Abstract
This study evaluated the impact of implementing a standardized institutional pathway on the evaluation and management of hypothermic infants, focusing on serious bacterial infection (SBI) workups, hospital admissions, antibiotic use, and re-admissions. We performed a single-institution subset study involving infants ≤60 days of age who presented to the emergency department or were hospitalized for hypothermia over a 5-year period before and after pathway implementation. Among 231 patients (157 pre- and 74 post-pathway), full SBI evaluations decreased from 61.1% to 48.5%, partial evaluations from 35.0% to 29.7%, and no evaluations increased from 3.8% to 21.6% (P < .001). Hospital admission rates dropped from 92.9% to 70.8% (P < .007), and antibiotic administration declined from 68.1% to 51.3% (P = .02). Re-admission rates were unchanged, and no SBI or IBI cases were missed. These findings suggest that a clinical pathway can safely reduce invasive evaluations, hospitalizations, and antibiotic use in this population.
Keywords
Background
Fever and/or hypothermia in young infants can be a sign of a serious infection.1 -9 Despite similar rates of invasive bacterial infections (IBI; bacteremia, and/or bacterial meningitis) in febrile and hypothermic infants, the majority of research has focused on febrile infants.2,10 -13 As a result, clinical practice guidelines and national efforts have been developed to standardize and improve the care of febrile infants, with no current work exclusive to the evaluation and management of hypothermic infants.14,15 Often hypothermic infants are included in febrile infant pathways, despite important physiologic and clinical differences.16,17 Physiologically, newborns are at increased risk of developing hypothermia due to decreased fat stores, thin skin, and larger body surface areas.18,19 Clinically when compared to febrile infants, hypothermic infants are more likely to be premature and to present for evaluation within the first week of life.5,20 Recently, repeated temperature instability, white blood cell count abnormalities, and thrombocytopenia were found to be associated with IBI in hypothermic infants. 2 Currently, no clinical practice guidelines exist to guide evaluation and management of the hypothermic infant population. As a result, variation has been demonstrated in serious bacterial infection (SBI; urinary tract infection [UTI], bacteremia, bacterial meningitis) evaluation, hospital disposition, and antimicrobial administration for well-appearing hypothermic infants. 21
While febrile infant decision tools can be applied to well-appearing hypothermic infants, these tools were not derived in this population. Application of febrile infant decision tools to hypothermic infants misclassifies many hypothermic young infants as higher risk for bacterial infection due to their age at presentation or history of prematurity resulting in unnecessary hospitalizations and invasive testing. 22 Specific pathways for hypothermic infants may improve care in this population.
Our institution developed a hypothermic young infant pathway to standardize evaluation and management of this population within our hospital. Our primary objective was to compare SBI evaluation of infants with hypothermia before and after the implementation of the pathway. Our secondary objectives were to compare the rates of admission, antimicrobial administration, and hospital re-admission rates before and after pathway implementation. We hypothesized the introduction of a hypothermic young infant pathway would reduce SBI evaluations, hospital admissions, and antimicrobial exposure, without leading to an increase in re-admission rates or missed cases of SBI after implementation of the pathway.
Methods
Study Design and Population
As part of a larger retrospective, cohort study conducted with the Hypothermic Young Infant Research Collaborative,2,21 -24 we performed a subset study at our institution consisting of young infants ≤ 60 days of age evaluated in the emergency department (ED) or hospital setting for hypothermia (≤36.0°C) over a 5-year time-period from September 1, 2016, to May 6, 2021 to assess the implementation of a novel clinical pathway. Our institution is a tertiary care, academic center with 27 400 annual visits to the pediatric ED. The emergency department (ED) primarily consists of pediatric fellowship trained Emergency Medicine physicians. There are 59 pediatric hospital beds and an 18-bed pediatric intensive care unit. The hypothermic infant pathway was created by a multidisciplinary team consisting of pediatric hospital medicine and emergency medicine physicians and implemented in our Children’s hospital as well affiliated outpatient clinics. The pathway was derived by modifying our institution’s febrile infant pathway and incorporating the best available evidence on hypothermic infants available at the time (Figure 1).3 -5,19 This pathway includes infants ≤ 7 days old and incorporates an infant’s response to rewarming. The pathway was introduced on May 1, 2020. The study protocol was approved by our institution’s institutional review board.

Hypothermic infant algorithm.
The optimal temperature to define hypothermia remains controversial, with no clear threshold identified for increased risk of a bacterial infection. 25 Most ED providers define hypothermia as ≤36.0°C. 16 As a result, we used the temperature (rectal, axillary, or other) of ≤36.0° C to define hypothermia consistent with definition of systemic inflammatory response syndrome 26 and consistent with previous hypothermic infant studies.2,4,5,21,22 With the different definitions of hypothermia and limited studies available at the time of the pathway introduction, a temperature of ≤36.1°C was used in the pathway.
Eligible patients were identified by 2 strategies as previously described.2,21 -23 First the International Classification of Diseases (ICD) versions 9 and 10 diagnosis codes were used to identify patients. These ICD 9 and 10 diagnosis codes corresponding to hypothermia and temperature disturbances have been used previously.2,3,21,25,27,28 However, due to the inaccuracy of relying on ICD diagnosis codes alone,2,21,25 a second search strategy to identify eligible patients pulled all young infants ≤ 90 days old seen in the ED or admitted to the hospital with documented hypothermia (≤36.0°C) within 24 hours of the initial patient encounter. Patients identified by an ICD diagnosis code were not included in this second search. Patients identified by ICD code or hypothermia within the first 24 hours had their charts manually reviewed for inclusion. Infants with documented hypothermia on initial vital sign measurement after manual chart review were eligible for inclusion.
Young infants ≤ 60 days of age with either historical or documented hypothermia were included. Historical hypothermia was defined as a temperature of ≤36.0°C reported by a caregiver or outside heath care facility. Documented hypothermia was a temperature of ≤36.0°C measured at a health care facility.
We excluded infants > 60 days of age, infants with no documented or reported hypothermia, infants with fever (≥38.0°C) within 48 hours of their encounter, patients with central lines, patient transfers from a referring hospital if unable to directly visualize culture results from SBI evaluation, and infants with known trauma (non-accidental trauma, fall, or motor vehicle collision) or cardiac arrest (Figure 2). Infants evaluated for hypothermia in the newborn nursery or neonatal intensive care unit during their birth hospitalization were also excluded as these infants represent a different patient population of study.29,30 Infants born in the NICU or nursery are often transitioning from the in utero environment to the outside world. Exclusion criteria applied is consistent with previous studies.2,3,5,16,17,20 -22 Patient charts were manually reviewed to ensure inclusion criteria were met.

Study patients.
Pathway Development
Our hospital participated in the Reducing Variability in the Infant Sepsis Evaluation (REVISE) through the AAP VIP network in 2017-18. 15 Prior to REVISE, our local practice had been to perform a full SBI evaluation of nearly every infant ≤60 days of life with fever or hypothermia, administer empiric antibiotics, and admit the infant while monitoring cultures results. Our participation in REVISE, resulted in decreased hospital admissions and antibiotic exposure for young infants with fever without missing any infants with SBI.
While participating in REVISE, we observed increasing variability of SBI evaluations in hypothermic infants. Given our success with improving outcomes in young infants with fever, we sought to do the same for hypothermic infants. At the time of pathway development, we found a paucity of studies on this population with most limited by small patient numbers and focusing on characteristics of hypothermic infants with SBI.3 -5,19 Despite limited evidence, our team believed a standardized approach could safely improve the quality of care to this population.
With no clear laboratory values to identify hypothermic infants with increased risk of bacterial infections, we relied on the laboratory values incorporated in the REVISE febrile infant algorithms. The incorporation of infants 0 to 7 days and the cut-off of >7 days to pursue a full SBI evaluation was based off of physiologic factors such as decreased fat storage contributing to environmental susceptibility to hypothermia18,19 and studies showing older infant age associated with SBI.5,19 Based off of our institution’s experiences, a rewarming procedure was incorporated into the pathway as a single episode of hypothermia compared to recurrent episodes of hypothermia seemed less likely due to an underlying pathologic condition. Re-warming consists of skin-to-skin, bundling (infant fully clothed wrapped in room temperature blanket), warmed blankets, heat lamp, neonatal isolette, and radiant warmer. Rewarming is addressed in a stepwise fashion if an infant is well-appearing. Infants requiring sustained rewarming by these measures are admitted to the inpatient floor, with the exception of the radiant warmer which requires pediatric intensive care unit admission for monitoring.
Data Collection and Definitions
De-identified data was extracted from the electronic health record (EHR) into a Research Electronic Data Capture (REDCap) at our institution and included patient demographics, clinical characteristics, laboratory evaluation, and treatment provided. Demographic data included age, gender, gestational age, and presence or absence of identified ICD code. Clinical characteristics included maternal history of Herpes simplex virus or Group B Streptococcus infection documented, hypothermia, lowest temperature recorded or documented, medical complexity, and ill or toxic appearance on presentation.
Ill or toxic appearance was defined by the following terms documented on physical examination: ill appearance, toxic, limp, unresponsive, gray, cyanotic, apnea, weak cry, poorly perfused, grunting, listless, lethargic, or irritable.2,21,31,32 Feudtner’s complex chronic conditions was used as the measure for medical complexity and defined as a severe medical condition expected to last ≥12 months and requiring subspecialty care or involving multiple organ systems. 33
Laboratory evaluation consisted of urine, blood, and cerebrospinal fluid cultures, complete blood cell count (CBC), C-reactive peptide (CRP), procalcitonin, HSV testing from skin, blood, or spinal fluid and a respiratory pathogen panel. Treatment consisted of administration of antibiotics or antiviral medications such as acyclovir. Partial SBI evaluation was defined as obtaining at least 1 culture from blood, urine, or CSF but <3 cultures. A complete SBI evaluation was defined as obtaining all 3 cultures from the urine, blood, and CSF. Infants with no SBI evaluation or partial SBI evaluation were considered negative for SBI or HSV if SBI was not subsequently diagnosed within 7 days on review of EHR. Temperature instability was defined as more than one occurrence of hypothermia in a 24-hour period of time. 2
SBI was defined as a diagnosis of urinary tract infection, bacteremia, and/or meningitis. UTI was defined as growth of a single bacterial organism from a catheterized sample treated as a pathogenic organism by the medical team with expected antibiotic course. Bacteremia was defined as growth of a single bacterial organism from a blood culture treated as a pathogenic organism by the medical team with expected antibiotic course. Bacterial meningitis was defined as growth of a single bacterial organism from a cerebral spinal fluid (CSF) culture or positive polymerase chain reaction (PCR) treated as a pathogenic organism by the medical team with expected antibiotic course. Neonatal herpes simplex virus (HSV) was defined as a positive PCR from a mucosal surface swab, blood, and/or CSF treated by the medical team with expected course of acyclovir.
Outcome Measures
The primary outcome was comparing SBI evaluations for hypothermic infants before and after implementation of the pathway. Secondary outcomes compared the rate of admission to the hospital, antimicrobial exposure, ED re-evaluation within 30 days from initial encounter, and hospital re-admission within 30 days from initial encounter before and after pathway implementation. Admissions included patients admitted from our ED and patients directly admitted to the hospital from an outpatient clinic or outside emergency department which we could visualize laboratory results.
Analysis
We used descriptive statistics to characterize the demographics, clinical characteristics, and outcomes of our cohort. Outcomes compared before and after pathway implementation included SBI evaluation, hospital admissions, HSV testing, empiric use of antibiotics, empiric use of acyclovir, ED re-evaluation within 30 days, and hospital re-admission within 30 days. Categorical variables were reported as frequencies (N) and percentages (%), and were analyzed using Chi-square or Fisher Test. The value of P less than .05 was considered statistically significant. All analyses were conducted using R statistical software (version 4.0.4).
We performed a post-hoc power calculation for the proportion of hypothermic infants with any SBI evaluation pre- versus post-pathway implementation. In order to detect a difference between the 96% of patients with any SBI evaluation pre-pathway and the 78% evaluated post-pathway with 80% power, we would require at least 53 infants in each group.
Ethical Approval and Informed Consent
This study did not require formal ethical approval from a review committee or IRB approval, as it involved the retrospective collection of de-identified medical data. All data were securely stored in a protected database to ensure confidentiality and privacy.
Results
Study Population
Of the total 1051 young infants identified, 231 met our inclusion criteria (Figure 2). The majority of infants were ≤21 days of age (87.9%), not medically complex (95.2%), and born at full term (52.4%). Of the infants born prematurely, no infants in the <34-week gestation were <30 weeks gestation. For clinical characteristics, the majority had documented hypothermia (92.2%), had repeated temperature instability (70.1%) and were well-appearing on presentation (81.0%). Only 6 infants did not require a rewarming procedure and 79 infants did not have a rewarming method documented. Young infants included in the pre-pathway group were more likely to be ≤21 days of age compared to the post-pathway group (93% vs 77%). Additional study demographics and characteristics can be found in Table 1. SBI, IBI, and HSV rates did not differ between pre- and post-pathway implementation. Of the infants with SBI, one infant was well-appearing and received a full SBI evaluation due to repeated temperature instability and was ultimately diagnosed with an Escherichia coli UTI.
Demographic and Clinical Characteristics of Cohort.
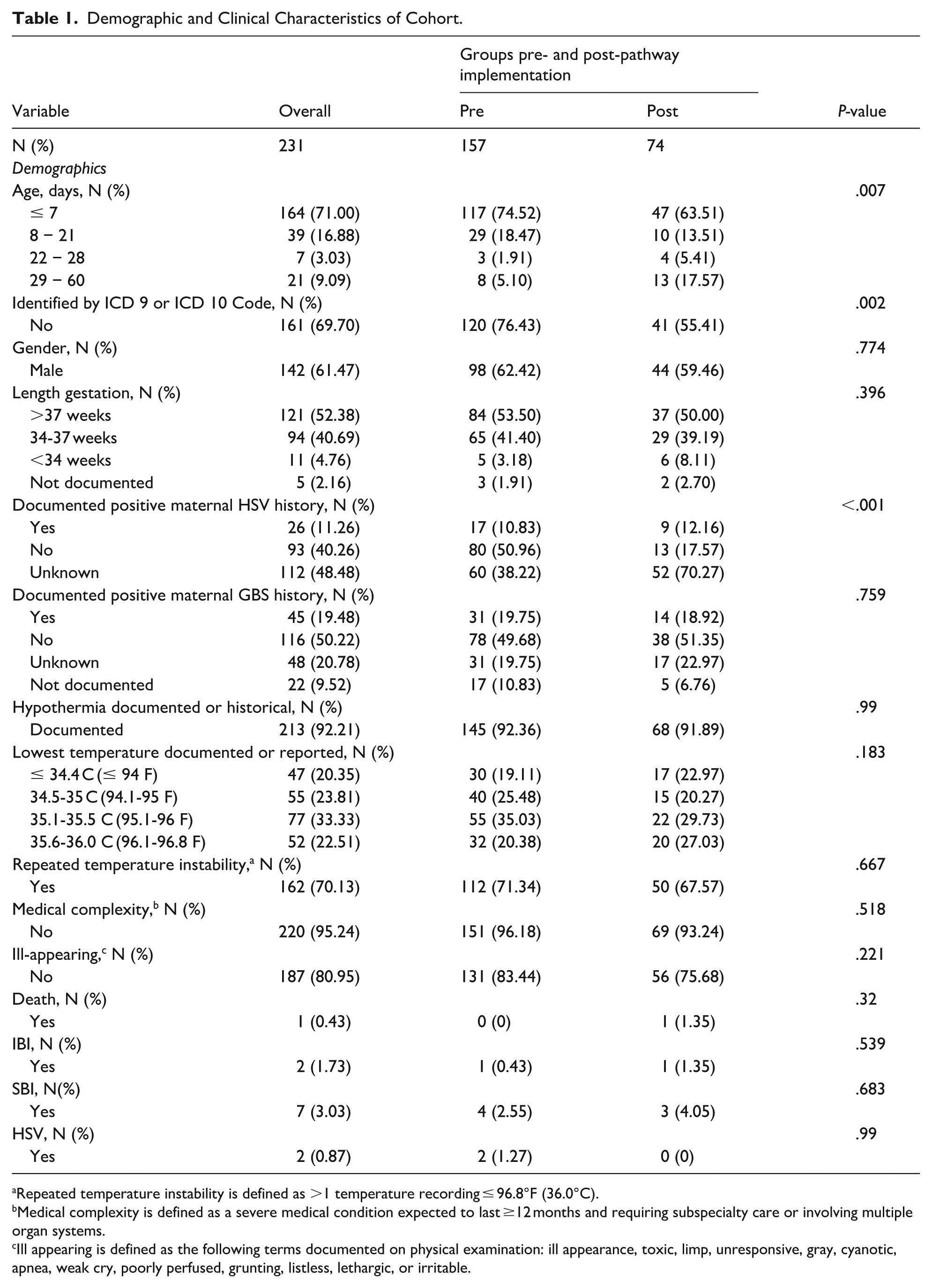
Repeated temperature instability is defined as >1 temperature recording ≤ 96.8°F (36.0°C).
Medical complexity is defined as a severe medical condition expected to last ≥12 months and requiring subspecialty care or involving multiple organ systems.
Ill appearing is defined as the following terms documented on physical examination: ill appearance, toxic, limp, unresponsive, gray, cyanotic, apnea, weak cry, poorly perfused, grunting, listless, lethargic, or irritable.
Outcomes
SBI Evaluation
Pre-pathway implementation, an SBI evaluation was obtained on 151 (96.2%) compared to 58/74 (78.4%) of the post-pathway group (P < .001). Of the SBI evaluations, a full SBI evaluation was obtained on 96/157 (61.1%) infants compared to 36/74 (48.6%) of the post-pathway group and a partial SBI evaluation was obtained on 55/157 (35.0%) infants compared to 22/74 (29.7%) infants’ post-pathway. No SBI evaluation was obtained on 6/157 (3.8%) infants in the pre-pathway group compared to 16/74 (21.6%) infants in the post-pathway group.
Hospital Admission and Management
Pre-pathway implementation, 52/56 (92.9%) of the hypothermic infants seen in the ED were admitted from the ED compared to 34/48 (70.8%) infants’ post-pathway (P < .007; Table 2). Of all infants included in the study, the percentage of direct admissions for hypothermic infants decreased from 101/157 (64.3%) to 26/74 (35.1%) during this same time frame.
Patient Evaluation, Disposition, and Management.

Full SBI workup defined as obtaining blood, urine, and CSF cultures.
Partial SBI workup defined as obtaining 1 or more, but less than 3 of blood, urine, and CSF cultures.
Regarding management, in the pre-pathway group 107/157 (68.1%) infants received empiric antibiotic therapy compared to 38/74 (51.3%) infants in the post-pathway group (P = .02). Empiric acyclovir was started in 28% of patients in the pre and post intervention group. A full list of patient disposition and laboratory work up obtained can be seen in Table 2.
Re-Evaluation and Re-admission
In the pre-pathway group 14/157 (8.9%) were re-evaluated in the ED compared to 6/74 (5.4%) in the post-pathway group. In the pre-pathway group 12/157 (7.6%) were re-admitted to the hospital compared to 4/74 (5.4%) in the post-pathway group. No cases of SBI or IBI were missed throughout the entire study period. One infant was diagnosed with mucosal HSV after initial hospitalization as the surface culture became positive 5 days after discharge. One infant death in the post-pathway group occurred in a 4-week-old infant with Trisomy 13 already on comfort care due to respiratory failure secondary to adenovirus.
Discussion
Implementation of a hypothermic infant pathway at our institution decreased SBI evaluations without missing any infants with SBI or IBI. The pathway also reduced hospital admissions and decreased antibiotic exposure. To our knowledge, our study is the first to demonstrate the impact of a clinical pathway specific to hypothermic young infants.
The majority of institutions do not have a hypothermic infant specific pathway and often include this population into febrile infant pathways. 16 While use of a hypothermic young infant pathway is novel, implementation of clinical practice guidelines into pathways for febrile infants is nothing new.15,34 -36 The REVISE national quality initiative found febrile infant management improved when variability in care was reduced. 15 Additionally, standardization of care led to significant reductions in invasive interventions (ie, lumbar puncture) for febrile infants at low-risk for SBI without missing cases of SBI.34,35 Mercurio et al found with the implementation of clinical practice guidelines, fewer febrile infants underwent laboratory testing and less required antibiotics. 34 Similar developments for hypothermic infants could further improve the evaluation and management of this population as demonstrated by our pathway.
Implementation of the hypothermic infant pathway also led to a significant decrease in obtaining complete blood cell counts (CBC) and a mild decrease in the use of the inflammatory markers (IM) CRP and procalcitonin for evaluation. The role of CBC use in this population is unclear, however, several studies have demonstrated platelet abnormalities are associated with SBI and IBI.2,5,37 While IMs are routinely used to risk stratify febrile infants,14,38 less is known about the role of IMs to risk stratify hypothermic infants. 39 IMs may be less reliable due to the immune system dysfunction which occurs during hypothermia.40,41 More studies are needed to better understand CBC values and IM thresholds to better predict risk of bacterial infection in hypothermic infants.
Additionally, use of a hypothermic infant pathway did not reduce HSV evaluation or the empiric use of acyclovir. There was an increase in use of respiratory pathogen panels (RPPs) after pathway implementation. The use of RPPs in febrile infants to determine risk of SBI in setting of a virus identified has been well described.42,43 Only one study has evaluated risk of SBI in hypothermic infants and did not reveal any associations with a positive RPP, however, this study was underpowered. 24 The increased use of RPPs in the post-pathway group may be related to the COVID-19 pandemic.
Important differences in patient age exist between the pre- and post-pathway implementation groups, which can influence SBI evaluation. Notably, 93% of young infants were ≤21 days of age in the pre-pathway group compared to the 77% in the post-pathway group. While clinicians may be inclined to perform a SBI evaluation in younger infants, IBI and SBI have been found to be associated with infants ≥ 21 days of age when presenting with hypothermia.2,5,37
Our study is not without limitations. While adequately powered to detect a difference between SBI evaluation rates in our pre- and post-pathway implementation cohorts, our overall number of SBI and IBI remained low. We also defined SBI and IBI as a positive specimen culture treated as expected by the medical team, which may differ across institutions resulting in differences in SBI and IBI rates. We used temperature ≤36.0°C as our criteria for hypothermia; other institutions may have different temperature thresholds for hypothermia, however, a recent study demonstrated this was the most common temperature used by clinicians. 16 Additionally, we may have missed any infants included in our pathway with a temperature of 36.1°C or infants who developed hypothermia during the hospital admission. Due to the retrospective nature of the study, we are uncertain how often clinicians followed the care pathway as designed instead of relying on their own clinical judgment. As noted, 79 infants had incomplete documentation of the specific rewarming method limiting recommendations for a specific approach to rewarming infants presenting with hypothermia. Finally, we are a single, academic, pediatric tertiary care site in the southeastern US which may limit generalizability. Wider use and study of this pathway through quality improvement measures could help to delineate its generalizability.
Since the development of our pathway, additional data has emerged on these infants, which further support optimization of our local pathway and the development of generalizable evidence-based clinical practice guidelines for this population.2,20,21,25,44,45 Future hypothermic young infant clinical practice guidelines should consider inclusion of platelet abnormalities and the development of subsequent fever, while recognizing the need to identify other pathologies outside of infection contributing to hypothermia on presentation.2 -4,37,46
Footnotes
Acknowledgements
We wish to acknowledge Dr. Elizabeth E. Halvorson who critically reviewed our manuscript. We also wish to acknowledge Dr. Grace Williams who contributed to the development of the pathway.
Abbreviations
IBI (Invasive bacterial infection), SBI (Serious bacterial infection), HSV (Herpes simplex virus), EHR (Electronic health record), ED (Emergency department), ICD (International classification of diseases), CSF (Cerebrospinal fluid), CBC (Complete blood count), CRP (C-reactive peptide), PCR (Polymerase chain reaction), IM (Inflammatory markers), UTI (Urinary tract infection), Reducing Variability in the Infant Sepsis Evaluation (REVISE).
Author Contributions
Dr. Saylor G. McCartor, contributed to the design of the data collection instruments, collected data, coordinated, and supervised data collection, conceptualized the study design, drafted the initial manuscript, critically reviewed, and revised the manuscript, and approved the final manuscript as submitted. Dr. Jennifer L. Raffaele designed the data collection instruments, collected data, conceptualized the study design, edited the initial manuscript, and critically reviewed and revised the manuscript. Dr. Xiyan Tan conceptualized the study design, carried out the initial and final analyses, and critically reviewed and revised the manuscript. Drs. Brittany M. Ross, Mark B. Krom, Elizabeth S. Tyson, and Kevin Polley, contributed to the design of the data collection instruments, collected data, coordinated, and supervised data collection, conceptualized the study design, edited the initial manuscript, and critically reviewed and revised the manuscript. Dr. Nicholas M. Potisek conceptualized and designed the study, designed the data collection instruments, collected data, carried out the initial analyses, edited the initial manuscript, and critically reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
