Abstract
Background:
This study aimed to determine the usefulness of the urine based malaria rapid diagnostic test (RDT) in clinical settings.
Methods:
All participants had blood and urine samples tested. To assess RDT performance, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated.
Results:
The malaria positivity rates were 67.4% for the SD Bioline RDT, 65.3% for the CareStart RDT, and 45.3% for the Fyodor UMT. There was an excellent agreement between the SD Bioline and CareStart malaria RDTs (Cohen’s kappa of .859). There was moderate agreement between the Fyodor UMT and the 2 blood-based RDTs, with a kappa of .491 (P < .001). The Fyodor UMT had sensitivity, specificity, PPV, and NPV of 58.6%, 100.0%, 100.0%, and 40.0%, respectively.
Conclusion:
The urine-based malaria test kit may have limited utility as a standalone diagnostic tool for malaria in febrile children, due to its relatively low sensitivity.
Introduction
Malaria remains a public health problem globally, with children and pregnant women disproportionately affected.1 -3 It is a major contributor to infant and under-5 mortality.1,3 In Nigeria, there were an estimated 194 000 deaths from severe malaria in 2021; 80% of these deaths were in children under-5 years of age. 3 In the same year, Nigeria recorded approximately 68 million cases of malaria representing 27% of the global burden. According to the 2021 Malaria Indicator Survey, the national malaria prevalence was 39.6% and 22.3% by Rapid Diagnostic Test and Microscopy respectively.3,4 There were state to state variations in the malaria prevalence rates with the lowest prevalence reported in Lagos (3.2% and 2.6% by RDT and Microscopy respectively) and highest prevalence (75.6% and 49.0% by RDT and Microscopy respectively) in Kebbi. 4
Prompt diagnosis and appropriate treatment of malaria are key components of malaria control and elimination. 1 In line with the World Health Organization, the Nigeria Federal Ministry of Health also recommends that all suspected cases of malaria be confirmed by parasitological diagnosis prior to treatment.2,5 -7 Blood film microscopy, although considered as the gold standard for the diagnosis of malaria, has some limitations. It requires personnel with high technical skills, expensive equipment, and electricity and access to water supply. These requirements are often inadequate in many low income countries, especially in rural areas of Nigeria.
The malaria rapid diagnostic test (MRDT) were introduced more than 20 years ago and gradually gained prominence because it was able to address many of the limitations of blood film microscopy.8 -10 It does not require the use of expensive equipment, high technical skills or electricity. Furthermore, it is easy to perform, requires only minimal training, rapidly provides results and is quite useful in remote, poorly resourced settings.8 -10 It therefore provides increased access to diagnostic testing for malaria and ultimately improves rational use of antimalarials. MRDTs work by detecting malaria parasite-specific antigens such as histidine rich protein 2 (HRP-2), parasite lactate dehydrogenase (pLDH) and plasmodium aldolase. MRDTs function through immunochromatographic antigen capture, 10 yet despite their attractive qualities, they remain subject to certain limitations. These include the need for blood sampling. usually a finger prick which can cause mild discomfort to patients. Most kits detects HRP-2 protein, which is specific to P. falciparum, and may therefore fail to identify non- falciparum infections. Also, a high prevalence of malaria parasites isolates with plasmodium falciparun HRP-2 and HRP-3 genes deletions resulting in false negative results have been reported in Nigeria. 11 In addition, blood MRDTs may remain positive for a few weeks even after effective treatment and, test kits have varying diagnostic accuracy and performance.12 -14 A review article on the efficiency of rapid diagnosis test kits in malaria diagnosis reported that accuracy and performance of the MRDT depends on factors such as the patients’ age, geographical location (malaria transmission rate) and genetic factors, among others. 15
To complement blood film microscopy and blood-based malaria rapid diagnostic tests (MRDTs), novel diagnostic technologies are being explored. One such innovation is the urine-based MRDT, which leverages on the detection of malaria parasite antigens excreted in urine.16,17 Urine-based MRDTs offer a less invasive alternative to blood-based tests, eliminating the mild discomfort associated with finger pricking for blood sample collection. Also, it can easily be performed by non-technical persons, which would aid community diagnosis and treatment of malaria. This has the potential to increase parasitological diagnosis of malaria, avoid unnecessary use of antimalarial drugs, reduce the risk of drug resistance and lower health care costs.
Fyodor Biotechnologies Baltimore USA recently developed a urine based malaria test kit (UMT) for the diagnosis of malaria. It is a recombinant monoclonal antibody based test that detects Plasmodium falciparum specific HRP-2 in urine.18,19 The urine MRDT being a new innovation needs to be validated for commercial use by many studies and for the kits to be improved upon where necessary. Few studies have examined this, and their reported sensitivity and specificity values for the urine MRDT vary.18 -22 The sensitivity and specificity values of urine MRDT (UMT by Fyodor Biotechnologies Baltimore USA) ranged between 65% to 93% and 83% to 90% respectively according to some studies in Nigeria.18 -20
The objectives of this study were to determine the agreement between urine MRDT and blood MRDT and evaluate diagnostic performance of urine malaria RDT and blood malaria RDT in comparison with blood microscopy.
Patients and Methods
Study Design and Site
This was an observational cross-sectional study conducted at the Children Emergency Ward (CEW) of the Ekiti State University Teaching Hospital, Ado-Ekiti (EKSUTH), Ekiti State. The Children Emergency Ward (CEW) has a capacity of 12 beds and admits both pediatric medical and surgical cases. The hospital functions as a referral center for 3 specialist hospitals, 18 secondary health facilities, and over 150 primary health care centers. The CEW is staffed by pediatric interns, resident doctors, and consultant pediatricians. The Teaching hospital also houses a microbiology laboratory, managed by trained microbiologists, which provides diagnostic support to the CEW and other hospital units. Malaria remains the leading cause of admission to the CEW. 23 The average temperature of the study location is 27°C; highest temperature (29°C) usually in March and lowest (25°C) in June. It enjoys 2 distinct seasons; wet season (April-October) and dry season (November-March). The mean annual total rainfall is 1367 mm.24,25 This environmental condition favors all year malaria transmission with highest transmission during the rainy season. The prevalence of malaria was 46.3% in the study location. 4
Study Population
The study involved all children younger than 18 years who presented with fever. Children who met the inclusion criteria and whose caregivers provided verbal and written consent (and assent for participants aged 8 years and above) were enrolled. Exclusion criteria included a history of antimalarial use within the preceding week, current or previous hematuria, urine dipstick confirmed hematuria, and symptoms suggestive of connective tissue disorders..
Data Collection and Study Procedure
A study proforma was used to document the hospital number, age, sex and results of malaria tests. All consecutive patients with fever who met the inclusion criteria were recruited into the study. Blood sampling via finger prick was performed on each participant, and rapid diagnostic testing was carried out using Carestat blood HRP2 RDT and SD Bioline HRP2 RDT in accordance with manufacturers’ instructions.
One milliliter of blood was taken from each study participants and sent to the microbiology laboratory of the hospital for assessment of blood film for malaria parasite and parasite density. Then a spot urine sample was taken into a universal bottle for urine malaria test.
Sample testing for film microscopy (thick and thin), blood RDT and urine malaria test were done according to standard procedures.12,17
Data Analysis
To evaluate the aggregate performance of the RDTs, point estimates of sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated. Statistical analyses were performed using Stata Statistical Software version 18 (College Station, TX: StataCorp LLC). Sensitivity and specificity were calculated from comparison of RDT outcomes with the gold standard of microscopy. Kappa statistics were also calculated to assess agreement between the RDTs and microscopy reference standard, controlling for chance agreement. Kappa values were interpreted as follows: <.00 represents poor agreement, .00 to .20 represents slight agreement, .21 to .40 represents fair agreement, .41 to .60 represents moderate agreement, .61 to .80 represents substantial agreement, and .81 to 1.00 represents almost perfect agreement.26,27
For all inferential statistics, a P-value < .05 was taken as statistically significant.
Ethical Approval and Informed Consent
This study was approved by the Ekiti State University Teaching Hospital Ethics and Research Committee (approval number EKSUTH A67/2020/04/002). Written informed consent was obtained from the caregivers of all participants prior to enrollment in the study. Also, we obtained assent from all participants aged 8 years and above.
Results
There were 95 study participants. The median age of the study participants was 36.0 (Interquartile range: 21-96) months. The majority of participants (58%) were under 5 years old with a slightly higher proportion of males (54%; Table 1).
Age and Sex Distribution of Respondents n = 95.

Malaria Test Results of the Study Participants
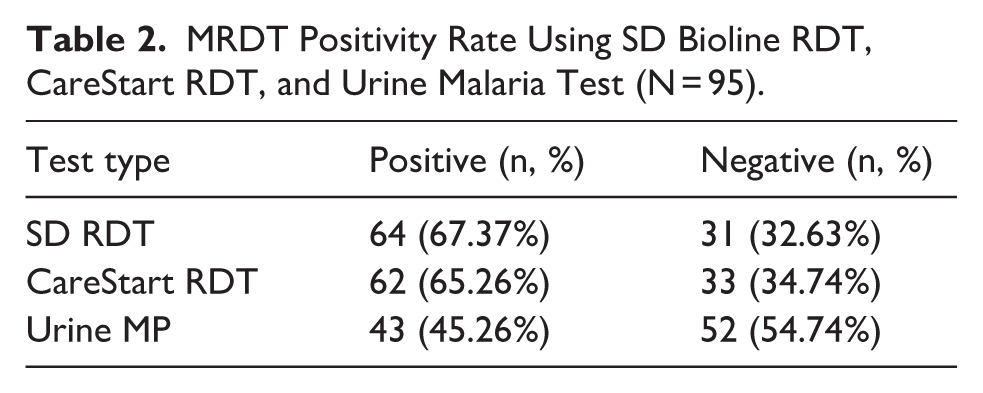
Among the 95 febrile children tested for malaria, the SD Bioline RDT had a positivity rate of 67.4%, while the CareStart RDT had 65.3% positivity rate. However, the urine-based malaria test kit showed a positivity rate of 45.3% (Table 2).
MRDT Positivity Rate Using SD Bioline RDT, CareStart RDT, and Urine Malaria Test (N = 95).
Performance Evaluation of the RDTs
Only 37 of the study participants had malaria parasite density done via microscopy. Of these, 8 (21.6%) had no parasitemia results, while 20 (54.1%) had moderate parasitemia., The SD Bioline RDT, CareStat and Urine MRDT results of these 37 participants were then compared with the microscopy result. Table 3 shows the age group and sex distribution of the study participants with malaria parasite density results.
Age Group and Sex Distribution of Children with Microscopy Results (N = 37).

Agreement and Kappa Statistics for Malaria Rapid Diagnostic Tests Among Febrile Children
There was strong agreement between the 2 blood-based rapid diagnostic tests (RDTs), the SD Bioline MRDT and the CareStart MRDT, with a Cohen’s kappa of .859, indicating near perfect concordance. In contrast, moderate agreement was observed between the urine malaria test and the 2 MRDTs, with kappa values of .491 for the comparison between the SD MRDT and the urine malaria test, and .570 for the comparison between the CareStart MRDT and the urine malaria test. All kappa values were statistically significant (P < .001; see Table 4).
Agreement and Kappa Statistics for Malaria Rapid Diagnostic Tests Among Febrile Children (n = 95).

Performance Evaluation of the RDTs
Using blood microscopy as the gold standard, the sensitivity and specificity of the SD Bioline MRDT was 72.4% and 100.0%, respectively. The positive predictive value (PPV) was 100.0%, while the negative predictive value (NPV) was 50.0%. In comparison, the CareStart MRDT had a sensitivity of 65.5% and specificity of 100.0%, with a PPV of 100.0% and an NPV of 44.4%. For the Urine MRDT, the sensitivity was 58.6% and specificity was 100.0%, with a PPV of 100.0% and an NPV of 40.0%, Details of these are provided in Table 5.
Sensitivity, Specificity, Positive Predictive Value and Negative Predictive Value of SD Bioline MRDT and CareStart MRDT.

When comparing the 3 MRDTs, the SD Bioline MRDT exhibited the highest sensitivity at 72.4%. All tests maintained a specificity of 100% and a PPV of 1.00 (100.0%). However, the NPV was highest for the SD Bioline MRDT at 50.0%, followed by the CareStart MRDT at 44.4%, and the Urine MRDT at 40.0% (Table 6).
Overall Diagnostic Performance of the 3 Tests Versus Microscopy.

Abbreviations: PPV, positive predictive value; NPV, negative predictive value.
Discussion
This study evaluated the performance of 3 malaria rapid diagnostic test kits in the clinical diagnosis of malaria among febrile children. The blood MRDT kits showed a higher detection rate of malaria than the urine MRDT kit. This agrees with previous studies in children by Ebuta et al and Ameh et al.28,29 Both studies reported a higher prevalence of malaria using blood RDT kits than UMT. Studies by Oyeniyi et al 30 and Okete et al 31 however reported a higher prevalence of UMT malaria positivity. These latter studies were conducted in only adults only and a mixed adult and pediatric population respectively. These findings suggest that the detection of malaria using the UMT may be dependent on the age of the patient. A meta-analysis by Danwang et al which included 18 studies also concluded that malaria rapid diagnostic test kits performed better on blood than urine samples. 32
The 2 blood MRDT kits showed excellent agreement with each other, but only moderate agreement with the UMT. This may reflect possible differences between the concentration of the HRP-2 antigen in the blood and urine. This may also suggest that the 2 blood MRDT kits do not confer any advantage over each other in the diagnosis of malaria and may be ready alternatives. However, the blood MRDT kits appear to perform better in malaria diagnosis compared to UMT.
The highest prevalence of malaria in this study occurred among children aged 2 to 4 years. This is consistent with previous findings that the prevalence of malaria is highest in the under-5 children. 28 Immunity to malaria among children improves with age, hence it is not surprising that the prevalence is highest among the under-fives. It may also reflect the higher number of participants from that age group in the study.
The sensitivity of SD-Bioline RDT in this study is 72.4%, suggesting that it can correctly detect about 72 out of 100 true malaria cases. This falls below the World Health Organization minimum recommendation for malaria test kits 33 and a sensitivity of 92.8% reported by Falade et al 34 in a multicenter study of 1860 under-5 children (aged 3 months to 5 years) in southwest Nigeria. It is however similar to the sensitivity of 75.0% reported by Nkenfou et al from Cameroon in a study done among children aged 6 months to 5 years. 33 Oyeniyi et al reported a much lower sensitivity of 25.4% among adults (>18 years) from southwest Nigeria. 30 The differences in the prevalence of malaria in the different study locations and the age of the study participants may be partly responsible for the variations in the sensitivities obtained from these studies, despite using the same SD-Bioline test kit. Generally, sensitivity of a test kit improves with prevalence of a disease and children are more prone to malaria than adults. The specificity is 100% in this study, and much higher than 49.1% and 40.7% reported by Nkenfou 33 and Falade 34 respectively. Neither of these 2 studies excluded children who were recently treated for malaria, and since MRDT would detect recently treated malaria up to 14 days post-treatment, 35 this would increase the false positive rate and lower the specificity.
Despite the moderate sensitivity of SD-Bioline in this study, the high positive predictive value of the SD-Bioline kit makes it a good screening tool for malaria, since predictive values are mostly thought to be more clinically useful than sensitivity and specificity.36,37 The combination of a high specificity and high positive predictive value makes the kit very reliable in detecting malaria. The lower sensitivity and negative predictive value would mean it is not a good tool to exclude malaria.
The CareStat MRDT kit has similar sensitivity (65.5%), specificity (100%), PPV (100%) and NPV (44.4%) as the SD-Bioline MRDT in this study. The degree of agreement between the 2 kits was also high (Cohen kappa .859). This suggests that under similar clinical conditions, both kits are likely to give similar results and could be ready substitutes in the diagnosis of malaria.
The urine malaria test kit (UMT) showed a low sensitivity of 58.6%. The low sensitivity suggests that the UMT would not be a reliable test to diagnose malaria and may miss many cases of malaria. This unreliability is further strengthened by the low NPV. The sensitivity result in this study is at great variance with reported sensitivities of 4.5%, 44.7%, 79.0% and 83.75% reported by different studies.18,19,28,29 Similarly low NPV of 43.2% and 55.5% were reported by Oyeniyi et al 30 and Okete et al 31 from Nigeria. The low sensitivity could also be a result of the timing of the urine sample collection. Studies showed that early morning urine sample tended to yield a better sensitivity, 19 since it is usually more concentrated thereby improving the likelihood of antigen detection, 38 However this is not practicable in clinical settings where diagnosis and treatment are usually required at the time of presentation. The high specificity and PPV of UMT in this study however suggests that a positive UMT screening result would be confirmatory of malaria. Reports by Ameh et al 29 and Okete et al 31 also showed high specificity and PPV.
Various factors could have influenced the diagnostic ability of the UMT in these various studies. These include the age of the participants, prevalence of malaria in the study location, the parasite density in the study participants, methodology (product manufacturer, duration of incubation of the UMT strip, rapid degradation of malaria antigens in urine samples), presence of urobilinogen and rheumatoid factor in the urine, genetic polymorphisms and PfHRP-2 deletions, and the prozone effect.19,22,39 Furthermore, the species of Plasmodium is an important factor as currently available UMT kits can only detect PfHRP-2, which is specific to P. falciparum. Thus, they could miss cases of non-falciparum malaria. In this study, the fact that only 3 out of the patients with blood microscopy and parasite density had high parasitemia (≥10 000 per µL of blood) might have contributed to the low performance of UMT recorded.
The UMT showed moderate agreement with the 2 MRDT kits used in this study (Cohen kappa of .49 and .57 for SD-Bioline and CareStart respectively). This may suggest some limitations in the use of UMT as an alternative diagnostic tool to the MRDT in the diagnosis of malaria.
All 3 diagnostic methods showed moderate agreement with microscopy in detecting malaria. The degree of agreement was lowest for UMT. Microscopy remains the gold standard for the diagnosis of malaria in clinical settings. The moderate agreement of the test kits with microscopy is a call for caution in their use to diagnose malaria. Confirmation with blood film microscopy should be sought where necessary. Ultra-sensitive MRDTs are also underway for use in clinical setting and they have been shown in a few studies to have higher sensitivity and specificity than the conventional MRDT. 40
A limitation of our study was the low number of participants with blood film microscopy result. Also, we did not measure urine specific gravity which would have helped to assess the concentration of the urine samples. Nevertheless, the study provides evidence that blood based MRDTs outperform UMT in febrile children in our setting.
Future studies may consider larger sample sizes to allow for a more representative population with results from blood film microscopy. This will allow for better comparisons between the different diagnostic modalities. Longitudinal studies can also be conducted to include comparison of MRDT and UMT performances across time, particularly between populations from different geographical settings. Investigating the compatibility of MRDTs with other diagnostic modalities, such as molecular techniques, can also give more insight into the role of MRDTs in malarial diagnosis and management. Also, comparing cost-effectiveness of MRDTs versus UMTs in various healthcare facilities could provide useful information for policymakers.
Conclusion
Blood-based malaria rapid diagnostic tests (RDTs) remain reliable tools for malaria diagnosis in clinical settings. The 2 evaluated blood-based RDT kits demonstrated comparable performance and may serve as suitable alternatives to each other. In contrast, the urine-based malaria test showed relatively low sensitivity and may have limited utility as a standalone diagnostic tool for malaria in febrile children in our setting.
Footnotes
Acknowledgements
The authors are grateful to the health care team working in the Children Emergency Ward of the Ekiti State University Teaching Hospital, Ado-Ekiti.
Authors Contributions
AOB was involved in conceptualization, data curation, literature review and drafted the manuscript. ATA was involved in data curation and critically reviewed the manuscript. TOO analyzed the data and critically reviewed the article. BFI was involved in data curation and critically reviewed the manuscript. OSO supervised data analysis and critically reviewed the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used/or analyzed during the current study available from the corresponding author on reasonable request
