Abstract
Introduction:
Laboratory request forms (LRFs) are pivotal to histopathologists as they are the only medium through which pathologists communicate with patients and clinicians. On many occasions, the LRFs are incompletely filled and do not paint the right picture of the patient.
Methods:
This was a cross-sectional study that audited 586 LRFs and specimen containers sent to the Mbarara University of Science and Technology (MUST) histopathology laboratory between July 2024 and September 2024 for their completeness and appropriateness. The main parameters considered were biodata, clinical history, clinician details, and hospital details.
Results:
Overall, only 3.6% (21/586) of LRFs were appropriately filled. Of the 4 main parameters, biodata was the most filled at 54.1%, hospital details at 18.1%, and clinical details at 7.5%, and the least completed was clinician details at 5.6%.
Conclusion:
The completion rate of the LRFs is unacceptably poor, and the used specimen containers are unbefitting. Patient age and name are the most completed parameters, while requesting the physician’s signature was the least completed variable. Clinicians need to be constantly reminded of their role in histopathology diagnosis through continuous medical education and the introduction of laboratory standard operating procedures (SOPs) on the wards and operating theaters.
Introduction
As the prevalence of cancer increases, the demand for pathology laboratory services is expected to surge. The services, however, must be of the right quality, timely, and accurate. The Timeliness, quality, and accuracy of laboratory results depend on several factors. These include procedures carried out before obtaining the sample, during sample acquisition, and proper requisition by the clinicians (pre-analytics).1,2 Pathologists or other laboratory personnel often do not meet with the patients in person. Their work relies on the information scripted on the request forms that clinicians send to the laboratory alongside the specimen. Unfortunately, research has indicated that the information is often incomplete,3,4 and this often delays diagnosis, creates avenues for errors, and puts patient safety at stake.1,5 According to the Centers for Disease Control and Prevention, up to 70% of clinical decisions depend on laboratory diagnosis 6 ; therefore, errors in laboratory diagnosis can have far-reaching consequences.3,7 False negatives, for instance, may cause delays in initiating treatment, leading to poor outcomes. On the other hand, false positives can result in permanent iatrogenic loss of body parts, which can catastrophically affect the quality of life and can lead to financial losses to the government and individuals. A meta-analysis done in African clinical laboratories revealed that 70% of the errors occurred in the pre-analytical phase. 3 If we are to reduce such errors in histopathology, the role of clinicians who obtain the samples and make the requisitions to the pathologists needs to be understood and emphasized.
Being the only avenue through which pathologists can communicate with both the clinicians and the patients, request forms should be impeccably filled. 8 Several studies in other countries have observed either 100% or close to 100% incompleteness in the filling of request forms.4,7,9 Every detail required on the request form is of critical importance (see Supplemental Appendix Ifor details needed on a request form). For example, a differential diagnosis may lessen the time the pathologists require to make a diagnosis. A clinician’s phone number may ease cross-consultation between the pathologist and the clinicians, which may shorten the turnaround time (TAT). Aside from laboratory request forms, information on the specimen container, the quality of the container, and the way the specimen is handled before histological examination is very critical. Tissue handling has a great bearing on the quality of immunohistochemistry, which is now a routine in the diagnosis of several cancers. Several studies have, however, observed that the containers are often inappropriate.2,9 A study conducted at Makerere Pathology Laboratories observed that errors occurred in all the reviewed laboratory request forms (an error rate of 100%). 9 Errors were mostly in the preprocessing examination of the specimens (grossing), patient/specimen identification, and errors in specimen containers, among others. There is no data about the prevalence of error rates in other histopathology laboratories in Uganda.
The MUST histopathology laboratory is a regional referral anatomical pathology laboratory, handling over 5000 cases yearly and serving the Mbarara Regional Referral Cancer unit. With over 30 years of service, it is important to audit the processes of the laboratory. This site was chosen because it is the only histopathology Laboratory in Southwestern Uganda and also serves as a training center for the region. Therefore, whatever is done at this laboratory is expected to be at an acceptable international standard. In this study, we assessed the appropriateness of specimen containers and the completeness of laboratory requisition forms sent to the MUST histopathology laboratory. We also determined the agreement between the clinical and histological diagnoses. The results from this study have the potential to change practice and improve the utilization of the laboratory. Ultimately, we hope to improve the communication between the clinicians and the pathologists, reduce the turnaround time (TAT), and improve patient care, hospital experience, and service satisfaction.
Methods
This was a prospective descriptive cross-sectional study. It involved specimen containers and histopathology request forms sent to the MUST histopathology laboratory from July 1st, 2024, to September 30th, 2024. Cytopathology specimens were not included in the study. The laboratory uses a purely paper system for requisitions of tests as well as the dispatching of results. These were audited for appropriateness and completeness, respectively. The container appropriateness was assessed using a pretested checklist that clearly outlined the qualities of an appropriate specimen container by international standards. Considered qualities included the container’s material, size of the container in relation to the size of the specimen, tightness of the seal, having a wide mouth, previous use of the container for other purposes, or not, and appropriate labeling. A container was considered suitable only if it was plastic, with a wide mouth, leak-proof, and not previously used for any purpose. These containers were also assessed for the presence of appropriate labels. The request forms accompanying the specimens were also assessed retrospectively with a checklist for the presence of the patient’s name, age, sex, address, clinical history, site of biopsy, examination findings, prior investigations done, differential diagnosis, and date of procedure as recommended by several internationally acceptable literature.10-12 A complete request form was one with complete biodata, clinical details, name of the requesting clinician, hospital, and ward. The audit included all specimens and the corresponding request forms received at the department during the study period. We excluded all cytology specimens and specimens received at the laboratory with already processed blocks requiring special stains.
The audit received approval from the MUST institutional review board under the number MUST-2024-1495. Consent was not required since the audit did not involve any patient intervention.
Data Collection and Analysis
Data was collected by pathology residents using pre-tested checklists (see Supplemental Appendices II and III) entered in Redcap, cleaned and then analyzed using STATA version 17. The proportion of appropriately filled request forms and suitable specimen containers were expressed as percentages with their corresponding 95% confidence intervals. The differential diagnosis provided by the requesting physician on the request form was compared to the final histological diagnosis by simple eyeballing to determine the level of agreement between the 2 and expressed as a percentage.
Results
A total of 586 lab request forms (LRFs) and specimen containers were reviewed/ audited. Only 3.6% (21/586) of the LRF were adequately filled (Figure 1). There were 4 (4) main parameters on the LRF; clinician details were the least filled (5.6%), followed by clinical details (7.5%), hospital details (18.1%), and Biodata (54.1%) was the most filled (see Tables 1–4).
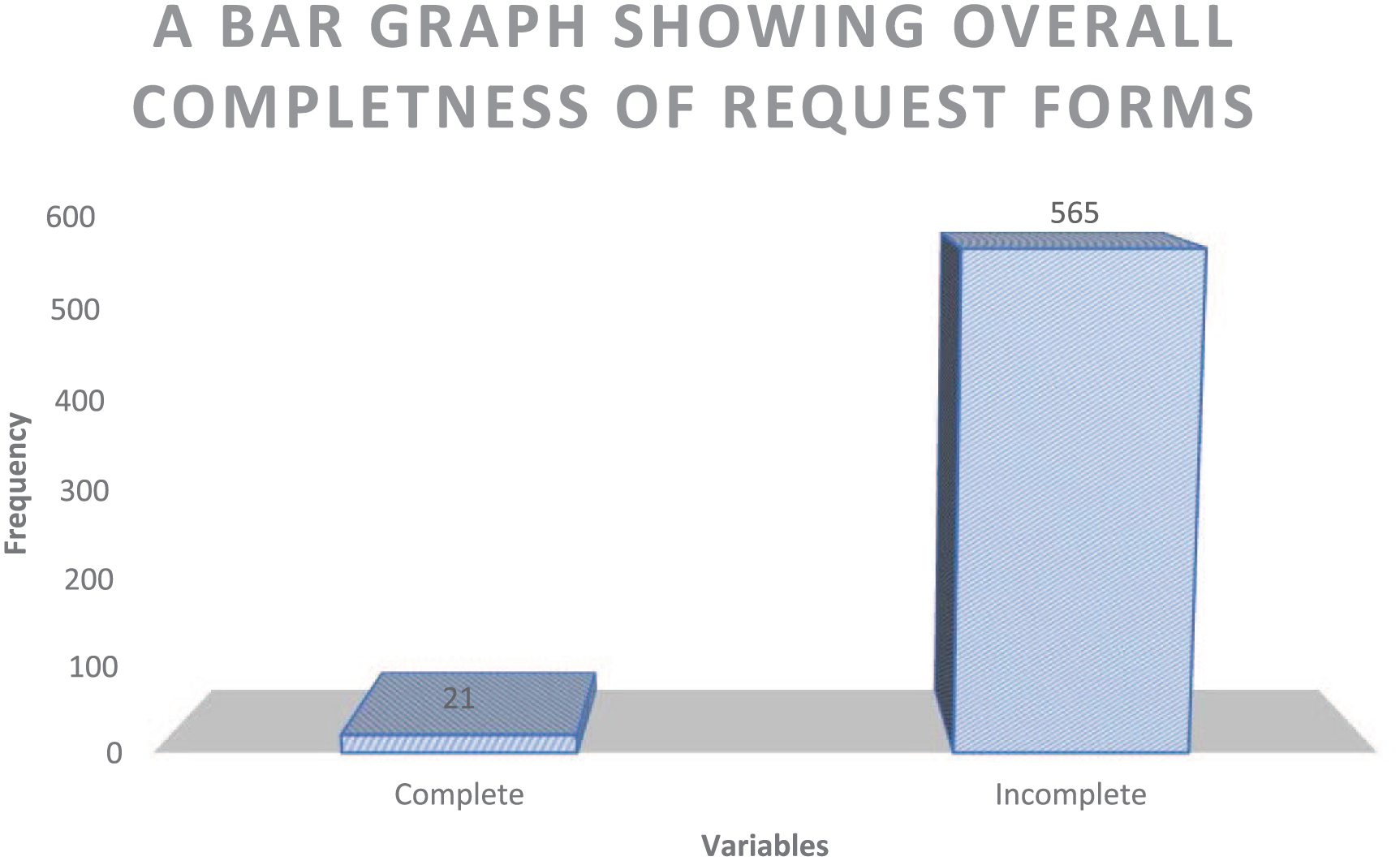
Bar chart showing the overall completeness of the laboratory request forms.
Biodata Details and the Proportions of Complete and Missing Hospital Details.

Proportion of Clinical Details as Presented on the LRFS.

Clinician Details and Their Breakdown.

Proportion of Container Properties and Labels that were Considered on a Specimen Container.

Patient biodata was appropriate in more than half (54.1%) of the LRFs. Name and age were the most filled sections at 99.50% (583/586), and the address (village and/or district) was filled in only 62.63% (367/586) and 76.79% (450/586), respectively (see Table 1). Hospital details were complete in only 18.14% (118/586) of the LRFs. The hospital name was present on 82.42% (483/586), the hospital ward 53.9% (316/586), inpatient/outpatient number 49.7% (291/586) of the LRF, and the unit was present only 40.6% (238/586) of all the LRFs (Table 1).
Clinical details were appropriately provided on only 7.5% of the LRFs. Previous investigations were the least provided details, 31.7%, followed by whether a biopsy had been done previously or not 44.4%, differential diagnosis 63.0%, nature of specimen 64.7%, site of specimen 89.1%, and clinical history was the most provided 90.3% (Table 2).
Among the clinician details, the name of the requesting clinician was the most filled on 89.8%, of the LRFs, only 11.8% of the clinicians signed the request forms, and their contacts information was only provided on 19.4% of the forms (Table 3). Agreement between clinical and final histopathological diagnosis was seen in only 47.1% of the reports that had clinical and final diagnosis.
Specimen Containers and Labeling
Up to 79.7% of the containers were appropriate for the specimens in terms of relative size, container material, and type of container mouth. Plastic containers were 91.3%, 94.9% had a wide opening/mouth, and 16.4% had been previously used for other purposes (Table 4). Only 1.2% of the containers were appropriately labeled. Specimen type was the most missed label (81.2%), followed by site of biopsy (67.1%), date of procedure (51.4%), sex (34.3%), age (25.3%) and the 2 names missed on (2.2%) of the containers (Table 4).
Discussion
The international organization for standards (ISO 15189:2022) for laboratories requires that LRFs contain enough information to identify patients, the requesting physician/nurse/health worker, as well as sufficient clinical information. 13 Despite these recommendations, Incomplete LRFs are still a major challenge in many Laboratories, especially in settings where electronic records are not available. So clinicians must scribble all the information on the request forms. In this audit, only 3.6% of LRFs had all the required information. This is similar to the earlier observations made in Nigeria, where only 3% of the forms were complete. 2 The LRFs in this study are more complete compared to 0% in Iran and 1.3% in a later study from Nigeria.14,15 Contrary to all the above, a study conducted in 2021 in Nigeria found that up to 32.4% of the forms were complete, 16 which may imply an improvement compared to earlier studies from the same country. With only 3.6% of forms appropriately filled, a lot of pre-analytical errors are bound to happen, and a lot has to be done if we are to improve our services.
Salient patient identifiers like name and age were missing on 0.51% of the forms, and gender was missing on 3.75% of the forms. These findings are similar to those observed in a tertiary hospital in South Africa, where gender was missing in 5.1% of the forms, date of birth or age was missing in 3.7%, and 0.2% of forms had no patient name. 17 A similar finding was also observed in Lagos, where the patient’s name was missed in 1% of the LRFs. Gender and age were also missing in 9.7% and 32% of the LRFs, respectively, 14 although these figures are much higher than those observed in the current study. A recent survey in Khartoum observed that up to 24.4% and 53.8% of the forms lacked age and gender 18 compared to 0.5% and 3.75% observed in the current study, respectively. It is, therefore, evident that these errors are a common occurrence, yet they are critical to a pathologist because some tumors tend to occur in certain age groups and genders. The name is also of major significance if sample mix-ups are to be avoided. It is unimaginable under which circumstances a clinician can forget to put the name on a LRF. From experience, forgetting a patient’s name can occur due to understaffing and working under too much pressure. Virtually, all samples with no name are rejected especially if the requesting clinician cannot be traced to complete the missing details. 19 Although missing the patients’ addresses may be inconsequential, it is critical when it comes to cancer geospatial mapping and epidemiological studies.
In the hospital details, the hospital name was the most filled (82.6%), followed by the ward (53.9%), the inpatient/outpatient number (49.7%), and lastly, the unit (40.6%). The missing inpatient/outpatient Identification numbers could be attributed to the independent nature of the different hospital clinics and lack of uniformity in allocating these identifiers in the clinics. This issue has currently been resolved by the introduction of some sort of electronic system in the hospital, although it is not yet fully rolled out in all the units, especially the laboratories. Our records are more incomplete compared to Nigeria, where the ward was filled in 82.8% of the forms and the unit/department was filled in 85.1%. 7 In another study still in Nigeria, the hospital number was provided in 66.0%, much higher than that observed in the current study. 8 A proper record of where a particular patient can be found in the hospital is cardinal. It eases patient follow-up and relay results to the patients. This would eliminate a common scenario where patients or caretakers are asked to go pick up their results but can’t find their way around the unfamiliar hospital environment. This creates unnecessary distress to already distressed patients/caretakers.
Only 7.5% of the forms had complete clinical information, 92.5% had incomplete clinical details and 0% of the forms had no clinical information at all. Clinical history was provided in 90.3%, site of biopsy (89.1%), nature of specimen (64.7%), and working diagnosis/differential diagnosis (63.0%). Information on whether there was any previous biopsy done or not was the least provided (44.4%), and other investigations done before the biopsy were only provided in 31.7% of the forms. In this study, it was oversimplified that presence of the presenting complaint, and a brief detail on the duration of symptoms, was considered enough clinical history. If a stricter protocol of clinical history as it is taught was to be followed as done by Ansar et al none of the forms would have been considered complete. 20 Despite the oversimplification, LRFs in our laboratory are still far more deficient in clinical information. Similar studies conducted in Nigeria and Sudan that had clinical information complete in 83.5% and 51.7% respectively. The 2 studies also reported that 89.0% and 89.2% of the request forms respectively had the nature of specimen (excision, incision, punch, shave biopsy etc.) indicated compared to only 64.7% in the current study.8,18 A more recent study in Pakistan recorded even more impressive results. It reported 100% documentation of dates of biopsy, site and size and 72% documentation of the nature of biopsy. 21 Clinical history is paramount to the pathologist since it is the only mirror through which they can see the patient. Adequate history reduces the diagnostic turnaround time and also helps in ruling out and ruling in some differentials which quickens the process of diagnosis and may ultimately improve overall patient care. The site from where the biopsy was picked was reported in 89.1% of the forms, much better than the 47% reported in Nigeria. 2 Clinical diagnosis was provided on 63.0% of the LRFs compared to 93.7% and 80.9% in studies conducted in Pakistan and in South Africa respectively.17,22 Only 47.1% (Table 3) of the clinical diagnoses were concordant with the final histological diagnosis, which is less than 69% observed in Nigeria. 2 This could be explained by several factors. It has been observed that many at times, the LRFs are filled by nurses, students, and on occasions interns. Most of the times these people are not aware of the exact diagnosis. On the other hand, it could also point to poor history gathering by internees who are usually the first people to interact with patients or even the managing doctors, thereby missing potential differential diagnoses. Unfortunately, this could also be telling of the quality of care that patients are receiving. In the current study, prior investigations were provided in 31.7% of the forms, much higher than the 18% reported in a Nigerian study. 2
Requesting clinician details were complete in only 5.6% of the forms, and the name was filled in 89.8% of them. This was closer to the 84.5% reported in Nigeria but less than the 98.3% observed in Pakistan 22 and yet better than the 32.8% reported in Sudan. 18 The requesting doctor’s contact was present on 19.5% of the forms, a finding similar to that observed in Pakistan (19.4%) 22 and much higher than 0.05% reported in South Africa and 6.8% reported in Sudan.17,18 Only 11.8% of the requesting physicians had signed their forms, a finding much lower than the 72.9% observed in a Nigerian study. 8 At least the clinician’s name and contact must be included in the LRFs. With such poor completion rates of request forms, these details would quicken cross-consultation between the pathologists and clinicians. In the end, it would lessen the TAT, improve the quality of reporting, and allow patients to receive care in time, but also reduce the backlog on the pathologist’s desk. Many times, if a pathologist requires more details about a case but cannot get in touch with the requesting clinician, such cases end up being pushed under the desk and are never signed out, which denies patients care.
Various types of specimen containers are used at the laboratory. From the current study, only 79.7% of the containers were appropriate for the specimen size, had a wide mouth, and were leak-proof. This was contrary to a recent study conducted in Khartoum, where 84.3% of the specimen containers were appropriate to the size of the specimen. 18 Among the specimen containers, whether appropriate or not, 91.3% were plastic, and the rest were either metallic (0.2%) or glass (8.5%). A significant number of the containers (16.4%) had previously been used for other non-laboratory purposes like food or spice storage, normal saline bottles, medicine bottles, syringes, and detergent containers, among other uses (See Figure 2). This is similar to the observations in Sudan, where some doctors were using jam bottles, saline containers, and injection bottles as specimen containers. 18 An appropriate container should allow the tissue to float in it freely without touching the edges of the container. The right container size allows for enough fixative to be added and achieve the recommended specimen to fixative ratio of (1:10-20) and enable proper fixation, which is pivotal to all the subsequent processes. 23 Every laboratory must ensure that the right container is used for the right purpose. Some containers may have remnants of chemicals that can alter the pH of the formalin, which might interfere with other subsequent processes like immunohistochemistry. Artifacts may also be introduced into the tissue, and this may lead to misdiagnosis, wrong diagnosis or even alter the antigenic properties of the tissue.

Micrographs demonstrating the different varieties of inappropriate specimen containers.
When it comes to the labeling of specimen containers, only 1.2% of the containers were labeled appropriately. Patient age, gender, procedure date, specimen type, and biopsy site were missing on 74.7%, 65.7%, 48.6%, 18.8%, and 32.9% of the forms, respectively. The patient’s name was either missing completely or only one name was written on 2.2% of the containers. The labeling of the specimen container is worse (1.2%) compared to 67.5% in Nigeria and 95.7% in Pakistan.2,24 Inappropriately labeled specimen containers create room for mixing up patient samples, which can be detrimental, especially in regions where people have similar names. This may cause a wrong diagnosis to be given to a wrong patient, which would ultimately result in the wrong treatment and undue side effects.
Conclusion
The completion rate of the LRFs in this study was unacceptably low and it was observed that specimen containers used at times are unprofessional. Clinician details were the least filled, followed by clinical details, hospital details, and lastly patient biodata as the most complete parameter. Of the individual variables, Physician signature was the least filled (11.8%) while patient name and age were the most filled (99.5%). Because of the incompleteness of LRFs, there is a high risk of errors in the laboratory.
From this study, we recommend the following:
Standard operating procedures (SOPs) about handling histology specimens, tissue rejection, and test requisition requirements should be populated in wards and theaters to act as constant reminders to clinicians.
Additionally, frequent multidisciplinary meetings and continuous medical education (CME) sessions should be introduced as constant reminders to clinicians. This is especially required whenever new intern doctors, residents, and nurses are recruited, since they are the ones mostly involved in investigating patients.
Lastly, more frequent audits of the LRFs should be done, and feedback should always be provided to the respective departments to encourage them to improve in areas where they might be lacking. Feedback about the findings in this audit will be given in the multidisciplinary hospital meetings and at the annual research dissemination conference organized by Mbarara University annually.
Limitations
In this audit, it was difficult to determine which departments had the most incomplete forms and we also could not determine the level of training of the requesting clinicians, as this could have a significant bearing on the completion rate of the LRFs. This was because of the high turnover rate of clinicians in our main hospital, which the laboratory serves. This study started at a time when a new lot of intern health workers had just been recruited, and this potentially affected the outcome. And being a cross-sectional study, it was difficult to analyze the behavior of clinicians over time regarding filling LRFs, and therefore, causality cannot be determined.
Also, because it was a cross-sectional study, we could not decipher the exact cause of such incomplete records. After dissemination of these results, we shall embark on the Plan-Do-Study-Act to determine the improvement in filling the request forms.
Supplemental Material
sj-docx-1-pat-10.1177_30502098251413817 – Supplemental material for Appropriateness of Histopathology Specimen Containers and Completeness of Laboratory Request Forms in Uganda: A Case of Mbarara Regional Referral Hospital, South Western Uganda
Supplemental material, sj-docx-1-pat-10.1177_30502098251413817 for Appropriateness of Histopathology Specimen Containers and Completeness of Laboratory Request Forms in Uganda: A Case of Mbarara Regional Referral Hospital, South Western Uganda by Yekosani Mitala, Raymond Atwine, Richard Dratu, Dianah Ishimwe, Francis Nsumba Kitenda, Joel Musayi, Isaac Keith Kasadha, Habert Tumwesigye, Vivian Naluzze, Simeon Eloba, Samuel Tumwesigire, Abraham Birungi and Brian Ssenkumba in Sage Open Pathology
Footnotes
Acknowledgements
We appreciate the help extended to us by the pathology department, especially the residents, for their effort during this audit. We also acknowledge the different Mbarara Regional Referral Hospital and all the surrounding healthcare facilities that provide us with the specimens. Lastly, the Faculty of Medicine of Mbarara University of Science and Technology provided financial support.
Ethical Considerations
The study received ethical approval from the MUST institutional review board, reference number MUST-2024-1495. Consent was not required for this audit to be done, but we obtained administrative clearance from the head of the laboratory.
Author Contributions
All authors made a significant contribution to this work, that is, in data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Faculty of Medicine of Mbarara University of Science and Technology under grant number MUSTFOM/SG/24/01.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
