Abstract
Around one-third of clinical drugs used today target G protein-coupled receptors (GPCRs). The action of hormones at GPCRs leads to downstream events predominantly mediated by cAMP through the activation of G proteins. The beta-arrestin (BA) pathway serves to recycle the receptor and desensitize the cell surface. Significant interest in manipulating GPCRs responsive to the incretins—glucagon-like polypeptide 1 (GLP1), gut insulinotropic polypeptide (GIP), and the glucose counter-regulatory hormone glucagon—has led to the development of agonists for these receptors. The development of molecules that act on more than one of these receptors has led to the field of unimolecular polypharmacy. The first of these agents to be commercially available for diabetes and obesity is tirzepatide (TZP). TZP is a full agonist at the GIP receptor and a partial agonist at the GLP1R. It is, however, able to elicit significant glucose reduction, increased insulin secretion, and weight loss; in addition, it improves insulin sensitivity independent of weight loss. The glucose reduction and weight loss are significantly greater than those achieved with semaglutide and are not fully explained by GIP action. Besides being an imbalanced agonist at the GIPR, TZP appears to be a biased agonist with reduced BA recruitment. This may in part explain the greater and prolonged action on the GLP1R of the molecule and its ability to reduce glucose. Other molecules in the pipeline appear to exploit this feature of biased agonism to overcome the limitations of dose escalation seen with traditional GLP1R agonists, aiming to reduce glucose and weight.
Keywords
Introduction
The activation of a receptor by a ligand or hormone sets in motion a series of events inside the cell. In specialized cells, such as those that secrete or respond to hormones, this could result in an enhancement of the principal function of that cell—agonism—or inhibition of its function—antagonism. A receptor may be activated by one or more related hormones. Similarly, a hormone may be active at more than one receptor. The interaction of a ligand (used interchangeably with hormone) with a receptor can lead to a quantitatively complete expression or inhibition of its functions—complete agonism or antagonism—or a partial expression or inhibition of its function—partial agonism or antagonism. In addition, there is mounting evidence that the ligand–receptor interaction, in some circumstances, can lead to a preferential stimulation or inhibition of one downstream effect of that interaction—this is called biased agonism.
The development of agents active against the neuroendocrine system, which mediated satiety, glycemia, and metabolism, has rekindled significant interest among clinicians in protein–ligand and ligand–receptor interactions.
G Protein-coupled Receptors
A third of clinically used drugs work at the G protein-coupled receptors (GPCRS). 1 These are a family of receptors located on the cell surface that receive signals from natural and synthetic ligands. Receptor phosphorylation evokes a series of downstream effects within the cell. GPCR activation also triggers a process of receptor internalization and desensitization by recruitment of non-G protein transducers. These include G protein receptor kinases (GRKs) and beta-arrestins (BAs). 2 The internalization is initiated by GRK phosphorylation followed by high-affinity binding to BA. BA scaffolds to endocytic proteins such as clathrin or adaptor protein 2 and promotes receptor internalization (Figure 1). While this is the predominant pathway, other mechanisms are also involved. 3 Traditionally, GPCR signaling was thought to be terminated by receptor phosphorylation followed by arrestin binding. However, receptor-bound arrestins appear to initiate a second round of signaling different from that mediated by G protein pathways.
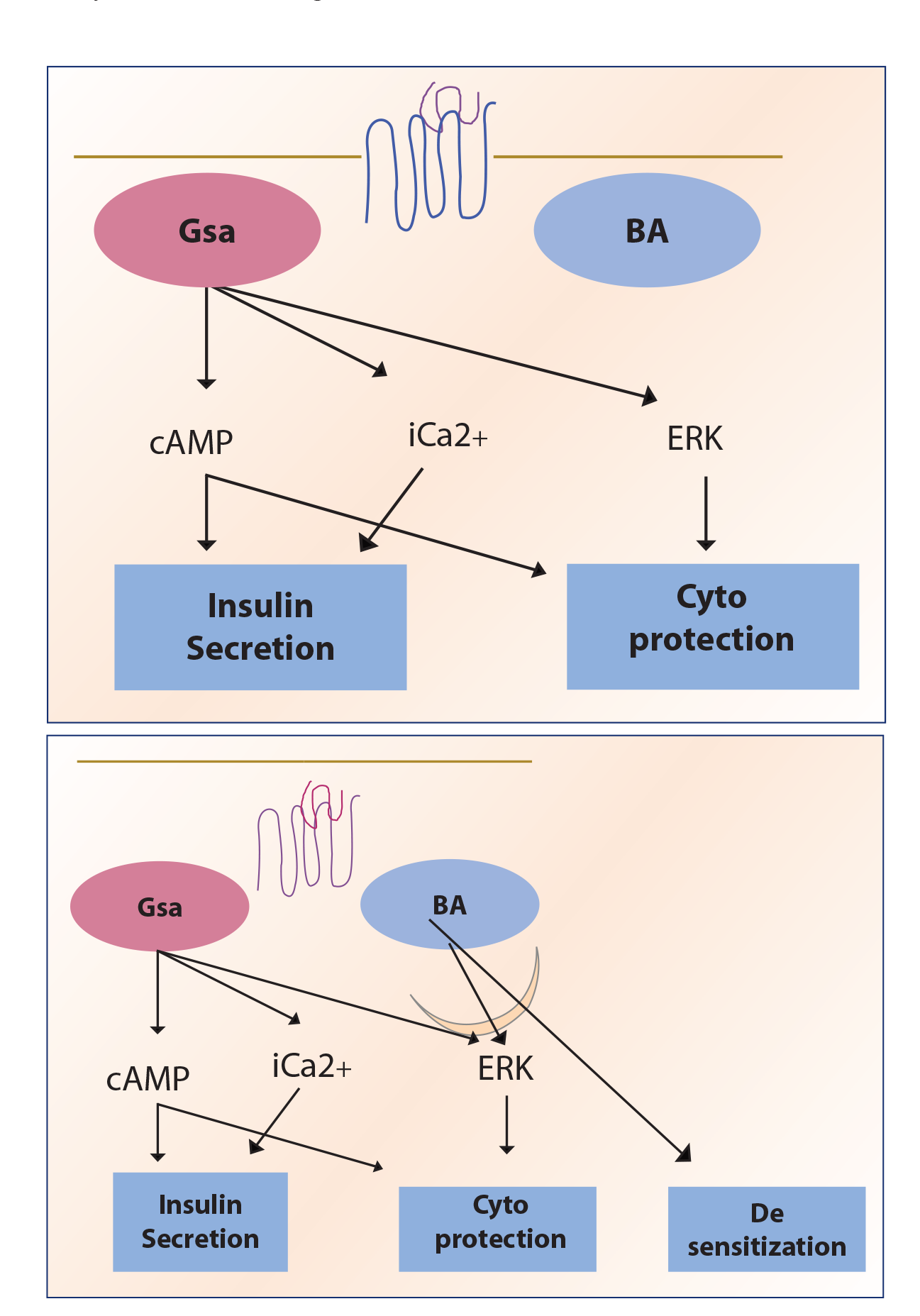
The glucagon receptor (GCGR), glucagon-like peptide 1 receptor (GLP1R), and gastric inhibitory polypeptide receptors are members of the Class B type of GPCRs. Their principal action appears to be mediated by Gαs, with a limited role for Gαq and Gαi. 4 Gαs coupling triggers an elevation in cAMP, protein kinase A (PKA), and exchange protein directly activated by cAMP (EPAC). GLP1R coupling also triggers internalization signals such as G Protein Receptor Kinase (GPRK) and BA.5,6
Postreceptor Action at the GLP1R
After GLP1 activates the receptor, the ligand–receptor complex is internalized fairly rapidly and is either recycled back to the cell surface or degraded. This action appears to be initiated by GRK. However, there is evidence suggesting that the activated receptor continues to be active while in the endosome. Ultimately, however, these are targeted by lysosomes. Internalization also reduces the amount of GLP1R available at the surface for activation (desensitization).
GLP1 action at its receptor is measured by either its ability to recruit Gα protein subunits and BA or its ability to signal intermediaries—cAMP, extracellular signal-regulated kinase 1 (ERK1), ERK2, or increase in intracellular Ca2+. cAMP-dependent pathways are crucial for the effects of GLP1R activation. 7 PKA is a target of cAMP secretion and may be responsible for GLP1R activation-mediated insulin secretion as well effects in the brain. EPAC2, a guanine nucleotide exchange factor, is another target for cAMP-mediated insulin secretion by GLP1. Intracellular calcium is particularly important in electrically excitable cells such as neurons and beta cells; intracellular Ca2+ increase may be mediated by Gαs and partly by Gαq.
The second pathway for GLP1R action involves recruitment of the BA—while it has been traditionally thought of as G protein-independent—recent evidence suggests that this may not be completely so. Its effects are measured by extracellular signal-regulated kinase 1/2(ERK1/2) phosphorylation. It must be noted that ERK phosphorylation may be partially dependent on PKA activation (see below)
There appear to be differences in the postreceptor activation of GLP1R and gut insulinotropic polypeptide receptor (GIPR). The GLP1R is rapidly internalized with a greater propensity for BA recruitment, endosomal activation, and lysosomal degradation; the GIPR, on the other hand, appears to have reduced coupling to G protein and BA, as well as reduced internalization endosomal activity, increased recycling, and an overall increase in beta-cell signaling despite highly reduced levels of endogenous surface receptor expression. 8 There are species differences in GIP action at receptors.
Beta Arrestins
The arrestins are a group of small cytoplasmic proteins—named for their function—which is to arrest signaling through GPCRs. There are four subtypes; two of these, BA1 and BA2, so named because they were originally studied in beta-adrenergic receptors, are important mediators at the GPCR. They have no known enzymatic function. Two isoforms of BA, BA1 and BA2 are present in the pancreatic cell and seem to have distinct and important roles. BA2 is GRK-independent and is important in mediating desensitization of GLP1R signaling, while BA1 is dependent on GRK and modulates downstream events, including insulin signaling and beta-cell proliferation.
Arrestins bind to the active, phosphorylated form of the receptor, thereby blocking the access of G proteins to the GPCR and terminating G protein-mediated signaling. At the same time, the receptor-arrestin complex initiates other signals. The most prominent of these is ERK1 phosphorylation. ERK1 phosphorylation sends a pro-proliferation, pro-survival signal. The mechanism of arrestin-mediated ERK stimulation is unclear.
Action of Pharmaceutical Agents at the GPCR
Synthetically engineered peptides and small molecules are able to engage the GPCRs and lead to actions that either mimic, enhance, or antagonize their function. In addition, some of these agents are able to achieve these outcomes at more than one receptor—a phenomenon known as unimolecular polypharmacy. A ligand can mimic the function of the native ligand completely or partially—acting as a complete agonist or partial agonist. An antagonist does not elicit a signal response but competes for receptor binding. An inverse agonist is able to suppress basal receptor signaling. When a molecule is active at more than one receptor, it can elicit a maximal signaling response at both (balanced agonist) or a maximal response at one and a submaximal response at the other (imbalanced agonist). After engaging the receptor, the interaction can either activate all pathways triggered by the native ligand or selectively initiate one or a few pathways, a phenomenon known as biased agonism (Figure 2).
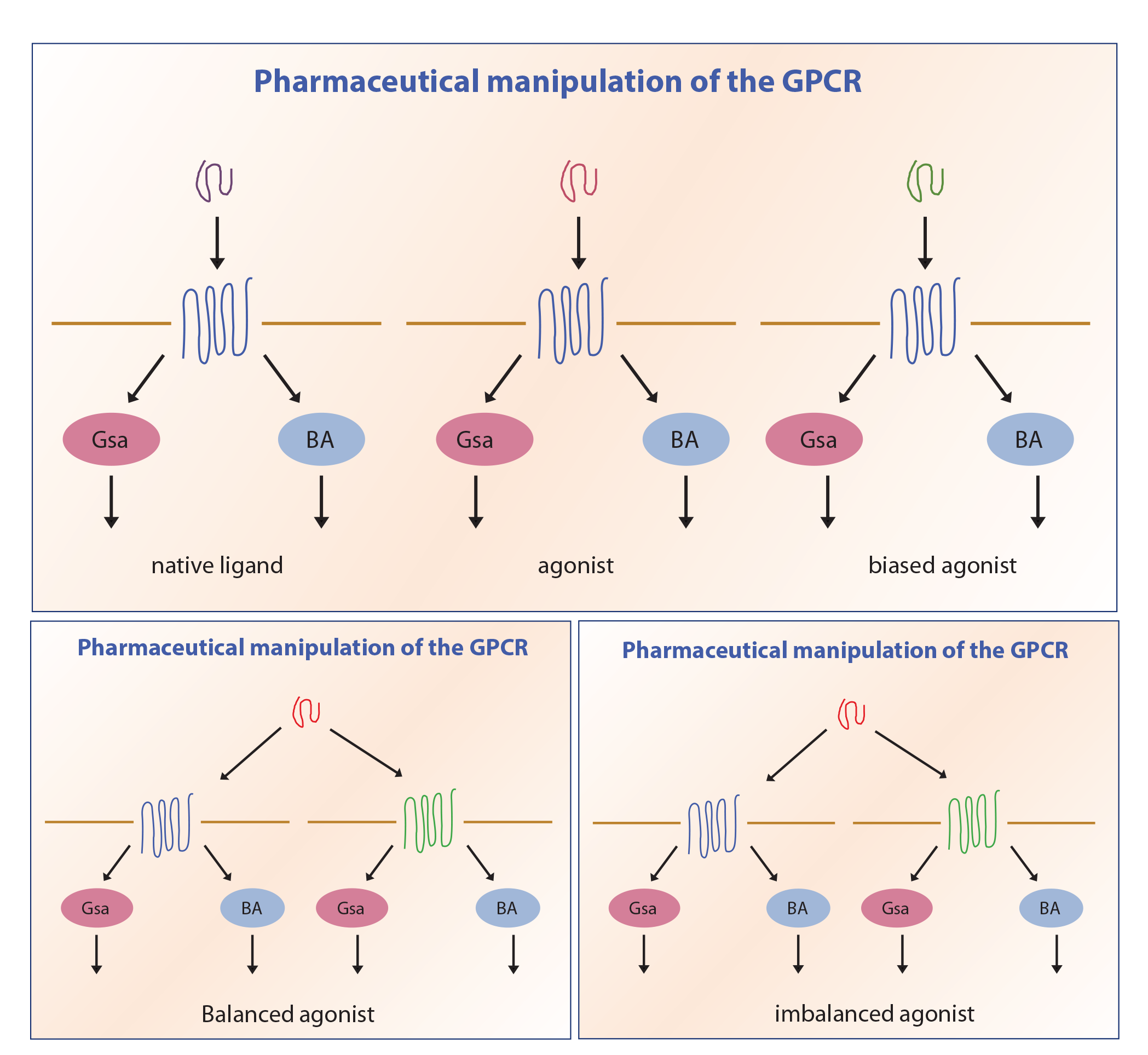
Biased Agonism
Biased agonists do not fit the classical paradigm of GPCR signaling. They signal with different efficacies in different pathways and hence can be agonists, partial agonists, or even antagonists in these pathways, thereby allowing the activation of selective responses. This appears to occur by stabilizing the receptors in conformational states other than the classical active or inactive states. 9
Biased agonism at the GLP1R with preferential activation of cAMP was first demonstrated in 2007. 5 It is difficult to quantify the effects of native GLP1-like peptides, however, because of cross-reactivity with, for example, GCGRs in the instance of oxyntomodulin. Similarly, there is no clarity about biased agonism for older agents such as lixisenatide, liraglutide, and dulaglutide—most appear to be either full agonists or with slight reduction in some pathways. 7
Exendin Phe1, in which the N-terminal histidine was switched to phenylalanine, showed full agonism at cAMP but markedly reduced BA recruitment and receptor endocytosis. There was greater insulin secretion by beta cells, faster dissociation kinetics suggesting less receptor recycling and desensitization. It also showed a greater effect on glucose lowering despite similar pharmacokinetics as exendin. 7 Interestingly, Exendin Phe has reduced efficacy of Gsα recruitment but is still able to generate cAMP signals. It appears that N-terminal modifications of amino acids of GLP1 seem to confer bias toward cAMP. On the other hand, modification of the mid-helical residues from alpha to beta amino acids seems to confer bias in favor of BA. 10
Tirzepatide and Biased Agonism
Tirzepatide (TZP) is a 39-amino acid linear peptide with agonist activity at both the GIPR and the GLP1R. The pharmacokinetic properties of TZP are affected through the conjugation to lysine located in the middle of the molecule to a C20 fatty di-acyl moiety via a hydrophilic linker.
Pharmacologically, TZP is an imbalanced agonist favoring GIPR over the GLP1R. The binding of GIPR is equivalent to native GIP. However, its binding to GLP1R is approximately fivefold weaker than native GLP1. 11 The strong GIPR activity is attested by a significant improvement in insulin sensitivity independent of weight loss. 12 Knockout models using GLP1r−/− and GIPr−/− demonstrate the ability of TZP to increase insulin secretion and reduce glucose concentrations through action at either receptor. Some of the weight loss of TZP can be attributed to GIP agonism in the adipose tissue and brain. 13 The additional action of the molecule on GIP does not, however, explain all the effects of TZP.
When compared with a weekly dose of 1 mg of semaglutide, the lowest dose of TZP has greater reduction in hemoglobin A1c. 14 In this study, the A1C reduction with the lowest dose of TZP was greater than semaglutide. Topline results from the SURMOUNT-5 study suggest greater weight loss with TZP than higher doses of semaglutide. Clamp studies demonstrate that there is significantly greater insulin secretion and insulin sensitivity with TZP than semaglutide, 15 similar to the effect of Exendin Phe1 (above).
While there is general similarity and orientation of TZP with GLP1, there is weaker binding of TZP to GLP1R. This could be explained by spatial shifts, N-terminal pocket density, and the C20 fatty acid moiety, overall resulting in a lower conformational fit of TZP at the GLP1R (but not the GIPR). Removal of the fatty chain, for instance, results in a fourfold increase in affinity of the molecule to the GIPR but no change in the affinity of the molecule at the GLP1R. 16 TZP features tyrosine at its N terminus, which is required for high GIPR affinity but has also been demonstrated to reduce BA recruitment efficacy at the GLP1R. 17
At the GIPR, TZP demonstrates comparable efficacy with GIP regarding GIP binding, cAMP generation, BA recruitment, and receptor internalization. In contrast, at the GLP1R, there is a fivefold lower affinity in competition binding and a 20-fold lower potency in cAMP accumulation, with a 20-fold rightward shift in TZP potency relative to GLP1R at all receptor densities. Additionally, BA recruitment was of low efficacy (Emax <10%).
cAMP generation and BA recruitment by TZP at the GIPR are comparable to native GIP. However, at the GLP1R, BA recruitment is reduced. Similarly, while TZP-induced internalization of the GIPR matches that of GIP, internalization at the GLP1R is significantly reduced by about 40%; this is consistent with its inability to recruit BA. 11
The reduction in BA recruitment in a cell with a high receptor reserve, such as the beta cell, would allow a low-efficacy agonist to generate a full cAMP response at the GLP1R. Reduced desensitization due to lower BA recruitment ensures that the resultant insulin secretion persists longer than with a higher-efficacy agonist. On the other hand, when GLP1R density is lower, such as in the central nervous system, the low-efficacy agonist does not elicit a full response; it still benefits from reduced desensitization, and its physiological effect matches that of the high-efficacy agonist.
Biased Agonism as a Pharmaceutical Strategy at the GPCR
The action of TZP at the GLP1R holds more than just a mechanistic curiosity. There are suggestions that non-recruitment of the BA pathway may reduce nausea, which is a rate-limiting factor in dose escalation of GLP1RAs. Although inconclusive as yet, biased agonism may provide a workaround.
There is significant interest in developing molecules that increase cAMP signaling at the expense of BA recruitment. The small molecule GLP1R agonist orforglipron is one such agent with unique binding distinct from other GLP1 agonists. It is a full agonist at high receptor densities but a partial agonist at lower densities. Oforglipron appears to benefit from partial agonism to achieve stimulatory potential at low receptor densities.
One of the concerns about biased agonism as a pharmacological approach is the potential long-term effects of reduced BA signaling. However, at this point of time, there is no evidence of an adverse long-term consequence from this approach.
With the increasing development of dual, triple, and poly-agonists, variations such as imbalanced and biased agonism may become the norm rather than the exception in one or more receptor–targeted therapies.
Conclusion
The decade has been characterized by the availability of highly potent agents active at the GPCRs of GLP1, GIPR, and glucagon. These agents seem to function in a mode that differs from our traditional understanding of the ligand–receptor downstream actions. Biased agonism, as demonstrated by TZP at the GLP1R, is one explanation for the increased glucose lowering and weight loss seen with it. Biased agonism may be useful in mitigating dose-limiting toxicity associated with these agents. Additionally, biased agonism may help achieve the full effect of the ligand in tissues with low receptor densities. Several molecules currently under investigated may help us understand the basis and therapeutic potential of this strategy.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
