Abstract
Gynaecomastia is the proliferation of male breast tissue and is most often treated effectively with surgical excision of the glandular component. Late recurrence following a seemingly complete excision is unusual, particularly when the original disease was related to anabolic androgenic steroid exposure. We describe a healthy Caucasian male who had a history of anabolic steroid use more than a decade ago. He underwent bilateral excision of tender gynaecomastia seven years previously, with full symptomatic relief. He recently re-presented with new bilateral painful subareolar masses that extended laterally toward the chest wall. On exploration, firm, ‘banana shaped’ fibrous cords of glandular tissue were discovered, adherent to the serratus anterior muscle and the pectoral muscle. Histopathological analysis confirmed gynaecomastia with dense stromal fibrosis. Following meticulous surgical excision, the pain resolved, and the patient returned to normal activities. This rare presentation of late, bilateral, fibrous recurrence demonstrates that even after apparently complete excision, persistent microscopic glandular foci may evolve over time. Clinicians should be aware of the fibrous phase of gynaecomastia, particularly in men with a history of anabolic steroid exposure. Recurrent fibrotic disease in patients who present with new painful chest wall masses many years after primary surgery should be considered and consented to during the preoperative consultations.
Keywords
Introduction
Gynaecomastia is characterised by the benign proliferation of breast tissue, caused by an imbalance between oestrogenic stimulation and androgenic inhibition. It has a variable prevalence depending on age, ranging from transient neonatal gynaecomastia to peaks in adolescence and again in older men. 1 One of the most notable causes is the use of anabolic androgenic steroids (AAS), which are sometimes used by bodybuilders and athletes seeking muscle hypertrophy. These agents, through aromatisation, may be converted to oestrogens while also suppressing endogenous production, leading to gynaecomastia. 2 These cases tend to consist of glandular tissue, which is much more fibrous and can be very painful for the individual. 3
Surgical excision is generally regarded as the definitive management for persistent, symptomatic gynaecomastia. The recurrence rate following thorough excision of glandular tissue is considered low, with some studies suggesting that only a small proportion of patients experience regrowth. Recurrence was more commonly observed in patients with gynaecomastia that mainly consisted of fatty tissue. 4
In the case of AAS-induced gynaecomastia, the evolution of gynaecomastia tissue into a fibrous or hyalinised phase may result in the reappearance of firm, painful masses that are easily mistaken for scarring or, more rarely, malignancy. 2 This case illustrates such a phenomenon; the unusual late, bilateral recurrence of dense fibrous gynaecomastia seven years after primary excision of steroid-induced disease. The recurrence extended laterally far beyond the boundaries of the primary gynaecomastia, which was much more localised. The extent, size and shape of the recurrent gynaecomastia were unusually large and extended laterally up to the serratus anterior muscle.
Case Presentation
The patient was a caucasian man in his early forties, otherwise healthy and a father of young children. He reported a remote history of AAS use approximately a decade before his initial presentation. He had ceased all steroid use prior to his primary surgery. At that time, he presented with bilateral tender gynaecomastia beneath the nipple areola complex (NAC). He underwent bilateral excision, with full resolution of symptoms and a satisfactory cosmetic outcome (Figure 1).
Initial Presentation and Primary Surgery of This Patient Presenting with AAS-induced Gynaecomastia.
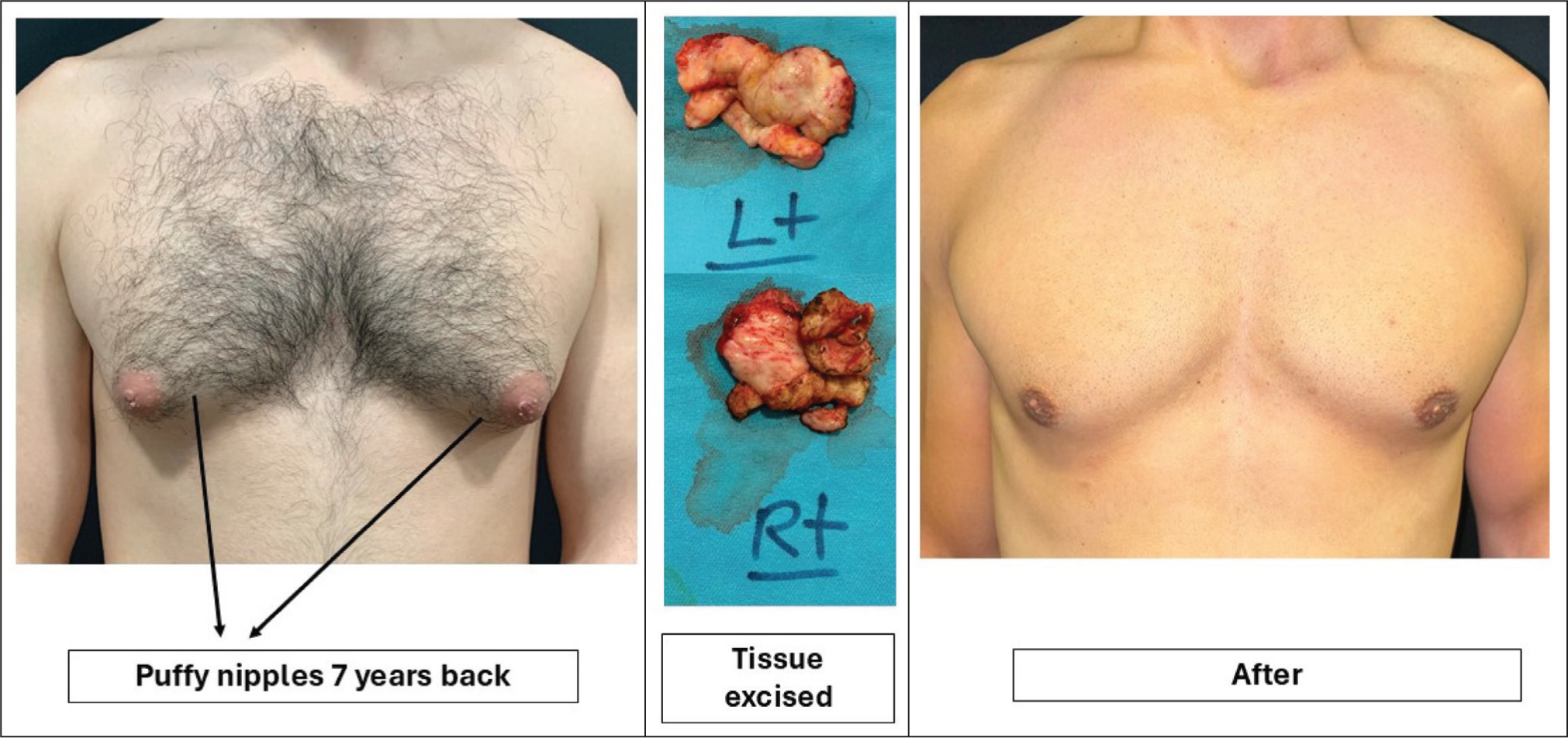
Seven years after his primary operation, he returned to the clinic with new complaints. He had noticed the development of painful lumps beneath both nipples, which extended laterally toward the outer chest wall. He had not been using AAS during this time. He described the pain as particularly bothersome when hugging his children, during physical exercise, or when his chest was compressed by clothing. He denied any systemic symptoms such as weight loss, fatigue, or nipple discharge.
On examination, bilateral firm subareolar masses were palpable, extending laterally and producing discomfort with pressure. There were no overlying skin changes, no axillary lymphadenopathy and no clinical features to suggest malignancy.
A clinical diagnosis of recurrent gynaecomastia was made, with a differential that included scar tissue, fatty regrowth and, less likely but important to consider, malignancy. The patient elected to undergo re-exploration to relieve his pain and clarify the underlying pathology.
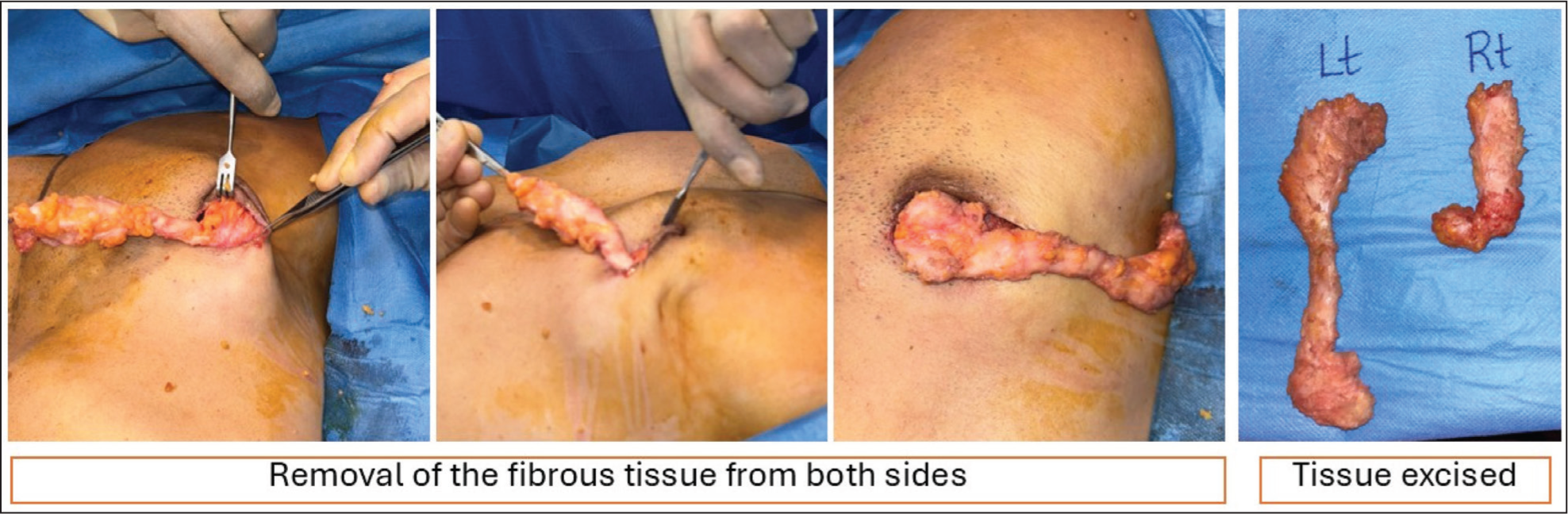
During surgery, the operative team identified thin, ‘banana shaped’ cords of firm fibrous tissue extending laterally from beneath the areolae towards the serratus anterior region (Figures 2 and 3). The tissue was described intraoperatively as rubbery and abnormally adherent to the chest wall. Careful dissection allowed complete excision of these cords while preserving surrounding structures. Haemostasis was secured, and the wounds were closed in layers via periareolar incisions.
Removal of Recurrent Fibrous Tissue Under Local Anaesthetic, Seven Years After the Primary Procedure. Thin, ‘Banana Shaped’ Cords of Firm Fibrous Tissue Were Excised.
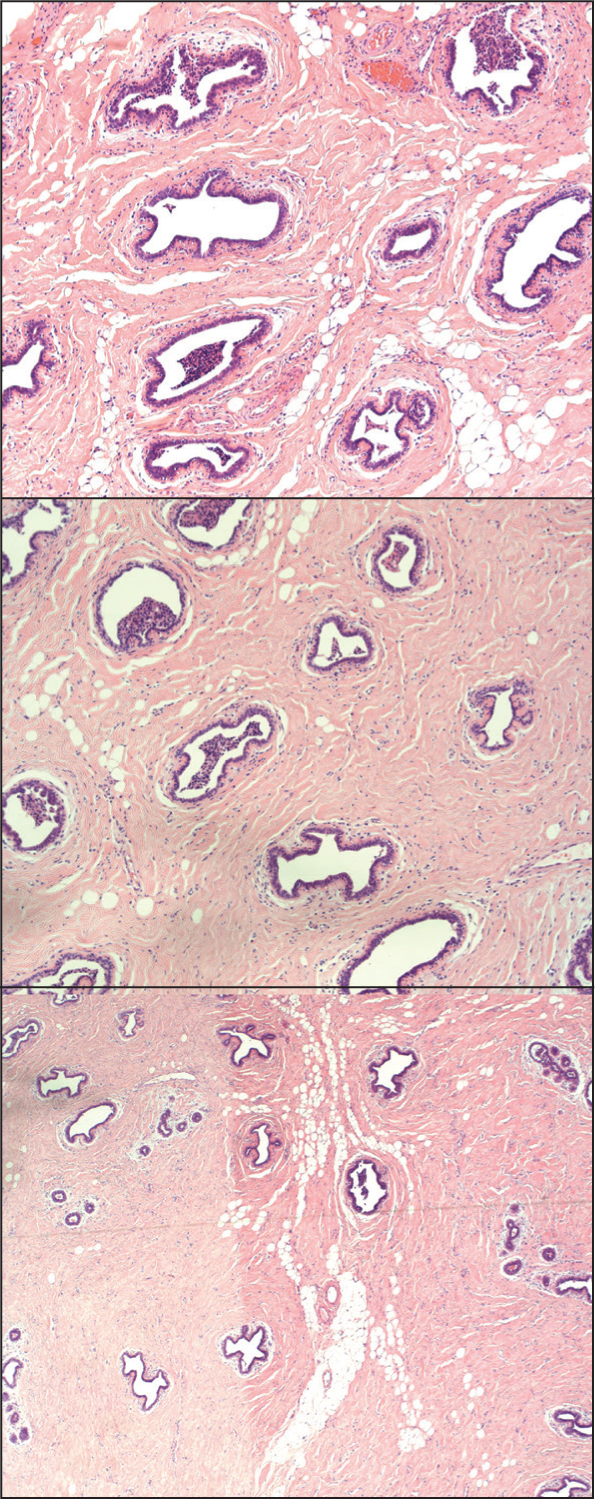
Histopathological examination (Figure 3) of the right breast showed scattered breast ducts with surrounding fibrotic stroma, and the left side also had scattered breast ducts with hyalinised fibrous stroma. No features of malignancy were seen.
Histological Images From This Patient Showing Scattered Breast Ducts with Hyalinised Fibrous Stroma. No Signs of Malignancy.
The patient recovered uneventfully. By three weeks post-operatively, his pain had resolved almost completely, and he had returned to normal daily activities. He was counselled on avoiding AAS or other medications known to trigger gynaecomastia and was scheduled for routine follow-up.
Discussion
This case illustrates a rare example of late, bilateral, fibrous recurrence of gynaecomastia after what had been considered a complete excision. The phenomenon is of clinical interest as it highlights the natural history of gynaecomastia tissue and the pathophysiological role of AAS use.
Recurrence After Surgical Excision
Recurrence of gynaecomastia following glandular excision is generally considered uncommon. A small long-term study by Fricke et al. 4 has reported recurrence rates of 12.5% of glandular gynaecomastia after more than a decade of follow-up. In comparison, they found a recurrence rate of 62.5% in those presenting with gynaecomastia predominantly caused by lipomatous growth. Use of liposuction alone, while effective in contouring the chest wall, may have a higher risk of recurrence because glandular elements are not always adequately removed. Innocenti et al. 5 found no recurrence. However, it must be noted that AAS-induced gynaecomastia is not mentioned in either of these studies.
In the present case, the original excision had achieved good symptomatic relief and cosmetic outcome, suggesting that most of the glandular tissue had been removed. The later development of fibrous cords strongly implies that small foci of microscopic glandular tissue, which over time underwent fibrotic transformation and became symptomatic.
We believe that correction of gynaecomastia is an aesthetic procedure to debulk the soft tissue bulge adequately enough to improve the chest wall appearance. Unlike in tumour excision, the aim is not to completely excise the gland and/or fat as ‘over excision’ causes an aesthetically unacceptable outcome. Overexcision can also compromise the vascularity of the nipple-areola complex (NAC).
Pathophysiology of AAS-induced Gynaecomastia
AAS-induced gynaecomastia arises from the disruption of the normal androgen-oestrogen balance. Many anabolic steroids are subject to aromatisation, producing oestrogens that directly stimulate breast tissue proliferation.6,7 At the same time, exogenous AAS suppresses the hypothalamic pituitary gonadal axis, reducing endogenous testosterone production and further shifting the balance toward oestrogenic activity. 6 The combination of increased oestrogen and decreased androgens creates an environment that favours the proliferation of ductal and stromal elements of breast tissue.
Histologically, gynaecomastia progresses through three overlapping phases. 8 The florid or proliferative phase is characterised by ductal hyperplasia, stromal oedema, and vascular proliferation. Patients often experience tenderness at this stage. Over time, the tissue transitions to an intermediate phase, where proliferation slows, and fibrosis begins. The final fibrous phase is marked by dense stromal fibrosis, hyalinisation, and fewer ductal structures. This phase tends to be less reversible and is often associated with firm, rubbery nodules or cords. 8
In the case described, the patient’s previous history of steroid exposure had led to the initial development of glandular gynaecomastia, which was treated surgically. The microscopic remnants of breast tissue, though clinically insignificant for years, eventually matured into the fibrous phase. This could explain the unusual intraoperative finding of ‘banana shaped’ cords adherent to the chest wall and the patient’s painful symptoms. Further study is required to clarify the mechanism underlying late fibrous recurrence of gynaecomastia, especially in patients with a history of AAS use.
Clinical Implications
The recognition of recurrent fibrous gynaecomastia is important for clinical practice. Men with a history of AAS exposure who present with recurrent chest wall pain years after surgery may be mistakenly reassured that recurrence is impossible. Complete excision of all breast tissue is not feasible, as it carries a significant risk of necrosis of the NAC. Resecting too much tissue can cause adherence of the NAC to the underlying pectoral fascia, producing an unsightly ‘saucer shaped’ deformity, which is cosmetically unacceptable. The surgeon must balance thorough excision with preservation of the NAC. A thin layer of soft tissue is deliberately left beneath the NAC to prevent deformities and avoid vascular compromise. The goal is adequate debulking for an improved chest contour, while minimising the risk of complications.
Clinicians should maintain suspicion for fibrous phase gynaecomastia, particularly when rubbery and adherent tissue is palpable. Imaging or biopsy may be considered when the presentation is atypical and when malignancy cannot be excluded. It could be suggested that medications such as tamoxifen could be used to prevent recurrence, but there is limited data on its use in the context of AAS-induced gynaecomastia. 9
Surgical re-excision remains the mainstay of management in symptomatic and chronic cases. 10 Meticulous dissection is required, especially when fibrous tissue adheres to the chest wall, as was seen in this patient. Outcomes are generally favourable, with significant symptomatic relief and low risk of further recurrence. From a public health perspective, surgeons should actively discourage the use of AAS, as this is not a sustainable means of bodybuilding and carries significant risks; it is well documented that AAS use increases the risk of cardiovascular disease and has negative impacts on mental health. 11
Conclusion
This case describes the unusual late recurrence of fibrous AAS-induced gynaecomastia seven years after prior excision. The patient’s history of AAS use contributed to the initial development of glandular proliferation before residual microscopic tissue underwent fibrotic transformation over time to produce painful, adherent cords. This case emphasises that recurrence, although rare, is possible after correction of gynaecomastia. Clinicians should consider the fibrous phase of gynaecomastia in men with prior steroid exposure who present with recurrent painful chest wall lesions. Surgical re-excision is effective in relieving symptoms and restoring quality of life. It is prudent for surgeons to counsel and consent patients that gynaecomastia surgery is not designed to completely excise the glandular tissue, but rather to adequately debulk the chest wall for aesthetic improvement and hence the procedure cannot be considered ‘recurrence free’.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
