Abstract
Objective:
To investigate netrin-1 (NT-1) and neuron-specific enolase (NSE) in the umbilical cord as biomarkers in HIE stage II/III.
Materials and Methods:
We included 100 infants with HIE stage II/III and 100 controls. Cord samples were taken to determine the levels of NT-1 and NSE.
Results:
The HIE group showed significantly higher NT-1 (343.3 ± 100.6 pg/mL) and NSE (55.17 ± 13.4 ng/mL; P = .037, P = .007, respectively) than the control group, which had NT-1 (268.1 ± 79.3 pg/mL) and NSE (31.3 ± 19.5 ng/mL). The cut-off values for NT-1 and NSE were 289.3 pg/mL and 31.9 ng/mL, respectively. NT-1 had a sensitivity of 92%, a specificity of 100%, a positive predictive value (PPV) of 94%, and a negative predictive value (NPV) of 100%. The sensitivity, specificity, PPV, and NPV for NSE were all 100%.
Conclusion:
NT-1 and NSE can be reliable biomarkers for predicting HIE stage II/III.
Introduction
Perinatal asphyxia (PA) is a condition observed in newborns as a result of hypoxia. 1 PA can cause brain damage, neonatal encephalopathy, and even death.2,3 Shortly after a hypoxic insult, energy failure impairs active membrane transport and depolarization, leading to the release of glutamate and subsequent neuronal damage through inflammatory and apoptotic mechanisms. 3 These events can later result in epilepsy, cerebral palsy, mental disabilities, speech disorders, motor and sensory impairments, or even death. 4 Currently, there are no accurate diagnostic tools with high sensitivity and specificity for neonatal hypoxic-ischemic encephalopathy (HIE). However, laboratory blood tests evaluating renal and hepatic function may assist in diagnosis but lack neuronal specificity. 5 Perinatal asphyxia is more likely if more than one of the following diagnostic criteria is met: (1) APGAR score < 5 at 5 and 10 minutes; (2) pH < 7.0 in an umbilical artery blood sample and/or a base deficit ≥ 12 mmol/l; (3) Evidence of acute brain injury seen on magnetic resonance imaging (MRI) or magnetic resonance spectroscopy (MRS); and (4) Multiple organ dysfunction. However, imaging modalities are not ideal for rapid diagnosis and are usually performed within the first 2 weeks of life.5,6
The treatment for hypoxic-ischemic encephalopathy (HIE) has a narrow therapeutic window within the first 6 hours of life before the inflammatory and apoptotic cascades take full effect. 7 Therapeutic hypothermia (TH) has become the gold standard intervention for neonatal HIE, especially in moderate to severe cases, through whole-body cooling, or selective head cooling. 8 However, a definitive diagnostic test for neonatal HIE is still lacking. This gap can lead to improper diagnosis and management.
Neuron-specific enolase (NSE) is a member of the enolase family of enzymes responsible for glycolysis. Enolases have 3 subunits (alpha, beta, and gamma) that combine to form 5 isoenzymes. Enolase 2 (γγ), also known as NSE, is specific to neurons and neuroendocrine cells.9,10 Previous studies have shown that plasma biomarkers, such as S100B, NSE, and cytokines, can be useful in assessing brain damage.11,12 Additionally, specific biomarkers in serum and urine may be helpful in predicting hypoxic insults to the kidneys, liver, and heart.12-15 Detection of serum NSE in infants with HIE is associated with brain injuries and damage to the blood-brain barrier (BBB). 16 NSE is a promising biomarker in neonatal and perinatal medicine, but studies evaluating NSE in infants with HIE are limited.17,18
Netrin (NT) is a member of the laminin family of extracellular matrix proteins, comprising 5 members: Netrin-1, -2, -4, G1a, and G1b. NT-1 plays a role in cell migration, morphogenesis, angiogenesis, axonal guidance, and adhesion of endothelial cells.19,20 In cases of cerebral ischemia, NT-1 contributes to astrocyte activation and inflammation. Thus, NT-1 may be a potential target for stroke therapy. 21 In traumatic and ischemic brain injuries, NT-1 helps maintain the integrity of the blood-brain barrier. 21
We investigated NT-1 and NSE as early umbilical biomarkers to predict and improve the outcome of early therapeutic hypothermia.
Patients and Methods
Our study is a case-control investigation conducted on 100 infants with HIE stage II/III, 35 infants with mild HIE, and 35 infants with non-HIE neonatal encephalopathy. Their gestational ages (GA) were ≥36 weeks. All cases received standard care, with infants with HIE stage II/III receiving moderate therapeutic hypothermia.
One hundred healthy infants without congenital anomalies, matched for gestational age and sex, served as a control group.
We determined the GA according to the last menstrual period and antenatal ultrasound.
Formal consent was obtained from the parents before enrollment into the study. Ethical clearance was obtained for the research study. Ethical guidelines of the 1964 declaration of Helsinki and its later amendments were followed.
Exclusion Criteria
Infants with GA < 36 weeks.
Major congenital malformations of the circulatory, respiratory, digestive, or renal systems.
Neonatal sepsis.
All participants were admitted to the Hospital. Infants in the control group were admitted to the well-baby nursery unit with their mothers until discharge. However, all cases were admitted to the NICU, where they received proper supportive care and management protocols according to the published guidelines.
All cases and controls underwent the following:
Blood gases were measured, and blood samples were analyzed using a blood gas analyzer (Siemens RAPIDLabVR 1200 Systems, Germany). NT-1 levels were measured using an enzyme-linked immunosorbent assay (ELISA; USCN Life Science Inc., Houston, TX). NSE concentrations were measured using ELISA (R & D Co., Marburg, Germany).
Neonatal seizures were identified through aEEG monitoring when there was an abrupt onset of rhythmic activity in the EEG lasting at least 10 seconds, with a change in at least 2 of the following: amplitude, frequency, or spatial distribution.
Active neonatal resuscitation was defined as the need for positive pressure ventilation (PPV) for at least 30 seconds at the time of birth.
The pathological findings on the aEEG monitoring included discontinuous low-voltage, discontinuous high-voltage, continuous pattern, burst-suppression pattern, continuous low-voltage, flat trace, or neonatal seizures. 1
Umbilical Cord Blood Samples
At birth, blood samples were taken from the umbilical cord and stored in simple tubes without any gel or preservatives. The blood samples were allowed to clot for 30 to 45 minutes at room temperature. Subsequently, centrifugation was performed for clotted samples at 3000 rpm for 10 minutes at room temperature. The supernatants were separated and stored at −80°C until further evaluation. Hemolyzed samples were discarded.
Diagnosis and Staging of HIE
To establish the diagnosis and severity of encephalopathy, a neurological examination was performed within 6 hours of birth according to the NICHD classification for modified Sarnat staging. 22
Mild Encephalopathy Group
This group consisted of newborns with 1 to 2 abnormal categories in the neurologic assessment, but who did not meet the criteria for hypothermia. They received supportive care, including an indwelling umbilical catheter. The mild HIE group consisted of 35 newborns who had fetal acidosis, mild abnormalities on neurologic examination within the first 6 hours of age, and an indwelling arterial catheter.
Moderate-Severe Encephalopathy Group
Cases categorized as lethargic with hypotonia, decreased suckling and Moro reflexes, overactive stretch reflexes, seizure activity, and miotic pupils. HIE stage III cases were categorized as stuporous and flaccid with absent stretch reflex, suckling or Moro reflexes, no seizures, mydriatic pupils with no reaction to light stimulus, and apnea. The decision for moderate therapeutic hypothermia was made according to the following criteria:
1. One or more of the following: prolonged positive pressure ventilation for up to 10 minutes after birth; 5-minute Apgar score ≤ 5; pH < 7.0 or base excess (BE) ≤−16 mmol/l within the first 1 hour after birth, and
2. Altered level of consciousness, with at least one of the following: abnormal reflexes, hypotonic, or neonatal seizures.
All cases of HIE stage II/III were treated according to the protocol for moderate therapeutic hypothermia and the guidelines of the “Total Body Hypothermia for Perinatal Asphyxia (TOBY)” study group. 15
Non-HIE Neonatal Encephalopathy Group
This group consisted of newborns with abnormal brain function due to causes other than hypoxic-ischemic encephalopathy (HIE). HIE should be distinguished from neonatal encephalopathy caused by other factors using data from history, physical and neurological exams, and further investigations. Infants in this group required immediate intubation, ventilation, and intravenous fluids. 23
Data from aEEG tracing were recorded using an Olympic CFM 6000 (Natus, Seattle, WA) device, and any abnormal waves were recorded and analyzed.
Treatment of HIE
Whole-body cooling was initiated as soon as possible within 6 hours after birth using the Tecotherm TS med 200 N (Inspiration Healthcare Ltd., Leicester, UK) device. Rectal temperature was maintained at 33°C to 34°C for 72 hours. After 72 hours of cooling, the patient’s rectal temperature gradually increased to normal body temperature. 15
Sample Size Calculation and Power of the Study
A sample size of 86 neonates per group is the minimum required to detect a 0.5-SD difference on the log scale with a single observation for neonates, with 80% power while controlling for a type I error of 0.0167.
Statistical Analyses
The data and variables were analyzed using the Statistical Package for Social Sciences (SPSS), version 16.0 (Chicago, IL, USA). The distribution of quantitative variables was assessed using the Kolmogorov-Smirnov test. Comparison between the studied groups regarding continuous variables was performed using an independent samples t-test. For non-normally distributed variables, comparisons between groups were made using the Mann-Whitney test. Continuous variables are presented as the median (interquartile range, IQR) and/or mean ± standard deviation (SD). Categorical variables and frequency distributions were presented as numbers and percentages. Categorical variables were analyzed using the chi-square test. A one-way analysis of variance (ANOVA) test was used to compare multiple means across the studied groups.
The relationship between NT-1, NSE levels, and the severity of HIE was analyzed using Pearson’s correlation coefficients. The Receiver Operating Characteristic (ROC) curve with area under the curve (AUC) was constructed to determine the sensitivity and specificity of NT-1 and NSE for diagnosing HIE II/III. Significance was considered at a P-value of less than .05.
Ethical Approval and Informed Consent
The study was conducted after obtaining written informed consent from the parents of all participants. Also, ethical clearance was obtained for the research study. The IRB of the Faculty of Medicine, Benha University, Egypt (RC: 38-11-2023) had approved the study. The study protocol and all procedures involving human participants were under the Revised Declaration of Helsinki (as in 2013).
Results
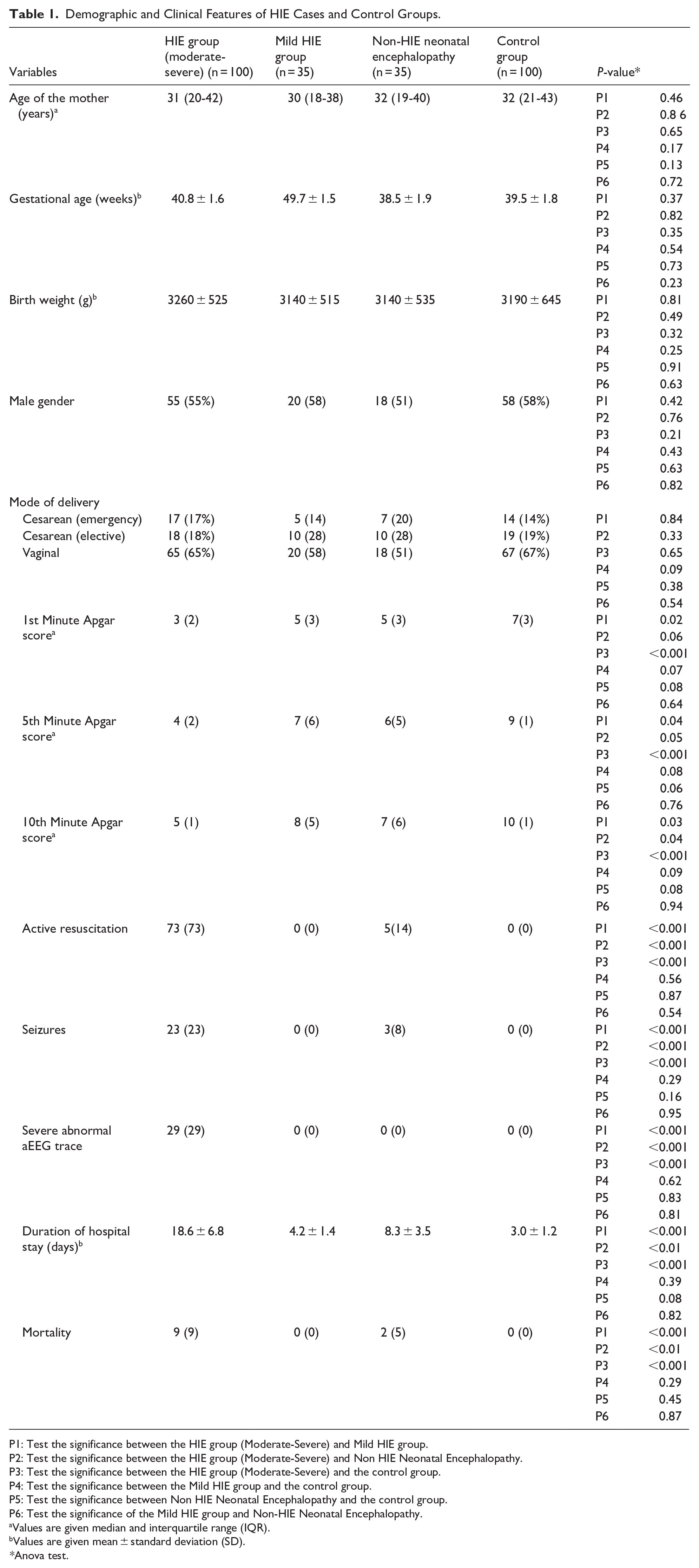
We included 100 infants with HIE stage II/III, 35 infants with mild HIE, 35 infants with non-HIE neonatal encephalopathy, and 100 control infants. Demographic data and clinical features of the studied groups are shown in Table 1. There were no significant differences between the cases and controls regarding maternal age, gestational age (GA), birth weight, gender, or mode of delivery.
Demographic and Clinical Features of HIE Cases and Control Groups.
P1: Test the significance between the HIE group (Moderate-Severe) and Mild HIE group.
P2: Test the significance between the HIE group (Moderate-Severe) and Non HIE Neonatal Encephalopathy.
P3: Test the significance between the HIE group (Moderate-Severe) and the control group.
P4: Test the significance between the Mild HIE group and the control group.
P5: Test the significance between Non HIE Neonatal Encephalopathy and the control group.
P6: Test the significance of the Mild HIE group and Non-HIE Neonatal Encephalopathy.
Values are given median and interquartile range (IQR).
Values are given mean ± standard deviation (SD).
Anova test.
However, the 1st, 5th, and 10th minute APGAR scores and the need for prolonged active resuscitation in the delivery room were significantly higher in the HIE (Moderate-Severe) group compared to the other groups (Table 1). Additionally, clinical seizures, EEG changes, duration of hospital stay, and mortality were significantly higher in the HIE (Moderate-Severe) group compared to the other groups (Table 1).
The results of the biochemical parameters for the cases and controls are shown in Table 2. Cases in the HIE (Moderate-Severe) and mild HIE groups showed significantly lower values of pH, HCO3, and BE compared to the non-HIE neonatal encephalopathy and control groups. However, pCO2 was significantly higher in both the HIE (Moderate-Severe) and mild HIE groups than in the control group (P < .05).
Biochemical Parameters of HIE Cases and Control Groups.

Values are given mean ± standard deviation (SD).
P1: Test the significance between the HIE group (Moderate-Severe) and Mild HIE group.
P2: Test the significance between the HIE group (Moderate-Severe) and Non HIE Neonatal Encephalopathy.
P3: Test the significance between the HIE group (Moderate-Severe) and the control group.
P4: Test the significance between the Mild HIE group and the control group.
P5: Test the significance between Non-HIE Neonatal Encephalopathy and the control group.
P6: Test the significance of the Mild HIE group and Non-HIE Neonatal Encephalopathy.
Anova test.
The non-HIE neonatal encephalopathy group showed significantly higher pCO2 than the control group. However, there were no significant differences in pH, HCO3, BE, and pCO2 between the non-HIE neonatal encephalopathy and HIE groups.
Liver function tests, coagulation profiles, cardiac enzymes, LDH, and uric acid were significantly higher in the HIE (Moderate-Severe) group than in the other groups (P < .05; Table 2).
The HIE (Moderate-Severe) group showed significantly higher NT-1 (343.3 ± 100.6 pg/mL) and NSE (55.17 ± 13.4 ng/mL) levels in the cord blood (P = .037, P = .007, respectively) than those in the mild HIE group (NT-1 275.4 ± 86.4 pg/mL, NSE 34.3 ± 18.5 ng/mL), the non-HIE neonatal encephalopathy group (NT-1 271.6 ± 82.6 pg/mL, NSE 35.7 ± 17.8 ng/mL), and the control group (NT-1 268.1 ± 79.3 pg/mL, NSE 31.3 ± 19.5 ng/mL).
There were no significant differences in NT-1 and NSE levels between the mild HIE group, the non-HIE neonatal encephalopathy group, and the control group.
Correlation Between NT-1, NSE Levels, and Severity of HIE
We performed Pearson’s correlation analysis between serum NT-1 and NSE levels and the severity of HIE. The analysis indicated that NT-1 and NSE levels were positively correlated with the severity of HIE. This correlation was statistically significant (P < .05). All the data results are shown in Figures 1 and 2.
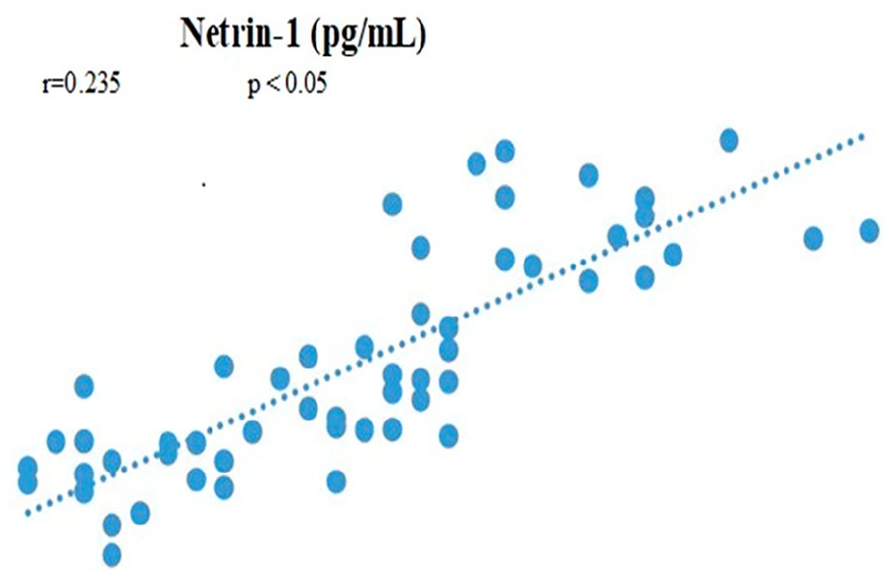
Correlation between NT-1, NSE levels, and severity of HIE.
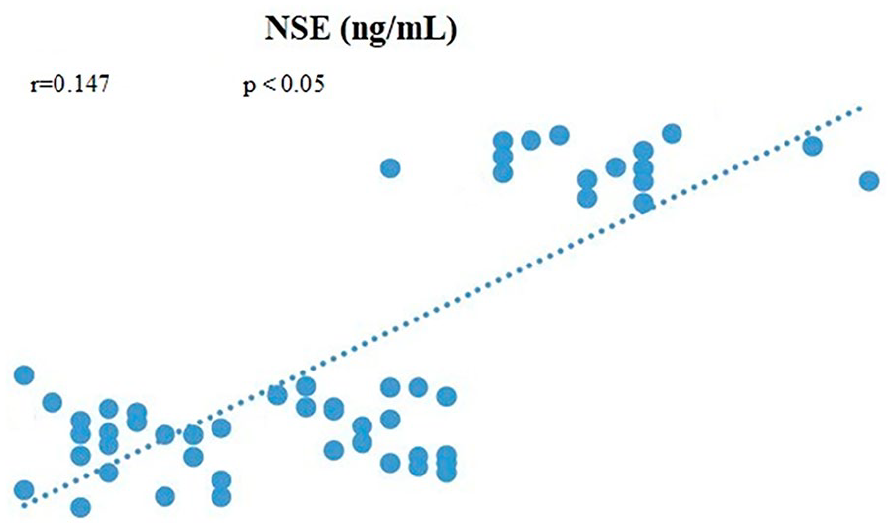
Correlation between NT-1, NSE levels, and severity of HIE.
Receiver-Operating Characteristic (ROC) Curves
We used ROC curves to identify the cut-off values for cord NT-1 and NSE. ROC curves for Netrin-1 and NSE for babies with HIE compared to controls are shown in Figure 3. The optimal cut-off levels for NT-1 and NSE were 289.3 pg/mL and 31.9 ng/mL, respectively. NT-1 had a sensitivity of 92% and a specificity of 100%. The positive and negative predictive values for NT-1 were 94% and 100%, respectively. However, for NSE, the sensitivity, specificity, positive predictive value, and negative predictive value were all 100% (Table 3).

Receiver-operating characteristic curves for Netrin-1 and neuron-specific enolase for babies with hypoxic-ischemic encephalopathy.
The Area Under the curve, Cutoff Level, Sensitivity, Specificity, PPV, and NPV of Serum Netrin-1 and NSE for HIE.

Discussion
Proper diagnosis and intervention are paramount importance for the long-term prognosis of neonates with HIE. The availability of an effective method for the early and accurate diagnosis of neonatal HIE is critical for subsequent therapeutic interventions.
Recently, newly discovered biomarkers have played a key role in neonatal neuroprotection. These biomarkers assist in screening infants for brain injury, identifying affected areas, monitoring the progression of disease, and assessing the extent of neuroprotection in clinical practice. In our study, infants with HIE stage II/III who received therapeutic hypothermia (TH) showed significantly higher umbilical cord blood levels of NT-1 and NSE compared to the control group. Early measurement of NT-1 and NSE from the umbilical cord was performed immediately after birth to establish an early diagnosis of HIE stage II/III. We recommend these potential biomarkers for early decisions regarding cooling therapy.
Despite ongoing research to identify possible biomarkers of HIE, no diagnostic biomarkers have been validated for early and accurate diagnosis, staging, or follow-up. 24
Previous studies have reported that serum is the most suitable sample for analysis, with proteins being the major candidate biomarkers. Among these biomarkers, NSE and S100B are considered the most important for HIE. Additionally, other proteins, such as UCHL-1 and GFAP, have been described as potential biomarkers involved in brain damage mechanisms. 25
Previous studies have reported significant elevations in NSE in neonates with HIE and perinatal asphyxia compared to controls.26-29 In our study, we found that umbilical cord blood levels of NSE were positively correlated with the severity of HIE. 26 The significant increase in NSE appears to be associated with substantial morbidity and mortality. Furthermore, prior research has indicated that NSE may be correlated with the severity of HIE. In contrast to our findings, Nagdyman et al conducted a study on 29 asphyxiated neonates and 20 controls and found no differences in NSE levels among the studied groups.27-30
Additionally, many studies have reported that serum NSE concentration correlates strongly with the severity of neonatal HIE, with higher NSE concentrations indicating more severe brain damage.18,31,32 A cut-off point of 40 mcg/L for NSE at 4 and 48 hours after birth has been suggested to distinguish between infants without HIE or with mild HIE (stage I) and those with moderate or severe HIE (stage II/III). 29
Additionally, a cut-off concentration of 45.4 mcg/L for serum NSE has been proposed as a predictor of poor prognosis in infants with HIE. 29
We found higher NSE levels in infants with HIE stage II/III compared to healthy controls. We determined a cut-off value of 31.9 ng/mL for NSE for therapeutic hypothermia, demonstrating high specificity and sensitivity for HIE stage II/III.
Serial measurement of NSE in the first 24 hours after birth may reflect different clinical scenarios and outcomes. Early measurement of NSE levels and monitoring over time are essential for obtaining more valuable insights.
Studies focusing on NSE as a biomarker for long-term outcomes of HIE are limited. Further large-scale longitudinal studies are recommended to evaluate NSE in the context of neonatal HIE, with extended follow-up periods.33,34
The full functions and role of NT-1 are not yet fully understood. 20 It has been reported that NT-1 has long-term negative effects in cases of transient focal ischemia. 35 NT-1 levels in infants have been studied in those appropriate for gestational age (AGA), intrauterine growth restriction (IUGR), and large for gestational age (LGA). However, no significant differences in NT-1 levels were observed among these groups in cord blood samples taken immediately after birth. 19
In our study, the umbilical cord NT-1 levels in the patient group (mean: 47.21 pg/mL) were significantly lower than in the control group (mean: 275 pg/mL). 19 We found that NT-1 levels in the umbilical cord were significantly higher in infants with HIE stage II/III than in the control group. A cut-off point for NT-1 concentrations was set at 31.9 ng/mL for infants with HIE stage II/III.
Some studies have concluded that NT-1 plays a vital role in maintaining the integrity of the blood-brain barrier (BBB) during experimental autoimmune encephalomyelitis, traumatic brain injury, and cerebral ischemic stroke in adults. 21 In cases of subarachnoid hemorrhage (SAH), NT-1 has shown benefits in early brain injury, including a reduction in brain edema, improved neurological status, and decreased neural apoptotic mechanisms in mice. 36 Further studies on NT-1 levels and long-term follow-up of infants with HIE will help determine the clinical uses and benefits of NT-1 in these infants. 21
Based on previous studies and theories regarding NT-1, as well as our findings, increased NT-1 may aid in the early diagnosis, and intervention of therapeutic hypothermia in infants with HIE stage II/III. Therapeutic implications for NT-1 in infants with HIE could be considered to reduce ischemic damage and protect the BBB.
These biomarkers can be used to differentiate between moderate-to-severe HIE and healthy controls, as the cut-off point for the decision to initiate therapeutic hypothermia (TH) was determined in our study. These biomarkers may also provide a means of assessing the severity of brain injury before initiating neuroprotective therapies.
A limitation of our study was the small sample size. Additionally, we did not evaluate the value of NSE and NT-1 in predicting the long-term sequelae of HIE, particularly the neurological outcomes. Another limitation is that we did not perform serial measurements of cord blood NT-1 and NSE, nor could we monitor serum NSE and NT-1 levels over the following days. We aimed to diagnose and decide on TH based on the HIE stages. Umbilical cord serum levels of NSE and NT-1 were studied for the early decision regarding TH. Further studies with larger patient samples are recommended to provide stronger evidence on this topic.
Conclusion
We conclude that NT-1 and NSE in the umbilical cord immediately after birth are significantly elevated in infants with moderate-to-severe HIE. However, these biomarkers did not show a significant increase in infants with mild HIE or non-HIE neonatal encephalopathy. Therefore, NT-1 and NSE may assist in making early decisions regarding TH in infants with HIE. NT-1 and NSE levels were positively correlated with the severity of HIE.
We recommend further long-term studies and validation on a larger sample of patients with HIE, using rapid-test techniques and prolonged follow-up periods, to detect the long-term sequelae of HIE, especially neurological ones. Such studies will help make early decisions regarding TH and provide stronger evidence to support our findings.
