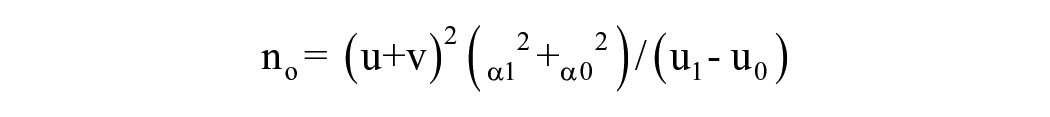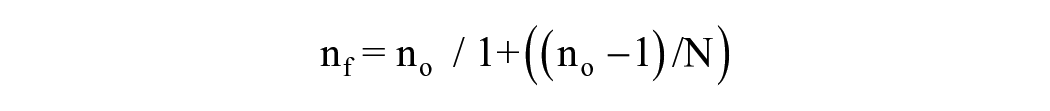Abstract
Objectives:
This study aimed to compare the haematological profiles of children with and without congenital heart defects at two tertiary institutions.
Methods:
The study was a comparative, cross-sectional study involving the consecutive enrolment of 30 children with acyanotic congenital heart disease (ACHD), 30 children with cyanotic congenital heart disease (CCHD) and 30 children without CHD.
Results:
The results revealed that the mean/median levels of haematocrit, PT, INR, TT, protein C, and D-dimer were significantly greater in CCHD patients (HCT: 42.51%, PT: 16.70 s, INR: 1.27, TT: 16.60 s, protein c: 9330.35, D-dimer: 1323.59) than in ACHD patients (HCT: 35.37%, PT: 14.25 s, INR: 1.09, TT: 14.15 s, protein C: 7833.03, D-dimer: 587.08).
Conclusion:
The mean/median levels of the haematological profiles were significantly greater in children with CCHD than those with ACHD and lowest in those without CHD. Haematocrit and oxygen saturation are haematological factors associated with an abnormal haematological profile.
Introduction
Congenital heart defects (CHDs) are structural abnormalities of the heart and/or great vessels that occur during intrauterine development.1 -4 They can present in isolation or as part of other syndromic anomalies. 1 They account for 28% of all congenital anomalies and 80% of all cardiac diseases. 2 Congenital heart defects have an estimated global prevalence of 8-12/1000 live births, with the highest burden in developed countries. 3 The prevalence in Africa is estimated at 1.9/1000 live births, whereas that in Nigeria varies between 3-14/1000 live births.3,4 In Nigeria, CHDs are beginning to gain recognition as important causes of mortality and morbidity. 1 Children with CHD are at risk of haematological dysfunction (bleeding diathesis and thrombotic events).5 -7 These haematological abnormalities may have asymptomatic, mild or severe manifestations.6,8,9 Bleeding complications may present as haemoptysis, epistaxis, and severe bleeding after trauma/surgery.9,10 Other major and life-threatening complications, such as cerebrovascular accidents, limb loss from ischemia and disseminated intravascular coagulation, may occur.9 -16
These haematological complications affect the quality of life of children with CHD and contribute to attributable morbidity and mortality. 5
Globally, studies have reported a greater prevalence of abnormal haematological profiles in children with CHD than in those without CHD. 11 The prevalence rate of haematological derangement in children with CHD varies from study to study. Henriksson et al 16 in Sweden and Arslan et al 17 in Turkey both reported very high prevalence rates of haematological derangement in children with CHD (85% and 70%, respectively). The high prevalence of haematological abnormalities reported by both studies could be due to the severity of the primary congenital heart defect, as both studies included subjects with complex cardiac defects. In contrast to the above studies, in a study conducted in the USA, Colon-Otero et al 18 reported a 19% prevalence rate of haemostatic derangement in children with CHD. The much lower prevalence reported by Colon-Otero et al 18 may be due to the larger sample size than that reported in the aforementioned studies.
This study aimed to investigate the haematological profiles of children with CHD compared with that of age- and sex-matched children without CHD and establish the relationship between haematocrit and oxygen saturation and the haematological profile. Understanding the haematological profile of children with CHD may help clinicians predict and mitigate bleeding risks, optimize perioperative management and improve patient outcomes. The findings from this study will contribute to the existing knowledge of paediatric haemostasis, providing valuable insights for the development of personalized treatment strategies for children with CHDs.
The majority of studies on haematological profiles in children with CHD are from the Western world, with only a few from Africa. 19 This is despite the established rich genetic diversity of the African population, which may suggest a probable difference in the haematological profile of children from other continents, as these factors are genetically mediated. 19
Research Questions
(1) Is there any difference between levels of haemostatic profile in cyanotic and acyanotic CHD and those without CHD?
(2) Is there any relationship between haematocrit levels and platelet count, bleeding time, PT/INR, activated partial thromboplastin time, thrombin time, D-dimer test, and serum protein C levels in children with cyanotic and acyanotic CHD?
Null Hypothesis
(1) There was no difference between the haematological profiles of the cyanotic and the acyanotic CHD and children without CHD.
(2) There was no relationship between haematocrit levels and platelet count, bleeding time, PT/INR, activated partial thromboplastin time, thrombin time, D-dimer test, or serum protein C levels in children with cyanotic CHD or those with acyanotic CHD.
Methods
Study Design
This was a cross-sectional comparative study. This study involved one-time contact with the study subjects, and no follow-up was performed. Two independent groups (children with CHD and those without CHD) were compared in the study. A subgroup analysis of those with ACHD and CCHD was also performed. The comparative groups were age- and sex-matched children without CHD who were receiving CHOP.
Study Area
This study was performed at two tertiary institutions.
Study Sites
This study was carried out in the Paediatric Cardiology Clinic, New born Special Care Units and Children’s Outpatient Clinic of two tertiary hospitals. An average of 8 and 35 children attended the cardiology clinics of the two tertiary hospitals per week, respectively, for a ratio of 1:4. These figures include both the old patients of the cardiology clinics and new referrals to the units.
Study Population
The participants in the study were children with CHD aged less than 1 month to less than 18 years attending the paediatric cardiology clinics and new born special care units of two tertiary hospitals. The comparative group included apparently healthy, age- and sex-matched children without congenital heart defects who presented at the children’s outpatient clinic at the two tertiary hospitals during follow-up after recovering from minor ailments.
Children from birth to 17 years of age with confirmed by 2-dimensional echocardiography (2D Echo) who have not undergone surgical correction were included in the study, whereas children with coexisting chronic disease that can cause derangement in haematological functions, e.g., chronic liver disease, chronic renal failure, known diagnosis of HIV (including clinical features and laboratory diagnosis), sickle cell anaemia, children with coexisting inflammatory conditions such as rheumatoid arthritis, those who have been on anti-inflammatory and anticoagulant medications such as aspirin in the preceding 2 weeks, those who have received blood transfusion in the preceding 3 months, those with a family history of bleeding disorders, and those with acquired heart defects were excluded. For the controls, children from birth to 17 years who do not have CHD, children who have not been hospitalized in the past 3 months, children who are not suffering from any chronic disease and children who do not have any inflammatory conditions were included in the study, while those with any chronic diseases that can cause derangement in haematological functions, for example, chronic liver disease, chronic renal failure, HIV (clinical and laboratory diagnosis), sickle cell anaemia, children who were on anti-inflammatory/anticoagulant medications such as aspirin in the preceding 2 weeks, children who had blood transfusions in the preceding 3 months, those who had a family history of bleeding disorders and those with acquired heart defects were excluded.
Determination of Sample Size
The formula below was used:
where no = the minimum sample size.
u = minimum power at 90% =1.28
v = significant level at 5% = 1.96. A formula for adjusting the calculated sample size for the study population size (Cochran correction formula) was used to reduce the sample size after a sample size of 43 was obtained. 20
where N = population of children with CHD in the two centres.
no = calculated sample size and nf = final sample size.
Therefore, a sample size of 30 per group (30 for CCHD, 30 for ACHD, and 30 for the control group) was sufficient to power this study. 21
Sampling Technique
Children with CHD who fulfilled the inclusion criteria were recruited consecutively by the researcher. Sub-jects were consecutively enrolled from the two tertiary hospitals. For the comparative group, age- and sex-matched controls who fulfilled the inclusion criteria were also recruited via simple random selection.
Subject Selection
The study was conducted over a period of 7 months. During this period, a total of 116 children and caregivers were approached for eligibility. Those who did not meet the exclusion criteria were excluded. A total of 90 children—30 children with CCHD, 30 with ACHD, and 30 children without CHD—were enrolled in the study. The selection of the subjects is shown in a schematic diagram in Figure 1.
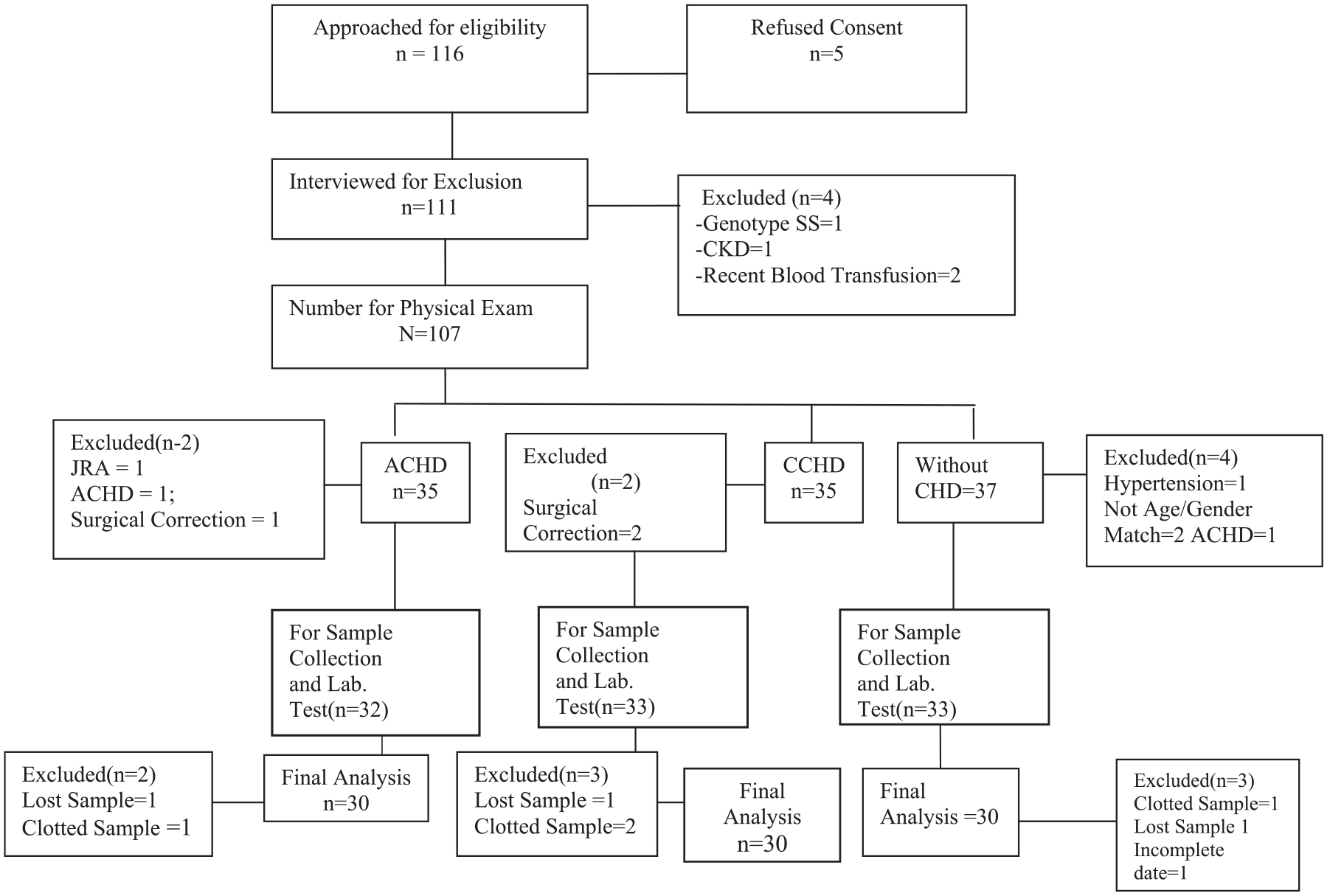
Flow chart diagram of participant enrollment.
Sociodemographic data were also obtained from the caregivers. A detailed physical examination, including measurements of oxygen saturation, blood pressure, and height, was performed.
Statistical Analysis
The data were analysed with the Statistical Package for Social Sciences (IBM—SPSS, Armonk, NY, USA) version 23. Descriptive statistics such as the means and standard deviations or medians and interquartile ranges were used to summarize the continuous variables as appropriate. Frequencies and percentages were used for categorical variables. Comparisons of the haematological profiles between those with cyanotic CHD and those with acyanotic CHD were performed via independent t tests or Mann‒Whitney U tests, as appropriate. Comparisons of haematological profiles among the three groups (CCHD, ACHD, and those without CHD) were performed via the Kruskal‒Wallis test or analysis of variance (ANOVA), as appropriate. Post hoc analysis (Dunnett’s T3 test and the Bonferroni correction) was used to determine the point of difference for significant variables. The relationship between haematocrit and the haemostatic profile was analysed via Pearson’s correlation test if the data were normally distributed or Spearman’s test if the data were not normally distributed. All tests of significance were two-tailed at the 95% confidence interval, and the P value was considered significant if it was less than 0.05. Multivariate linear regression was used to determine the predictors of the haematological profile in the study participants.
Results
Sociodemographic Characteristics
A total of 90 children from birth to 17 years of age, comprising 30 children with CCHD, 30 with ACHD, and 30 without CHD, were recruited for this study. Table 1 does not show any statistically significant differences in age, sex, or socioeconomic class.
Sociodemographic Characteristics of the Study Participants (N = 90).

According to the table, the sociodemographic characteristics of the participants, such as age group (P = 0.119), sex (P = 0.270) and socioeconomic status (P = 0.667), were similar.
There was a statistically significant difference in PT (P = 0.020), INR (P = 0.001), TT (P = 0.002), PCV (0.005), protein C (0.030) and D-dimer (P = 0.010) between the subjects with cyanotic CHD and those with acyanotic CHD. However, the bleeding time, platelet count and activated partial thromboplastin time did not differ significantly between the two groups (Table 2).
Comparison of Haematological Indices Between Participants with Cyanotic and Acyanotic Congenital Heart Diseases (N = 60; N = no of ACD + CCHD).
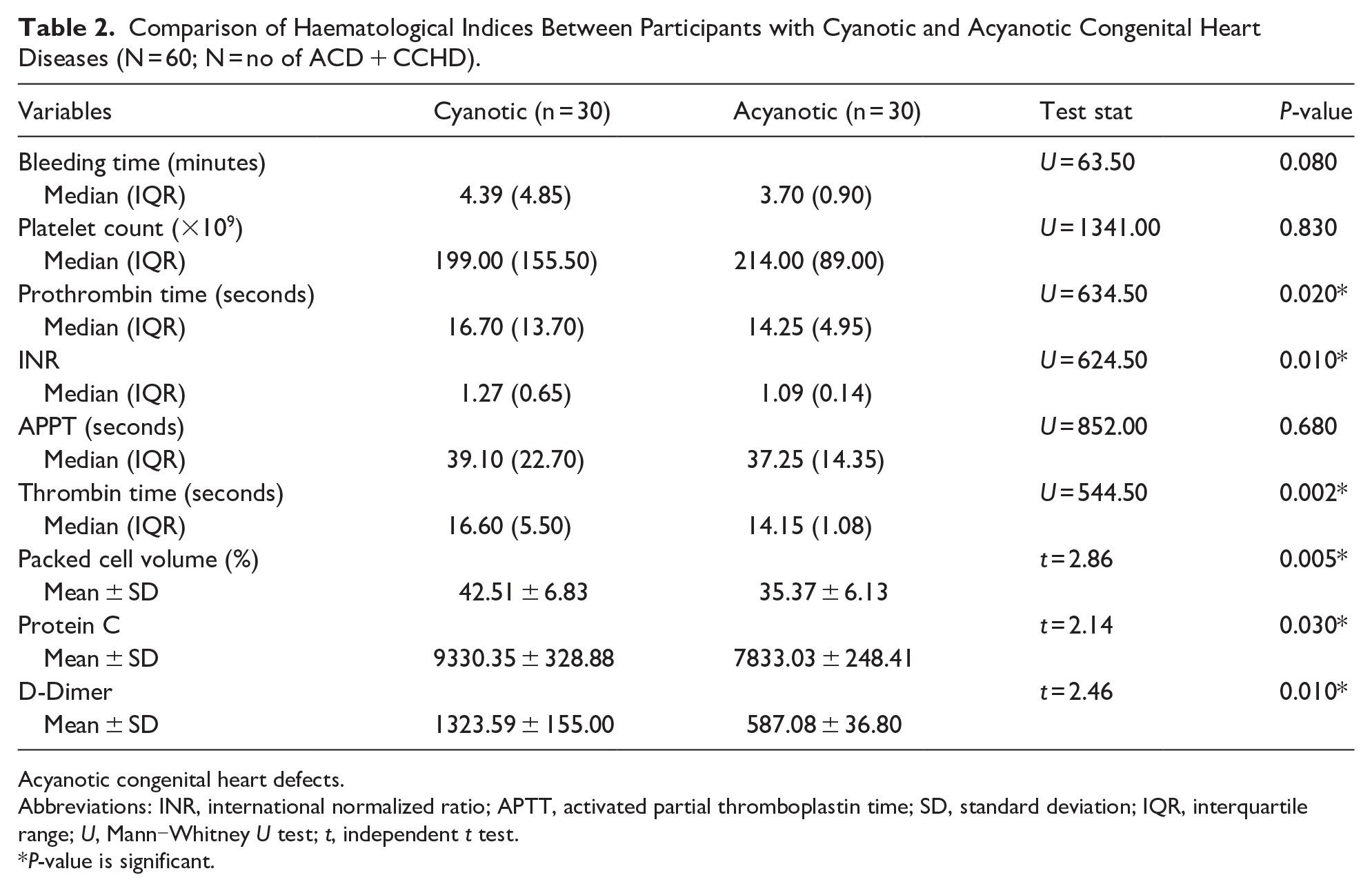
Acyanotic congenital heart defects.
Abbreviations: INR, international normalized ratio; APTT, activated partial thromboplastin time; SD, standard deviation; IQR, interquartile range; U, Mann‒Whitney U test; t, independent t test.
P-value is significant.
Table 3 shows significant differences in BT, platelet count, PT, INR, TT, PCV, protein C, and D-dimer among the three groups (P-value < 0.05).
Comparison of Haematological Variables Among Participants in the Cyanotic CHD, Acyanotic, and Healthy Control Groups (N = 90).

Abbreviations: INR, international normalized ratio; APPT, activated partial prothrombin; SD, standard deviation; IQR, interquartile range; KWT, Kruskal‒Wallis test; F, analysis of variance (ANOVA).
P-value is significant.
Post hoc analysis (Table 4) revealed differences between the cyanotic and acyanotic, cyanotic and control groups but not between the acyanotic and control groups.
Post Hoc Pairwise Comparisons of the Acyanotic, Cyanotic, and Healthy Control Groups.

Abbreviation: INR, international normalized ratio.
Significant P value. **Dunnett’s T3 test. ***Bonferroni correction.
The mean/median levels of BT, PT, INR, TT, HCT, protein C, and D-dimer were most prolonged/highest in CCHD, followed by ACHD. Those without CHD had the lowest values. Children with CCHD had lower median platelet counts than did those with ACHD and those without CHD.
Table 5: There was no significant relationship between haematocrit and the haemostatic profile in ACHD patients, and there was a significant negative correlation between platelet count and oxygen saturation in ACHD patients.
Correlations Between Haematocrit (HCT) and SPO2 Levels and Haemostatic Variables in Patients with Acute CHD (N = 30).

Table 6 shows a significant positive but weak correlation between HCT and APTT among children with cyanotic CHD (r = 0.447, P = 0.013). There was a significant negative but weak correlation between HCT and protein C among children with cyanotic CHD (r = −0.459, P = 0.028). However, no significant relationships were found between HCT and the platelet count, prothrombin time, thrombin time or D-dimer test among children with cyanotic CHD (P > 0.05). There was a significant positive but weak correlation between SPO2 and protein C among children with cyanotic CHD (r = 0.430, P = 0.041). No significant correlations were detected between SPO2 and the platelet count, bleeding time, prothrombin time, activated partial thromboplastin time, thrombin time or D-dimer test in children with cyanotic CHD (P > 0.05).
Correlations Between Haematological Indices, SPO2, and Haematocrit (HCT) Among the Cyanotic Participants.

Table 7 shows the multivariate linear regression analysis of the predictors of coagulation variables and several other parameters. For bleeding time, of the three independent variables, HCT, SPO2, and age, only HCT was a significant predictor of bleeding time (t = 2.56, P = 0.02 β = 0.66; coefficient of multiple determination (R2) = 0.266). In the multivariate analysis of the platelet count, HCT, SPO2, and age were entered into the regression equation, and only age was a significant predictor of the platelet count (t = −0.75, P = 0.02 β = −0.30). R2 (coefficient of multiple determination) = 0.203. In the multivariate linear regression analysis of the predictors of the INR, HCT, SPO2, and age were entered into the regression equation. Among these three independent predictive variables, SPO2 was not a significant predictor; however, there was a trend (P = 0.05). R2 (coefficient of multiple determination) = 0.074. In the multivariate linear regression analysis of the predictors of P, HCT, SPO2, and age were entered into the regression equation. There was no significant predictor; however, SPO2 tended to be a predictor (P = 0.05). R2 (coefficient of multiple determination) = 0.122. In the multivariate linear regression analysis of the predictors of aPTT, PCV, SPO2, and age were entered into the regression equation, and only HCT was a significant predictor of aPTT (t = 2.58, P = 0.01, β = 0.39). R2 (coefficient of multiple determination) = 0.235.
Multivariate Linear Regression Analysis of the Predictors of Bleeding Time Among Participants with CHD (N = 60).

In the multivariate linear regression analysis of the predictors of TT, HCT, SPO2, and age were entered into the regression equation. Among these three variables, there was no significant predictor. R2 (coefficient of multiple determination) = 0.048
In the multivariate linear regression analysis of predictors of protein C. PCV, SPO2, and age were entered into the regression equation. Among these three variables, there was no significant predictor. R2 (coefficient of multiple determination) = 0.174
In the multivariate linear regression analysis of the predictors of D-dimer, HCT, SPO2, and age were entered into the regression equation, and there was no significant predictor. R2 (coefficient of multiple determination) = −0.03.
Discussion
The findings from this study, which revealed a significant difference in the median/mean levels of haematological profiles in children with CHD (cyanotic and acyanotic) compared with those in children without CHD, are consistent with a previous report by Majiyagbe et al 21 and reports from Africa and the western world.22 -25 Although there are some methodological differences between the index study and the aforementioned studies, a similar conclusion can be drawn that there is a significant alteration in the haematological profile in children with CHD compared with those without CHD and that, among children with CHD, this alteration in the haematological profile is more pronounced in children with CCHD than in those with ACHD. There were a significant number of children with TOF in the CCHD subgroup in this study. Unlike some other types of cyanotic CHD, children with TOF have higher rates of survival into later childhood, adolescence and even adulthood and are associated with chronic hypoxia and hyperviscosity, which are the major risk factors for the development of haemostatic derangement in children with CCHD. Additionally, a significant proportion of the index study participants were less than 5 years old. In keeping with the concept of developmental haemostasis, there are physiologically lower levels of coagulation factors in younger children. 26
This study also reported lower median platelet counts in CCHD and ACHD patients than in children without CHD. This finding agrees with earlier African reports by Ismail and Youssef 27 and Majiyagbe et al 21 in Egypt and Lagos, respectively. Similar findings were also reported by Olgun et al 28 and Horigome et al, 29 contrary to these findings. Maurer et al, 30 in the USA, reported relatively normal platelet counts in those with congenital heart defects. Maurer et al 30 argued that derangement in platelet function and not necessarily low platelet count may be responsible for some of the coagulation challenges observed in children with CHD. This theory by Maurer et al, 30 however, could not be tested by the index study, as platelet function was not studied.
The median bleeding time in children with cyanotic CHD was significantly greater than that in children with acyanotic CHD and that in children without CHD. This finding was similar to that of Maurer et al 30 who noted a prolonged bleeding time in cyanotic CHD patients than in acyanotic CHD patients compared with those without CHD. 30 This prolonged bleeding time has been linked to low platelet count and/or abnormal platelet function, which are prevalent in children with CHD, especially in the cyanotic subgroup. The median PT was significantly longer in CCHD patients (17.40 s) than in acyanotic CHD patients and those without CHD. These findings are similar to those of Arslan et al, 31 Colon-Otero et al, 32 Dittrich et al, 33 Ismail and Youssef, 27 and Majiyagbe et al. 21 Similarly, the median INR was significantly greater in cyanotic patients than in acyanotic patients and was much lower in those without CHD. The median thrombin time was also significantly longer in cyanotic CHD and acyanotic CHD patients than in children with CHD.
In this study, the median APTT was greater in CCHD patients than in ACHD patients and those without CHD, although the difference was not statistically significant. Majiyagbe et al 21 reported similar findings. This result was different from those of other studies, such as those that reported significantly greater APTT in CCHD and ACHD patients than in those without CHD.
This study also revealed that D-dimer in CCHD patients was significantly greater than that in ACHD patients and that children without CHD had lower D-dimer levels. This is in keeping with research by Dittrich et al 33 and Ismail and Youssef 27 The elevation of D-dimer levels in CHD, especially CCHD, is due to hypoxia-induced polycythaemia, which causes hyperviscosity and excessive slugging in the peripheral vasculature, resulting in thrombosis. Higher levels of coagulation parameters in CCHD patients suggest greater vulnerability to thromboembolism. On the other hand, Horigome et al 29 and Majiyagbe et al 21 did not find any significant differences in the D-dimer levels between CCHD and ACHD patients.
Protein C consists of haematological proteins and natural anticoagulants produced in the liver. There is documented evidence of protein C deficiency in children with CHD. This deficiency is considered a major contributor to the development of thrombosis in children with CHD.
In contrast to popular reports, this study revealed that protein C was higher in children with CCHD than in those with ACHD and those without CHD. This study was corroborated by several other studies. For example, Macdonald et al 34 studied protein C and several other coagulation factor levels in 29 term babies with symptomatic CHD; 28% of them had low levels of protein C; of these 28%, 50% had evidence of thrombosis, and a fraction of them had thrombotic complications. Medical and surgical interventions did not improve protein C levels in the subjects. Odegard et al reported similar protein C deficiency in children with CHD. Protein C deficiency was noted to be worse in children younger than 2 years.
In another study, Horigome et al 29 investigated soluble P selectin and the thrombomodulin-protein C-protein S pathway in children with CCHD and secondary erythrocytosis. This study aimed to establish the role of the following in the pathogenesis of thrombosis in CCHD patients. All the components of the protein C pathway were markedly reduced in children with CHD. This reduction was greater in the CCHD group. The finding of higher protein C levels in this study could be attributed to the body’s compensatory mechanism in hypercoagulable states. 35 Ganapathyraman et al 36 noted elevated protein C levels in patients with ischaemic heart disease, which is another hypercoagulable state. Taking the D-dimer and protein C results obtained in this study into account, it could be assumed that many of the CCHD subjects were already in a hypercoagulable state and hence had high levels of protective protein C. More studies need to be done to establish this assumption. More research is needed to establish the benefits of protein c in the prevention of thromboembolic complications in children with CHD. If the above is established, there may be a need for advocacy to the government and philanthropists to finance prophylactic protein replacement for children with CHD at high risk of thromboembolic events.
Haematocrit correlated positively with bleeding time, platelet count, and aPTT and correlated negatively with protein C in congenital heart defects. These findings were more pronounced in the subgroup with cyanotic congenital heart defects. This infers that haematologic derangement occurs more in the setting of a high haematocrit. These findings suggest that polycythaemia is a major culprit in the development of haemostatic derangement in children with CHD. Therefore, routine monitoring of haematocrit in children with CHD may identify patients at risk of developing haemostatic complications. This finding may also imply that periodic haematocrit-lowering procedures such as saline exchange transfusions may remarkably reduce the risk of altered haematological profiles and haemostatic complications. The above finding from this study is similar to other studies that reported findings of greater derangement in haematologic profiles with higher haematocrit levels. Horigome et al, 29 Colon-Otero et al, 32 Ismail and Youssef, 27 and Majiyagbe et al 21 reported similar correlations between haematocrit and haematological profiles.
This study reported a positive correlation between oxygen saturation and protein C. This finding is consistent with the hypothesis that tissue hypoxia impairs hepatic synthesis of pro-coagulants and anticoagulants. These findings agree with those of Aslan et al, 31 Colon-Otero et al, 32 and Majiyagbe et al, 21 who reported correlations between SPO2 and abnormal haematologic functions. In contrast to other reports, this study revealed a negative correlation between the SP02 concentration and the PLT. This finding is rather surprising considering that low oxygen levels have been described as stimulants for erythropoiesis with resultant polycythaemia, which is thought to be responsible for the cascade of hyperviscosity and abnormal haemostasis observed in CHD patients. However, as opposed to the findings of Maurer et al, 30 the abnormal coagulation observed in CHD patients may be the result of abnormal platelet function and not necessarily due to a low platelet count, which may explain the negative correlation between SPO2 and the platelet count reported in this study. Moreover, the fact that this study reported a very significant relationship between haematocrits and almost all the haematological profiles and that there was no correlation between oxygen saturation and those haematological profiles may suggest other reasons for high haematocrits in children with CHD in addition to chronic hypoxia. Additionally, pulse oximetry, as used in this study, has been shown to overestimate oxygen saturation in persons with hypoxemia compared with arterial blood oxygen saturation measured by invasive methods.
In conclusion, the findings of this study demonstrate that children with CHD have higher median/mean haematological profiles than do children without CHD, and these altered haematological profiles are related to high haematocrit levels and low oxygen saturation.
Limitations of the Study
The small sample size of study participants was a major limitation of this study. However, this may be explained by the low prevalence of congenital heart defects. A larger sample size would have increased the precision of the study findings. The cross-sectional study design used could not allow for follow-up of the subjects, and a prospective study design would have been better; however, it was not used because of time and financial constraints.
Conclusion
Compared with children without CHD, children with congenital heart defects (CHDs) have altered haematological indices; therefore, there is a need for routine evaluation of the haematological profile in children with CHD. Thus, children with CCHD are at greater risk of developing haematologic complications than are those with ACHD.
Recommendation
Develop and implement enhanced monitoring protocols for haemostatic indices in children with CHD, with particular attention to those with cyanotic congenital heart defects (CHDHs), given their higher median values of haemostatic variables. Haematocrit levels should be incorporated into risk assessment models for bleeding and thrombosis in CHD patients. To develop evidence-based guidelines for the haemostatic management of children with CHD and initiate educational programmes for healthcare providers involved in the care of children with CHD, with a focus on the implications of altered haemostatic indices and the importance of their management in the context of CHD.
Footnotes
Acknowledgements
We are grateful to the research assistant who helped in data entering.
Ethical Considerations
Ethical approval to carry out the study was obtained from the Health Research and Ethics Committee of both ESUTTHP and UNTH. The code registration for the ethical clearance is NHREC/05/01/2008B-FWA00002458-1RB00002323. Individualized counselling was carried out on each participant/ caregiver pair. Written informed consent was obtained from literate caregivers of the participants while the non-literate ones were asked for thumb print. Assent was obtained from children aged 7 years and above before enrolling them into the study.
Consent to Participate
Written informed consent will be obtained from the literate caregivers of the participants, while the non-literate caregivers will be asked for a thumb print. Informed consent will be obtained from children aged 7 years and above before they are enrolled in the study. Participation in the study will be voluntary, and participants will be free to withdraw from the study at any time, even after providing initial consent, and this will not affect their medical care in any way. The parents, participants and managing team will be informed of the findings of the study, and if any previously undiagnosed condition is discovered in the course of the study, the participants will be counselled on how to seek care. The data obtained will be stored on an encrypted laptop to protect the confidentiality of the data.
Author Contributions
CJM conceived and designed this study while NCS, AEA, EU, and CJM helped in the critical revision of the article. CJM also did the Data analysis/interpretation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was not funded by any organization. We bore all the expenses that accrued from in study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is provided within the manuscript or supplementary information files