Abstract
Objectives:
To investigate the effectiveness of Botulinum Toxin A (BTX-A) intravesical injections in improving quality of life (QoL) among pediatric patients.
Introduction:
Overactive bladder (OAB) in children impacts their QoL. Current treatments often have limitations, necessitating the exploration of alternative therapies.
Methods:
A quasi-experimental design was conducted in four Jordanian hospitals, with participants being parents of pediatric OAB patients aged 5 to 18 years. Statistical analysis was utilized for data analysis.
Results:
Results showed that BTX-A injections significantly improved QoL scores for pediatric OAB patients, with marked reductions in symptoms and enhanced well-being in the intervention group. Key demographic factors like household income and age at symptom onset were linked to QoL outcomes, and pre-treatment QoL varied by healthcare provider type.
Conclusion:
This study highlights BTX-A as a promising therapeutic and advocates for its broader adoption in pediatric urology to enhance the well-being and daily functioning of children with OAB.
Introduction
Overactive bladder (OAB) is a condition that affects children, causing urinary urgency, increased voiding frequency, and possibly urinary incontinence. 1 OAB can be defined as a distinct form of lower urinary tract dysfunction caused by involuntary detrusor contractions during the bladder’s filling phase. 2 The pathophysiology of OAB in pediatric patients is complex and not fully understood, with a significant hypothesis suggesting the role of dysbiosis in the urinary microbiome. 3 The prevalence data on pediatric OAB varies widely, with estimates ranging from 1.5% to 36.4%. 4 The peak incidence of OAB is noted between 5 and 7 years of age, with a higher occurrence reported in males. 2
Pediatric OAB presents unique challenges compared to adult cases, necessitating an age-specific approach to diagnosis and treatment. Children with OAB often develop compensatory behaviors, such as holding maneuvers to delay urination, which can lead to complications such as bladder overdistention, recurrent urinary tract infections, and vesicoureteral reflux. 5 This condition impacts daily life, affecting the child’s emotional, social, and developmental well-being, including educational performance and familial relationships. 2
The quality of life (QoL) implications for children with OAB are profound and multifaceted. They often experience significant social embarrassment and isolation, leading to withdrawal from peer interactions and extracurricular activities.6,7 Additionally, pediatric patients with OAB have significantly lower scores in health-related QoL assessments, particularly in domains related to emotional and social functioning. 8 The chronic nature of OAB symptoms also contributes to ongoing anxiety and a decrease in overall life satisfaction. 9
The burden extends to caregivers, as highlighted by Nambiar et al, 10 who face psychological stress and disruptions within the family dynamic. This extensive impact underscores the need for treatments that address the condition from both the patient’s and caregivers’ perspectives. Existing treatment modalities, such as antimuscarinic drugs, are accompanied by side effects that are particularly concerning in children, such as dry mouth, constipation, and blurred vision. 11 These side effects not only diminish the QoL but also can lead to poor adherence to medication, undermining the effectiveness of the treatment.
The existing research on pediatric OAB and BTX-A treatment, including Uçar et al 12 and Mahjani et al 13 studies has been predominantly centered on adult populations. This adult-centric approach creates a significant gap in pediatric-specific data. For instance, while Uçar et al 12 provided valuable insights into BTX-A treatment, the study’s relevance to pediatric patients is limited due to a sample composition where a minimal percentage of participants were children. Such adult-focused research fails to address the unique physiological and developmental considerations pertinent to pediatric OAB treatment.
There is a critical need for longitudinal and experimental studies to explore the long-term effects of BTX-A in children with OAB. Most existing research follows patients for relatively short durations, often only a few months. This limited timeframe is insufficient for assessing the prolonged efficacy and safety of BTX-A in the pediatric population, particularly considering the ongoing developmental changes in children. Comprehensive studies with extended follow-up periods are necessary to gain a thorough understanding of the long-term impact of BTX-A treatment in this vulnerable group. Furthermore, a significant research gap exists in comprehending the effects of pediatric OAB and BTX-A treatment from the viewpoints of parents or caregivers in the Jordanian context, due to the lack of previous similar studies. Therefore, this study focuses on the QoL as perceived by parents, offering a new dimension to the existing body of research. By incorporating parental perspectives, the study seeks to enhance our understanding of the impact of OAB on family life and daily functioning. The research aims to contribute significantly and offer insights that could lead to more effective and holistic treatment strategies for children suffering from OAB. The insights from this study could guide the development of healthcare policies and resource allocation, emphasizing OAB treatments that consider the QoL outcomes for the pediatric population.
Aim of the Study
The main aim of the study is to evaluate the effectiveness of BTX-A intravesical injections in improving the quality of life in pediatric patients from the parents’ perspective.
Research Questions
What is the impact of BTX-A intravesical injections on the QoL of pediatric patients with OAB?
What is the relationship between demographics and QoL in pediatric patients with OAB before and after BTX-A intravesical injections?
Methods
Design
The study adopts a quasi-experimental design. This design comprises 2 groups: parents whose children are receiving BTX-A intravesical injections and a control group whose children are undergoing standard OAB care. This approach is selected for its applicability in real clinical settings. 14 The quasi-experimental design allows for the comparison of perceptions and experiences between the 2 groups, 15 offering valuable insights into the impact of BTX-A treatment versus standard care on the overall QoL from the parental perspective.
Setting
The study was conducted in four Jordanian hospitals, including 1 governmental, 1 educational, and 2 privates, which are known for their specialized urology services and BTX-A injections, particularly for pediatric patients. These hospitals provide a diverse sample of participants and treatment environments. They have been selected for their distinct operational frameworks and their significant contributions to pediatric urology, providing a comprehensive setting for this study.
Sample
A convenient sampling technique was used to ensure easy access to participants, particularly in busy clinical settings where recruiting participants might be challenging and facilitate rapid data collection. This method may yield diverse perspectives from parents across different socio-economic backgrounds, enhancing the generalizability of findings. However, convenience sampling may introduce selection bias. The sample size was calculated using G*Power software for a 2-group t-test, with a power of 0.8, an effect size of 0.35, and an alpha of 0.05. We adjust the final required sample size to 80 participants to account for potential dropouts and ensure reliable and valid results.
In the targeted hospital, OAB in children was diagnosed by a physician following clinical guidelines, and eligible participants were parents of pediatric patients aged between 5 and 18 years who had been professionally diagnosed with OAB, were candidates for BTX-A intravesical injections according to clinical guidelines and were willing to participate in the study. The children’s age range of 5 to 18 years was selected to encompass a broad spectrum of developmental stages, from early childhood through adolescence, capturing variations in symptom presentation and treatment response.16,17 The study excludes parents of pediatric patients with contraindications for BTX-A treatment, those whose children have undergone BTX-A injections in the past 6 months, and parents with severe cognitive or communication impairments that could impede their effective participation in assessments.
Instrumentation
This study used 2 questionnaires. The first questionnaire gathered sociodemographic data, including the child’s age and gender, the caregiver-child relationship, household income, education, and employment status, as well as disease-related information such as the duration and onset of OAB symptoms, treatment history, symptom severity, comorbidities, current medications, surgical interventions, frequency of healthcare visits, and provider specialty. It also included contextual factors like family size, restroom access, location, transportation, insurance, cultural views on pediatric OAB, support resources, and OAB’s impact on the child’s school performance and caregiver’s life. This comprehensive tool aimed to capture the child’s condition and its effects on their family. The second tool, the Pediatric Incontinence Questionnaire (PinQ), assesses the impact of bladder dysfunction, including OAB, on pediatric quality of life (QoL) from both child and parent perspectives. 18 It includes 20 items in 2 subscales—intrinsic factors (eg, self-worth, emotional well-being, internalized concerns) and extrinsic factors (eg, social interactions, parental awareness), with Likert-scale responses. Higher scores indicate a greater impact on QoL. Many studies have shown that the PinQ is highly reliable and valid.18-20
Procedures
Participants were allocated into 2 groups, an intervention group and a control group, based on the treatment choices made by parents in consultation with their healthcare providers, see Figure 1. This study was conducted across multiple centers, with all centers adhering to a standardized treatment protocol to ensure consistency in diagnosis, intervention, and outcome assessment. The intervention group consisted of parents of pediatric patients who received BTX-A intravesical injections as part of their treatment for OAB. The BTX-A treatment involved administering a specified dose (eg, 100 units) directly into the detrusor muscle of the bladder using cystoscopic guidance. The procedure was performed under general anesthesia or sedation to ensure patient comfort. A pediatric urologist or trained specialist carried out the injection process, which involved multiple small injections across different sites of the detrusor muscle to maximize the therapeutic effect. The procedure took approximately 20 to 30 minutes, with patients being observed post-procedure for any immediate adverse reactions. The treatment aimed to inhibit involuntary detrusor muscle contractions, thereby reducing symptoms of urinary urgency, frequency, nocturia, and urge incontinence. Patients were monitored through follow-up visits at 1-month post-procedure.

The CONSORT flow diagram of the study.
The control group received standard management for OAB, consisting of behavioral modifications and medication to alleviate symptoms and improve quality of life. Behavioral interventions included fluid restriction, where patients were advised to limit fluid intake to prevent excessive bladder activity, and timed voiding, which involved scheduled urination every 3 to 4 hours to establish a consistent bladder-emptying routine and reduce urgency and incontinence episodes. In addition to these strategies, patients were prescribed anticholinergic medications, such as Solifenacin (Sulfi) and Desmopressin (Minirin), to help control bladder muscle spasms and improve bladder function. Since this was a multicentric study, all centers followed a standardized treatment protocol to ensure consistency in diagnosis, intervention, and outcome assessment. Medication adherence and any side effects were assessed during follow-up appointments at 1-month post-initiation.
The primary outcome was the improvement in quality of life (QoL) of pediatric patients with OAB, as perceived by parents. QoL was assessed using standardized tools, including the Pediatric Incontinence Questionnaire (PinQ). The secondary outcome was the reduction in OAB symptoms, including urinary urgency, frequency, nocturia, and urge incontinence. Additionally, the study examined the relationship between demographic factors and QoL outcomes, identifying potential influences on treatment effectiveness. These outcomes were evaluated using the PinQ at baseline and 1-month post-intervention.
Recruitment and Data Collection
Recruitment took place from December 2023 to February 2024. Study implementation began in March 2024, with participants followed for 1 month post-treatment. Follow-up assessments were completed in April 2024. The research team exclusively handled participant recruitment and data collection. Eligible parents received detailed information on the study’s objectives, procedures, risks, benefits, and participant rights before providing informed consent. Parents were assigned to 1 of 2 groups; the intervention group, which consisted of parents of pediatric patients eligible for and choosing BTX-A treatment, and the control group, which included parents of children ineligible for or opting out of BTX-A treatment. Baseline data collection included sociodemographic and disease-related information and responses to the Pediatric Incontinence Questionnaire (PinQ). The intervention group received educational materials and consultations with healthcare providers about BTX-A treatment, while control group parents were informed that their children would continue standard care without BTX-A intervention. To maintain consistency with pediatric OAB follow-up practices, the intervention group had a 1-month follow-up to assess BTX-A’s effects. Questionnaires were distributed during hospital visits, with parents given options to return completed forms via drop-off points, mail, or electronic submission. Both the intervention and the control groups reported no severe adverse events or unintended effects.
Data Analysis
The derived data were analyzed utilizing statistical analyses, SPSS version 26, with a significance level set at .05 to determine statistical significance. Initially, descriptive statistics are employed to summarize the demographic and clinical characteristics of the participants. To compare these characteristics between the 2 groups, independent t-tests are utilized for continuous variables, and chi-square analyses are applied for categorical variables. Additionally, to evaluate the differences within each group before and after the intervention, paired t-tests are employed. Also, to explore the relationships between the post-intervention QoL scores and the demographic characteristics, Pearson correlation analysis is used. One-way ANOVA was utilized for subgroup analyses.
Ethical Approval and Informed Consent
Institutional Review Board (IRB) approval was obtained from Applied Science Private University (reference #2023-2024-2-1). Informed consent was secured from participating parents, ensuring their full understanding of the study’s objectives and their right to voluntary withdrawal at any stage. Strict privacy and confidentiality measures were implemented to protect participants’ personal and medical data. All collected information was de-identified and stored securely using encrypted databases with restricted access limited to authorized research personnel. The study documents were kept in locked cabinets within secured facilities. Data sharing and dissemination followed rigorous protocols, ensuring that no personally identifiable information was disclosed. Any electronic communication or transfer of data was conducted through secure, password-protected platforms to prevent unauthorized access. Additionally, participants were assigned unique identification codes to further safeguard anonymity. The study adhered to the principles of beneficence and non-maleficence by maximizing benefits and minimizing risks for participants. Recruitment and reporting processes were guided by equity, fairness, and transparency.
Results
Demographic Characteristics
The demographic characteristics of the study participants, displayed in Tables 1 and 2. The study participants in both the control group (CG) and intervention group (IVG) had similar mean ages for children (around 8.5 years) and caregivers (around 34 years), yet differed notably in income, with the IVG reporting higher average household income. Children in the IVG generally exhibited symptoms of pediatric OAB at a younger age (mean age 4.94) compared to the CG (mean age 6.41). Hospital types varied, with more IVG participants treated in private hospitals and CG participants in government or teaching hospitals. Both groups had a predominantly female caregiver demographic, though educational levels and employment rates were higher in the IVG. Symptom duration also differed, with more children in the IVG experiencing symptoms for over 4 years. The IVG had a greater history of prior treatments, higher rates of comorbid conditions (UTIs, constipation), and more frequent healthcare visits than the CG. In terms of OAB treatment, the IVG participants were more likely to be prescribed anticholinergic medications. Geographic location was largely urban across both groups, though the IVG had less health insurance coverage and higher accessibility to transportation.
Demographic Characteristics of the Study Participants Based on Group (Continuous Variables).

Abbreviations: CG, control Group; IVG, intervention group; M, mean; SD, standard deviation.
Demographic Characteristics of the Study Participants Based on Group.

Abbreviations: CG, control group; IVG, intervention group; N, number.
The Impact of BTX-A Intravesical Injections on the QoL
The QoL scores for the CG and the IVG both before and after receiving BTX-A intravesical injections are displayed in Table 3. Before treatment, the IVG had a higher QoL score (73.95) compared to the CG (68.42), and this difference was statistically not significant (P = .31), indicating a worse QoL due to more severe OAB symptoms. After the treatment, the QoL scores in the IVG dramatically improved, dropping to an average of 14.70, which indicates a significant enhancement in their QoL (P = .00). This substantial reduction in the score reflects the effective alleviation of OAB symptoms, leading to a better daily living experience and overall well-being. In contrast, the CG saw a slight decrease in their QoL score to 61.60, suggesting a marginal improvement, possibly due to natural variations over time rather than the impact of any intervention. These results effectively demonstrate that BTX-A injections have a significant positive influence on the QoL in pediatric patients with OAB, markedly reducing their discomfort and enhancing their ability to engage more fully in daily activities.
QoL Scores Pre- and Post-Treatment in Control and Intervention Groups.

Abbreviations: CG, control group; IVG, intervention group; N, number; M, mean
Results are significant at the 0.05 level.
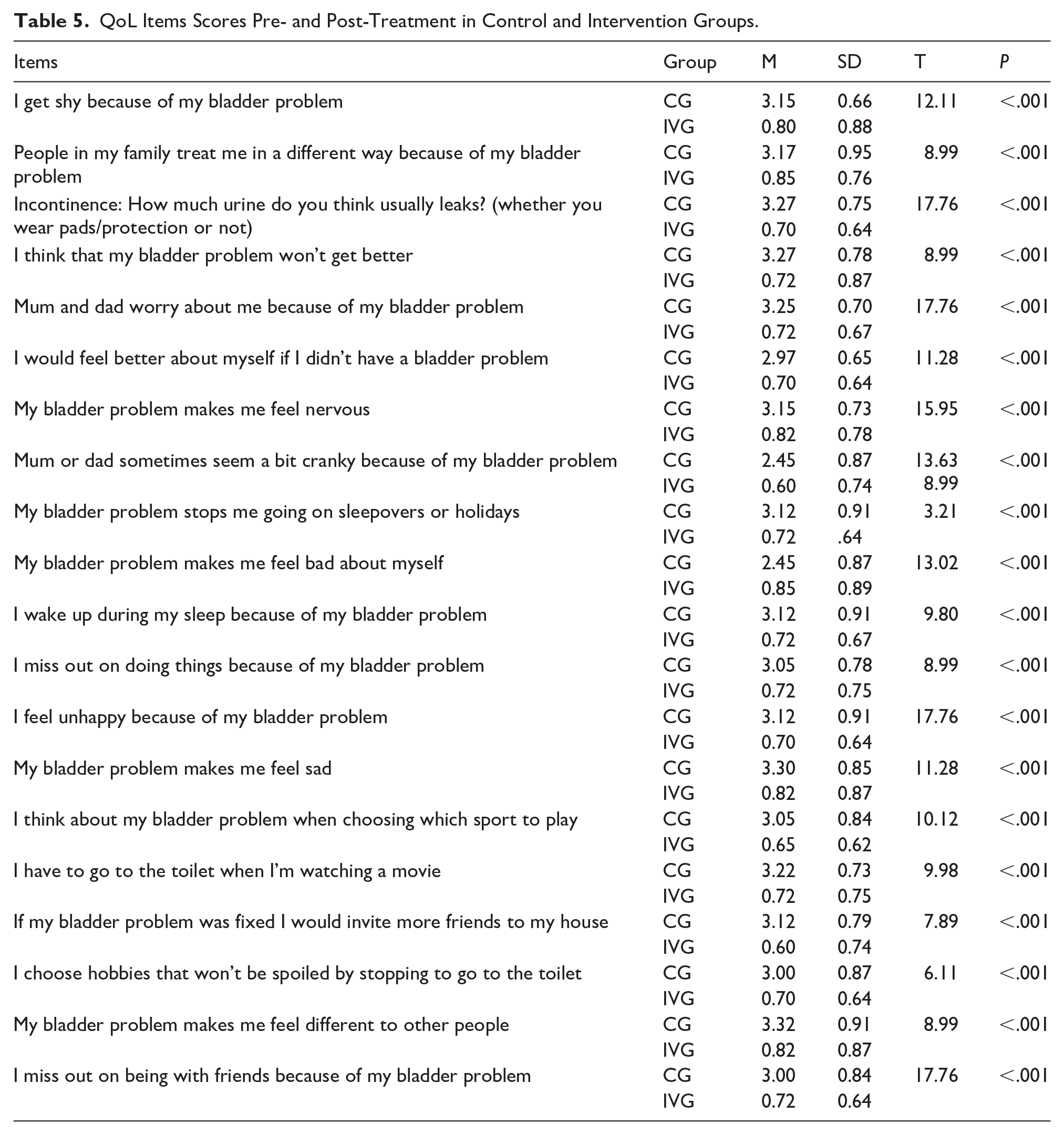
Table 4 shows the comparison of QoL scores pre- and post-intervention within the IVG, employing a paired t-test. Prior to the intervention, the mean QoL score was 73.95 (SD = 5.06), which significantly decreased post-intervention to 14.70 (SD = 9.69). This change was significant (t = 32.89, P < .05), indicating a substantial impro-vement in QoL following the intravesical Botox injection treatment among pediatric patients with OAB. In addition, Table 5 displays the QoL scores related to the personal and social impacts of OAB problems in pediatric patients, comparing the CG and the IVG both before and after BTX-A treatment. The data show dramatic improvements in the IVG post-treatment across a range of QoL items. Scores significantly decreased in areas concerning feelings of shyness, being treated differently by family, incontinence severity, fears about the permanence of the bladder problem, and the psychological impacts such as nervousness, sadness, and low self-esteem. For example, the average score for feeling shy due to bladder problems dropped from 3.15 in the CG to 0.80 in the IVG, and similar substantial reductions were observed in other psychosocial domains. These results indicate a notable enhancement in the overall QoL and social well-being of the children in the IVG following BTX-A treatment, demonstrating the treatment’s effectiveness in reducing the personal and social burdens of OAB.
Comparison of Pre- and Post-Treatment QoL Scores Within the IVG.

Abbreviations: IVG, intervention group; N, number; M, mean.
Results are significant at the 0.05 level.
QoL Items Scores Pre- and Post-Treatment in Control and Intervention Groups.
Relationship Between Demographics and QoL Before and After BTX-A
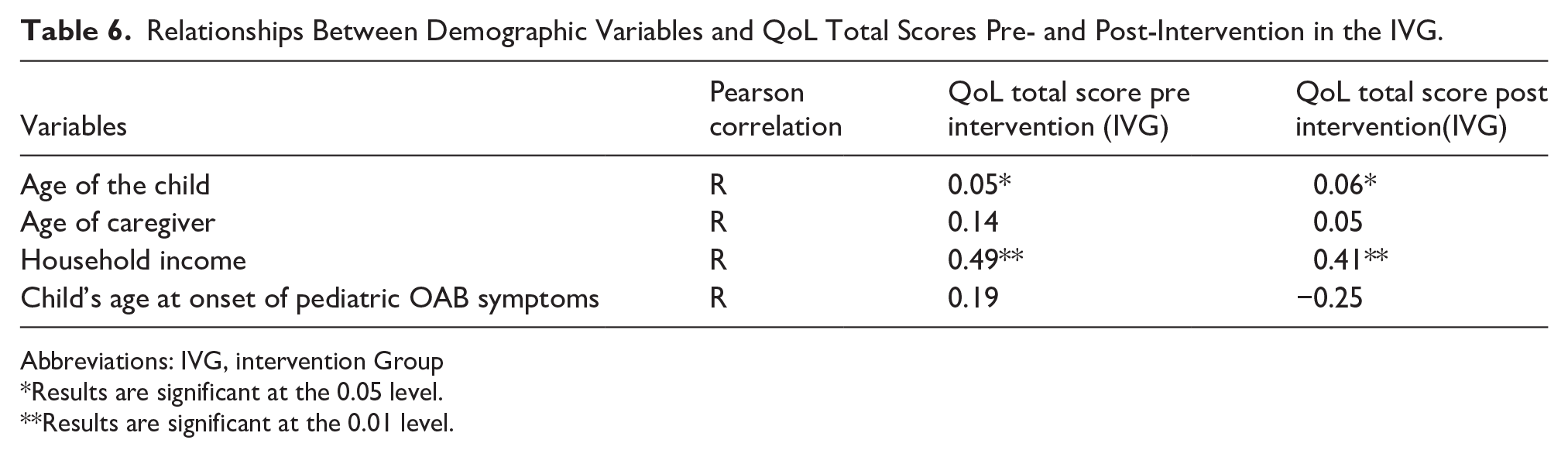
The Pearson correlation coefficients between demographic variables and QoL total scores pre- and post-intervention within the IVG are displayed in Table 6. Among the variables, household income showed a significant positive correlation with both pre-intervention (r = .49, P < .01) and post-intervention (r = .41, P < .01) QOL scores. Additionally, age of the child demonstrated weak positive and significant relationships with both pre- and post-intervention QoL scores (P < .05). Notably, the age at onset of pediatric OAB symptoms exhibited a weak positive correlation with pre-intervention QoL scores (r = .19) and a negative weak correlation with post-intervention QoL scores (r = −.25). The asterisks denote statistical significance, at the .05 level and at the .01 level.
Relationships Between Demographic Variables and QoL Total Scores Pre- and Post-Intervention in the IVG.
Abbreviations: IVG, intervention Group
Results are significant at the 0.05 level.
Results are significant at the 0.01 level.
Moreover, Table 7 presents the results of a one-way ANOVA examining the variation in QOL total scores based on healthcare providers within the IVG. The analysis revealed a statistically significant difference in pre-intervention QoL scores among different healthcare provider categories (F = 3.44, P = .04). Post-hoc comparisons indicated that the mean pre-intervention QoL score for pediatric Urologists (M = 74.60, SD = 4.42) was significantly higher compared to that of Pediatri-cians (M = 71.66, SD = 4.93) (P < .05), while no significant differences were observed between Urologists (M = 66.00, SD = 11.31) and the other groups. These findings suggest that the choice of healthcare provider may have an impact on the pre-intervention QoL of pediatric patients with OAB.
Variation in QoL Total Scores Based on Healthcare Providers.

Abbreviations: IVG, intervention Group; N, number; M, mean.
Results are significant at the .05 level.
Discussion
This study aimed to evaluate the effectiveness of BTX-A intravesical injections on pediatric patients diagnosed with OAB in Jordanian hospitals and understand the relationship between demographic factors and treatment outcomes. The research adopted a quasi-experimental design involving 4 hospitals, providing robust data comparing the intervention group receiving BTX-A treatment to a control group undergoing standard OAB care. The study found that BTX-A intravesical injections significantly improved the QoL of pediatric patients with OAB, which can relate to the alleviation of physical symptoms and the reduction in psychological distress. This improvement is consistent with previous studies, such as Al Edwan et al 21 and Austin et al, 22 which reported significant enhancements in QoL scores following BTX-A treatment in children with OAB. The study findings are also supported by other studies.23,24 However, Fan and Kuo 25 presented mixed results regarding the impact of BTX-A on QoL in pediatric patients with refractory OAB. While some patients experienced substantial improvements, others reported only minimal changes, highlighting the variability in treatment response. This inconsistency underscores the need for further research to identify factors that predict positive outcomes and optimize treatment protocols for individual patients.
The improvements in the emotional and social aspects of QoL post-treatment with BTX-A intravesical injections are substantial and noteworthy in the current study. Pediatric patients in the intervention group (IVG) reported marked reductions in emotional distress and significant enhancements in social interactions following BTX-A treatment. These outcomes underscore the profound psychosocial benefits that come with effective symptom management in pediatric OAB patients. The psychosocial benefits observed in the current study are supported by previous research.21-24,26 However, Santos et al 20 suggested that while parasacral TENS treatment for OAB improved urinary symptoms, it did not significantly change psychological symptoms. This suggests that individual patient factors, such as the severity of symptoms and baseline emotional state, play critical roles in determining the overall psychosocial impact of the treatment. 27
The current study found a significant positive correlation between household income and QoL outcomes following BTX-A intravesical injections. Children from higher-income households showed greater improvements in QoL scores, which can be explained by better access to healthcare resources among families with higher incomes. 28 However, other studies reported minimal impact of household income on treatment efficacy,20,29 suggesting that while financial resources facilitate treatment access, treatment efficacy itself may not be significantly influenced by household income.25,27
The current study highlights the importance of early intervention in managing pediatric OAB by focusing on the neuroplasticity and adaptability of younger patients. The findings align with previous research, indicating that younger patients with an earlier onset of symptoms respond more favorably to treatments like BTX-A. 30 Early intervention during critical developmental periods can mitigate the long-term impact of OAB on a child’s physical, emotional, and social well-being, as observed in the current study and supported by Wu et al. 23 However, Fernandez-Ibieta and Ayuso-Gonzalez 31 provided a contrasting perspective, suggesting that treatment outcomes for pediatric OAB are not significantly influenced by age or age at onset. Their research indicated that while early intervention is beneficial, the primary determinant of treatment success is the intrinsic efficacy of the therapeutic intervention itself, rather than the age of the patient.
This study found that patients treated by pediatric urologists experienced the most significant improvements in QoL compared to those treated by general urologists or pediatricians. Pediatric urologists, with their specialized training and experience in treating pediatric bladder disorders, are better equipped to tailor BTX-A treatments to the unique needs of children, resulting in superior QoL outcomes. This finding is consistent with Ramsay and Bolduc, 3 Sharifiaghdas et al, 32 and Al Edwan et al 21 However, Kroll 33 found no significant difference in treatment outcomes based on healthcare provider type, suggesting that standardized treatment protocols ensure consistent outcomes regardless of provider specialization. Nonetheless, the comprehensive evaluation and tailored treatment strategies employed by pediatric urologists likely contribute to the superior QoL outcomes observed in the current study and reported by Wu et al 23 and Lambregts et al 34 The involvement of pediatric urologists leads to more effective symptom management and enhanced QoL, underscoring the value of specialized care in managing pediatric OAB.
Implications and Recommendations
The study found that BTX-A intravesical injections significantly improve the quality of life for children with pediatric OAB. Socioeconomic factors also play a role in healthcare access and outcomes, with higher household income correlated with better QoL improvements. This suggests that financial barriers may hinder access to advanced treatments like BTX-A, highlighting the need for policies ensuring equitable healthcare access regardless of socioeconomic status. Recommendations for clinical practice, healthcare policy, and future adjustments andintegrating BTX-A into standard treatment protocols, providing appropriate training for healthcare providers, improving access to advanced therapies for lower-income families through subsidized treatment programs or insurance coverage adjustments, and focusing on the long-term effects of BTX-A treatment on academic performance and psychosocial development. Future research should focus on the long-term effects of BTX-A treatment in pediatric OAB, identifying predictors of positive treatment outcomes and refining dosing protocols to enhance efficacy and minimize variability in patient responses.
Limitations
This study has several limitations. The non-randomized allocation of participants may introduce selection bias, as treatment choice was based on parental and provider decisions rather than random assignment. Although efforts were made to control confounding variables, inherent differences between the intervention and control groups may still influence the results. Additionally, the use of convenience sampling may limit the generalizability of findings. This approach, while practical, increases the risk of selection bias and sampling imbalances, potentially affecting the external validity of the study. Future research employing randomized controlled trials (RCTs) or broader recruitment strategies would enhance applicability and reproducibility.
The short follow-up period presents another limitation. While the study demonstrated improvements in symptoms and quality of life (QoL) at 1-month post-treatment, the long-term sustainability of these benefits remains unclear. Pediatric patients may experience fluctuations in symptom severity and QoL over time, requiring extended follow-up to assess the durability of BTX-A effects. The study also relied on self-reported QoL measures, which are subject to response bias. Parents might overestimate or underestimate their child’s symptoms or improvements, introducing potential imprecision in the assessment of treatment effects. Combining self-reports with objective clinical measures in future studies could enhance data accuracy. Finally, the sample size may have limited the study’s ability to detect subtle variations in treatment outcomes. A larger, more diverse sample would improve the statistical power and generalizability of the findings, ensuring broader applicability to pediatric OAB populations. Despite these limitations, the study provides valuable insights into the short-term impact of BTX-A intravesical injections on QoL and symptom management in pediatric patients with OAB. Future research should focus on randomization, extended follow-up, and larger sample sizes to strengthen the evidence base and confirm the long-term benefits of this intervention.
Conclusion
This study found that BTX-A treatment significantly improved the QoL for pediatric patients with OAB. Patients reported improvements in daily functioning and well-being post-treatment. Demographic factors like household income and age at symptom onset were associated with QoL outcomes, and treatment outcomes varied based on healthcare provider type. The study suggests BTX-A has strong potential for improving QoL in pediatric OAB patients. Future research addressing the study’s limitations will further solidify the role of BTX-A in treating pediatric OAB and improving the overall well-being of affected children.
Footnotes
Acknowledgements
We would like to express our sincere gratitude and thanks to all the parents that took part in this study.
Ethical Considerations
Ethical approval was obtained from the Institutional Review Board at Applied Science Private University (2023-2024-2-1) and the targeted hospitals.
Author Contributions
SM A-R: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
IA: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MMI: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
BO: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
AME: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
HK: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
ZFA: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
ARS: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
