Abstract
Introduction. Viral suppression rates among children and young adolescents living with HIV (CYALHIV) has been reported to be lower compared to adults. Follow up through viral load monitoring may influence their health outcomes. Objective. In this cross-sectional study we determined viral load monitoring practices and correlates of viral non-suppression of all CYALHIV in the main public hospitals in Kiambu County, Kenya. Data was abstracted from electronic medical records. Methods. Multivariable log binomial regression was used to estimate prevalence ratios (PR) and assess correlates of viral non-suppression. Adherence to viral load testing guidelines was also assessed. Viral non-suppression was defined as a viral load (VL) >1000c/ml. Results. Of the 252 CYALHIV, the median age was 11 (IQR: 7-13) years. At the most current VL, 14% were non-suppressed. Correlates of non-suppression included a history of Tuberculosis, [aPR = 4.25; 95% CI = 1.41-12.8; p = .01], antiretroviral drug side effects [aPR = 3.01; 95% CI = 1.37-6.62; p = 0.006] and having received enhanced adherence counselling (EAC) [aPR = 5.32; 95% CI = 2.00-14.15; p = .001]. Although the timing of baseline VL tests improved over the years, routine VL monitoring as per the guidelines remained suboptimal. Conclusions. There is need for adherence to VL monitoring guidelines for prompt action among non-suppressed children.
Keywords
Introduction
In 2022, approximately 1.5 million children under 15 years were living with HIV (human immunodeficiency virus) globally, with the number of new infections in this age group projected to be 130,000, of which 45% were predicted to occur in Eastern and Southern Africa. 1 In Kenya 7% of people living with HIV (PLHIV) are aged 15 years and below. 2 It is imperative to target this age group with efforts aimed at reducing morbidity and mortality in order to safeguard their right to thrive.
Regionally, viral suppression rates amongst children and young adolescents living with HIV (CYALHIV) has been reported to be low.3 -6 In Kenya, prevalence of viral non-suppression among children was higher (51.8%) than that observed in adults (28.4%). 7 Viral non-suppression is associated with increased risk of opportunistic infections and mortality.4,5
Low level viremia has recently been shown to be associated with neurocognitive developmental delays 8 which could affect children’s learning capacity. To achieve the 2030 United Nations program on HIV/AIDS (UNAIDS) 95-95-95 targets where 95% of PLHIV should know their HIV status, 95% PLHIV should be on antiretroviral therapy (ART) and 95% of those on ART should have viral suppression, it is important to understand correlates of viral non-suppression to guide clinical practice and design of HIV care interventions.
Previously reported correlates of viral non- suppression among CYALHIV include being male, history tuberculosis(TB), HIV status disclosure at an older age, experiencing ART adverse drug events, all of which have been associated with non-adherence.4,9,10 Some caregiver characteristics associated with viral non-suppression include one who isn’t the mother, caregiver substance abuse, poor socio economic status, inadequate social support.11,12 However, previous studies using routine programmatic data have been reported out of high HIV prevalence settings. It is important to explore whether these factors remain relevant in a relatively low HIV prevalence setting.
WHO recommends routine viral load (VL) monitoring as the preferred approach to ART monitoring. Routine VL monitoring allows for early detection of treatment failure, with subsequent drug switches for better treatment outcomes. Given the life-long nature of ART, treatment failure and drug resistance testing (DRT) is critical especially among CYALHIV, to preserve their drug options in future. In Kenya, the 2018 guidelines on the use of ART for treating and preventing HIV recommended VL monitoring at treatment initiation and every 6 months thereafter. In case of virological non-suppression (VL > 1000 copies/ml) while on ART, enhanced adherence counselling (EAC) was recommended, followed by a repeat VL after 3 months. However, the level of adherence to these guidelines among CYALHIV is not well documented locally.
In Kenya, through the support of PEPFAR (U.S President’s Emergency Plan for AIDS Relief), HIV records are digitized. The electronic medical record system (EMR) has a clinical decision support functionality that is aligned to the national guideline, prompting the user when VL monitoring is due. To an extent, this should influence the timing of VL testing.
We set out to evaluate the prevalence and correlates of viral non-suppression and describe adherence to VL testing guidelines among CYALHIV in Kiambu County, Kenya, a region with low HIV prevalence relative to other parts of the country, and high access to VL testing.
Aim
To evaluate correlates of viral non-suppression and adherence to viral load testing Kenyan guidelines in monitoring CYALHIV in the main public referral hospitals of Kiambu County, Kenya
Methods
Study Design
This was a cross-sectional study using data abstracted largely from electronic medical records of CYALHIV in Kiambu, Kenya, with some foundational demographic data pulled from archive paper records.
Study Setting
Kiambu County is largely peri-urban and borders Nairobi, the capital city of Kenya. With an estimated population of 684,616 children below 15 years. It has a HIV prevalence of 2.1% and an estimated 45,919 persons living with HIV compared to a national prevalence of 3.7% and 1,376 789 PLHIV. 13 It has 52 facilities offering comprehensive HIV care and treatment services serving both rural and urban populations, including the urban poor from neighboring slums in Nairobi.
We purposively selected the three main public referral hospitals because they have a high patient volume on ART and are located in a distinct geographical semi urban setting with diverse populations. Clinical care is supported by an EMR at all service delivery points, except the laboratory.
For VL testing, samples are drawn at the hospital lab, batched and transported 17km to a reference laboratory in Nairobi. When the results are ready, they are relayed back to the hospital via email and routinely updated in the EMR within a week after receipt. Samples for DRT are referred using the same system. VL and DRT are offered at no cost to the patient.
Starting in 2020, Kenya had embarked on treatment optimization among children and adolescents, recommending use of Dolutegravir (DTG) as a first-line drug in this population. An addendum guideline was released, recommending VL testing within three months of the empirical switch to DTG.
Eligibility Criteria
The inclusion criteria were CYALHIV aged 15 years and below, who had been active in care for at least 6 months, marked by at least one clinic visit in the 3 months preceding data abstraction in October 2022. The exclusion criteria was being on ART for less than 6 months.
Study Procedures
Variables of interest were structured into an electronic data capture tool (Kobo©). Childs demographic characteristics, treatment /clinical factors and caregiver characteristics for caregivers who had HIV were abstracted from the EMR system. Information for HIV negative caregivers was supplemented from paper records. Data abstraction was done between 3rd October and 31st October 2022. Research assistants were trained on the use of data collection instrument and on data abstraction from Definitions and correlates of viral non-suppression.
Baseline viral suppression was defined as a VL < 1000 copies/ml within 7 months of treatment initiation, while viral non-suppression was having ≥1000 copies/ml after being on ART for 6 months. 14 Undetectable VL was defined as having 0 copies/ml while 1 to 999 copies were classified as low-level viremia, both of which were considered viral suppression.
Covariates of viral non-suppression considered were the demographic characteristics including age, sex, education level, having siblings with HIV positive siblings and clinical/ treatment factors such as age at diagnosis, duration on ART, reported ART side effects, tuberculosis (TB) infection. Lack of adherence was denoted by having received enhanced adherence counselling (EAC). Caregiver characteristics evaluated included sex, relationship to the child and HIV status. Additionally, age, education level, employment and viral suppression status were included for caregivers who had HIV since this information was available.
Statistical Analysis
We summarized categorical variables using counts and proportions while continuous variables were described using medians and interquartile ranges (IQR). Log binomial regression analysis was used to assess factors associated with viral non-suppression. Measures of association in form of prevalence ratios (PR) and a 95% confidence interval (CI) were obtained. Significant factors in bivariate analysis (p-value < .1) were then included in the multivariate analysis to obtain adjusted PRs.
Analysis was done using STATA version 16 (Stata Corp®, College Station, Texas, USA).
Adherence to VL Monitoring Guidelines
Adherence to viral load testing was based on 2012, 2016 and 2018 national Kenyan guidelines for HIV treatment and prevention using the following four criteria’s: Baseline VL within 6 months of ART initiation, six monthly VL monitoring, enhanced adherence counselling and follow up VL monitoring in case of viral non-suppression and drug resistance testing after two consecutive non-suppressed VLs with EAC.
Ethical Approval and Informed Consent
The study was conducted in accordance with the Declaration of Helsinki and was approved by the University of Nairobi/Kenyatta National Hospital Ethics and Research Review Committee (P268/03/2022) and National Commission for Science, Technology and Innovation (NACOSTI/P/22/20290). Permission to carry out research was granted by County Department of Health Research Unit and the administration of participating hospitals.
The Ethics committee gave waiver for individual patient consent as routinely collected data was anonymously abstracted from the medical records. Authors had no access to any information that could identify individual participants during or after data collection
Results
Of the 281 CAYLHIV active in care at the time of abstraction, we abstracted records for 252 who met the inclusion criteria (Figure 1). The median age was 11 (IQR: 7-13) years. Majority 198 (79%) had been diagnosed at the age of 0 to 4 years with 135 (54%) being male. A high percentage (87%) of the caregivers were females and about a half 88(51%) were less than 40 years old. In addition, 174 (70%) caregivers were living with HIV (Table 1).

Prevalence of viral non-suppression.
Characteristics Associated With Current Viral Non-suppression.

Only sex and relationship were available for caregivers who did not have HIV.
Prevalence of viral non-suppression
A quarter (63/252) CYALHIV had viral non-suppression at the first available VL. Prevalence of non-suppression at the most current VL test was 14% (Figure 1).
Correlates of Viral Non-suppression
As at October 2022, nearly all (94.4%) had been switched to Dolutegravir (DTG) and had a lower likelihood of non-suppression compared to those on a protease inhibitor (PI)-based regimen [adjusted PR [aPR]: 0.35 (CI:0.15-0.85: P = .021)]. Having reported ART side effects [aPR]:3.01; (CI:1.37-6.62: P = .006), history of TB [aPR]:4.25 (CI:1.41-12.8: P = .010) and having received EAC, a proxy for poor adherence [aPR]: 5.32 (CI: 2.00–14.45; P = .001) were significantly associated with a higher risk of viral non-suppression. Children with caregivers 40 years and older who were living with HIV were less likely to have non-suppression [aPR]: 0.44(CI:0.89-0.99; p-value: .05) (Table 1)
Adherence to VL Testing Guidelines
Baseline VL Within 6 months of ART Initiation
For the entire cohort, only 36 (14%) children and young adolescents had a baseline VL test within the recommended 6 (5-7 months) after treatment initiation. Before 2010, nearly all (80%) had their first VL tests done after 24 months. However, as time went on, more children were getting VL tests done after ART initiation. After 2018, 61% of CAYLHIV were tested within 6 months of ART initiation as recommended (Table 2).
Time to Baseline VL Tests Over the Years.

Six monthly VL monitoring
Restricting the analysis to viral load tests done after 2018 (n = 169), routine monitoring for majority of the children was outside of the recommended follow up VL time at every 6 months. The number of those who received VL testing every 6 months reduced over time. Non-compliance was at 90% for the most recent VL compared to 69% at the earliest VL (Figure 2).
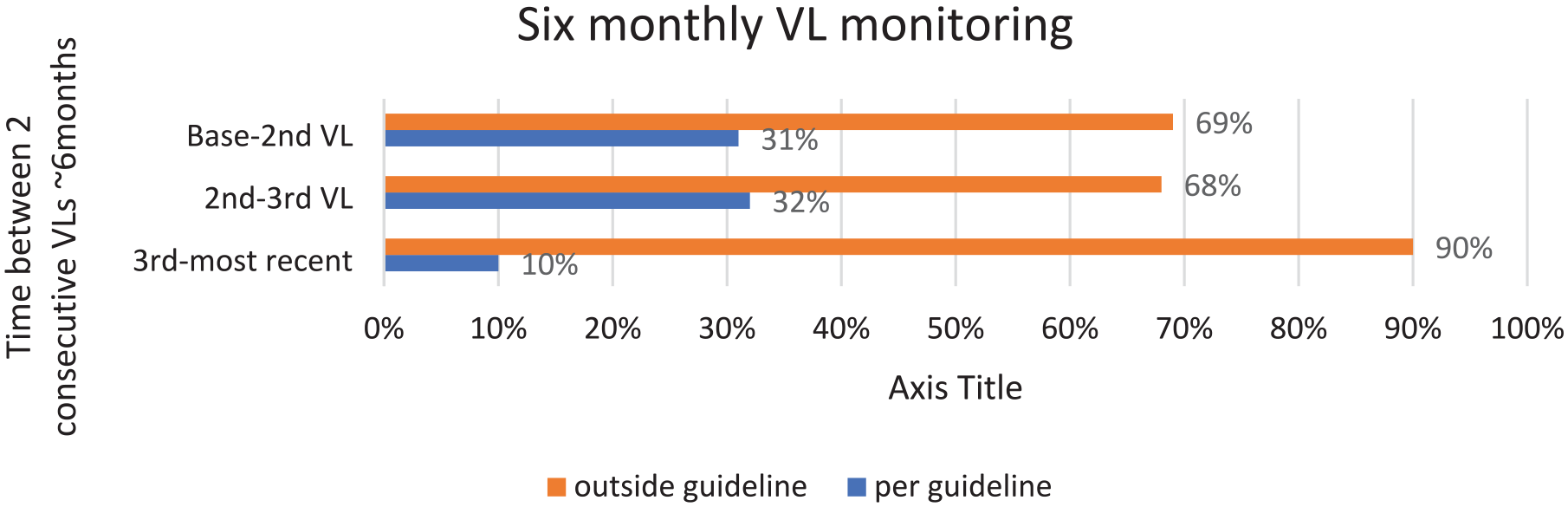
Routine VL monitoring within 6 months as per the 2018 guideline recommendation.
Enhanced adherence counselling and follow up VL monitoring
Of the entire cohort, ninety-six CYALHIV had at least one non-suppressed viral load at one point in their medical history. Of these, 80% consequently received enhanced adherence counselling. Nevertheless, only 4 (5%) children had a repeat VL test after the recommended 3 months of EAC (Figure 3).
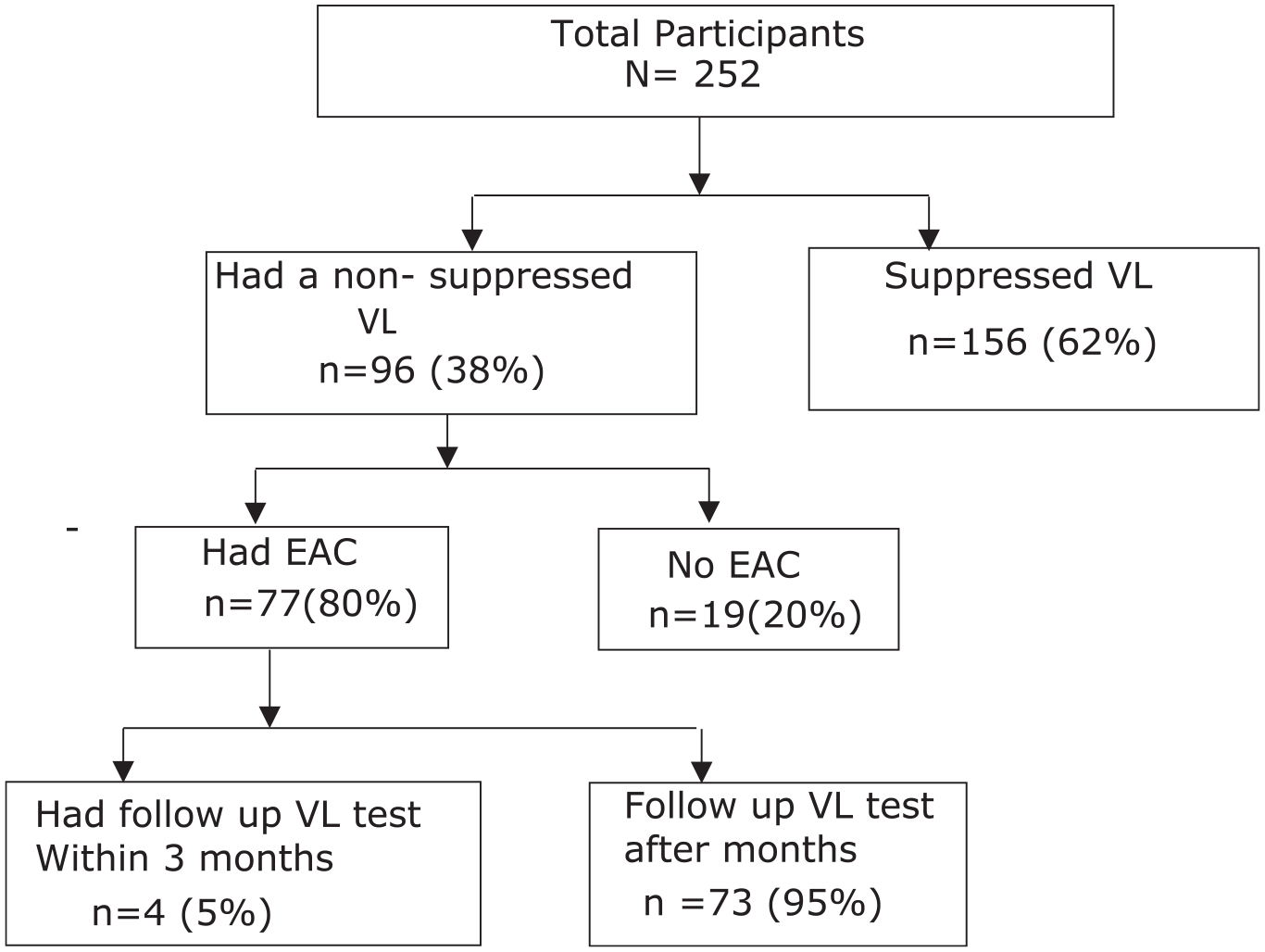
EAC and followup VL testing.
VL testing within DTG optimization
As at October 2022, 238(94%) children had been switched to DTG. Of these (77%) had obtained a follow up VL after the switch. However, only 21(11%) were retested within the recommended timeframe of 3 months (Figure 4).
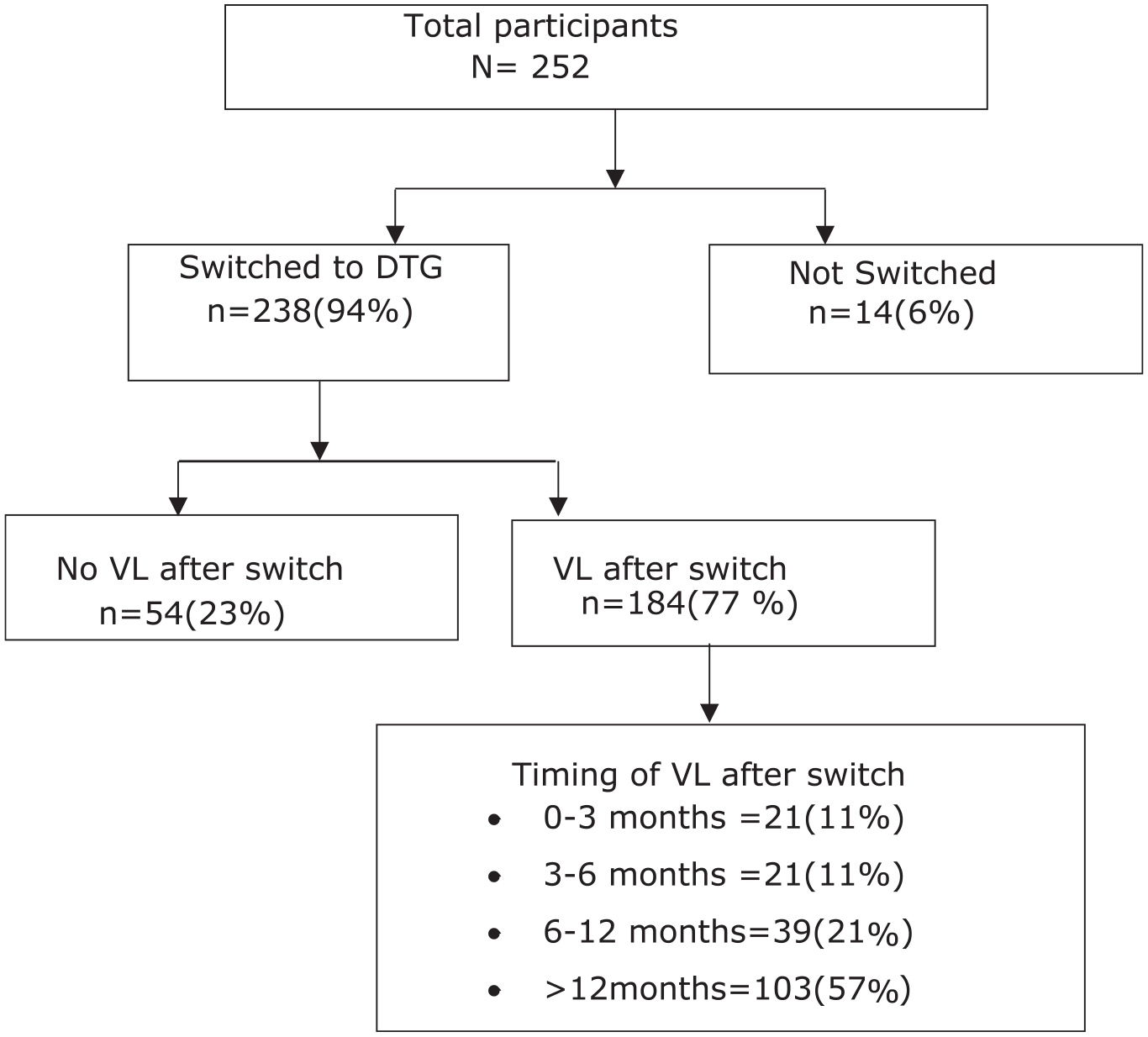
VL testing following DTG optimization.
ART drug resistance testing
There were twelve CYALHIV who had been switched from a PI to a DTG-based regimen due to viral non-suppression. Of these, only four received drug resistance testing (DRT) as recommended in the Kenya 2018 guidelines. However, none of these DRT results were available in the EMR.
Discussion
At most current VL, 14% of the CYALHIV had viral non-suppression. Correlates associated with higher likelihood of non-suppression included ART related side effects, history of TB and having received EAC as a proxy for poor adherence. Having a caregiver 40 years and older who was living with HIV was less likely associated with non-suppression.
Viral suppression is crucial in reduction of negative outcomes among CYALHIV but the rates are worryingly low compared to adults regionally.4 -9 Similarly, in our study viral suppression was way below the UNAIDS 2030 targets that aims at achieving viral suppression in 95% of PLHIV on treatment. However, prevalence of non-suppression from this study was lower in comparison to the 2018 Kenyan national estimates which was 51.7%. 7 This could be due to the continued roll out and training for health workers on the use of new treatment guidelines, including roll out of DTG as first-line therapy in children which is associated with viral suppression. 15 Additionally, among the CYALHIV with a VL <1000 c/ml, 14% had low level viraemia that could increase the risk of virological failure 16 and neurocognitive developmental delays 8 which could subsequently affect their learning capacity. Hence the need for close follow for these children.
Consistent with other studies though not statistically significant, younger children( 0-4 years) had a higher likelihood of viral non-suppression compared to older (10-14 years) CYALHIV.9,17 -19 Younger children have issues during treatment such as non-pleasant medication, dosing inaccuracies, treatment administration problems resulting to insufficient drug levels, risk of drug resistance and eventual virologic failure.20 -23 Additionally it could be due to the positive effects of disclosure among older children. 18
Similar to other studies, we found CYALHIV who had history of TB had a higher likelihood of non-suppression.4,24 This may be due to the increased pill burden, drug interactions, or active TB’s immune activation effects resulting in HIV viremia. 25 ART-related side effects were also significantly associated with non-suppression. While the large aPRs for these associations can be explained by our relatively small sample size, it could also underpin the strength of these associations, highlighting the need to specifically support CYALHIV and their caregivers who report ART side effects as well as those with TB.
ART adherence contributes to achievement of viral suppression thus EAC is recommended for PLHIV who are non-adherent or have detectable VL.11,26 In our study, 44% of participants had received EAC, a proxy for non-adherence and were more likely to be non-suppressed. Further implementation research is needed to optimize adherence counselling for CYALHIV, including the timing, frequency, content and perhaps nature of the counselling. 27
Studies have described the interaction between the child and their caregiver as a defining factor in the level of viral suppression for CYALHIV especially in resource limited situations. 28 We found that CAYLHIV with caregivers living with HIV aged 40 years and older had a lower risk of non-suppression compared to younger caregivers living with HIV. Age here could be a proxy of adherence or stability in the home set up.
WHO guidelines from 2016 recommended VL testing as the preferred monitoring approach for all individuals on ART in order to assess treatment response, detect treatment failure and determine the need to switch to second line regimen in a timely manner. The guidelines also recommended VL testing within 6 months of initiating ART. 14
Routine VL monitoring has been reported as vital in early detection of ART failure, prevention of unnecessary switches thus improving treatment care outcomes.29-33 In this study, the trend of time to the first VL test indicates improvement over time, as VL testing became more accessible (Table 2). However, starting 2018 when the WHO guideline was adopted in Kenya, only 61% CYALHIV were tested within 6 months of treatment initiation, which was lower than expected. This is particularly concerning in this region because of the proximity to the testing lab, availability of donor support services for patient care and the few patient numbers compared to other regions in Kenya with higher HIV prevalence and are less resourced.
Gaps in the VL cascade of care such as delayed follow up VL, delayed or lack of regimen switch in virological failure has been reported as a hinderance to ART treatment success.34,35 In our study, only 5% CYALHIV had a follow up test to confirm treatment failure (Figure 3). This is disconcerting as it indicates failure of proper data use, that unsuppressed test results do not result in any action or follow up. Our findings further support the call for models of action that ensure VL results are used for action. 36 It also calls for evaluation of clinical decision support functions with EMR systems and their contribution to adherence to guidelines for improved patient outcomes. 37
Up to 60-90% children who don’t attain viral suppression tend to have drug resistance which may hinder success of HIV care interventions and therefore drug resistance testing is a vital component in management of treatment failure.38–40 Kenya guidelines recommend DRT in CYALHIV who are on a PI based regimen and are unsuppressed. Among the twelve CYALHIV who had been switched from a PI based (first line therapy for CYALHIV) to DTG based regimen due to non-suppression (outside of the empirical switch in 2020), only 4 (33%) had received drug resistance testing (sample had been referred for testing). However, the EMR did not have any DRT results. This further supports the need for interoperability within health information systems, which would ensure direct transmission of results from the lab to the clinician. 41
Some of the limitations in our study include its cross-sectional nature and use of data abstracted from the EMR, whose variables are constrained in nature, limiting our ability to enquire deeper into some of the findings. Given its retrospective nature, our study could not evaluate other factors that could have influenced the VL monitoring practices, such as availability of testing reagents and timeliness of sample transport. Nonetheless, our findings give credence to use of EMR data for research, with findings that support the need to interrogate how data in these systems is utilized for better care at patient-level, beyond the global targets.
Conclusion
At the most recent VL, 14% children were non-suppressed, higher than the 5% UNAIDS 2030 target. Routine VL monitoring as per the guidelines was suboptimal despite access to VL testing, suggesting other barriers to VL monitoring which need to be explored further, including use of EMR data at patient-level for action.
Footnotes
Acknowledgements
We acknowledge the study hospitals and Kiambu county research department for allowing us to collect data.
Author contributions
L.N developed the study materials. L.N and A.N conceptualized and did data analysis.
L.N wrote the first draft of the manuscript. A.N provided feedback and approved the final submitted manuscript.
Data availability statement
The data that supported the findings of this study has been shared to the journal editor. The data are not publicly available due to privacy restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Routine data collection of this study was supported by University of Nairobi department of paediatrics through Health-Professional Education Partnership Initiative (HEPI)-Kenya NIH funded project-R25TWO11212. There is no funding support for publication. Funders had no role in designing of the study, data collection and analysis, manuscript preparation nor decision to publish
Ethical approval
The study was approved by Kenyatta National Hospital/University of Nairobi Ethics and Research Review Committee (P268/03/2022) and National Commission for Science, Technology and Innovation (NACOSTI/P/22/20290)
