Abstract
Pediatric neurological disorders such as autism spectrum disorder (ASD) and attention-deficit/hyperactivity disorder (ADHD) are increasingly understood to arise from complex interactions between genetics and environmental factors. Emerging evidence highlights the role of epigenetic mechanisms, particularly DNA methylation, histone modification, and non-coding RNAs, in mediating how early-life exposures shape brain development. This review synthesizes findings from 48 recent studies, including human cohorts and animal models, linking prenatal stress, maternal nutrition, endocrine disruptors, and air pollution to epigenetic alterations that disrupt neurogenesis, synaptic plasticity, and neural differentiation. These mechanisms offer insight into clinical phenomena and open potential for early screening and epigenetic interventions in pediatric neurology. The main finding reveals that early environmental exposures can leave lasting epigenetic marks that significantly influence neurodevelopment and contribute to pediatric neurological disorders.
Keywords
Introduction
Pediatric neurological disorders such as Autism Spectrum Disorder (ASD) and Attention-Deficit/Hyperactivity Disorder (ADHD) have seen a noticeable rise in global prevalence over the past few decades.1,2 While genetic predisposition remains a central factor in these conditions, emerging research underscores the pivotal role of environmental influences during early development, particularly through epigenetic modifications.3,4 These changes do not alter the DNA sequence itself but regulate gene expression, shaping neural outcomes in critical periods of brain development.
The developing brain is especially vulnerable to environmental cues during prenatal and early postnatal life. Factors such as maternal stress, poor nutrition, endocrine-disrupting chemicals, and air pollution can alter epigenetic marks, including DNA methylation, histone modifications, and non-coding RNA expression.5 -7 These molecular alterations may lead to dysregulation of neurodevelopmental genes, potentially increasing susceptibility to disorders like ASD and ADHD.8 -10
Despite the growing evidence, the exact pathways through which environmental exposures influence pediatric neurological outcomes remain complex and often underexplored in clinical contexts. Many studies remain siloed within molecular biology or epidemiology, with limited integration into pediatric neurology. Thus, a focused synthesis is needed to link environmental epigenetics with clinically observable pediatric disorders.
This review aims to bridge that gap by exploring how environmental factors impact epigenetic regulation during early brain development and how these changes may contribute to pediatric neurological disorders. By integrating evidence from human cohorts, animal models, and molecular studies, this review highlights emerging clinical implications, such as early screening strategies, epigenetic biomarkers, and therapeutic interventions, with a specific emphasis on their relevance to child neurology.
Methodology
This review was conducted using a structured narrative approach to synthesize current knowledge on the interplay between environmental exposures, epigenetic mechanisms, and pediatric neurological disorders such as autism spectrum disorder (ASD) and attention-deficit/hyperactivity disorder (ADHD). The emphasis was placed on connecting molecular-level epigenetic changes to clinical outcomes in children.
We performed a comprehensive literature search using databases including PubMed, Scopus, and Web of Science. Search terms included combinations of keywords such as epigenetics, brain development, maternal stress, autism, ADHD, DNA methylation, histone modification, non-coding RNAs, and pediatric neurology. Peer-reviewed studies published in English between 2000 and 2025 were considered, with a focus on the most recent decade to capture the latest advances.
In total, 48 relevant papers were selected after screening based on relevance to early-life environmental exposures (eg, maternal stress, pollution, endocrine disruptors, and nutrition), epigenetic regulatory mechanisms (including DNA methylation, histone modifications, and non-coding RNA expression), and their associations with neurodevelopmental outcomes in children. Both human cohort studies and animal model research were included, particularly those providing translational insights or longitudinal findings. Screening was conducted based on predefined inclusion and exclusion criteria detailed in Table 1, ensuring relevance to early-life environmental exposures, epigenetic regulatory mechanisms, and pediatric neurodevelopmental outcomes.
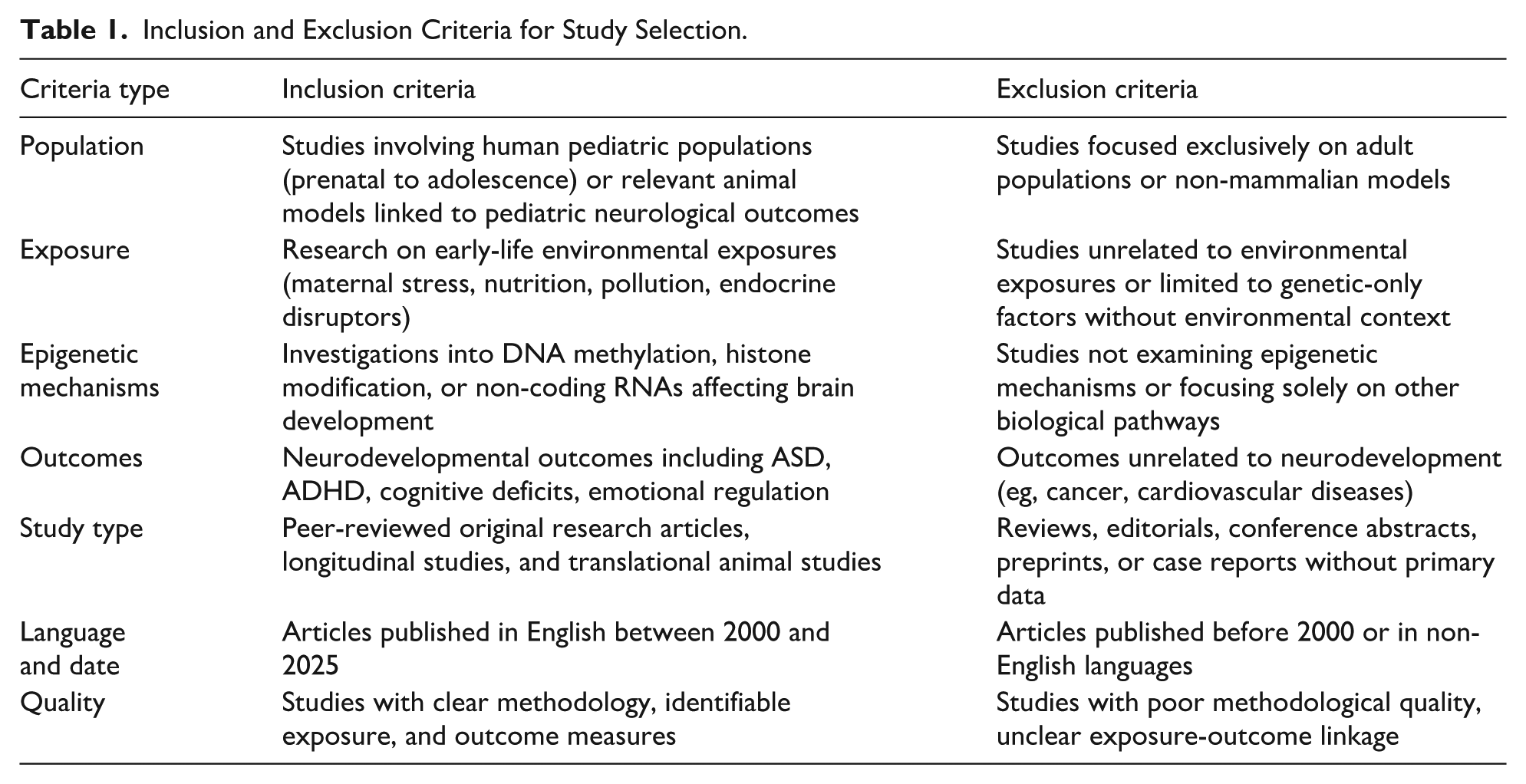
Inclusion and Exclusion Criteria for Study Selection.
The selected studies were categorized and analyzed according to core thematic areas: epigenetic mechanisms in brain development, environmental risk factors and their epigenetic effects, translational evidence from human and animal studies, and clinical implications for pediatric neurology. Rather than conducting a meta-analysis, we synthesized the findings narratively, aiming to highlight biological mechanisms and clinical relevance, and to inform future directions for early diagnosis, risk stratification, and potential epigenetic interventions in child neurology. Methodological flow of this research is presented in Figure 1.
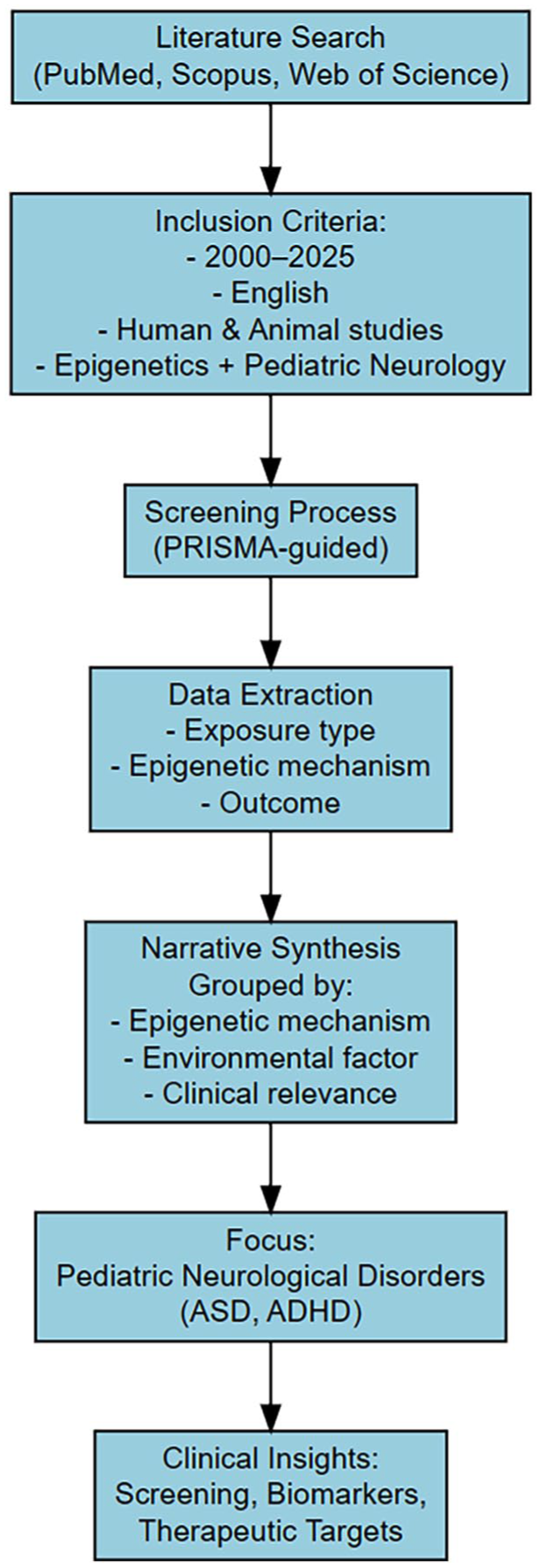
Methodological flow.
Ethical Approval and Informed Consent
Not applicable as this study didn’t include animals or human participants.
Results
The interaction between epigenetic regulation and environmental exposure plays a pivotal role in shaping early brain development. 3 This section explores how specific epigenetic mechanisms influence neurogenesis and neural differentiation, and how environmental risk factors may disrupt these processes. We also examine supporting evidence from human and animal studies and discuss clinical implications for pediatric neurology.
Epigenetic Mechanisms and Brain Development
DNA Methylation and Neurogenesis
DNA methylation plays a central role in regulating gene expression during embryonic brain development, particularly by silencing or activating neural-specific genes at key developmental windows. This mechanism is essential for neurogenesis, where neural progenitor cells differentiate into neurons and glial cells. 11 Aberrant DNA methylation patterns have been observed in disrupted neurogenesis and linked with neurodevelopmental disorders. For instance, studies have shown hypermethylation of promoters in Wnt and Notch signaling pathways, both essential for neuronal differentiation, correlating with altered brain volume in children diagnosed with autism spectrum disorder (ASD). 9 In utero environmental factors, such as maternal stress and nutrient deficiencies, can induce long-term methylation changes in neurodevelopmental genes. Case studies in ASD and ADHD have revealed abnormal methylation in key regulatory genes like MECP2 and SHANK3, suggesting epigenetic dysregulation may underlie observed phenotypes.10,12
Histone Modification in Synaptic Plasticity
Histone modifications, such as acetylation and methylation, critically regulate chromatin accessibility and thus gene transcription in the developing brain. These modifications are especially important for synaptic plasticity, learning, and memory.6,7 Histone acetylation promotes a relaxed chromatin structure conducive to the expression of synaptic proteins and neuronal signaling molecules. Dysregulation of histone acetyltransferases (HATs) or histone deacetylases (HDACs) can impair synaptic pruning, a process crucial for cortical maturation. Abnormal pruning has been implicated in ASD and ADHD, where both hypo- and hyperconnectivity are observed. 13 Genetic studies have linked mutations in histone-modifying enzymes like KDM5C and EHMT1 to intellectual disability and behavioral impairments, highlighting histone regulation as a key epigenetic contributor to pediatric neurological disorders.14,15
Non-coding RNAs in Neural Differentiation
Non-coding RNAs (ncRNAs), including microRNAs (miRNAs) and long non-coding RNAs (lncRNAs), play essential roles in regulating early neural differentiation, neuronal migration, and connectivity. These molecules modulate gene expression post-transcriptionally and contribute to chromatin remodeling. For example, miR-124 and miR-9 are involved in neuronal lineage specification, and disruptions in their expression can lead to disordered neuroarchitecture. 16 Emerging studies have found altered expression of miRNAs such as miR-137 and miR-132 in children with ASD, linking them to deficits in synaptic function and plasticity. 17 Furthermore, the potential for these ncRNAs to serve as non-invasive biomarkers for early diagnosis in pediatric neurology is gaining attention, as they are detectable in cerebrospinal fluid and even peripheral blood. 18 Emerging evidence now highlights circular RNAs (circRNAs) as stable and regulatory non-coding species whose expression in the developing brain can be altered by environmental exposures, such as PM2.5 in rodent models, underscoring their potential role in epigenetic mediation of neurodevelopmental disorders. 19
Environmental Risk Factors and Epigenetic Alterations
Prenatal Stress and Cortisol Exposure
Maternal stress during pregnancy has significant effects on fetal brain programing through epigenetic changes, particularly involving the hypothalamic-pituitary-adrenal (HPA) axis. Elevated cortisol levels can cross the placenta and affect fetal brain development by altering DNA methylation in the glucocorticoid receptor gene (NR3C1).20,21 These epigenetic alterations can impair stress response systems in the child, contributing to behavioral dysregulation and increased risk for ADHD. 22 Prenatal stress has also been associated with changes in brain structure, particularly in the hippocampus and amygdala, which are key regions involved in emotion regulation and executive function. 23
Maternal Nutrition and Micronutrient Availability
Maternal intake of methyl-donor nutrients such as folate, vitamin B12, and choline is essential for proper DNA methylation during fetal neurodevelopment. Deficiencies in these nutrients can disrupt neurodevelopmental pathways and are associated with increased risks of cognitive and behavioral disorders.24,25 For instance, low maternal folate has been linked with hypomethylation of the BDNF gene and impaired neural connectivity. 26 Intervention studies suggest that prenatal supplementation can mitigate some of these effects, offering a potential preventive strategy for populations at risk. 27 Moreover, nutrient status can influence not just the immediate offspring but also epigenetically program susceptibility in future generations. 28
Endocrine-Disrupting Chemicals (eg, BPA, Phthalates)
Exposure to endocrine-disrupting chemicals (EDCs) like bisphenol A (BPA) and phthalates during pregnancy can have lasting effects on the child’s neurodevelopment by altering epigenetic regulation of hormone-sensitive genes. BPA, for example, has been shown to modify DNA methylation at the estrogen receptor (ESR1) gene locus, disrupting brain sexual differentiation and increasing risk for ASD-like behaviors.29,30 These findings emphasize the need for stricter public health measures and regulations surrounding prenatal exposure to EDCs.
Air Pollution and Particulate Matter Exposure
Airborne pollutants, particularly fine particulate matter (PM2.5), have been implicated in neuroinflammatory processes and oxidative stress, which may mediate their neurotoxic effects via epigenetic modifications. 31 Children in urban environments exposed to high levels of air pollution have shown changes in DNA methylation patterns of genes involved in neural signaling and inflammation.32,33 Longitudinal studies, such as the Columbia Center for Children’s Environmental Health cohort, have linked early exposure to traffic-related pollutants with lower IQ scores and increased ADHD risk.34,35 Moreover, socioeconomic disparities exacerbate these effects, as marginalized communities are often disproportionately affected, raising concerns around environmental justice and pediatric neurological health.
Translational Evidence from Human Cohort and Animal Studies
Notable Longitudinal Cohort Studies
Longitudinal cohort studies provide critical insight into how prenatal and early-life exposures interact with the epigenome to shape neurodevelopment. The Avon Longitudinal Study of Parents and Children (ALSPAC) in the UK has identified DNA methylation signatures associated with maternal smoking, diet, and stress, revealing links to later cognitive and behavioral outcomes in children.36,37 Similarly, the CHAMACOS cohort in California has demonstrated that prenatal exposure to organophosphate pesticides correlates with reduced IQ and increased ADHD risk at school age, with associated changes in methylation patterns in neurodevelopmental genes.38,39 The Generation R study in the Netherlands has linked urban air pollution with altered brain morphology and reduced white matter integrity, implicating inflammation-driven epigenetic changes in children from highly polluted environments. 40 These large-scale, population-based studies underscore how environmental factors leave molecular imprints that may serve as early indicators of neurodevelopmental vulnerability.
Translational Insights from Rodent and Primate Models
Animal models, especially rodents and non-human primates, have played a crucial role in establishing cause-and-effect relationships between environmental exposures and epigenetic changes. Controlled experiments in rodents have shown that prenatal stress leads to methylation of the NR3C1 promoter, blunting HPA axis feedback and producing anxiety-like behavior in offspring. 20 Similarly, BPA exposure in pregnant mice alters methylation of estrogen receptor genes in the offspring’s brain, impairing social behavior. 41 In primate studies, maternal stress has been shown to alter expression of genes involved in synaptic plasticity, providing a closer translational link to human neurodevelopment. 42 However, the extrapolation of animal findings to humans remains limited by species differences in brain development timelines, environmental complexity, and social behavior, warranting cautious interpretation.
Clinical Implications for Pediatric Neurology
Epigenetic Biomarkers in Early Screening
The identification of epigenetic markers in peripheral tissues such as blood or saliva offers a promising route for early detection of neurodevelopmental disorders. Several studies have identified differential methylation patterns in genes like MECP2, OXTR, and NR3C1 in children later diagnosed with ASD or ADHD, suggesting their potential utility in screening. 12 These biomarkers could support pediatric neurologists in assessing risk before the full clinical phenotype emerges, facilitating early intervention. However, challenges persist regarding the tissue specificity of epigenetic changes, the reproducibility of biomarker panels, and ethical concerns around predictive screening in children. 43
Risk Stratification Based on Exposure History
Incorporating a detailed record of prenatal and early-life exposures into pediatric neurological assessments could improve risk stratification for disorders like ASD, ADHD, and intellectual disability. Exposure to environmental toxicants, maternal stress, and nutritional deficiencies can all be used alongside family history and developmental milestones to flag children for closer monitoring. This approach could help allocate limited resources to those at highest risk, supporting more equitable preventive care. 44 However, such strategies must be developed with attention to privacy, consent, and potential stigmatization, particularly in underserved populations.
Opportunities for Epigenetic Therapy and Intervention
Given the plasticity of the epigenome, there is growing interest in interventions aimed at reversing or mitigating early-life epigenetic disruptions. Nutritional interventions, such as folate, choline, and omega-3 fatty acid supplementation, during pregnancy have shown promise in modulating DNA methylation and improving neurodevelopmental outcomes. 45 Pharmacological agents targeting HDACs and DNA Methyltransferase (DNMTs) are under investigation for neurodevelopmental disorders, though their use in children remains experimental due to safety concerns. 46 Epigenetic counseling and personalized maternal care plans could become integral parts of pediatric neurological practice in the future.
Future Directions
There is an urgent need for longitudinal, multi-omics studies that combine epigenomic, transcriptomic, proteomic, and metabolomic data to understand the complex biological underpinnings of pediatric neurological disorders. Such integrated approaches could identify causal pathways linking environmental exposures to altered brain development. Establishing causality versus mere association remains a critical challenge, and methods like Mendelian randomization and intervention-based animal models will be essential in addressing this gap.47,48
Developing clinically usable epigenetic screening tools requires standardized protocols, cross-tissue validation, and large normative datasets. Pediatric neurologists may eventually be able to incorporate epigenetic risk profiles into clinical algorithms, much like genetic testing today. Ultimately, personalized medicine in pediatric neurology could evolve to include individualized prevention and treatment plans based on a child’s unique epigenetic landscape, bridging molecular insights with clinical care. Table 2 presents a summary of the results of this review paper.
Summary of the Results.

Abbreviations: ADHD, attention deficit hyperactivity disorder; ALSPAC, Avon Longitudinal Study of Parents and Children; ASD, autism spectrum disorder; BDNF, brain-derived neurotrophic factor; BPA, Bisphenol A; CHAMACOS, Center for the Health Assessment of Mothers and Children of Salinas; CSF, cerebrospinal fluid; DNMT, DNA Methyltransferase; EDCs, endocrine-disrupting chemicals; EHMT1, Euchromatic Histone Lysine Methyltransferase 1; ESR1, Estrogen Receptor 1; HAT, Histone Acetyltransferase; HDAC, Histone Deacetylase; HPA axis, Hypothalamic–Pituitary–Adrenal axis; IQ, intelligence quotient; KDM5C, Lysine Demethylase 5C; MECP2, Methyl-CpG Binding Protein 2; miR, MicroRNA; NR3C1, Nuclear Receptor Subfamily 3, Group C, Member 1 (Glucocorticoid Receptor); OXTR, oxytocin receptor; PM2.5, particulate matter with diameter ≤ 2.5 μm; SHANK3, SH3 and Multiple Ankyrin Repeat Domains 3.
Discussion
This review highlights the complex and dynamic interplay between environmental exposures, epigenetic regulation, and early brain development, offering a mechanistic lens through which to understand the origins of pediatric neurodevelopmental disorders. The findings reinforce that epigenetic mechanisms such as DNA methylation, histone modifications, and non-coding RNAs play essential roles in neurogenesis, synaptic plasticity, and neuronal connectivity, processes that are highly sensitive to prenatal and early-life environmental influences. Disruptions in DNA methylation patterns, notably in pathways like Wnt and Notch, are consistently associated with ASD and ADHD phenotypes, while alterations in histone acetylation and specific microRNA expression further illustrate how fine-tuned gene regulatory networks can be disturbed by external insults. A consistent theme emerging from human cohort studies and translational animal research is the critical window of vulnerability during gestation and early infancy, when environmental exposures such as maternal stress, micronutrient deficiencies, endocrine-disrupting chemicals, and air pollutants can imprint lasting epigenetic changes that predispose to cognitive, emotional, and behavioral dysfunctions later in life.
The convergence of evidence from large-scale longitudinal studies like ALSPAC, CHAMACOS, and Generation R strengthens the argument for environmental exposures leaving molecular “scars” detectable as early epigenetic biomarkers. However, despite the wealth of associative evidence, causality remains difficult to establish in human studies due to confounding factors and ethical limitations. Rodent and primate models provide indispensable insights into causative pathways but must be interpreted cautiously due to species-specific differences in brain development and environmental contexts. Clinically, the emerging field of epigenetic biomarkers offers exciting prospects for early detection and risk stratification of neurodevelopmental disorders, though challenges around tissue specificity, reproducibility, and ethical deployment in pediatrics remain significant hurdles. Moreover, the evidence suggests that targeted interventions, particularly nutritional supplementation during pregnancy, may offer promising routes to mitigate adverse epigenetic programing, though pharmacological strategies remain largely experimental and must be approached carefully given the risks of altering epigenetic landscapes in a developing brain.
Looking forward, advancing the field will require comprehensive, integrative multi-omics studies that can map the dynamic interplay between environmental exposures and biological systems across developmental trajectories. Standardization of epigenetic assays, establishment of normative pediatric epigenetic reference databases, and rigorous causal modeling using tools like Mendelian randomization will be critical to translate these molecular insights into clinical practice. Ultimately, a paradigm shift toward personalized pediatric neurology, incorporating environmental exposure history, epigenetic risk profiling, and individualized intervention strategies, holds the potential to transform early diagnosis and prevention of neurodevelopmental disorders. This vision, however, demands interdisciplinary collaboration, sustained investment in longitudinal cohorts, and a commitment to addressing social and environmental inequities that disproportionately burden vulnerable populations. Future research should also address the influence of paternal environmental exposures on offspring epigenetic programing, as emerging evidence suggests that sperm-derived epigenetic marks may significantly contribute to neurodevelopmental outcomes. A conceptual framework summarizing these relationships is depicted in Figure 2, highlighting how diverse environmental exposures converge upon epigenetic regulatory pathways to influence pediatric brain development.
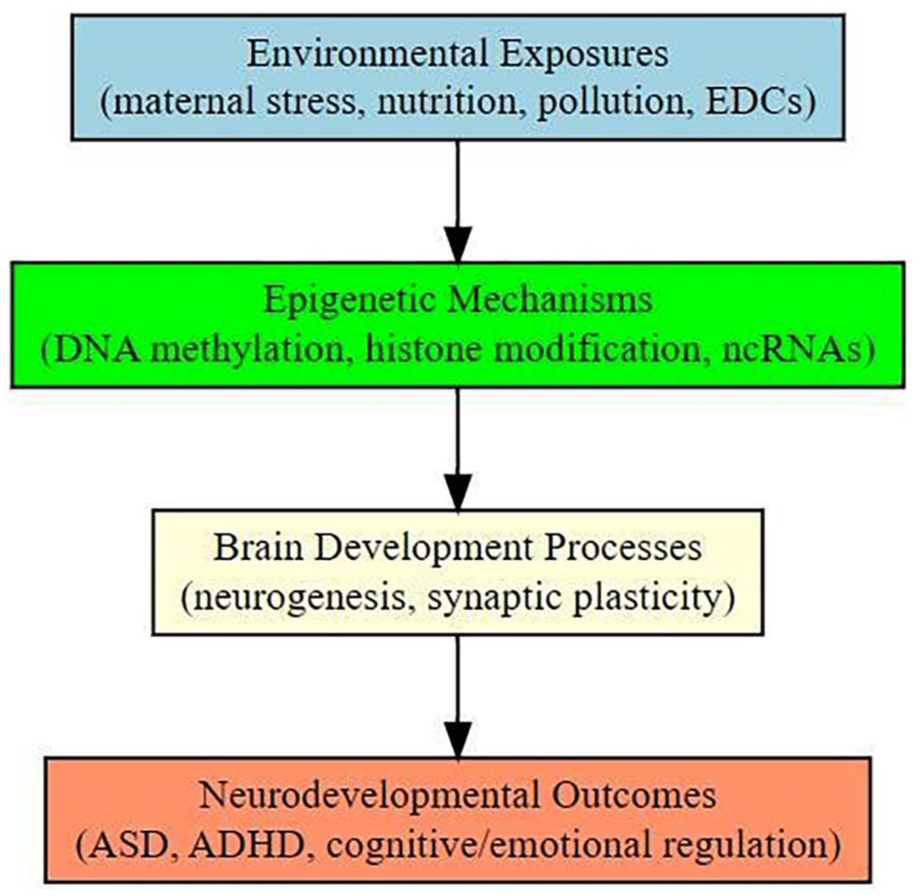
Conceptual framework linking environmental exposures, epigenetic mechanisms, and pediatric neurodevelopmental outcomes.
Conclusion
The intricate interplay between environmental exposures and epigenetic regulation during early brain development is emerging as a critical area in understanding pediatric neurological disorders. From prenatal stress and nutritional imbalances to environmental pollutants and endocrine disruptors, these external influences can leave lasting epigenetic marks that shape neurodevelopmental trajectories. Mechanisms such as DNA methylation, histone modifications, and non-coding RNAs are pivotal in mediating these effects, potentially disrupting neural differentiation, synaptic plasticity, and cognitive functions.
Evidence from longitudinal human cohort studies and controlled animal models underscores both the complexity and significance of these epigenetic-environment interactions. Importantly, this growing body of research opens new clinical possibilities, from early risk screening using peripheral biomarkers to novel therapeutic avenues aimed at reversing epigenetic dysregulation.
However, challenges remain. Distinguishing causation from correlation, translating findings across species, and integrating epigenetic tools into standard pediatric neurology practices will require interdisciplinary collaboration and longitudinal, multi-omics studies. As we advance, a precision medicine approach, guided by a child’s epigenetic and exposure profile, could fundamentally change how we understand, prevent, and treat childhood neurological disorders. The opportunity to intervene earlier and more effectively has never been greater.
Footnotes
Acknowledgements
None.
Ethical Considerations
Ethical approval was not required.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Hassan Jubair: Conceptualization, literature search, data extraction, analysis, writing (draft and editing), visualization, referencing, final approval.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable.
Trial Registration Number/Date
Not applicable.
