Abstract
Growth hormone deficiency (GHD) is a key differential diagnosis in children with proportionate short stature. While brain magnetic resonance imaging (MRI) is recommended for all confirmed GHD cases to rule out hypothalamic-pituitary anomalies, its universal application remains debated We conducted a retrospective, single-center study including 84 pediatric patients diagnosed with isolated GHD Brain MRI revealed hypothalamic-pituitary abnormalities in 27.4%. Compared to those with normal MRIs, patients with abnormalities were younger (6.78 ± 2.72 vs 8.34 ± 3.38 years, P = .038), shorter (height SDS −2.83 ± 0.58 vs −2.56 ± .64, P = .032), and had lower IGF-1 SDS (−1.07 ± .66 vs −.78 ± .70, P = .041). Multivariate logistic regression confirmed age, height SDS, and IGF-1 SDS as independent predictors of MRI anomalies. ROC analysis identified cutoff values with moderate predictive accuracy: age <7.1 years, height SDS <−2.7, and IGF-1 SDS <−.9.
Keywords
Introduction
Short stature is a clinical condition defined by a height that is more than 2 standard deviations below the mean, or below the third percentile, for age and sex, relative to the patient’s reference ethnic population. 1 In pediatric practice, among the possible differential diagnoses of a child presenting with proportionate short stature,1 -3 growth hormone deficiency (GHD) must be considered. 4 GHD is characterized by a complete or partial, organic or idiopathic impairment of the pulsatile secretion of growth hormone (GH), 5 leading to reduced circulating levels of IGF-1 and a deceleration in linear growth velocity. 6 The incidence of GHD varies according to the ethnicity of the population studied, but it is estimated to range from approximately 1 in 28 800 to 1 in 46 700 children, with a prevalence of 1 in 4000 to 1 in 10 000 children.1,2
The majority of GHD cases are idiopathic in origin, 7 although it is well established that any significant anatomical, traumatic, neoplastic, congenital, or inflammatory insult affecting the hypothalamic-pituitary region can result in secondary GHD of known etiology.8,9 The exact incidence of organic forms of GHD is not fully known1,2; the majority of patients present with isolated GHD, which is classified as idiopathic, while fewer than 10% of patients without other clinical symptoms exhibit an anatomical alteration, either congenital or acquired, of the hypothalamic-pituitary region.9,10
GH secretion follows a circadian rhythm and may be partially or completely impaired, either temporarily or permanently 11 ; growth hormone is the primary regulator of bone growth and enables linear stature increase until the complete closure of the growth plates, a process that occurs under the stimulation of sex hormones such as testosterone and estrogens.11,12 According to current national guidelines, the diagnosis of GHD requires an abnormal response—defined as a GH peak <8 ng/mL—in at least 2 different GH stimulation tests. 13 These tests are generally performed following the observation of growth deceleration, particularly when auxological parameters fall below the target range based on parental heights, in the presence of a proportionate phenotype and reduced serum IGF-1 levels. 14 IGF-1, the main peripheral mediator of GH activity, is routinely measured in children with suspected growth failure. 15
In distinguishing idiopathic from organic forms of GHD, it is recommended that brain MRI with a detailed assessment of the hypothalamic-pituitary region be performed after diagnosis and prior to the initiation of GH replacement therapy. 16 This imaging modality is critical for the early detection of anatomical abnormalities and for the exclusion of neoplastic lesions, which, if active, currently constitute a contraindication to GH treatment.17,18 Although brain magnetic resonance imaging (MRI) is regarded as an indispensable diagnostic tool and is endorsed by current guidelines for all confirmed cases of growth hormone deficiency,19,20 even in the absence of neurological symptoms or clinical suspicion of intracranial pathology, 1 it remains a costly procedure. Furthermore, in children, MRI is often challenging to perform while awake, frequently necessitating hospitalization and sedation. Notably, many patients with GHD exhibit completely normal brain MRI results, lacking hypothalamic-pituitary abnormalities such as pituitary hypoplasia, interruption or absence of the pituitary stalk, or ectopic or absent posterior pituitary lobe. 21 Consequently, several authors have questioned the routine utility of this imaging technique in children without neurological signs or strong clinical suspicion of an organic etiology. 22
Despite these considerations, brain MRI currently remains crucial in the diagnosis of idiopathic and organic GHD,1 -5 and it is recommended for all patients at the time of diagnosis.6 -9 There are no clear predictive factors of hypothalamic-pituitary abnormalities that can definitively identify patients with such alterations, except for those with multiple pituitary hormone deficiencies who become symptomatic early due to corticosurrenal or thyroid insufficiency.1 -7
Based on these considerations, we propose this retrospective, single-center study aimed at analyzing the incidence of hypothalamic-pituitary abnormalities in a cohort of children diagnosed with GHD.
Materials and Methods
This is a retrospective, single-center study conducted at the Pediatric Endocrinology Unit of Ospedale Perrino—Azienda Sanitaria Locale di Brindisi. The study included all patients diagnosed with growth hormone deficiency based on the results of at least 2 GH stimulation tests showing a pathological peak response (<8 ng/mL), and who underwent brain MRI with detailed evaluation of the hypothalamic-pituitary region.
Following diagnosis, all patients were started on standardized growth hormone replacement therapy, with dosages ranging from .025 to .035 mg/kg/day, in accordance with national guidelines. 12 A minimum follow-up period of 12 months after diagnosis was required for inclusion.
Characteristics of Growth Hormone Stimulation Tests (GHSTs)
All patients underwent at least 2 morning growth hormone stimulation tests, performed under standardized conditions.
The first GHST conducted in all cases was the Insulin Tolerance Test (ITT), which is considered the gold standard for the diagnosis of growth hormone deficiency and was utilized in accordance with current national and international guidelines.12,19 The ITT was carried out using a weight-based dose of intravenous insulin to induce hypoglycemia (target blood glucose <40 mg/dL), with close clinical monitoring throughout the procedure. 23
If the ITT produced a pathological response, defined as a GH peak below 8 ng/mL, a second GH stimulation test was subsequently performed using an alternative pharmacological stimulus, either arginine or clonidine, selected based on clinical feasibility and patient tolerance. In all cases, the choice between arginine and clonidine was determined by the pediatric endocrinologist managing the patient, taking into account the clinical characteristics and age of the child. The diagnostic cutoff value remained consistent across all tests, as it was standardized according to our national protocols.
Arginine was administered at a dose of .5 g/kg, while clonidine was dosed at 100 to 150 μg/m2, both adjusted for weight and body surface area. 24 In all cases, blood samples for GH measurement were collected at baseline (T0) and at 15, 30, 60, 90, and 120 minutes post-stimulus, for a total of 6 time points.
No patient received sex steroid priming prior to testing, as this is not part of the standard protocol at our center.
The interval between the 2 GHSTs for each patient was greater than 24 hours but less than 30 days, ensuring clinical and auxological stability (height, weight, and BMI) across tests.
A diagnosis of GHD was confirmed when both stimulation tests demonstrated subnormal GH responses (peak GH <8 ng/mL). 12 In cases of discordant results—that is, 1 positive and 1 negative test—the patients were monitored longitudinally. If subsequent auxological evaluation revealed a further decline in growth velocity or worsening of height percentile, a third GHST was performed, using one of the available stimuli (arginine, clonidine, or growth hormone-releasing hormone [GHRH] at 1 μg/kg) to confirm or exclude the diagnosis of GHD. 24
Data Collection and Analyzed Clinical Characteristics
We included in our study sample all patients under the age of 16 with a diagnosis of idiopathic GHD who were followed at our hospital. Patients with genetic syndromes (such as Noonan syndrome), despite having a confirmed GH deficiency, and those with non-idiopathic forms of GHD (eg, secondary to organic causes) were excluded.
Medical records of all enrolled patients were retrospectively reviewed in order to extract relevant clinical, auxological, biochemical, and radiological data collected during routine endocrinological evaluations. The following parameters were systematically retrieved: anthropometric measurements (weight, height, body mass index [BMI]), chronological age, bone age (as assessed by left-hand radiography using the Greulich and Pyle atlas), and longitudinal growth rate. Biochemical assessments included serum levels of (IGF-1). Detailed results of GHSTs were also collected, including type of test, stimulation agent used, peak GH concentrations, and timing of sample collection. Additionally, all available brain MRI reports were analyzed, with specific focus on the hypothalamic-pituitary region to identify structural abnormalities.
All data were entered into a standardized and anonymized digital database, which served as the foundation for subsequent statistical analysis. This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines to ensure transparency and methodological rigor in the presentation of observational data. All relevant components, including study design, participant selection, data collection methods, statistical analyses, and interpretation of results, were structured following the STROBE checklist. 25
To ensure data uniformity and comparability, auxological measurements and IGF-1 values were normalized by calculating corresponding SDS and percentiles based on age- and sex-specific reference data from national growth charts 26 and IGF-1 reference ranges. Patient sex and age at diagnosis were also recorded for subgroup analysis.
Neuroimaging findings were interpreted qualitatively based on established pathological features commonly associated with growth hormone deficiency. A standardized grading scale, was not applied; instead, abnormalities were defined according to deviations from normative values for pituitary size (height, width, and volume) 27 reported in the literature, with particular attention to age- and sex-specific reference ranges. 28 Pathological findings included reduced pituitary size, ectopic posterior pituitary, hypoplastic or interrupted pituitary stalk, and the presence of hypothalamic–pituitary tumors (eg, craniopharyngiomas, germinomas). All brain MRI scans were reviewed by a single senior radiologist with specific expertise in pediatric neuroradiology. In cases of uncertain or ambiguous findings, the images were discussed collegially with the pediatric endocrinology and radiology teams to reach a consensus diagnosis.
Ethical Considerations
Formal approval from an institutional ethics committee was deemed unnecessary for this retrospective study in accordance with the provisions set forth by the General Authorization to Process Personal Data for Scientific Research Purposes (Authorization No. 9/2014) issued by the relevant national data protection authority. This regulatory framework explicitly exempts retrospective research that relies solely on the analysis of previously collected, coded data from requiring formal ethical review, provided that such data do not permit the direct identification of individual subjects.
Despite this exemption, all clinical evaluations, diagnostic procedures, and therapeutic interventions performed within the cohort strictly adhered to established standards of good clinical practice. Standardized protocols routinely employed in the diagnosis and management of growth hormone deficiency were consistently followed to ensure the highest quality of patient care and data integrity.
The study was conducted in full compliance with the ethical principles outlined in the Declaration of Helsinki and all its subsequent amendments, ensuring respect for the rights, safety, and well-being of the subjects involved. Importantly, throughout the entire course of the investigation, no personally identifiable information was collected or stored, thereby guaranteeing the confidentiality and privacy of the patients.
Furthermore, prior to inclusion in the study, the parents or legal guardians of all pediatric participants were informed in detail about the aims, procedures, and potential implications of the research. After addressing any questions or concerns, they voluntarily provided written informed consent, authorizing the use of their child’s anonymized clinical data for research purposes. This process ensured compliance with ethical standards regarding informed consent and the protection of vulnerable populations.
Statistical Analysis
All statistical analyses were conducted using Python software (version 3.11). A 2-tailed P-value of less than .05 was considered indicative of statistical significance. Prior to group comparisons, the normality of continuous variables was assessed using the Shapiro–Wilk test. Depending on the distribution of the data, group comparisons for continuous variables were performed using either parametric one-way analysis of variance (ANOVA) or the non-parametric Kruskal–Wallis test when normality assumptions were not met.
To compare clinical and biochemical parameters between patients exhibiting abnormal findings on brain MRI and those with normal MRI results, independent samples T-tests were applied. The variables analyzed in these comparisons included age, height, height SDS, weight, weight SDS, serum concentrations of IGF-1 and its SDS, insulin-like growth factor-binding protein 3 (IGFBP-3) and its SDS, as well as peak growth hormone levels obtained from stimulation tests.
Results
A total of 84 children (56 males and 28 females) with a confirmed diagnosis of isolated growth hormone deficiency were included in the analysis. The mean chronological age at diagnosis was 7.92 ± 3.27 years, and the mean bone age, assessed by the Greulich and Pyle method, was 6.56 ± 3.23 years.
Anthropometric data at diagnosis showed a mean height SDS of −2.64 ± .63 and weight SDS of −1.75 ± .95. Serum IGF-1 levels had a mean value of 158.33 ± 81.45 ng/mL, with a corresponding IGF-1 SDS of −.87 ± .70. More information are reported in Table 1.
General Characteristics of Our Study Population.

Two GH stimulation tests were performed for each patient. The first, using the insulin tolerance test, showed a mean GH peak (Peak 1) of 5.21 ± 1.93 ng/mL. The second test, conducted using ITT, arginine, or clonidine (based on the patient’s characteristics), yielded a mean GH peak (Peak 2) of 6.38 ± 1.45 ng/mL. All patients met the criteria for IGHD with GH peaks <8 ng/mL in both tests, and none were diagnosed with multiple pituitary hormone deficiency (MPHD).
Brain MRI, focused on the hypothalamic-pituitary axis, was performed in all patients. Anomalies were detected in 23 of 84 children (27.4%), while the remaining 61 (72.6%) showed normal imaging. The anomalies identified included pituitary hypoplasia (n = 20) and ectopic posterior pituitary (n = 3). In all cases, aside from short stature and the clinical suspicion of GHD, no associated symptoms were present.
When comparing patients with and without MRI abnormalities, those with anomalies had a significantly lower chronological age (6.78 ± 2.72 vs 8.34 ± 3.38 years, P = .038), significantly lower height SDS (−2.83 ± .58 vs −2.56 ± .64, P = .032), and lower IGF-1 SDS (−1.07 ± .66 vs −.78 ± .70, P = .041). No significant differences were found for BMI SDS, GH peak values, or birth-related variables. More information are reported in Table 2.
Comparison Between Patients With and Without MRI Abnormalities.

Statistically significant p-values are highlighted in bold.
Before performing the multivariate logistic regression, we conducted univariate logistic regression analyses for each variable to assess their individual association with the presence of MRI abnormalities.
The selection of predictors for the regression analyses was based on both clinical relevance and the results of preliminary univariate comparisons. Chronological age, height SDS, and IGF-1 SDS were chosen as candidate variables given their known associations with structural abnormalities of the hypothalamic-pituitary axis in patients with GHD, as reported in previous literature. Additionally, these variables showed statistically significant differences between patients with and without MRI abnormalities in our cohort.
Chronological age at diagnosis (OR: 1.24, 95% CI: 1.01-1.52,), height SDS (OR: 1.89, 95% CI: 1.05-3.40), and IGF-1 SDS (OR: 1.67, 95% CI: 1.02-2.74) were all significantly associated with abnormal MRI findings. Subsequently, a multivariate logistic regression model including these 3 variables confirmed their independent predictive value: lower age (OR: 1.31, 95% CI: 1.04-1.66, P = .024), lower height SDS (OR: 2.15, 95% CI: 1.12-4.15, P = .021), and lower IGF-1 SDS (OR: 1.81, 95% CI: 1.03-3.17, P = .038) remained significantly associated with MRI abnormalities.
To assess the diagnostic performance of these variables, ROC curve analyses were performed. For chronological age, the optimal cut-off to predict MRI anomalies was <7.1 years, with an area under the curve (AUC) of 0.69 (95% CI: .57-.80, standard error .058, P = .005), sensitivity 74%, and specificity 63%. For height SDS, a cut-off of <−2.7 yielded an AUC of 0.67 (95% CI: .55-.79, SE: .061, P = .008), with 70% sensitivity and 61% specificity. For IGF-1 SDS, the best cut-off was <−.9, showing an AUC of .65 (95% CI: .53-.77, SE: .062, P = .015), sensitivity 68%, and specificity 59%. These findings suggest that younger age, more severe height impairment, and lower IGF-1 levels at diagnosis may help identify patients at higher risk for underlying hypothalamic–pituitary structural abnormalities. More information are reported in Table 3.
ROC Curve Analysis with Sensitivity and Specificity for Each Variable.
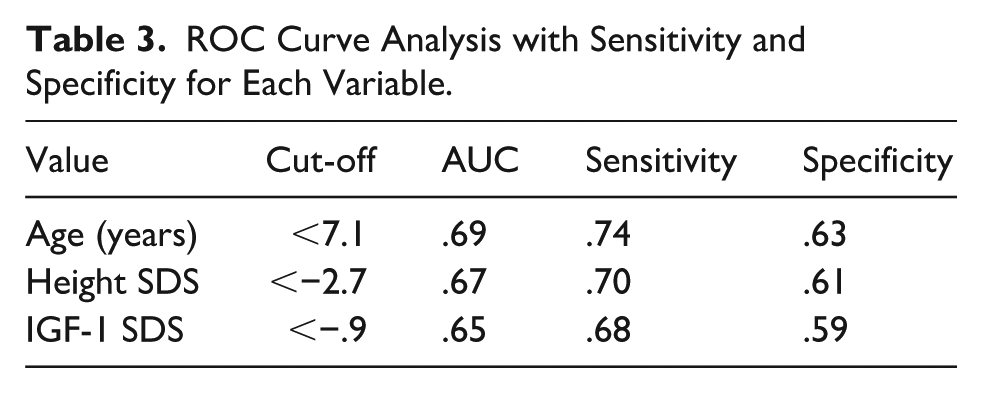
In summary, based on the results from our cohort of patients with GHD, a younger age at diagnosis, markedly reduced height, and significantly low IGF-1 levels relative to age- and sex-specific reference ranges demonstrated fair sensitivity and specificity in identifying patients with abnormalities of the hypothalamic-pituitary region.
Discussion
Our retrospective study, conducted on a cohort of children diagnosed with growth hormone deficiency, confirmed that the majority of cases are idiopathic in nature, consistent with the current definition of GHD. 29 With regard to the analysis of baseline laboratory data, lower growth velocity and height standard deviation scores at the time of diagnosis, as well as reduced IGF-1 levels and IGF-1 SDS, were found to be associated with the presence of abnormalities on brain MRI. While the identified predictors showed statistically significant associations with MRI abnormalities, the ROC curve analyses revealed only modest discriminatory capabilities, with AUC values ranging from .65 to .69. These values, although indicative of some predictive value, fall below the commonly accepted threshold of .70 for acceptable diagnostic accuracy. Therefore, the predictive performance of these variables should be interpreted with caution. Rather than suggesting strong clinical utility, these findings may serve as preliminary indicators to guide further investigation or closer imaging surveillance in selected patients. Future studies with larger cohorts and additional predictive markers are warranted to improve the accuracy and clinical applicability of MRI abnormality prediction in children with idiopathic GHD.
The use of brain MRI in patients diagnosed with growth hormone deficiency remains a matter of debate. 30 As previously discussed, although current clinical guidelines consistently recommend neuroimaging of the hypothalamic-pituitary region in newly diagnosed GHD cases, 31 it is well recognized that the majority of GHD forms are idiopathic in nature and are frequently associated with normal brain MRI findings, without structural abnormalities in the hypothalamic-pituitary axis. 32
Although it is commonly assumed that severe growth hormone deficiencies—characterized by low GH responses during stimulation tests—are more frequently associated with structural abnormalities of the hypothalamic-pituitary region, evidence indicates that GH peak levels are not reliably correlated with the presence of intracranial anomalies. 33
The findings of our retrospective study align with previously published data, as no correlation was observed in our cohort of GHD patients between peak GH levels during stimulation tests and the presence of hypothalamic-pituitary abnormalities on subsequent neuroimaging. However, some authors have reported an association between brain MRI abnormalities and GH peak levels, noting a higher incidence of intracranial anomalies in patients with a GH peak below 3 ng/mL. 34 The inconsistency of these findings across studies further emphasizes the need to establish clear, evidence-based criteria for the use of radiological investigations in patients with GHD. Indeed, although the GH peak is valuable for diagnosing GHD, it neither predicts the type of deficiency nor the response to growth hormone therapy. 11 This is despite the fact that severe anatomical alterations of the hypothalamic-pituitary region may cause pituitary parenchymal destruction and GH levels approaching zero. Therefore, prospective studies are warranted to clarify this potential correlation.
The widespread use of radiological investigations, even in the absence of strict clinical indications, may have a negative impact on the healthcare system in terms of increased costs. 35 Moreover, it can lead to a higher detection rate of incidental findings that are not clinically significant, often resulting in additional diagnostic procedures and follow-up evaluations that may ultimately prove unnecessary.35,36 A more individualized diagnostic approach would therefore be desirable, restricting the use of radiological investigations to cases in which there is a strong clinical suspicion of an underlying organic disease or anatomical abnormality. However, specific criteria to reliably identify this subset of patients have yet to be defined. As a result, brain MRI is currently recommended for all patients following a confirmed diagnosis of growth hormone deficiency. It should also be acknowledged that the use of brain MRI, beyond its diagnostic role in growth hormone deficiency, 37 often holds significant prognostic value—particularly when complex hypothalamic-pituitary anatomical abnormalities are identified. Such findings can support the classification of GHD as permanent from the time of diagnosis. 38
One of the most recent and comprehensive meta-analyses on the topic 21 evaluated data from 32 clinical studies involving a total of 39 060 children. The findings confirmed that patients with isolated GHD often exhibit normal neuroimaging studies. The cumulative incidence of hypothalamic-pituitary abnormalities was reported to be 58%, with a significantly higher prevalence among patients presenting with multiple pituitary hormone deficiencies and in those whose GH peak during stimulation tests was ≤5 μg/L.The discrepancy between the rate of abnormalities reported by the authors and our findings may be attributed to the different cut-off values used for the diagnosis of GHD; specifically, our study included patients with a GH peak below 8 ng/mL. Additionally, our cohort consisted exclusively of Caucasian patients, and it cannot be excluded that inclusion of individuals from different ethnic backgrounds might yield divergent results.
Another potential indicator for hypothalamic-pituitary abnormalities warranting evaluation by brain MRI is low basal IGF-1 levels. A recent study 39 demonstrated a correlation between baseline IGF-1 levels and patient height—both expressed as standard deviation scores—and the frequency of pituitary abnormalities, specifically reduced pituitary height. Our statistical analysis revealed a similar association, linking the incidence of pituitary structural alterations to hormonal IGF-1 levels as well as to various auxological parameters such as height and growth velocity. It is therefore plausible to hypothesize that brain MRI should be recommended based on markedly reduced basal hormonal values rather than solely on GH peak responses during stimulation tests.
Our statistical analysis did not reveal a significant correlation between peak GH levels during growth hormone stimulation tests and the incidence of hypothalamic-pituitary abnormalities. Although organic lesions may cause destruction or hypofunction of somatotroph cells, resulting in GH levels approaching zero, there is generally no consistent correlation between GH levels and the severity of GHD, either in terms of etiopathogenesis or temporal stability. Indeed, according to current clinical guidelines, both near-zero and borderline GH responses during GHSTs are considered equivalent, as both fall below the diagnostic threshold of 8 ng/mL. Nevertheless, some studies in the literature have reported associations between pituitary gland dimensions and the dynamic response of growth hormone. 40
This study has several limitations that should be acknowledged. First and foremost, it is a retrospective, single-center analysis, which inherently limits the generalizability of the findings. Prospective studies are warranted to confirm and validate our results in broader and more diverse populations. Additionally, the study cohort consisted exclusively of Caucasian patients from Southern Italy; as such, the findings may not be generalizable to the entire Italian population, nor to global pediatric populations with differing genetic, environmental, or socio demographic backgrounds. It is also important to note that none of the patients included in this study had a diagnosis of organic GHD due to neoplastic causes, limiting the applicability of our conclusions to that specific subgroup of children.
Nonetheless, this study benefits from a relatively large and well-characterized cohort of children with idiopathic GHD, all of whom were diagnosed and managed in accordance with national clinical guidelines. Our analysis identified a significant association between certain baseline auxological and hormonal parameters and the presence of structural abnormalities on brain MRI. Although the individual predictive performance of these variables was limited, they may still aid clinicians in identifying patients who could benefit from early or prioritized neuroimaging, particularly in settings where MRI availability is limited. These findings support a more individualized approach to neuroimaging, rather than recommending routine MRI for all patients with idiopathic GHD.
Moreover, our results highlight the potential for developing more refined and clinically useful prediction models through the integration of additional clinical, biochemical, or genetic markers. If confirmed by future prospective, multicenter studies with larger populations, these findings could assist pediatric endocrinologists in optimizing neuroimaging strategies, improving diagnostic efficiency and minimizing unnecessary procedures in children with GHD.
Conclusion
Our retrospective analysis of a pediatric cohort diagnosed with growth hormone deficiency revealed that reduced height and growth velocity, expressed as standard deviation scores, alongside decreased absolute IGF-1 levels and corresponding SDS, are significantly associated with hypothalamic-pituitary abnormalities identified via brain MRI. In contrast, GH peak values obtained during stimulation tests did not correlate with intracranial anomalies. Clinically, these findings suggest that anthropometric measures and IGF-1 levels may serve as more reliable indicators for identifying patients at risk of structural hypothalamic-pituitary abnormalities than GH peak alone. Nevertheless, brain MRI remains an indispensable diagnostic modality for all patients with GHD to ensure timely detection of potential intracranial pathology. Future prospective studies are essential to refine risk stratification criteria, which could help optimize the use of neuroimaging by identifying patients who might safely avoid unnecessary radiological evaluations, thereby reducing costs and procedural burdens.
Footnotes
Acknowledgements
We sincerely thank all the pediatric patients and their families who participated in the Pediatric Endocrinology Unit at Perrino Hospital.
Consent to Participate
Informed consent was obtained from the parents or legal guardians of all participants included in the study. The study protocol was conducted in accordance with the Declaration of Helsinki.
Consent for Publication
All authors have given their consent for publication. Additionally, parental consent for publication of anonymized data was obtained.
Author Contributions
The study design, data analysis, and manuscript drafting were performed by Giorgio Sodero. All other authors contributed to data collection and critically revised the manuscript for intellectual content. All authors approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
