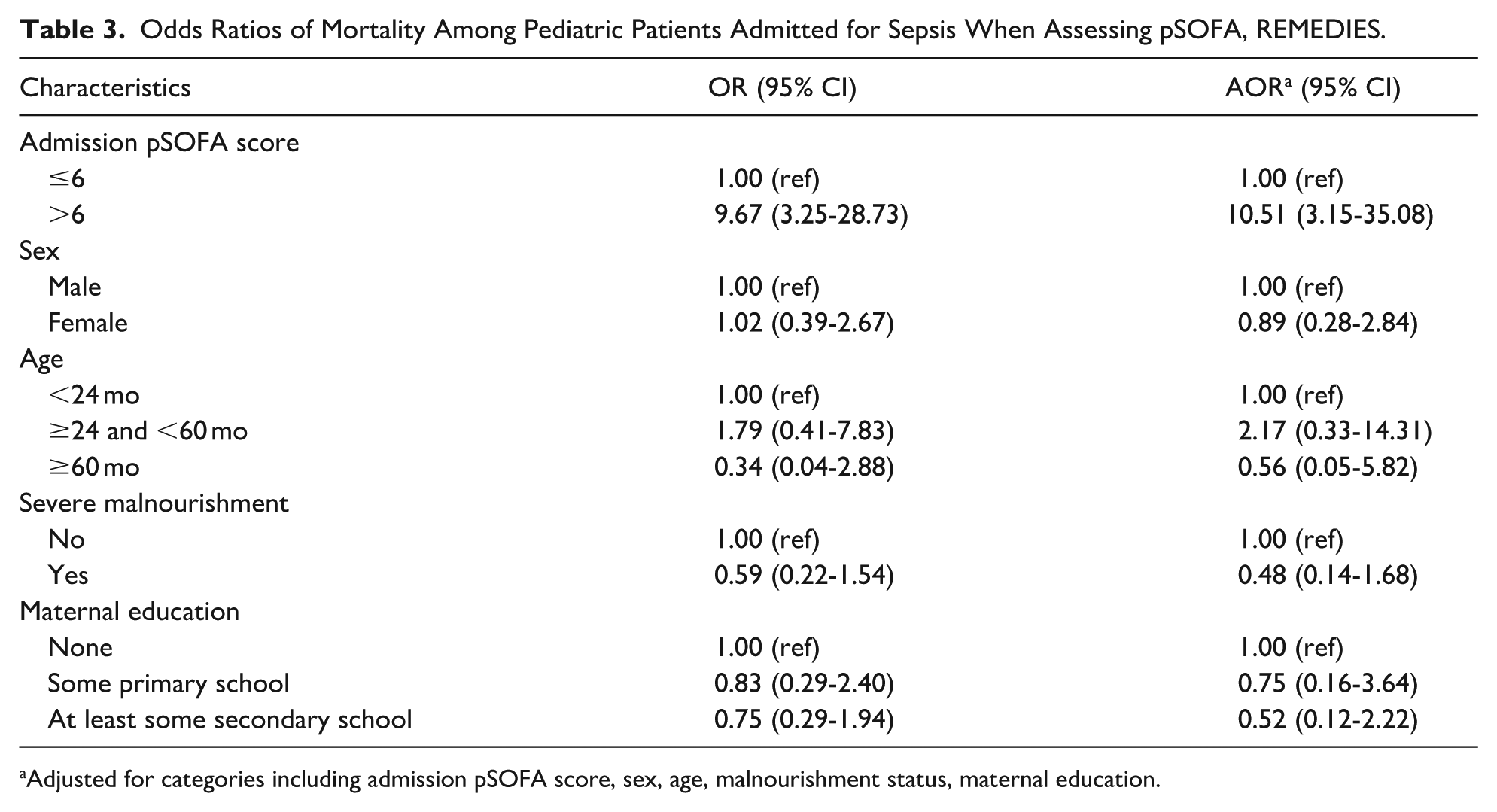
Adjusted for categories including admission pSOFA score, sex, age, malnourishment status, maternal education.
Odds Ratios of Mortality Among Pediatric Patients Admitted for Sepsis, REMEDIES.

Discussion
This study found that PELOD-2, pSOFA, and Phoenix Sepsis Scores at admission all had strong performance and could all similarly discriminate mortality among this cohort of septic children admitted to an ICU in an LMIC, Dhaka, Bangladesh. Given the lack of feasibility of routine use of arterial blood gas measurements (required in the PELOD-2) and the Society of Critical Care Medicine’s recent publication and recommendation for use of the Phoenix Sepsis Score to identify pediatric sepsis, the Phoenix Sepsis Score is perhaps the more practical, recognized, and preferred pediatric sepsis stratification tool compared to PELOD-2 and pSOFA score globally.
Recently published studies have validated the use of Phoenix Sepsis Score in high income countries, such as Bulgaria, Australia, and New Zealand.33,34 However, to the authors’ knowledge, this remains the first study to assess the new Phoenix criteria in an LMIC setting (outside of the original Phoenix research study), with comparison to other widely used pediatric organ dysfunction scores. Though our dataset lacked the availability of Phoenix Sepsis Score variables of fibrinogen and D-dimer 23 which is a common limitation in many LMIC clinical settings, the AUC in this study compared closely to that of both internal and external validation sets in the development and validation study of the Phoenix Sepsis Score (0.82) even without these datapoints. 23 This suggests that the Phoenix Sepsis Score appears valid for use even in LMICs where not all components of the score may be available, although further validation particularly in larger and more diverse cohorts is greatly needed.
Since their development, both the PELOD-2 and the pSOFA score have been validated in LMICs such as India, Egypt, and Pakistan.5,35,36 These studies found the score to be useful for prognostication of patients with sepsis. They also noted the importance of externally validating these scoring systems in populations outside of their origin. 23 It was noted that pSOFA was easier to implement in resource-denied settings as it utilized oxygen saturation rather than the partial pressure of oxygen, the latter of which would require arterial blood gas. The discriminatory thresholds of both scoring systems varied, suggesting that we should not establish a universal cutoff.5,15,27,28
Additionally, it was found that PELOD-2 scores >3 and pSOFA scores >6 at admission were associated with significantly increased odds of mortality compared to PELOD-2 ≤3 and pSOFA ≤6 at admission in this cohort. A specific cut-off point was selected to optimize both sensitivity and specificity, using Youden’s index, also ensuring it also aligned with clinical relevance to comprehensively capture sufficient relevant cases. These discriminatory thresholds exhibit significant variance from those delineated in previous research, indicating that establishing universal cutoffs is not advisable.6,19 -22 The sensitivities, specificities, positive predictive values, and negative predictive values for differing PELOD-2 and pSOFA score cutoffs (Supplement eTables 1 and 2) described in this study, can help providers decide how to risk stratify patients early in their hospital course and better direct scarce hospital resources and clinician attention while also prioritize preventing pediatric patient death within LMIC settings. For this reason, the cut-offs in this study, should be used as guidance rather than a universal discriminatory threshold. Similar to findings with the Phoenix Sepsis Score, it may also be beneficial to interpret these scores in conjunction with remote organ failure.
Limitations
The study results were limited by a small sample size, resulting in wide confidence intervals. Further studies with a larger sample size may expand this preliminary data with increased power to identify which of the 3 scoring systems demonstrate the best predictive value and allow for stronger applicability to other global contexts. In addition, the study site, icddr,b primarily treats patients with sepsis of gastrointestinal origin, though there were patients with pneumonia and other sources of infection. This may affect the generalizability of these results among the general pediatric sepsis population. 37 Another limitation is that all patients in this analysis were missing d-dimer and fibrinogen values which are used to calculate the Phoenix Sepsis Score. The original Phoenix study has been critiqued for high rates of missingness of these labs at some study sites. 38 Nevertheless, our study remains impactful as these labs are not routinely available in many LMICs, demonstrating that the Phoenix Sepsis score may still be considered in these contexts. Lastly, while there were 4 patients missing initial laboratory data needed for the calculation of PELOD-2 and pSOFA, sensitivity analysis suggests that the potential impact of the missing data is negligible, so, the missing data could have been ignored in the analysis rather than imputing 39 (Supplement eFigure 1).
Future Directions
Research with aims to understand the discriminative power of PELOD-2, pSOFA, and Phoenix Sepsis Scores at admission to predict the need for in-hospital vasopressor medications and in-hospital ventilatory support will add to the understanding of these scores. These scores could be valuable to assist decision-making regarding referrals from smaller hospitals to tertiary care hospitals with an ICU such as Dhaka Hospital upon early identification of elevated scores indicating organ dysfunction and greater risk of mortality. In addition, future studies could incorporate these scores into clinical decision support tools such as on mobile devices to increase the ease and accessibility of use in busy clinical environments, particularly in LMICs.
Conclusion
This study compared the use of the PELOD-2, pSOFA, and Phoenix Sepsis Scores to discriminate mortality in an LMIC setting, with the scores having similar discrimination. Additionally, this is the first study to compare the new Phoenix criteria outside of the original research study in an LMIC setting where several coagulation score components were not available. The Phoenix Sepsis Score and pSOFA may be more practical compared to PELOD-2 considering the latter requires arterial blood gas measurements which are often not feasible to obtain in pediatric settings globally.
Supplemental Material
sj-docx-1-gph-10.1177_30502225261424157 – Supplemental material for Predicting Pediatric Sepsis Outcomes: A Comparison of 3 Sepsis Risk Scores in Bangladesh
Supplemental material, sj-docx-1-gph-10.1177_30502225261424157 for Predicting Pediatric Sepsis Outcomes: A Comparison of 3 Sepsis Risk Scores in Bangladesh by Nidhi Kadakia, Siraj Amanullah, Annie Gjelsvik, Gazi Md. Salahuddin Mamun, Shamsun Nahar Shaima, Monique Gainey, Abu Sayem Mirza Md. Hasibur Rahman, Alicia Genisca, Atin Jindal, Monira Sarmin, Farzana Afroze, Jaejoon Shin, Md. Tanveer Faruk, Abu Sadat Mohammad Sayeem Bin Shahid, Tahmeed Ahmed, Mohammod Jobayer Chisti, Adam C. Levine and Stephanie Chow Garbern in Sage Open Pediatrics
Footnotes
Acknowledgements
We extend our deepest gratitude to the dedicated clinical and research staff at icddr,b in Dhaka, Bangladesh whose commitment and efforts were instrumental in the success of the REMEDIES study. Your professionalism, expertise, and passion have contributed immensely to advancing our understanding in this field. We would also like to express our heartfelt appreciation to all the patients and their families whose participation and cooperation made the REMEDIES dataset possible. Your willingness to be involved and your invaluable insights have enriched our work and will undoubtedly impact future research and patient care.
Author Contributions
All authors made significant contributions to data acquisition, study design, data analysis, interpretation, and/or manuscript revision; they stand by the integrity and accuracy of the work and approved the final version for publication.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This secondary research analysis received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. The parent REMEDIES study was funded by the National Institutes of Health Fogarty International Center (R33TW012211) which helped to support the publication fees of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
