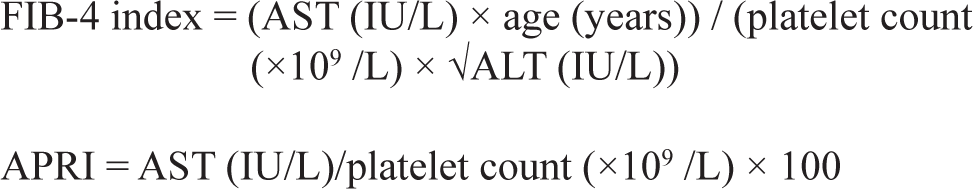Abstract
Aims:
Several studies support type 2 diabetes mellitus (T2DM) as one of the risk factors of metabolic dysfunction associated steatotic liver disease (MASLD). Recently, the use of pioglitazone and sodium-glucose co-transporter 2 inhibitors (SGLT2i) has been on the rise considering its beneficial effects in reducing liver dysfunction. We aim to determine the clinical effectiveness of these drugs in improving MASLD and T2DM.
Materials and methods:
We conducted a retrospective study of 279 T2DM patients with MASLD who were prescribed pioglitazone and/or SGLT2i from two clinics in India. The primary endpoint was change in fibrosis (FIB-4) index and the aspartate aminotransferase to platelet ratio index (APRI) score at one year follow-up. Appropriate statistical tests were applied to assess the significance of the outcomes.
Results:
Of the total (276) patients, 46% of patients were at low risk of advanced fibrosis, 11% were at high risk for advanced fibrosis, and the majority (50%) were on SGLT2i treatment. At follow-up, there is a statistically significant reduction in the mean (SD) FIB-4 index (1.92 [1.22] vs. 1.68 [0.99]) and APRI score (0.42 [0.34] vs. 0.33 [0.21]); p < .001. Among treatment groups, there is a significant difference in the mean FIB-4 index both at baseline (F: 4.23; p < .05) and follow-up (F: 3.45; p < .05). These scores were highly significant in the combination treatment with SGLT2i and pioglitazone (p < .05).
Conclusions:
Our results indicate a combination therapy with SGLT2i and pioglitazone might be beneficial in managing MASLD, especially in reducing the FIB-4 index and APRI score in Indian T2DM patients with MASLD.
Keywords
Introduction
Metabolic dysfunction associated steatotic liver disease (MASLD) previously called non-alcoholic fatty liver disease (NAFLD) is a chronic liver disorder having a prevalence of 25% globally. 1 The major risk factors of MASLD are T2DM, metabolic syndrome (MS), and obesity. The increased prevalence of these metabolic disorders can contribute to the increased incidence of NAFLD. 1 In June 2023 a multi-society Delphi consensus statement suggested that the use of new terminology MASLD, would be appropriate than NAFLD. 2
MASLD emphasizes liver steatosis along with at least one of five cardiometabolic risk factors, such as central and overall obesity, high blood glucose levels, elevated blood pressure, and abnormal lipids.2,3 It is characterized by liver steatosis and it may also manifest severe conditions such as liver cirrhosis, hepatocellular carcinoma, and non-alcoholic steatohepatitis (NASH). 4 The major predisposing and risk factor of MASLD is T2DM (Figure 1). Hyperinsulinemia and insulin resistance induce lipolysis in adipose tissue and enhance the absorption of fatty acids by hepatocytes. This leads to increased levels of fatty acids and triglycerides in the liver and causes steatosis, the diagnosis of which is done usually by ultrasound (liver elastography, i.e., fibro scan), liver biopsy, liver function test (LFT) and transient elastography (TE), etc. 1
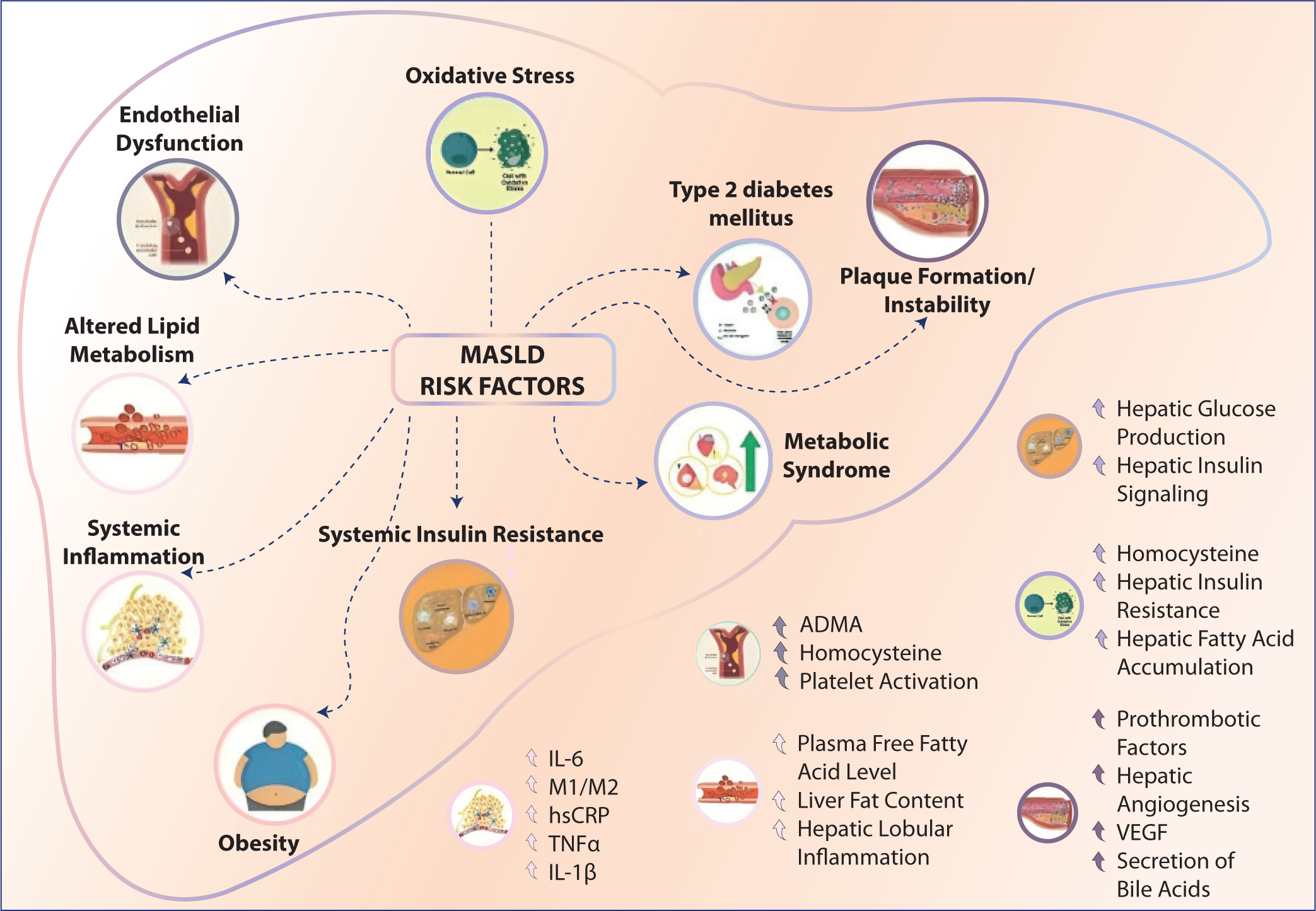
The SPRINT study conducted in Indian cities (n = 101) reported the prevalence of MASLD (56.5%) in T2DM patients aged between 25 and 84 years. 5 The prevalence was highest among northern states (72.4%), while the lowest prevalence was recorded in western states (44.1%).5,6 MASLD identifies patients with an elevated risk of hepatic steatosis progression. The use of pioglitazone appears to improve symptoms of MASLD such as hepatic steatosis, inflammation in steatohepatitis, and ballooning necrosis in patients with T2DM and prediabetes when used for six months and further reduced hepatic fibrosis when used for 18 months.7-10
Pioglitazone is a thiazolidinedione derivative and acts as an agonist of peroxisome proliferator-activated receptor γ (PPARγ), a nuclear receptor. 11 In patients with T2DM, pioglitazone improves insulin sensitivity. 12 It primarily exerts its action by reducing the systemic lipid concentration and redistributing visceral fat, thus reversing lipotoxicity and elevating plasma adiponectin levels. 11 It also exerts anti-fibrotic and anti-inflammatory effects in patients with MASLD and T2DM. 13 Thus, it improves both glucose and lipid metabolism.
There is an increased focus on the sodium-glucose co-transporter 2 inhibitors (SGLT2i) for the treatment of T2DM and MASLD7,10 as their pharmacological action is by controlling the reabsorption of glucose by renal proximal tubules and promoting the excretion of glucose through urine (Figure 2). 4 This reduces hyperglycemia and improves insulin resistance. 14 In addition they also help in reducing body weight and liver fat. 15 Studies suggest that SGLT2i positively impacts oxidative stress and inflammation in T2DM patients and improves non-invasive hepatic steatosis and fibrosis. 16
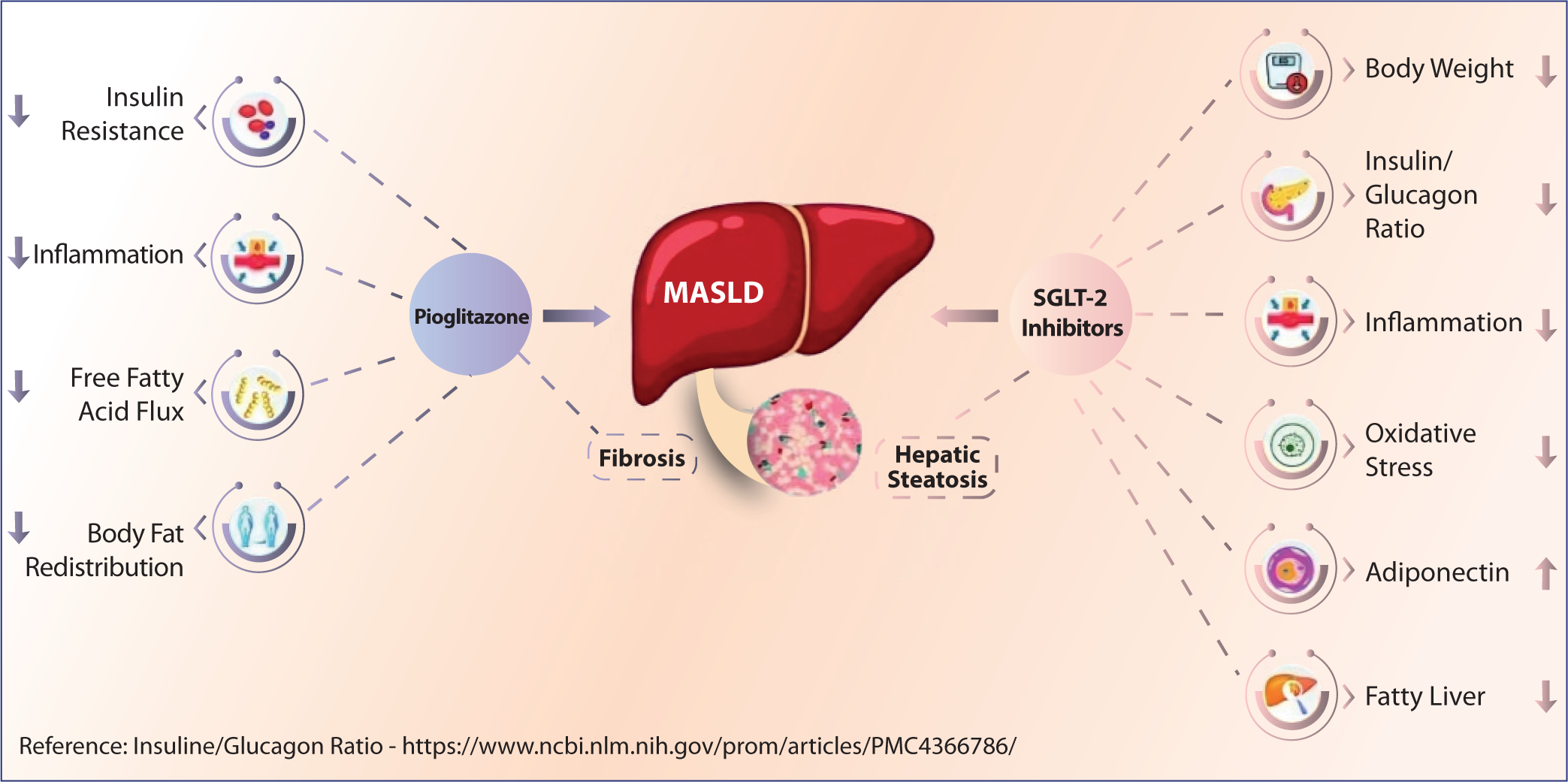
Akuta et al. conducted a five-year follow-up study on the use of the SGLT2i, canagliflozin in MASLD patients with T2DM and observed a long-term improvement in the histopathology. 17 The primary mechanisms through which SGLT2i regulates MASLD are targeting lipid metabolism, inhibiting de novo lipogenesis in the liver, and acetyl-coenzyme A (CoA) carboxylase (ACC) inhibition. 18 Few observational studies reported the use of SGLT2i namely ipragliflozin, canagliflozin, and luseogliflozin in patients with T2DM and NAFLD and they found that this class of anti-diabetes drugs reduced the liver enzyme levels, liver fat, and liver fibrosis indices. The investigators also observed that the SGLT2i improved glycemic parameters, insulin resistance, obesity, and dyslipidemia. 19
There are very few studies that evaluated the use of pioglitazone and SGLT2i in T2DM associated MASLD. Hence, our study aims to evaluate the clinical effectiveness of pioglitazone and SGLT2i, SGLT2i alone or in combination in improving the MASLD symptoms mainly in the improvement of fibrosis 4 (FIB-4) score and aspartate aminotransferase to platelet ratio index (APRI) score along with glycemic variables in Indian patients.
Materials and Methods
The present observational retrospective study included (n = 279) T2DM patients with MASLD who were on treatment with anti-diabetes drugs from two specialty clinics in south and western India. The study was conducted as per the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) - guideline for Good Clinical Practice (GCP) and approved by the independent ethics committee. The individuals who were under medication with pioglitazone and/or SGLT2i for <3 months and with no hepatic steatosis were excluded from the study. The hepatic steatosis diagnosis was done using ultrasonography. T2DM was diagnosed using blood glucose levels and glycated hemoglobin (HbA1c). All these assessments used to diagnose T2DM and hepatic steatosis were considered to define MASLD.
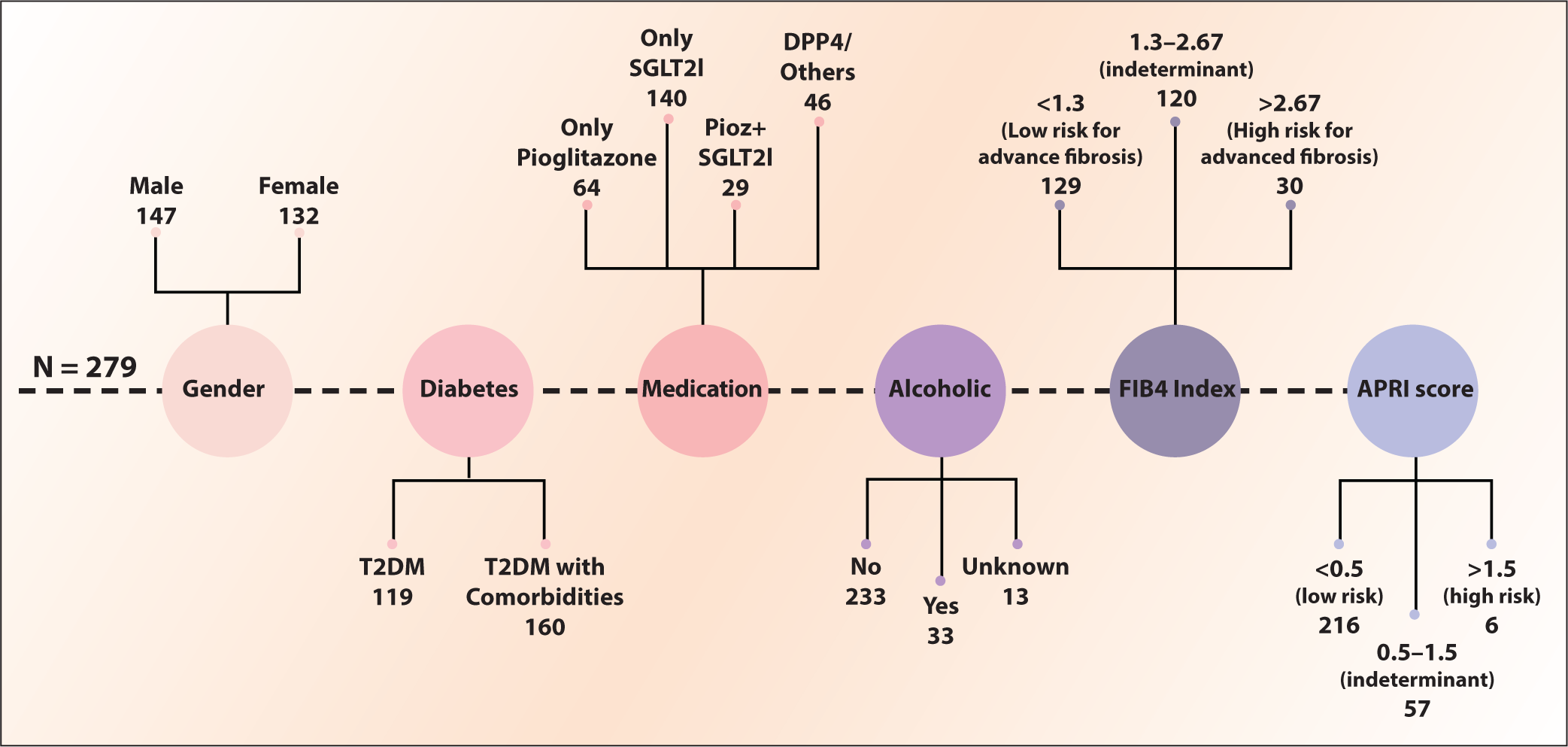
To understand the anti-diabetic drug SGLT2i and pioglitazone effectiveness, we stratified the individuals into three groups (a) the SGLT2i group (n = 140) (b) the Pioglitazone group (n = 64) and (c) the SGLT2i +Pioglitazone group (n = 29). There are 46 patients who were prescribed other anti-diabetes drugs (ADDs). The vitals and laboratory data obtained from the participants in each group were analyzed and compared between each treatment group. The data included weight, body mass index (BMI), fasting blood glucose (FBG), HbA1c, total cholesterol (TC), triglyceride (TG), low-density lipoprotein-cholesterol (LDL-C), and high-density lipoprotein-cholesterol (HDL-C), glutamic-oxaloacetic transaminase (SGOT), glutamic-pyruvic transaminase (SGPT), platelets, creatinine, blood urea nitrogen (BUN), and uric acid (UA).
Two markers of hepatic fibrosis, namely the FIB-4 index and APRI were also calculated. The standard formulas were used to calculate FIB-4 index 20 and APRI score. 21
Based on the FIB-4 index score, the patients were categorized into three groups: patients with FIB-4 index <1.3 as low risk for advanced fibrosis, between 1.3 and 2.67 as indeterminant, and >2.67 as high risk for advanced fibrosis. Similarly, based on the APRI score, the patients were stratified into three groups low risk (<0.5), indeterminant (0.5–1.5), and high risk (>1.5).
Statistical Analysis
Descriptive statistics was used to present MASLD data at baseline and follow-up. The data represented as the mean ± standard deviation (SD) for continuous variables, frequency and percentages for categorical variables. The comparison of baseline data between the groups was performed using an unpaired t-test. The categorical datasets among the SGLT2i, pioglitazone, and SGLT2i+pioglitazone groups were compared using Fisher’s exact test. The paired student’s t-test was used to analyze the change in the FIB-4 and APRI scores obtained at baseline and at follow-up treatment. Among the treatment groups, the changes in the APRI score and FIB-4 index were analyzed by analysis of variance (ANOVA). The statistical significance was considered to be at p < .05.
Results
Demographic and Baseline Characteristics of Participants
A total of 279 individuals diagnosed with T2DM and MASLD were included, their mean (SD) age was 53.7 (13.4), 147 (53%) were males and 132 (47%) were females. Mean (SD) weight was 73.5 (18.7) kg and BMI was 25.2 (9.9) kg/m 2 . The majority 233 (83%) did not report alcohol consumption and the others had an alcohol use within the limits of consideration of MASLD. Of these patients, 23% (64) were on pioglitazone, 50% (140) were on SGLT2i, 10% (29) were on a combination of SGLT2i+pioglitazone, and 16% (46) were on other ADDs. At the time of diagnosis or baseline, 46% of patients were at low risk of advanced fibrosis and 11% at high risk for advanced fibrosis. The complete demographic and baseline data of the participants are reported in Figure 3.
Baseline and Follow-up Characteristics of MASLD and Glycemic Variables
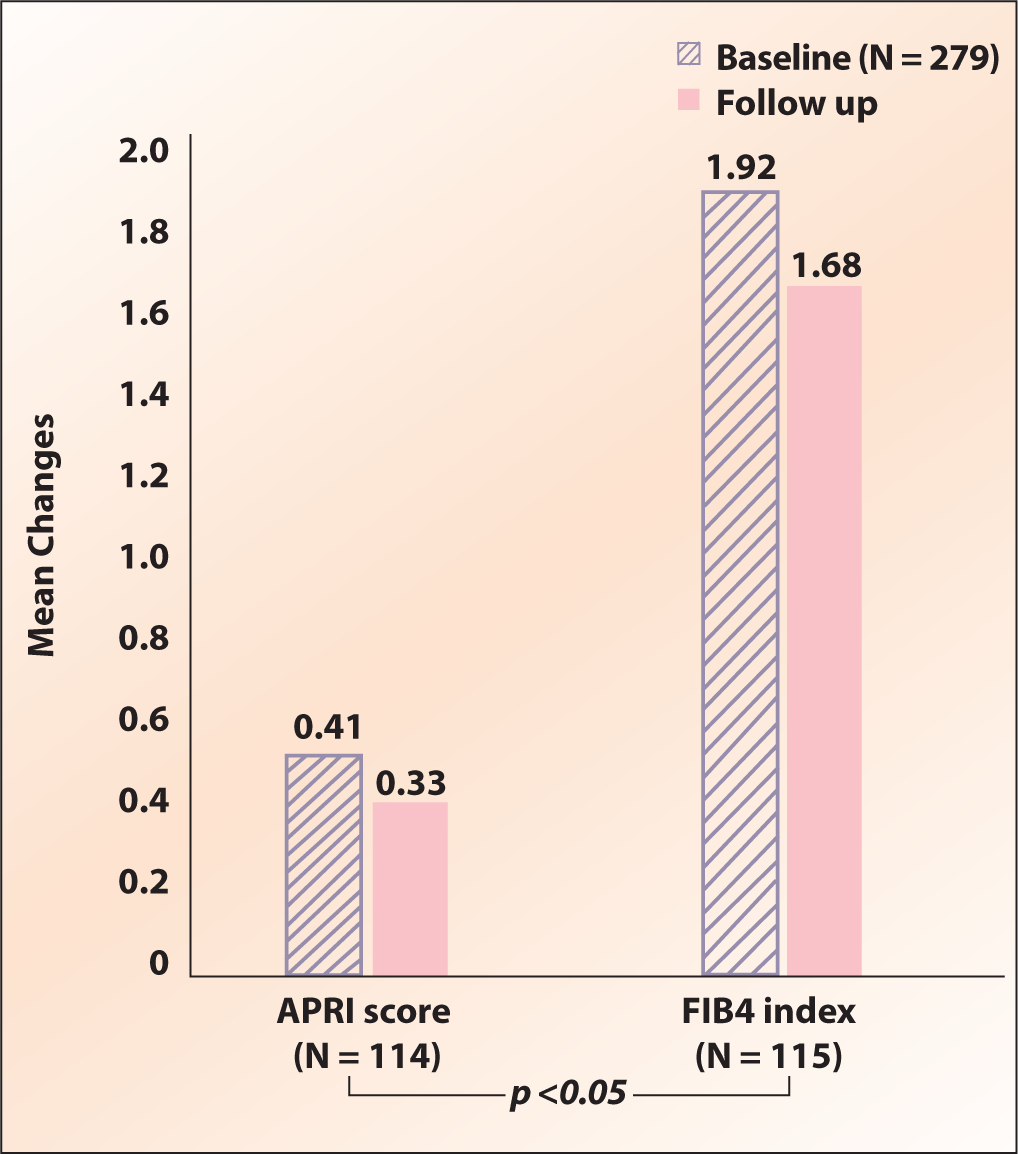
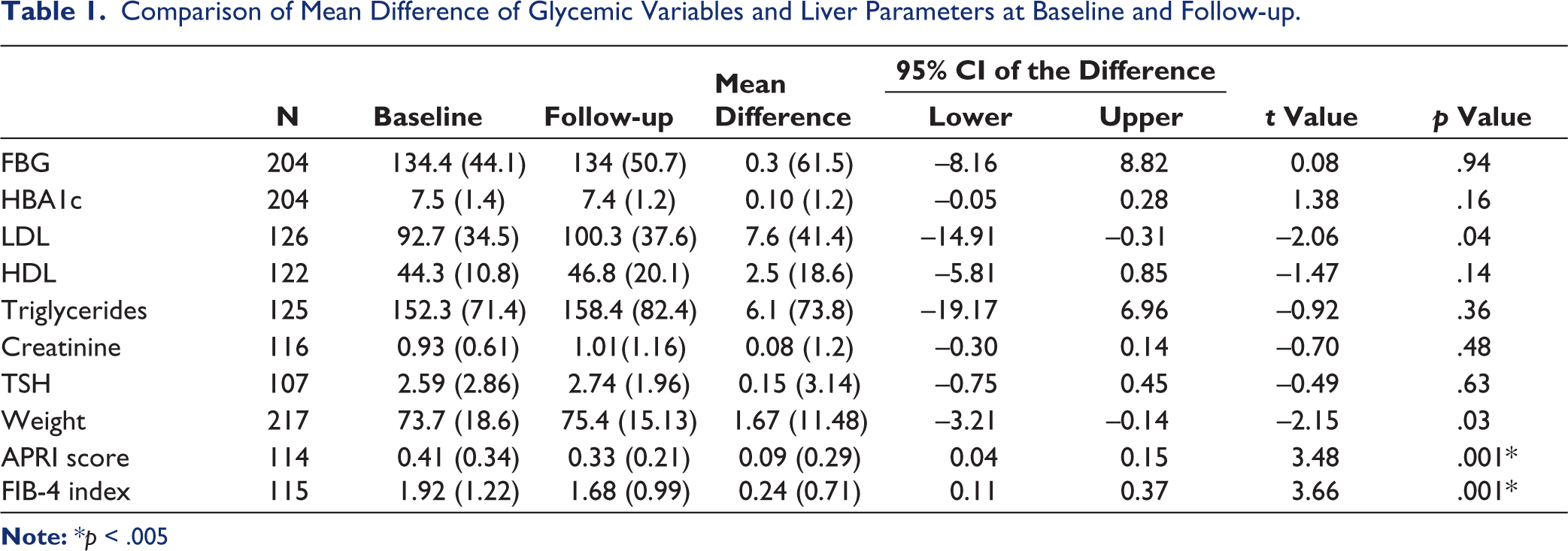
Paired analysis of MASLD and glycemic variables at baseline and follow-up, a significant (p < .05) reduction (0.1) in the mean APRI score (n =114) was observed at follow-up 0.33 (0.21) from baseline score 0.41 (0.34). Similarly, a significant (p < .05) reduction (0.24) in the FIB-4 index (n =115) was also observed during follow-up 1.68 (0.99) compared to the baseline 1.92 (1.22) (Figure 4). There was no significant difference observed with respect to other variables (Table 1).
Mean of Aspartate Aminotransferase to Platelet Ratio Index (APRI) and Fibrosis 4 index (FIB 4) Score at Baseline and Follow-up.
Comparison of Mean Difference of Glycemic Variables and Liver Parameters at Baseline and Follow-up.
Comparison of APRI Score and FIB-4 index Within Treatment Groups
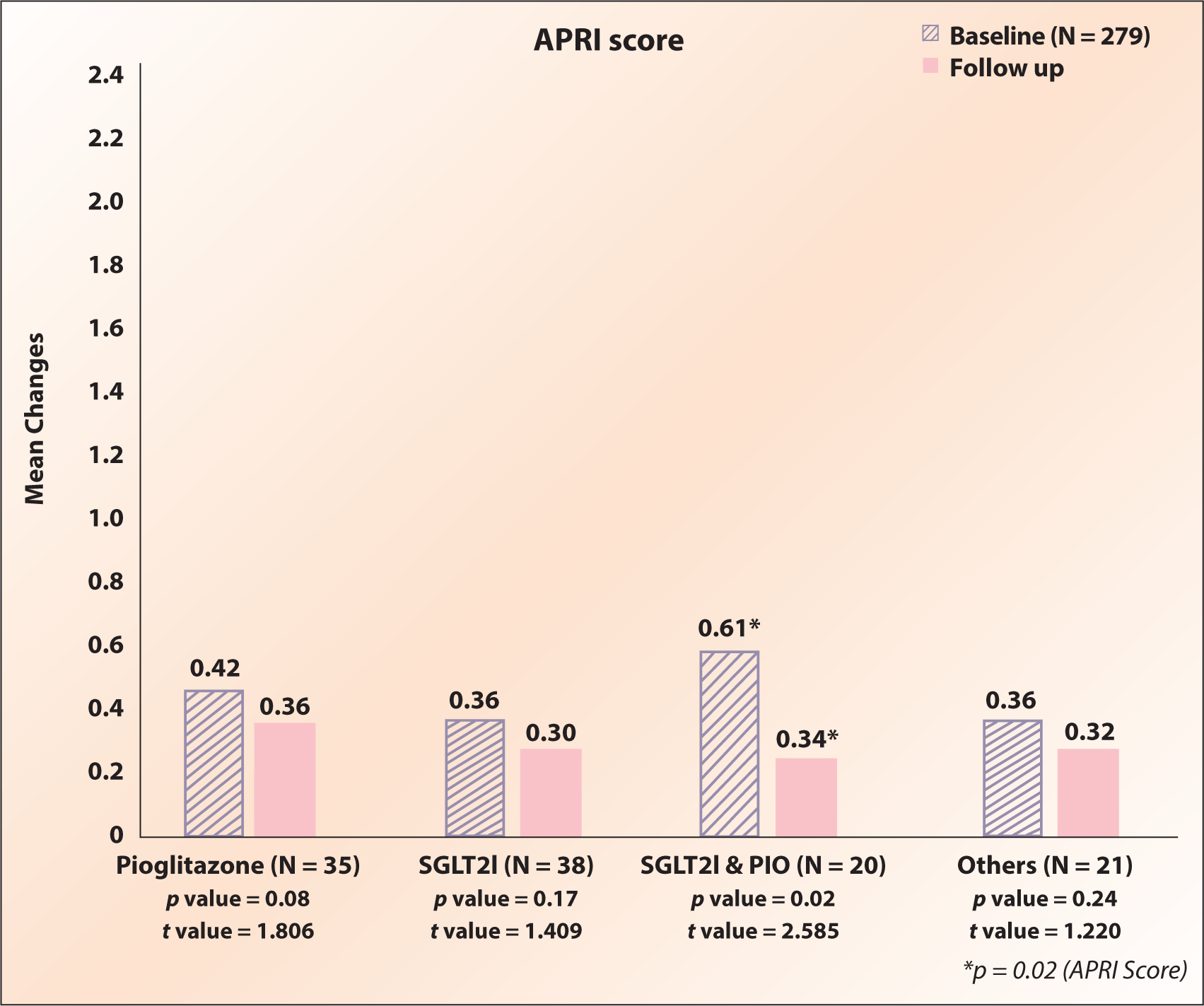
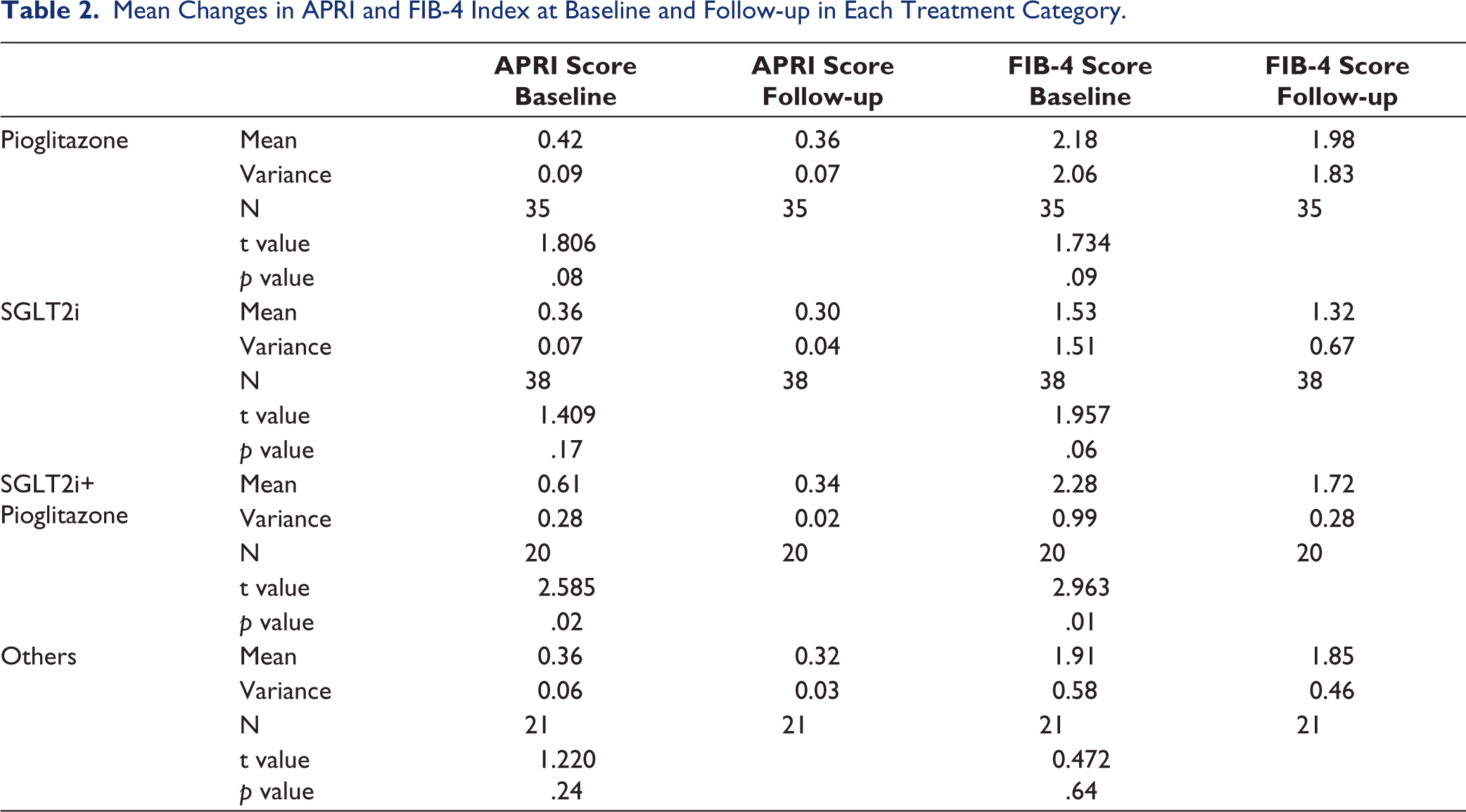
Within treatment groups (pioglitazone, SGLT2i, SGLT2i +pioglitazone, and others), there was numerical but not statistically significant improvement in mean APRI score (Figure 5) and FIB-4 index (Figure 6) from baseline to follow-up. Compared to individual drug treatment, in the combination treatment with SGLT2i+pioglitazone, both APRI score and FIB-4 index improved and were statistically significant (APRI score [p = .02] and the FIB-4 index [p = .01]) (Table 2).
Aspartate Aminotransferase to Platelet Ratio Index (APRI) Score in Piogliatazone, SGLT2i and Combination of Pioglitazone and SGLT2i Group at Baseline and Follow-up.
Fibrosis 4 Index (FIB 4) Score in Piogliatazone, SGLT2i and Combination of Pioglitazone and SGLT2i Group at Baseline and Follow-up.
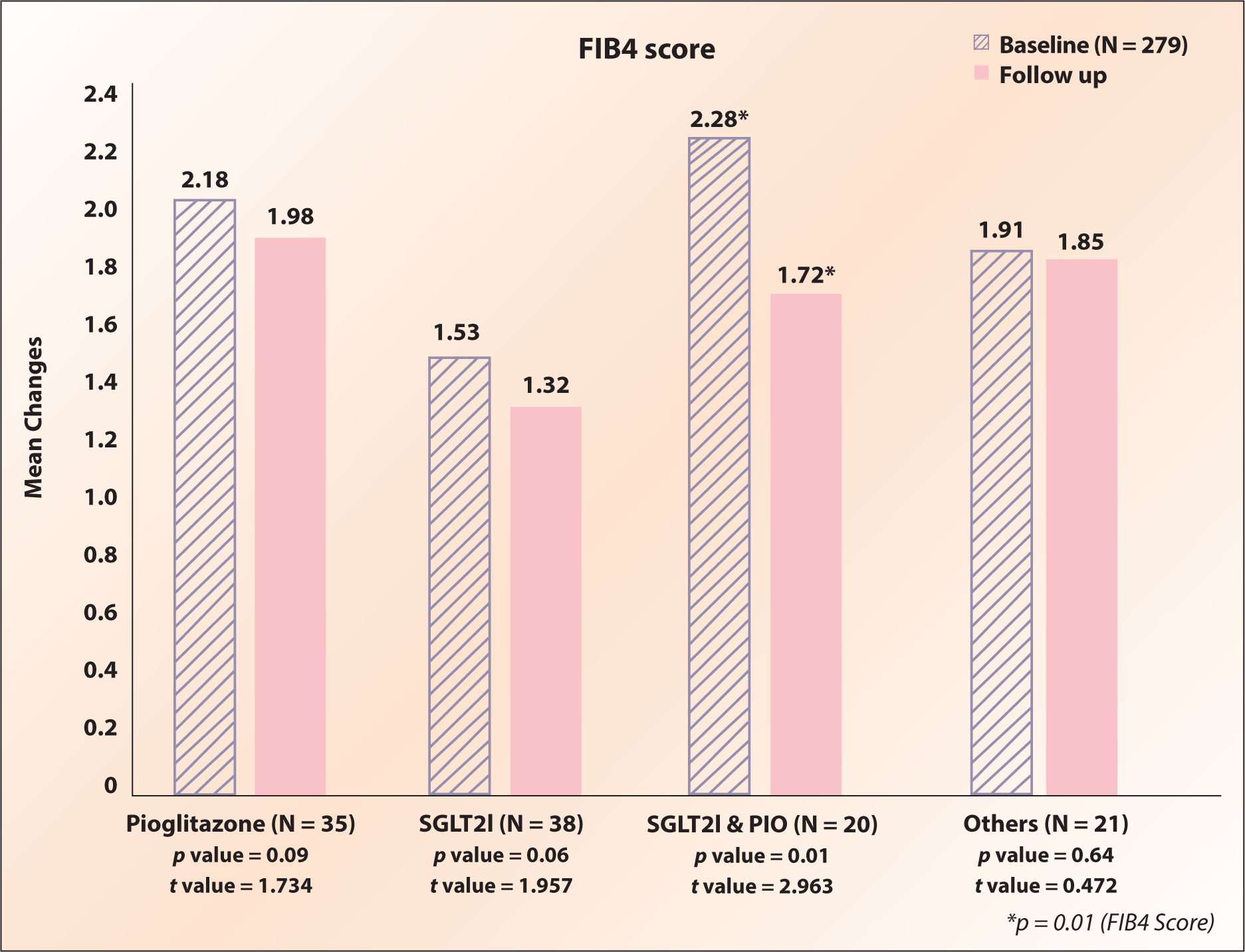
Mean Changes in APRI and FIB-4 Index at Baseline and Follow-up in Each Treatment Category.
Discussion
Ours is the first Indian study to evaluate the effect of SGLT2i and pioglitazone on fibrosis scores in patients with T2DM with NAFLD.
Mino et al. found a reduction in FIB-4 index in patients treated with SGLT2i alone but not pioglitazone, but the APRI score was decreased in both the groups suggesting that the treatment with SGLT2i had a greater effect on FIB-4 index in MASLD patients than with pioglitazone. 4 Some randomized clinical trials also reported that treatment with pioglitazone can help in non-alcoholic steatohepatitis (NASH) patients.22-25
Colosimo et al., study analyzed FIB-4 index in T2DM patients prescribed with dipeptidyl peptidase 4 inhibitors (DPP-4is), glucagon-like peptide-1 receptor agonists (GLP-1RAs), and SGLT2i. The effect of these drugs on steatosis and fibrosis after 6 and 12 months of treatment was assessed and observed a reduction in FIB-4 index in T2DM patients treated with GLP-1RAs and SGLT2i compared to others. 26
In the CANVAS (NCT01032629) and CANVAS-R (NCT01989754) trials, patients with MASLD using canagliflozin, an SGLT2i had improved APRI scores at 2.4 years follow-up. 27
Chehrehgosha et al., a placebo-controlled clinical trial evaluated the efficacy of empagliflozin in T2DM with MASLD compared to pioglitazone. They observed no significant difference in FIB-4 index in the empagliflozin group, however, the number of individuals classified as indeterminant category based on FIB-4 index was reduced. Similar results were also observed with respect to APRI score. 28
In the current study, we observed an overall improvement in FIB-4 and APRI scores in all the treatment groups. A statistically highly significant improvement was observed in the combination treatment of SGLT2i+pioglitazone. Our results suggest that T2DM patients with MASLD on treatment with more than one anti-diabetic drug may have an additive effect in controlling fatty liver and hence steatosis. Other similar studies have also suggested that the combination of SGLT2i and pioglitazone treatment showed significant improvements in MASLD symptoms. 29
In contrast to our results, a controlled study (TOPIND study) conducted by Yoneda and co-investigators found no difference in baseline and follow-up FIB-4 index in T2DM patients with MASLD treated with tofogliflozin, an SGLT2i and pioglitazone. 30
A single-center survey conducted by Das et al. did not observe any difference in FIB-4 index and APRI score in T2DM patients with MASLD when treated with dapagliflozin, an SGLT2i. 31 Likewise, a meta-analysis by Zhang et al., also did not observe improvement in FIB-4 or APRI score upon treatment with empagliflozin. 32
The limitations of our study are worth mentioning—it is a retrospective study of real-world clinical data from only two centers. There could be a possibility of bias in the sample selection, sample size, and short duration of follow-up. The anti-fibrotic effects using other liver biomarkers, and fibrosis scoring systems were not assessed due to the non-availability of data. Long-term prospective observational studies with a large sample size are warranted to establish the current study outcomes.
In summary, our results indicate that combination treatment with SGLT2i and pioglitazone has a better impact on the FIB-4 index and APRI score rather than individual drugs in T2DM patients with MASLD. Therefore, combination therapy with SGLT2i and pioglitazone might be beneficial in managing MASLD symptoms, especially in reducing FIB-4 score and APRI score in T2DM patients with MASLD from India.
Footnotes
Acknowledgements
Editorial assistance was provided by Medswan Global Healthtech Private Limited.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
The study was conducted as per the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) - guideline for Good Clinical Practice (GCP) and approved by the independent ethics committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was sponsored by USV Private Limited.