Abstract
Esophageal stenosis is a very rare entity. Most cases of esophageal stenosis are asymptomatic in neonatal life. This is a case series of 2 babies with esophageal stenosis presenting in neonatal period with same clinical sign but different etiology. Of the two, one is congenital and other is acquired in etiology.
Keywords
Key Clinical Message
● It is not uncommon for babies with esophageal stenosis to present in the neonatal life.
● Infective causes should also be kept in mind in neonates presenting with esophageal stenosis.
Introduction
Esophageal stenosis in children is commonly caused by congenital or acquired causes. Acquired causes includes traumatic, inflammatory, gastro esophageal reflux (GER), and post-surgery. 1 Congenital esophageal stenosis (CES) accounts for only 4% cases of esophageal stenosis. CES is a very rare disease with incidence of 1 in 25 000 to 50 000 live births. It is associated with malformations in 16% to 33% cases. 2 It typically becomes evident in infancy, often coinciding with the introduction of solid foods, and presents with symptoms such as vomiting, failure to thrive, dysphagia, wheezing, food impaction, and respiratory issues.2,3 In this series, we are discussing 2 cases of esophageal stenosis presenting within 28 days of life.
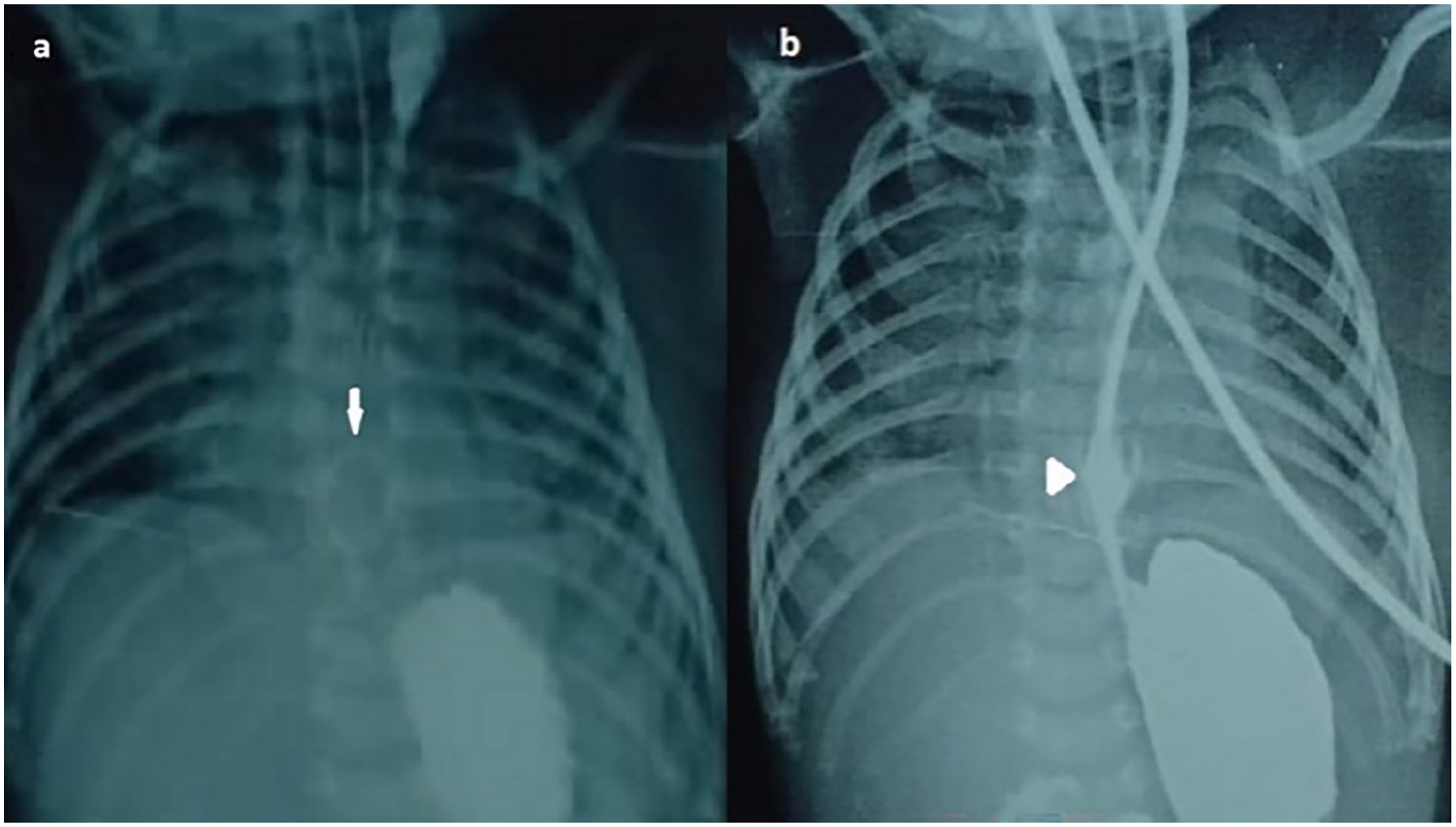
However, on the 24th day of life, she was admitted to our hospital due to episodes of apnea and decreased feeding. Upon admission, her weight was recorded as 1 kg, and her vital signs were stable. Initial screening for sepsis and cerebrospinal fluid (CSF) analysis yielded normal results. The following day, due to recurrent apnea, the baby required intubation. However, attempts to initiate enteral feeding were unsuccessful, as the nasogastric tube (Size-5 F) became coiled in the lower esophagus, as depicted in Figure 1a on the chest X-ray, which also indicated right-sided collapse. Subsequent esophagography revealed a very thin lower esophageal segment, with dye gradually entering the stomach, as depicted in Figure 1b.

(a) CXR showing RUZ collapse with coiling of NG tube (arrow). (b) Esophagogram showing very thin lumen of NG tube(Size 5 F) lower one-third of esophagus (arrow head).
Despite the prompt initiation of broad-spectrum antibiotics following a positive sepsis screen, the infant’s condition continued to deteriorate, and unfortunately, she did not respond to treatment and passed away.
By the fourth day of life, the baby developed acute kidney injury, presenting with hyperkalemia (maximum potassium level of 10.4 mEq/L) and metabolic acidosis. However, by the fifth day, the baby no longer required inotropic support. Enteral feeding commenced the following day, gradually progressing to full feeds via 7F size NG tube. Despite feeding tolerance and regular bowel movements, the baby showed minimal spontaneous respiratory efforts. Additionally, oral candidiasis was diagnosed on the 10th day and treated with candid mouth paint.
On the 21st day of life, difficulty in passing an orogastric tube prompted further investigation with an esophagogram and contrast-enhanced computed tomography (CECT) of the chest. The esophagogram revealed a circular mass lesion in the lower esophagus causing partial obstruction, with dye trickling into the stomach, as depicted in Figure 2a. The CECT showed thickening of the lower esophageal wall and features suggestive of aspiration pneumonia in the bilateral lung fields. A repeat dye study performed after 2 days showed a significant reduction in the size of the lower esophageal mass, as shown in Figure 2b. Orogastric feeds were resumed on the 25th day of life.
(a) Esophagogram showing circular contrast enhancing lesion in lower esophagus (arrow). (b) Repeat Esophagogram shows decrease in size of mass lesion (arrow head), NG tube size-7 F.
Due to severe thrombocytopenia, oral candidiasis, and an esophageal fungal ball, the baby was started on Amphotericin B. Unfortunately, the baby passed away on the 29th day of life due to pulmonary bleeding. Blood culture revealed Serratia marcescens, while cerebrospinal fluid culture was sterile. Postmortem esophageal mass biopsy was not conducted as the parents declined autopsy.
Discussion
Esophageal stenosis can result from congenital or acquired factors. Congenital esophageal stenosis (CES) typically becomes apparent in infancy, often with symptoms such as vomiting, poor growth, swallowing difficulties, wheezing, and food impaction.2,3 While CES is rarely diagnosed in newborns, it may be detected alongside esophageal atresia.
An interesting case reported by Mai et al 4 highlighted CES associated with duodenal atresia during the neonatal period. Similarly, D’Alasenndro et al 5 documented a case of a 28-day-old newborn experiencing difficulty with orogastric tube insertion, later diagnosed with esophageal stenosis via barium esophagogram. In our series, Case 1 exhibited an inability to negotiate a nasogastric tube, leading to a diagnosis of congenital esophageal stenosis due to the absence of history suggestive of an acquired etiology. Recurrent apnea and failure to thrive in a baby may be attributed to CES, which can cause feed intolerance and repeated microaspirations leading to right lung collapse. Therefore, it’s important to consider CES as a potential cause in infants experiencing both failure to thrive and recurrent apnea.
CES arises from congenital malformations of the esophageal wall, with 3 primary types: fibromuscular stenosis (FMS), tracheobronchial remnant (TBR), and membranous stenosis (MS). 6 Each subtype displays varying locations within the esophagus, with FMS being the most prevalent, particularly in the middle/lower thirds, as indicated by a study by Michaud et al. 7
Diagnosis of esophageal stenosis typically involves esophagogram, endoscopy, or biopsy. Treatment approaches vary based on subtype, with segmental resection favored for TBR due to the risk of perforation with endoscopic dilation. Endoscopic dilation is preferred for the other types. Terui et al’s 8 systematic review revealed an incidental CES incidence of 9.6% during or post-repair of esophageal atresia and/or tracheoesophageal fistula. Additionally, Kim et al’s 9 retrospective study highlighted differences in diagnosis and treatment outcomes based on EA-TEF association.
In our case series, Case 2 encountered difficulty in negotiating an NG tube on day 21 of life, indicating a different etiology despite similar clinical presentation. The acquired nature of the disease was suggested by the challenge of inserting an NG tube in a neonate already receiving enteral feeds via nasogastric tube for nearly 2 weeks. The presence of circular contrast-enhancing esophageal lesions, oral candidiasis, and urine fungal hyphae positivity further pointed toward an esophageal fungal ball.
Candida, particularly C. albicans, is a leading cause of fungal sepsis in neonates, notably among very low birth weight infants. Candida esophagitis, characterized by white mucosal plaque-like lesions, can also present as an esophageal mass. While cases like that reported by Al-Shoha et al 10 in a 70-year-old male with esophageal candidiasis mimicking malignancy exist, neonatal cases of esophageal fungal masses have not been documented. Therefore, not only preterm infants but also term babies who have had a difficult course in the NICU can develop fungal infections, which may subsequently lead to esophageal stenosis. However, our series is limited by the lack of histological confirmation in both cases.
Conclusion
In summary, congenital esophageal stenosis can present in neonates, supported by existing literature, though mostly occurs after introducing solid food. Clinicians should be vigilant for possible infectious causes, such as fungal infections, in moderately to deeply stuporous infants, as candidiasis is treatable. Ensuring proper oral hygiene in these infants may help prevent such infections.
Footnotes
Author Contributions
Dr.Nidhi Jain-manuscript writing, Dr.Susrita-editing of manuscript, Dr.Ravindra - helped with proofreading of final manuscript, Dr.Aparna-initial draft making.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethics
Parennts of the subjects have given their written informed consent to publish their case (including publication of images).
