Abstract
Objectives:
This study investigated the clinical features, treatment approaches, and outcomes of severe Bacille Calmette-Guérin (BCG) vaccine complications in Chinese children.
Introduction:
BCG is the only available vaccine for tuberculosis (TB) prevention but can cause serious complications such as local abscesses, lymphadenitis, and disseminated BCG. Research on their management in China is limited.
Methods:
This observational study reviewed 95 children with severe BCG-related complications treated at a tuberculosis-specialized hospital in Guangdong, China (2016-2020). Of these, 33 (34.7%) had injection site reactions, 55 (57.9%) developed lymphadenitis with 31 suppurative cases, and 7 (7.4%) had disseminated BCG. All deep abscesses and suppurative lymphadenitis were successfully treated with surgery and anti-tubercular therapy. Two disseminated BCG cases had primary immunodeficiency, and 1 of them died.
Conclusion:
Most local reactions respond to conservative treatment. Surgery combined with anti-tubercular therapy is effective for suppurative lymphadenitis. Early immunological evaluation is essential for disseminated disease.
Introduction
The Bacille Calmette-Guérin (BCG) vaccine, a live-attenuated strain of Mycobacterium bovis, has been widely used and is considered one of the safest vaccines for tuberculosis (TB) since its introduction in 1921. 1 BCG vaccination significantly prevents miliary pulmonary TB and TB meningitis, especially in children. 2 The vaccine shows 78% efficacy against disseminated TB and 64% efficacy against TB meningitis. Severe tuberculosis incidence can be decreased by 92%.2,3 The BCG vaccine offers protective efficacy against leprosy and non-tuberculous mycobacteria. 4
Each year, approximately 100 000 newborns in Guangzhou, China, are vaccinated with BCG. The commonly utilized strain, D2PB302, originates from the Statens Serum Institute Copenhagen strain 823 and exhibits genetic variations compared to the internationally employed Danish 823 strain. 5 A common vaccination response involves redness or a small lump at the injection site, potentially forming an ulcer under 1 cm in diameter within 2 to 3 weeks. The ulcer usually heals with a small flat scar within 2 to 3 months. 6 Systemic reactions are uncommon, and axillary lymph nodes under 1 cm on the injection side are deemed a normal response. 7
Severe BCG-related adverse reactions are typically categorized as local or systemic events. 8 Local reactions encompass injection site reactions and lymphadenitis, whereas systemic reactions mainly present as disseminated BCG disease.8,9 These adverse reactions have been associated with the specific strain of BCG administered. 10 The current incidence of severe BCG-induced adverse reactions in China is reported to be 48.86 per million, which is lower than the incidence reported by the WHO’s adverse event surveillance system in the Western Pacific region. 11
There is a lack of international consensus on managing BCG-related adverse events, and research on severe BCG-induced reactions in China is limited. This study presents our experience with severe reactions in children referred to a tuberculosis treatment center in Guangdong province, China.
Methods
This study is a retrospective observational study. The study population included children who presented with severe BCG-related complications, such as injection site abscesses, regional lymphadenitis, or disseminated BCG. The study encompassed all children experiencing severe BCG vaccination complications from January 2016 to December 2020 at the Guangzhou TB Prevention & Treatment Center (Guangzhou Chest Hospital), Guangdong, China. Approximately 500,000 infants in Guangzhou were administered the BCG vaccine during this time. BCG vaccination is typically administered within the first day of life in birth hospitals, although conditions such as neonatal pneumonia or pathological jaundice may cause delays. BCG related complications in children are usually treated by general pediatricians, with more severe cases referred to our hospital.
Children were identified via referral letters from general pediatricians or infectious disease specialists, and their medical records and laboratory data were reviewed. Children initially referred to other specialties, such as general or orthopedic surgery, and later referred to our clinic were also included in the study. Information on baseline demographics, presentation age, symptoms, complication types, microbiological and radiological findings, intervention methods, and outcomes was collected using a structured format. Inclusion criteria were a confirmed diagnosis based on clinical, histological, and microbiological findings. Patients with incomplete medical records or unclear diagnoses were excluded from the study. Also, children with BCG site reactions due to Kawasaki disease were excluded. Patient records were examined for any history of illnesses indicative of immunodeficiency, such as severe combined immunodeficiency (SCID), chronic granulomatous disease (CGD), Mendelian susceptibility to mycobacterial disease (MSMD), HIV infection, and other cell-mediated immune deficiencies.
Our study categorized severe complications as injection site reactions, lymphadenitis, and disseminated BCG disease. Injection site adverse reactions were generally characterized by local abscesses or ulcers larger than 1.0 cm, requiring more than 12 weeks to heal. These reactions were further classified into 4 types: ulceration, lichenoid lesions, granulation hyperplasia, and deep abscesses, as shown in Figure 1.

Severe adverse reactions in injection site were classified as 4 types. (A) Ulceration. (B) Lichenoid lesions. (C) Granulation hyperplasia. (D) Deep abscesses.
BCG lymphadenitis is characterized by lymphadenopathy exceeding 1.0 cm on the same side as the injection site, potentially with suppuration, impacting the ipsilateral axillary, supraclavicular, or cervical nodes. In our study, BCG adenitis was diagnosed based on a combination of clinical, microbiological, and molecular findings. These included a history of recent BCG vaccination, the absence of pulmonary or systemic manifestations typical of tuberculosis (TB) disease, and culture results confirming the presence of the Mycobacterium bovis BCG strain, characterized by resistance to pyrazinamide (PZA), a hallmark feature of M. bovis strains. Additionally, molecular biology results positive for Mycobacterium tuberculosis complex (MTBC) further supported the diagnosis. Systemic BCG infection was defined by the presence of miliary tuberculosis (clinical presentation consistent with active tuberculosis associated to a typical miliary pattern on chest imaging), hepatitis, nephritis, lymphocytic meningitis, arthritis, disseminated lymphadenopathy or osteomyelitis, after BCG vaccination, responding to anti-tuberculosis treatment, and with no alternative diagnosis. Microbiological (positivity for M. tuberculosis complex by culture or by nucleic acid-based methods) and/or histopathological evidence of mycobacterial infection (ie, caseating granulomas in biopsy specimens) supported the diagnosis. 12 Molecular biology techniques (MBT) were employed using the QIAamp DNA Micro Kit (Hugobiotech, Beijing, China) to extract DNA from pus or tissue samples. Metagenomic sequencing (mNGS) involved preparing DNA libraries with the QIAseq Ultralow Input Library Kit and sequencing them on the Illumina NextSeq 550 platform (SE75bp). For targeted next-generation sequencing (tNGS), DNA libraries were constructed with the General Kit for Identification and Drug Resistance Gene Detection of MTBC (Hugobiotech, Beijing, China). Sequencing data were analyzed against a curated database containing Mycobacterium tuberculosis complex (MTBC) species and resistance genes. This analysis successfully identified M. bovis and its associated resistance gene (pncA).
Treatment methods encompassed conservative app-roaches such as watchful waiting, the use of topical or systemic medications, and surgical interventions. Patient outcomes were classified as complete recovery (notable scarring at the injection or surgical site), clinical improvement (organ lesions resolved), or death.
The Medical Ethics Committee of Guangzhou Chest Hospital approved our study (approval no. KYSB-2024-056). Informed consent was obtained directly from the children’s families or legal guardians. Written informed consent for publication was obtained retrospectively from the legally authorized representatives of all subjects involved in this study.
Statistical Analysis
Statistical analysis, encompassing both descriptive and inferential statistics, was conducted using IBM SPSS version 22.0. Normality was assessed using the Shapiro-Wilk test and normal Q-Q plots. Data satisfying the conditions of normality were presented as means and standard deviation (±SD). Data with a skewed distribution was characterized using the median and interquartile range (IQR). The Wilcoxon rank-sum test (Mann-Whitney U test) or Kruskal-Wallis test was used to compare non-normally distributed data. Categorical variables were analyzed using the chi-square test or, when appropriate, Fisher’s exact test. A P-value less than .05 was deemed statistically significant.
Results
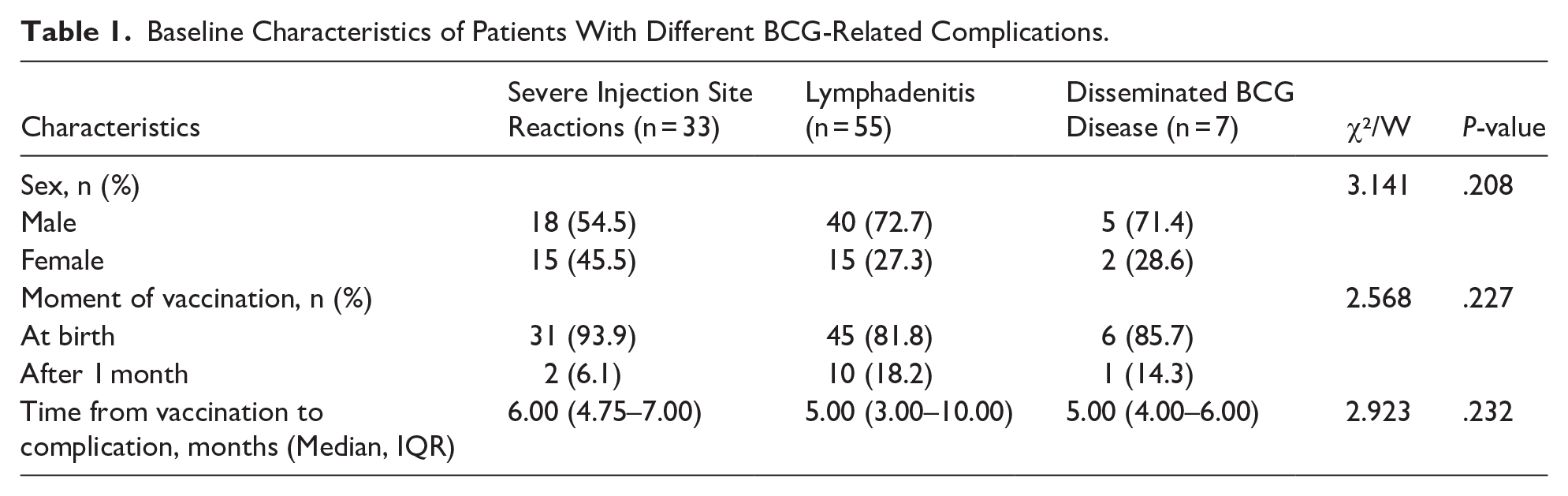
A total of 95 patients with severe reactions to the BCG vaccine were identified. The median age at diagnosis was 6.00 months (IQR 4.00-11.00), with 66.3% (63) of the patients being male. Among them, 83 individuals (87.4%) were vaccinated at birth, with a median time to adverse event of 6.00 months (IQR 4.00-10.00). Among the adverse events, 33 patients (34.7%) developed severe injection site reactions, 55 (57.9%) experienced lymphadenitis, and 7 patients (9.4%) presented with disseminated BCG disease. We compared the characteristics of patients across the severe injection site reactions group, lymphadenitis group, and disseminated BCG disease group. Our analysis indicated no statistically significant differences among the 3 groups concerning the timing of vaccination (at birth vs after 1 month), the median time from vaccination to complication, or sex (P > .05, Table 1).
Baseline Characteristics of Patients With Different BCG-Related Complications.
Injection Site Adverse Reactions
Injection site adverse reactions were observed in 33 patients, accounting for 34.7% of the total. Of these, 18 children (54.5%) were males and 15 (45.5%) were females. The median time from vaccination to complication was 6.00 months (IQR 4.75-7.00). Among them, 16 patients (48.5%) were younger than 6 months, 12 (36.4%) were between 6 months and 1 year, and 5 (15.1%) were older than 1 year. All subjects were administered the BCG vaccination at birth, except for 2 who received it at 5 months due to pathological jaundice. None of the patients were born prematurely, and all had normal birth weights. Local adverse reactions included ulceration in 12 children (36.4%), lichenoid lesions in 3 (9.1%), granulation hyperplasia in 3 (9.1%), and deep abscesses in 15 (45.4%). None of the patients exhibited fever, weight loss, or systemic symptoms.
All children tested negative for the interferon-γ release assay (IGRA). All patients had normal CD4 and CD8 counts and tested negative for HIV. Culture, AFB smear, and MBT were performed on tissue samples from lesions at the injection site for microbiological evaluation, and only 4 samples returned positive results. Resistant to rifampicin or isoniazid was not found. Interventions, guided by dermatology recommendations, were tailored to the type of local adverse reaction and included topical and oral medications as well as surgical treatments (Figure 2). For cases of deep abscesses, the median time of oral isoniazid was 2.00 months (IQR 1.75-2.25). During the subsequent 5.00 months (IQR 4.50-6.00) follow-up, all children showed complete recovery within 3 months and had no relapses.

Management and outcome of children with injection (BCG) site adverse reactions.
Lymphadenitis
Of the 55 children with lymphadenitis, 72.4% were male and 27.3% were female. Preterm births occurred in 6 patients, accounting for 10.9% of the total. Of the children studied, 46 were vaccinated with BCG at birth, 7 at 1 month, 1 at 2 months, and 1 at 6 months. Demographic characteristics are presented in Table 2. The average age was 7.91 ± 5.97 months. Among the subjects, 32 (58.2%) were under 6 months old, 41 (74.5%) were under 12 months old, 12 (21.8%) were aged between 12 and 24 months, and 2 (3.6%) were older than 24 months old. The median time after vaccination was 5.00 months (IQR 3.00-10.00). Among the 58 nodes involved, 52 (94.5%) were axillary, 5 (9.1%) were supraclavicular, and 1 (1.8%) was cervical. Three children exhibited both axillary and supraclavicular involvement, while only one patient showed both axillary and cervical involvement. Among 55 children, 27 (49.1%) had suppurative lymphadenitis with spontaneous rupture, 4 (7.3%) had suppurative lymphadenitis without rupture, and 24 (43.6%) had non-suppurative lymphadenitis (Table 2).
Characteristics of Children with Lymphadenitis.

All children tested negative for both whole-blood IGRA and HIV. All patients exhibited normal CD4+ and CD8+ counts in whole blood. Fourteen children (58.3%) with non-suppurative lymphadenitis received a 2-month course of oral isoniazid based on their legal guardians’ consent, while the other 10 children (41.7%) were under observation. During the follow-up, 2 patients from both the oral isoniazid group and the observation group developed lymph node suppuration. Once suppuration occurred, surgical interventions were performed to remove the pus and granulation tissues in affected lymph nodes. Isoniazid and rifampin were administered to those who underwent lymph node surgery (Figure 3). The median time of antibiotic treatment was 2.00 months (IQR 2.00-3.00). The Microbiological evaluations, including culture, AFB smear, and MBT, were conducted, with 30 cases (54.5%) testing positive. No rifampicin or isoniazid resistant was identified. All cases completely recovered within 6 months, and the mean follow-up period was 9.25 ± 1.95 months.

Management and outcome of children with BCG lymphadenitis.
Disseminated BCG
Among the 7 patients, 2 (28.6%) were female and 5 (71.4%) were male. All patients were vaccinated with BCG at birth, except for 1 who received it at 1 month old due to neonatal pneumonia. Out of 7 patients, 6 (85.7%) had normal birth weights, whereas 1 had a birth weight under 2500 g. Among the children, 57.1% (4 individuals) exhibited symptom onset at 3 months of age, while the remaining children showed onset at 4, 6, and 9 months. In 3 cases (42.9%), diagnosis was made 1 month after symptom onset, while for the others, the time to diagnosis ranged from 1 week to 2 months. The median time from vaccination to the development of the disseminated disease was 5.00 months (IQR 4.00-6.00). The most common symptoms or signs observed were anemia (6 cases, 85.7%), generalized lymphadenopathy including axillary nodes (5 cases, 71.4%), fever (5 cases, 71.4%), weight loss (4 cases, 57.1%), difficulty in raising the head and sitting (1 case, 14.3%), and left cervical generalized lymphadenopathy (1 case, 14.3%; Table 3).
Baseline and Clinical Characteristic of Seven Children With Disseminated BCG.

Abbreviations: H, isoniazid; R, rifampicin; E, ethambutol; Lzd, linezolid; Amk, amikacin; PZA, pyrazinamide; CXR, chest x ray; MRI, magnetic resonance image; U/S, Ultrasound.
All children tested negative for both whole-blood IGRA and HIV. Imaging revealed lesions in the lungs in 6 cases (85.7%), mediastinal and abdominal lymphadenopathy in 3 cases (42.9%), brain foci in 2 cases (28.6%), bony destruction in 1 case (14.3%), and hepatomegaly in 1 case (14.3%). Two children (28.6%) showed reduced CD4+ T cells in peripheral blood, diagnosed with chronic granulomatous disease (CGD) and severe combined immunodeficiency (SCID), respectively. The other 5 patients exhibited normal CD4+ T cell counts. Specimens from left axillary nodes in 4 patients were positive for Mycobacterium tuberculosis on AFB smear and culture, all showing resistance to pyrazinamide (PZA) but were susceptible to rifampicin and isoniazid. A positive PCR was found in 5 children (71.4%), with specimens collected from gastric wash in 2 cases (28.6%), left axillary nodes in 2 cases (28.6%), and left cervical nodes in 1 case (14.3%). Biopsies in all children exhibited chronic granulomatous inflammation, as detailed in Table 3.
All patients received anti-tuberculosis treatment consisting of at least 3 drugs, with treatment lasting 12 to 18 months. An appropriate, personalized anti-tuberculosis regimen was selected based on the clinical condition and the family’s preferences. All children showed improvement except for 1, who was diagnosed with severe combined immunodeficiency (SCID) and died from multi-organ failure. After 5 years of follow-up, no recurrence was observed.
Discussion
Despite the widespread and free administration of the BCG vaccine to millions of newborns in China on their first day of life, reports of severe adverse reactions remain rare. 11
This study examined the clinical features of 95 children in China who experienced severe complications from the BCG vaccine over a 5-year span. Among them, the most common complication was lymphadenitis, followed by site infection. Disseminated BCG was uncommon, identified in only 7 cases.
We compared patient characteristics among those with severe injection site reactions, lymphadenitis, and disseminated BCG disease, and we found no statistically significant differences concerning the moment of vaccination (at birth vs after 1 month), median time from vaccination to complication. These findings suggest that these factors may not independently determine the type of BCG-related complications. Other potential contributors, such as host immune responses, genetic predisposition, or BCG strain, may play a role. Future studies with larger sample sizes and more comprehensive immunological assessments are needed to better understand risk factors associated with different BCG-related complications.
In contrast with other series, like the one by Turnbull et al, 13 we found that the median time from vaccination to complication in both the severe injection site reactions group and the lymphadenitis group was longer in our cohort. This may be due to mild BCG-related complications being managed in community or other hospitals, while patients referred to our specialized center often had persistent or worsening symptoms despite prior treatment, leading to a longer observed time to complication in our cohort. Severe adverse reactions at the injection site are relatively rare events caused by BCG vaccines. 14 Most of the patients in our cohort received their BCG vaccination at birth. However, previous studies have reported a higher frequency of severe local reactions in vaccinees older than 6 months (2.4%) compared to infants under 6 months of age (0.5%). 13 BCG revaccination could also increase the incidence of abscess formation at the injection site. 15 Due to pathogenesis and pathological changes similar to true cutaneous tuberculosis, these events can manifest as abscesses, purulent discharge, crusts, eczematous lesions, or lichen scrofulosorum-like lesions. 16 In our study, we have observed ulceration (36.4%), lichenoid lesions (9.1%), granulation hyperplasia (9.1%), and deep abscesses (45.4%) as local complications. The clinical manifestations of these local complications are influenced by the patient’s inherent susceptibility, the vaccination dose, and the technique used. 17
The age of onset is similar to previous studies, 18 with the majority occurring within the first year of life. However, rather than being solely attributed to immune immaturity of infants, severe local reactions are often linked to hypersensitivity responses and vaccination techniques, particularly inadvertent subcutaneous injection in neonates. 19 Previous studies have shown that most severe local reactions occur in children older than 6 months, when immune function is more developed.13,18 This contrasts with disseminated BCG disease, which has a well-established association with immunodeficiency. We hypothesize that deep abscesses are mainly affected by vaccination technique, whereas the formation and progression of other lesions are likely linked to the patient’s immune response to the BCG vaccine, especially delayed-type hypersensitivity.
No consensus exists on treating cutaneous complications after BCG vaccination. Research indicates that oral isoniazid, alone or combined with rifampicin, effectively treats injection site ulcers and abscesses over 1 cm, with a complete healing rate exceeding 90%.18,20 In our study, patients with deep abscesses received oral isoniazid, alongside essential surgical removal of abscesses and chronic granulomatous tissue. Other lesions were primarily treated with oral chlorpheniramine and topical medications to control infection and reduce skin inflammation, resulting in complete healing of all cases.
The incidence of BCG vaccine-induced lymphadenitis varies between 0.1% and 0.01%. 8 In this study, lymphadenitis was the predominant type, with half of the cases occurring within the first year of life, consistent with previous studies [19, 20]. This is likely associated with a weakened Th1 immune response during infancy. Consistent with prior studies,21,22 the axillary lymph nodes were most frequently affected (94.5%), followed by supraclavicular (9.1%) and cervical nodes (1.8%). Our study observed a notably high incidence of axillary lymphadenitis, potentially attributable to demographic variations and the specific vaccine strain employed.
Lymphadenitis caused by the BCG vaccine is classified into suppurative and non-suppurative types. 22 This study found that suppurative lymphadenitis accounted for 56.4% and non-suppurative lymphadenitis for 43.6%, aligning with earlier research.21,23 Studies indicate that conventional anti-tuberculous treatment (ATT) fails to prevent the progression of non-suppurative lymphadenitis to its suppurative form, with watchful observation typically advised. 24 Rermruay et al followed 12 infants with non-suppurative lymphadenitis (<2.5 cm) for 6 months, and all cases resolved spontaneously. 25 In our study, 14 cases (58.3%) of non-suppurative lymphadenitis were managed with oral isoniazid, contrasting with existing literature. The lack of research on BCG lymphadenitis treatment in China may lead physicians to depend on clinical experience and family preferences. In our cohort, the progression to suppurative lymphadenitis was limited to 2 cases in both the oral isoniazid and observation groups, indicating that oral anti-tuberculosis medication does not decrease the incidence of suppurative lymphadenitis.
Numerous strategies for managing suppurative lymphadenitis have been documented, such as observation, anti-tubercular therapy, needle aspiration, incision and drainage, and lymph node excision.6,21,23,25 -27 Observation is not advised due to the rarity and potential year-long duration of spontaneous recovery. 22 Incision and drainage are neither recommended because they can lead to delayed wound healing, poor scarring, and ongoing discharge. 28 Needle aspiration could be a safe and effective procedure,28,29 though it may necessitate multiple sessions and extend the disease duration, potentially heightening the risk of chronic sinus formation. Surgical excision of the affected lymph nodes and abscess is recommended to accelerate healing and avert sinus formation.6,30 In our study, surgical removal of pus and granulomatous tissue in affected lymph nodes was performed on all patients with suppurative lymphadenitis, with no complications arising from general anesthesia or the procedure. Moreover, all patients received administered oral isoniazid and rifampin for 2 to 4 months to eliminate residual bacilli and prevent transmission. Ultimately, all patients made a full recovery.
Disseminated BCG is a rare yet serious complication, occurring in 1 out of every 230 000 to 640 000 vaccinated individuals, 8 with an even lower incidence reported in China. 11 Disseminated BCG infection, though uncommon, is a serious complication of BCG vaccination in immunocompromised children with an unknown immunodeficiency at the moment of vaccination, frequently resulting in severe consequences. 31 Studies indicate that immunodeficiency is present in two-thirds of patients with disseminated BCG. 32 Primary immunodeficiencies commonly linked to disseminated mycobacterial infections include severe combined immunodeficiency (SCID), chronic granulomatous diseases (GCD), Mendelian susceptibility to mycobacterial disease (MSMD), Wiskott-Aldrich syndrome, complete DiGeorge syndrome, and HIV infection. 33 In our study, 2 of the children that presented with disseminated BCG (28.6%) exhibited reduced peripheral blood CD4+ T cells and were diagnosed with GCD and SCID, respectively. The presence of PID in the other children was unrecognized, potentially due to inadequate testing methods or the families’ financial constraints. Our study observed that symptoms typically appeared within 3 months post-BCG vaccination, with anemia, left axillary generalized lymphadenopathy, fever, and weight loss being the most prevalent signs and symptoms, aligning with previous studies.34,35 The mortality of disseminated BCG in our study was 14.3%, which is significantly lower than the 54 to 80% reported in previous studies. 31 This may be attributed to the higher proportion of immunosuppressed patients in those cohorts (up to 60%), whereas in the current cohort, only 2 patients had a recognized immunodeficiency. Prior studies have demonstrated a strong correlation between immunosuppression and poor outcomes in disseminated BCG disease. 36 The delay in the diagnosis and onset of anti-tuberculosis treatment could also explain the high mortality. Resistance to isoniazid or rifampicin of BCG strains, could also contribute to the high mortality. 37 In our cohort, we found no evidence of resistance to isoniazid or rifampin, which may be related to differences in the strains used for BCG vaccine production.
Our study has some limitations. First, we cannot estimate the overall complication rate of BCG vaccination, since we cannot access the total number of vaccinated children in the community during the study period. Moreover, it is possible that the number of local site complications was higher since most children with mild injection site reactions were treated in community or other hospitals. As a retrospective study, the laboratory tests for immunodeficiency screening were not exhaustive, especially in children presenting with injection site reactions and lymphadenitis. This may have been influenced by the mildness of the condition or economic factors. Data on serum immunoglobulin levels and the nitro blue tetrazolium test were unavailable for our patients with disseminated BCG.
Conclusion
Adverse reactions after BCG vaccination appear to be rare. However, clinicians may be aware of these complications, since they can be potentially severe. Severe adverse reactions at the BCG injection site present with varied cutaneous manifestations. Our findings indicate that deep abscesses require a combination of surgical and anti-tubercular treatment, whereas other mild local site complications most cases can be managed conservatively with topical therapy and oral antihistamines. For suppurative BCG lymphadenitis, surgical interventions, including drainage, combined with oral anti-tubercular therapy, were effective and well-tolerated in our cohort. Disseminated BCG disease is rare but warrants immunological evaluation. Early recognition and appropriate management contribute to favorable outcomes of BCG vaccination complications.
Footnotes
List of Abbreviations
BCG Bacille Calmette-Guérin
TB Tuberculosis
PID primary immunodeficiency
SCID severe combined immunodeficiency
MSMD mendelian susceptibility to mycobacterial disease
GCD chronic granulomatous disease
PCR polymerase chain reaction
AFB acid fast bacillus
IGRA interferon-γ releasing assay
Author Contributions
SH: contributed to analysis and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
FC: contributed to interpretation; drafted manuscript; gave final approval.
HG: contributed to acquisition; gave final approval.
SKZ: contributed to conception and design; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
