Abstract
Neonatal pneumonia often leads to respiratory failure (RF), with severity varying widely. This retrospective study of 107 neonates examined predictors of severe RF, focusing on prenatal, perinatal, and hematological factors. Neonates were grouped by RF severity: mild, moderate, and severe, based on respiratory support needs. Key findings revealed that early gestational age, low birth weight, and maternal fever significantly increased the likelihood of severe RF. Hematological markers, including elevated white blood cell counts, low hemoglobin, and high C-reactive protein, also emerged as strong predictors. The CRP-to-platelet ratio showed moderate promise as a diagnostic tool for identifying at-risk neonates. These results highlight the importance of early risk assessment in neonatal pneumonia, offering clinicians actionable insights to optimize care. This study underscores the need for targeted interventions to improve outcomes in this vulnerable population, aligning with efforts to reduce neonatal morbidity and mortality.
Introduction
Neonatal pneumonia remains a leading cause of morbidity and mortality in the first 28 days of life, contributing significantly to the global burden of neonatal deaths. Globally, neonatal pneumonia accounts for ~10% of under-5 mortality, with an estimated 750 000 annual deaths in low- and middle-income countries, where access to advanced neonatal care is often limited. 1 In Vietnam, neonatal pneumonia is a significant health challenge, with studies reporting incidence rates of 12% to 18% among hospitalized neonates and mortality rates ranging from 4% to 10% depending on the availability of intensive care. 2 Respiratory failure (RF), a critical complication, varies in severity from mild hypoxemia requiring oxygen therapy to severe cases necessitating invasive mechanical ventilation. 3 Understanding the factors driving this progression is essential for improving neonatal outcomes.
Prenatal and perinatal factors, such as gestational age, birth weight, and maternal infections, have been implicated in neonatal respiratory distress. 4 Premature and low-birth-weight infants are particularly vulnerable due to immature immune systems and underdeveloped lungs. 5 Maternal fever, for instance, may signal intrauterine inflammation, potentially exacerbating lung injury via cytokine release. 6 Hematological biomarkers, including white blood cell (WBC) counts, hemoglobin levels, and C-reactive protein (CRP), reflect systemic inflammation and oxygen transport capacity, both critical in determining pneumonia severity. 7 Recent studies have explored composite indices like the CRP-to-platelet ratio (CPR) as prognostic tools in neonatal sepsis, but its role as a supplementary predictor in assessing RF severity remains underexplored due to its moderate diagnostic accuracy. 8
Given the significant morbidity and mortality associated with neonatal pneumonia and its progression to severe RF, identifying reliable predictors is crucial for early risk stratification and timely intervention. This study aims to address this gap by analyzing prenatal, perinatal, and hematological factors associated with severe RF in neonates with pneumonia at a regional hospital in Vietnam. We hypothesize that a combination of clinical and laboratory markers, with CPR as a supplementary tool, can effectively stratify risk, enabling targeted strategies to mitigate severe RF and improve outcomes in this vulnerable population.
Material and Methods
Study Design and Population
This retrospective cohort study was conducted at a region hospital in Vietnam between May 2023 and June 2024. The hospital features a Level III neonatal intensive care unit (NICU), equipped to provide advanced respiratory support, including continuous positive airway pressure (CPAP) and invasive mechanical ventilation, as well as comprehensive critical care for neonates. As a tertiary referral center for the South Central Coast region, the hospital receives referrals from surrounding regional hospitals, serving a diverse population of neonates with complicated medical needs. The study population comprised all 107 neonates diagnosed with pneumonia within the first 28 days of life during the study period. Pneumonia was defined based on clinical signs (eg, tachypnea, cyanosis) and laboratory evidence of infection (eg, elevated CRP), with chest X-ray findings (eg, pulmonary infiltrates, consolidation, or air bronchograms) used to support the diagnosis when present; radiographic evidence was not strictly required for inclusion. Neonates were followed from the time of pneumonia diagnosis through their hospital course to assess the development and severity of respiratory failure (RF) as the primary outcome, establishing a temporal relationship between exposure variables (prenatal, perinatal, and hematological factors) and the outcome, consistent with a retrospective cohort design. Neonates were excluded if they had congenital anomalies affecting the respiratory system, identified through clinical evaluation, chest X-ray findings, electrocardiographic screening to rule out congenital heart diseases, and medical record review for documented congenital conditions. Neonates were also excluded if they lacked complete medical records or had pneumonia secondary to non-infectious causes (eg, meconium aspiration).
Data Collection
Data were extracted from medical records using a standardized case report form (CRF) to ensure consistency in data collection. Data were collected by trained pediatric research staff, with variables defined according to clinical guidelines from the Vietnam Ministry of Health and the study protocol to standardize measurements (eg, gestational age [weeks], birth weight [g], laboratory values [specified units]). Quality control was performed by having a second researcher independently double-check the extracted data for accuracy. Data were categorized into 3 domains: demographic/clinical characteristics, prenatal/perinatal risk factors, and hematological biomarkers. Demographic variables included gestational age (weeks), birth weight (g), and sex. Clinical presentation was assessed through symptoms such as fever, tachypnea, bradypnea, and cyanosis, as well as respiratory support modalities (oxygen therapy, continuous positive airway pressure [CPAP], or invasive ventilation). Prenatal factors encompassed maternal conditions (eg, urinary tract infection, fever ≥38 °C), while perinatal factors included mode of delivery, APGAR scores at 5 minutes, and rupture of membranes. Hematological biomarkers comprised WBC count (k/µL), neutrophil and lymphocyte counts (k/µL), hemoglobin (g/L), platelet count (k/µL), and CRP (mg/L). Chest X-ray findings were reviewed by a pediatric radiologist blinded to clinical outcomes.
Classification of Respiratory Failure
RF severity was classified based on guidelines from the World Health Organization (WHO) 9 :
Mild RF: The child has a respiratory rate of 60 to 90 breaths/minute, accompanied by the absence of chest indrawing and no grunting.
Moderate RF: Presence of one of the following:
Respiratory rate of 60 to 90 breaths/minute with chest indrawing and/or grunting.
Respiratory rate >90 breaths/minute with the absence of chest indrawing and no grunting.
Severe RF: Presence of one of the following:
Respiratory rate >90 breaths/minute with chest indrawing and/or grunting.
Respiratory rate <20 breaths/minute; or not breathing at all, even when stimulated; or gasping.
Statistical Analysis
Continuous variables were reported as means ± standard deviations (SD) or medians (95% confidence intervals [CI]) and compared using ANOVA or Kruskal-Wallis tests, as appropriate. Categorical variables were analyzed with chi-square or Fisher’s exact tests. Univariate logistic regression identified factors associated with severe RF, with significant variables (P < .05) entered into a multivariate model to calculate odds ratios (ORs) and 95% CIs. The CPR’s predictive value was assessed via ROC analysis, yielding area under the curve (AUC) with 95% confidence intervals, sensitivity, and specificity. The CPR was calculated as CRP (mg/L) divided by platelet count (k/µL), using laboratory values obtained on admission. The cut-off point was determined using Youden’s Index to maximize the sum of sensitivity and specificity. The ROC analysis was performed using IBM SPSS Statistics 20.0 (IBM, New York, NY, USA), consistent with other statistical analyses. Statistical significance was set at P < .05. Analyses were conducted using IBM SPSS Statistics 20.0.
Ethical Approval and Informed Consent
Written informed consent was obtained from the patient’s parent(s) or guardian(s). The study protocol was approved by the Ethics Committee at Hue University of Medicine and Pharmacy (IRB #2023-045).
Results
Clinical Characteristics
Table 1 summarizes the demographic, clinical, and laboratory characteristics of the 107 neonates across mild, moderate, and severe RF groups. Gestational age differed significantly among groups (P = .018), with the severe RF group having a mean of 36 ± 3 weeks, compared to 35 ± 3 weeks for mild and 34 ± 3 weeks for moderate RF groups; this trend may reflect a subset of severe RF cases influenced by resistant bacterial infections rather than solely prematurity-related fetal pneumonia. Birth weight did not differ significantly among groups (mild: 2561 ± 794 g; moderate: 2302 ± 550 g; severe: 2618 ± 775 g; P = .626). Hemoglobin levels were significantly lower in the severe RF group (mean 151 ± 31 g/L) compared to the mild (mean 171 ± 26 g/L) and moderate (mean 164 ± 24 g/L) RF groups (P = .018). CRP levels showed no significant differences across groups (mild: 1.4 mg/L, IQR 0.6-22.5; moderate: 1.7 mg/L, IQR 0.9-21.9; severe: 1.9 mg/L, IQR 1.0-65.7; P = .170). Radiographic findings consistent with pneumonia were observed in 84.8% of mild, 95.7% of moderate, and 100% of severe RF cases, with significant differences across groups (P = .041). Respiratory support differed markedly: 96.4% of severe RF cases required invasive ventilation versus 0% in mild RF (P < .001).
Clinical Characteristics of Neonatal Pneumonia Cases.

Predictors of Severe RF
Table 2 indicates the factors associated with severe RF. Perinatal predictors included gestational age (GA) <32 weeks (OR 8.45, 95% CI 1.02-88.19, P = .048), birth weight <1500 g (OR 4.12, 95% CI 1.15-14.76, P = .030), and maternal fever ≥38 °C (OR 3.67, 95% CI 1.09-12.38, P = .036). Hematologic predictors included white blood cell count >25 k/µL (OR 5.32, 95% CI 1.44-19.67, P = .012), hemoglobin <135 g/L (OR 2.89, 95% CI 1.03-8.11, P = .044), and C-reactive protein (CRP) >10 mg/L (OR 4.78, 95% CI 1.33-17.19, P = .016). Platelet count <150 k/µL was significant in univariate analysis (OR 4.04, P = .034) but not multivariate (P > .05).
Factors Associated With Severe Respiratory Failure.

Diagnostic Value of CPR
Figure 1 present the diagnostic performance of the CPR for predicting severe RF. The area under the curve (AUC) was 0.644 (95% CI 0.532-0.756), indicating fair predictive accuracy. The optimal CPR cut-off value of 0.016 was determined using Youden’s Index, yielding a sensitivity of 53.6% and specificity of 78.5%. The positive predictive value (PPV) was 44.7%, and the negative predictive value (NPV) was 84.2%.
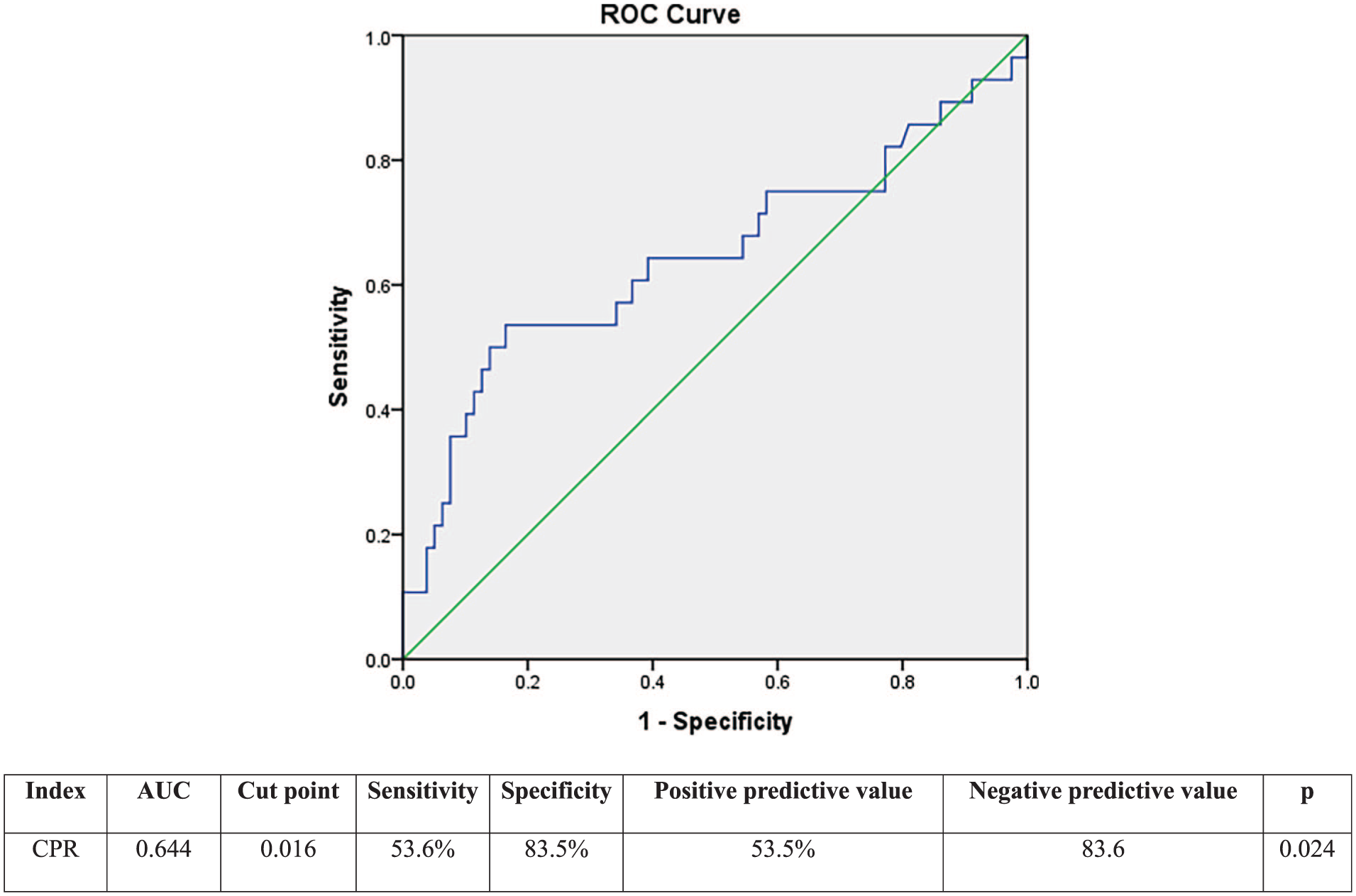
ROC curve showing the predictive value of the CRP-to-platelet ratio for severe respiratory failure.
Discussion
This study delineates key predictors of severe RF in neonatal pneumonia, emphasizing the roles of prematurity, maternal inflammation, and hematological dysregulation. Gestational age <32 weeks and birth weight <1500 g emerged as potent risk factors, aligning with prior research linking preterm birth to underdeveloped alveoli and surfactant deficiency. 10 These neonates are predisposed to severe infections due to immature immune responses. 11 Maternal fever ≥38 °C, a proxy for chorioamnionitis or systemic infection, likely exacerbates lung injury via inflammatory cascades, as noted by Redline. 6
Hematological biomarkers further refined risk stratification. Elevated WBC (>25 k/µL) and CRP (>10 mg/L) indicate robust inflammatory responses, consistent with findings in neonatal sepsis. Although CRP levels showed no significant distributional differences across groups in univariate analysis (P = .17), their significance in the regression model (OR 4.78, 95% CI 1.33-17.19, P = .016) likely reflects their interaction with other predictors, such as gestational age and maternal fever, in the multivariate context. Low hemoglobin (<135 g/L) may impair oxygen delivery, compounding respiratory distress—a mechanism supported by studies on anemia in critical illness. 12 The CPR’s fair predictive value (AUC 0.644, 95% CI 0.532-0.756) for severe RF, with a prevalence of 26.2% in our cohort, mirrors its utility in differentiating neonatal late-onset sepsis in neonates with pneumonia, 8 reflecting the interplay between inflammation and thrombocytopenia in severe infections. 13
Clinical implications are significant for improving neonatal outcomes in resource-constrained settings like our hospital. The finding that 100% of neonates with severe RF exhibited abnormal chest X-rays (P = .041), compared to 84.8% in mild and 95.7% in moderate RF groups, underscores the importance of radiographic evaluation in confirming severe pneumonia and guiding escalation of respiratory support. Neonates with these risk factors warrant close monitoring and early escalation of respiratory support. Elevated WBC (>25 k/µL), CRP (>10 mg/L), and low hemoglobin (<135 g/L) can guide the initiation of targeted interventions, such as timely antibiotic therapy or blood transfusions for severe anemia, to mitigate progression to severe RF. Although birth weight showed no significant distributional difference across groups in Table 1 (P = .109), its significance in the regression model (OR 4.12, 95% CI 1.15-14.76, P = .030) suggests that low birth weight (<1500 g) contributes to severe RF risk when adjusted for other factors, such as gestational age. The CPR, calculable from routine blood tests, offers a practical adjunct to risk assessment, though its modest sensitivity (53.6%) suggests it should complement, not replace, clinical judgment. Compared to studies like Kushnareva et al, which focused on microbial etiology, our emphasis on host factors provides a broader prognostic framework. 14
Limitations include the retrospective design, which may introduce selection bias, and the inclusion of all 107 eligible neonates during the study period without a formal sample size calculation, potentially limiting statistical power. Despite electrocardiographic screening, potential misclassification of subtle congenital anomalies affecting the respiratory system could have occurred, as not all anomalies may be detectable through routine clinical and radiographic evaluations. The initial lack of detail on data collection methodology, now clarified in the Methods section with the use of a standardized case report form and quality control by double-checking, may have limited transparency in the original submission. The absence of a priori sample size determination, due to the exploratory nature of the study and the relatively low incidence of neonatal pneumonia, may contribute to the wide confidence intervals observed (eg, WBC OR 2.88-166.44). The wide confidence intervals observed in the regression analysis (eg, GA <32 weeks: 1.02-88.19) further suggest limited statistical power, which may affect the precision of the estimated odds ratios. Additionally, including both CRP and platelet count as separate predictors in the regression model may introduce multicollinearity, and future studies could explore CPR as a composite predictor to enhance prognostic utility. Unmeasured confounders, such as specific pathogens or antibiotic timing, could also influence outcomes. 4 Future research should validate these predictors in larger, multicenter cohorts and explore microbial contributions to RF severity.
Conclusion
Severe RF in neonatal pneumonia is driven by prematurity (gestational age <32 weeks, birth weight <1500 g), maternal fever, and hematological markers (WBC >25 k/µL, hemoglobin <135 g/L, CRP >10 mg/L). The CPR offers moderate diagnostic utility, enhancing risk stratification. Early recognition of these factors could guide timely interventions, potentially reducing morbidity and mortality in this vulnerable population.
Footnotes
Acknowledgements
We would like to acknowledge all individuals and the hospital involved in the implementation of the survey.
Ethical Consideration
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of Hue University of Medicine and Pharmacy (approval date: January 21, 2023, approval number: 107/NCKH-DHY). The personal information of the patient (name, phone number, address) was not collected. Other data was anonymized and maintained with confidentiality.
Author Contributions
S.B.-B.-B. designed and supervised the entire project. T.N.-T.-H. designed the study, contributed data analysis, and participated in writing the manuscript. All authors read, commented on, and approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
