Abstract
Autologous fat grafting (AFG) is a versatile and widely adopted technique in both aesthetic and reconstructive surgery. However, the high cost of specialised patented equipment limits its utility in resource-constrained settings. In this article, we introduce a cost-effective and efficient method for fat harvesting and processing using readily available tools. An Inter-Costal Drainage jar is used for the collection of large-volume fat through a suction apparatus in a closed-loop setup. A household plastic strainer with a nylon mesh is then used for the purification of fat. Our technique has proven to be clinically effective with no adverse outcomes in more than 200 cases and hence, posits a cost-effective solution for AFG whilst maintaining high surgical standards.
Keywords
Plain Language Summary (PLS)
PLS Title: A Simple and Affordable Way to Process Fat for Grafting
Autologous fat grafting (AFG) is a common volume restoration technique used in aesthetic and reconstructive surgery, offering long-lasting results. However, the high costs of specialised equipment preclude its usage in resource-constrained settings. Our study introduces a low-cost, effective method that repurposes everyday medical and household tools to streamline fat harvesting and purification.
Instead of utilising expensive fat collection systems to harvest fat in a sterile, closed-loop setup, we adapted an inter-costal drainage (ICD) jar for the same. An ICD jar is a readily available, sterilised medical-grade container. After fat collection, a simple plastic strainer with a fine nylon mesh is used to efficiently filter fat, removing unwanted fluids and debris. These plastic strainers are commonly used in Indian households for filtering milk.
Without compromising on the quality, sterility, and viability of harvested fat, our AFG setup, consisting of an ICD jar and a simple strainer, costs under $20 as compared to commercial systems priced between $500 and $900. Our technique has proven to be clinically effective with no adverse outcomes in more than 200 cases and hence, posits a cost-effective solution for AFG whilst maintaining high surgical standards.
Introduction
Autologous fat grafting (AFG) is a widely employed technique in both aesthetic and reconstructive surgery, valued for its biocompatibility and lack of immunogenicity. Although reported graft retention rates vary significantly from 20% to 80%, retained fat tends to exhibit greater longevity compared to synthetic fillers. 1 However, ensuring predictable graft survival remains a challenge, as outcomes are influenced by the techniques used for harvesting, processing, and injection.
Despite its numerous advantages, the high cost of equipment for fat collection and processing poses a considerable barrier, particularly in settings with limited access to specialised consumables. This article presents a cost-effective and efficient method for fat collection and processing using readily available tools, integrating insights from existing literature with our clinical expertise to deliver consistent and reproducible results.
Innovations
Adapting an ICD Jar for Fat Harvesting
ICD jars are commonly utilised in the management of pneumothorax and pleural effusions. In a typical setup, one end of the jar is connected to a chest tube and placed under a water seal, while the other end serves as an air outlet.
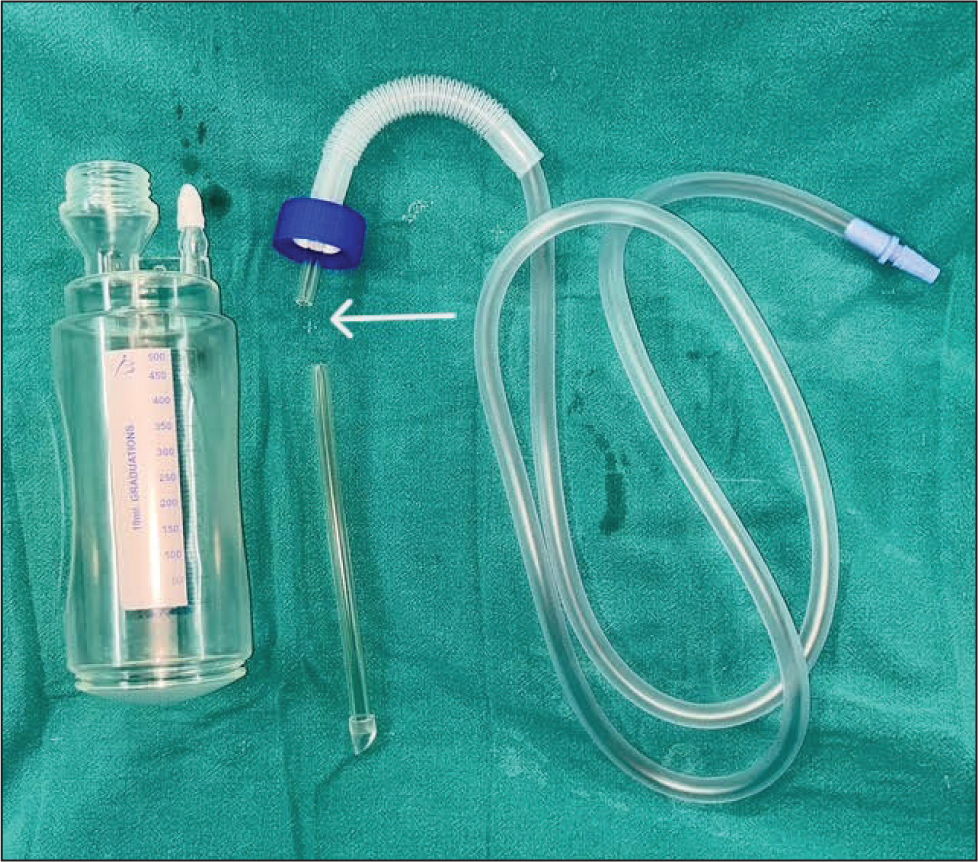
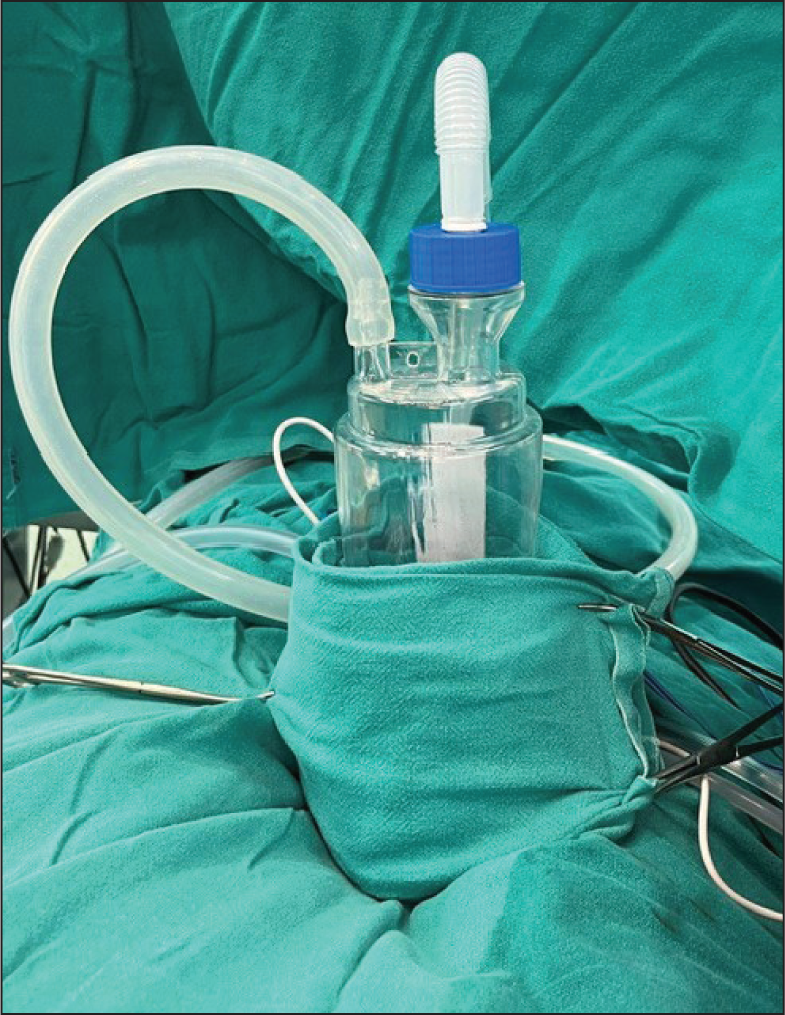
Recognising its potential beyond its conventional use, we used a Romoseal→ 1200 ICD jar (Romsons Group, New Delhi) as an efficient fat collection system (Figure 1). One end of the jar is connected to a fat aspiration cannula, while the other end is attached to a negative pressure machine, creating a makeshift sterile closed-loop setup for fat harvesting. Since a water seal is not required for this application, the internal tubing is removed to optimise fat collection (Figure 1). To ensure stability, the jar is secured using towels and towel clips (Figure 2).
The ICD Jar for Fat Collection. During Graft Harvesting, the Wide End Is Attached to a Suction Cannula and the Narrow End Is Connected to a Negative Pressure Machine to Create a Functional, Makeshift Closed-loop System. Note That the Long End of the Underwater Seal Tube Has Been Cut Short (White Arrow) to Facilitate the Collection of the Fat Graft and Prevent Fat Aspiration.
Arrangement of the ICD Collection Jar on the Operation Table. The Jar Is Kept Stable by Wrapping It With a Towel and Fixing It to the Table by Using Towel Clips.
A disposable sterile ICD jar at a cost of less than $20 is not only cheap but also easily available, and being made of medical-grade plastic makes it inert. Connecting the jar to a negative pressure machine forms a closed loop, which minimises the aerial exposure of adipocytes, thus improving their viability on AFG. 2 A negative suction pressure of 250 and 300 mmHg is maintained throughout to ensure minimal trauma to adipocytes and prevent damage to the ICD jar due to excessive negative pressure.
Repurposing a Common Household Strainer for Fat Purification
We repurposed a plastic strainer, commonly used in Indian households for straining milk, for fat purification (Figure 3). With its fine nylon mesh (pore size = 500 µm), it efficiently filters the aspirate. Priced at less than $1, the strainer is economical enough for single use but can also be sterilised with ethylene oxide for reuse. This makes it a practical and cost-effective alternative to commercial filtration devices.
a) Plastic Strainer With a Nylon Sieve Commonly Used in Indian Households for Straining Milk, Repurposed for Fat Purification. b) A Close-up Image Demonstrating the Strainer’s Pore Size of 500 Microns.

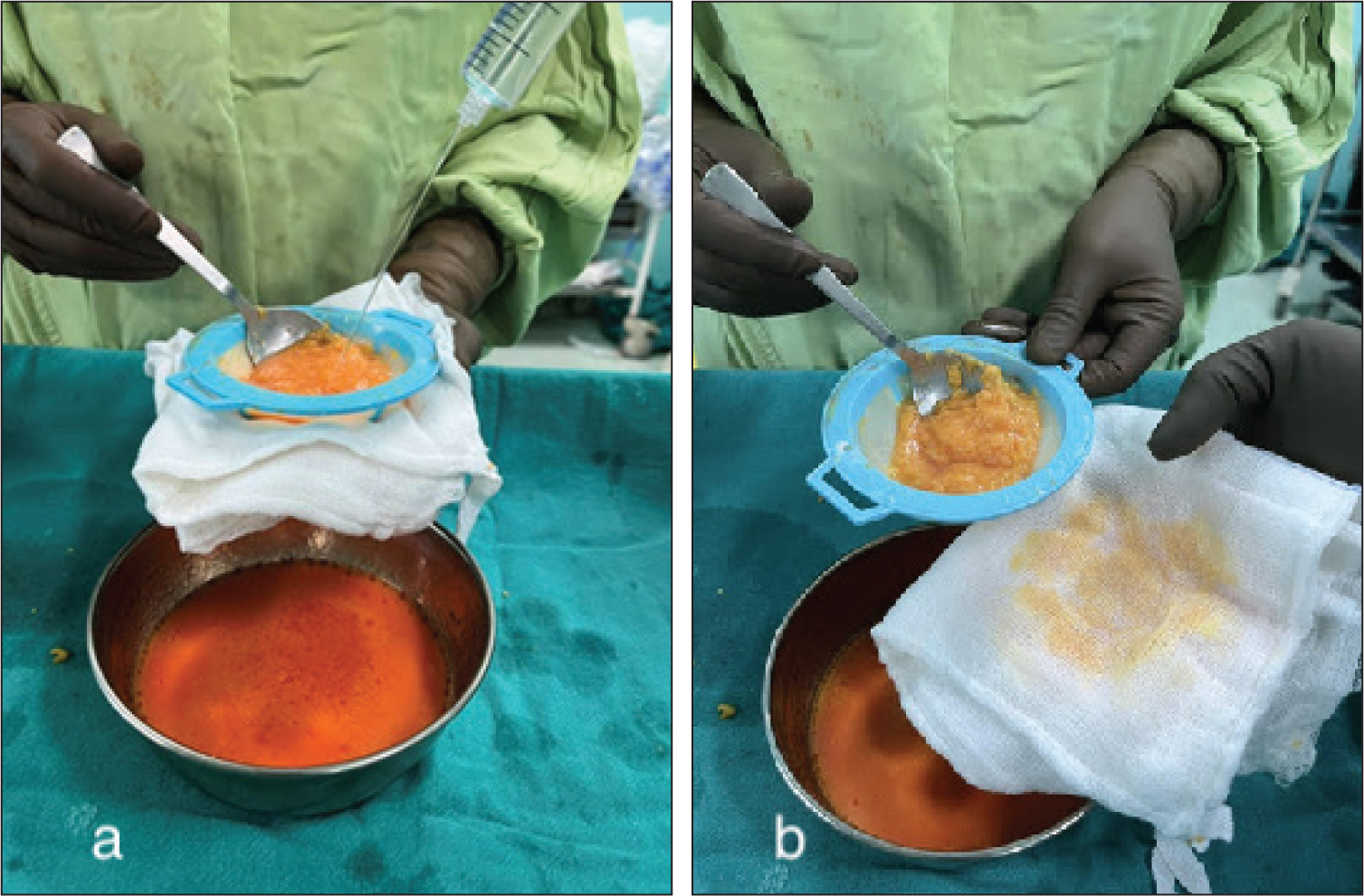
Using a no-touch technique, the aspirate is first transferred from the ICD jar to the strainer in small quantities (around 40–50 mL at a time). Second, it is washed with an equal quantity of sterile saline poured into the strainer whilst carrying out manual gentle churning of the fat with a sterile stainless steel spoon. Third, a sterile gauze mop is applied to the bottom of the strainer to absorb excess oil and fluid via capillary action (Figure 4). This effectively removes blood, residual tumescent solution, oils, and cellular debris, leaving behind pure and viable fat.
a) Aspirated Fat Is Washed with Sterile Saline and Strained. b) A Sterile Gauze Mop Is Kept in Contact with the Bottom of the Strainer to Effectively Absorb Excess Fluid and Oil Passing Through It via Capillary Action.
Finally, this purified fat is fractionated and transferred to smaller 1–2 cc syringes for injection using standard techniques for the required indication. This entire process of straining, washing, and drying is easily completed within 5 minutes, thus minimising the aerial exposure of adipocytes and reducing their damage.
Discussion
Numerous fat harvesting and processing techniques have been described in the literature; however, no single method has been consistently proven to achieve superior graft retention. 3 Much of the existing research has been conducted in vitro. The lack of standardisation across various processing methods makes direct comparisons challenging and hinders the establishment of a consensus on an ‘optimal technique’. Furthermore, variability in lipo-injection techniques amongst practitioners adds another layer of complexity, as these differences can influence graft retention even when extraction methods are consistent.1,4
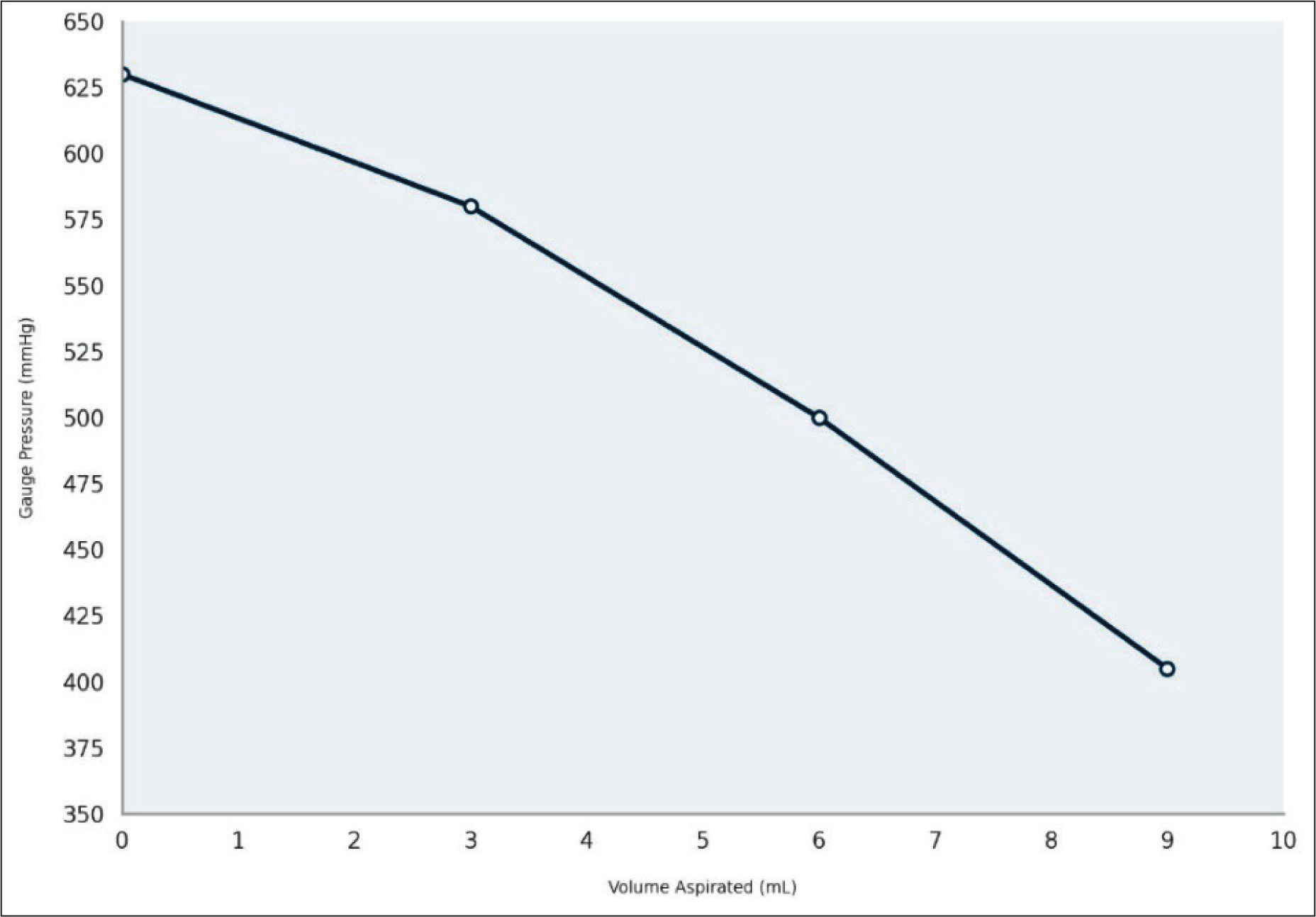
Use of syringe-based manual aspiration for fat harvesting is based on a common belief that it is associated with less adipocyte trauma, reduces fat contamination with fascia and tissue debris, and perhaps ensures better fat viability. Although we broadly agree with these advantages, we believe that syringe-based fat suction has a low efficiency and is time-consuming. We have substantiated this belief with a simple simulation experiment in which we manually measured negative pressures with a connected barometer at various stages of syringe plunger retraction with increasing volumes of coloured fluid. As more and more liquid accumulates in the syringe (equivalent to fat), we observed a significant drop in negative suction pressure (Figures 5 and 6). This observation clearly indicates that the efficiency of syringe-based fat suction decreases as the suction progresses manually. Our results are also consistent with findings from other researchers,5–8 who demonstrated that syringe-generated negative pressure is determined by the plunger’s position rather than the syringe’s size.
Measurement of Negative Pressure with a Connected Barometer, Generated by Manually Pulling the Syringe Plunger at Varying Fluid Volumes. As More and More Fluid Accumulates in the Syringe Barrel, It Causes a Drop in the Generated Negative Pressure at the Same Intensity of Manual Suction, Demonstrating an Inverse Relationship Between the Aspirate Volume and Negative Suction Pressure.

Graph Illustrating the Relationship Between Negative Gauge Pressure (in mmHg) and Volume Aspirated in the Syringe (in mL). The Results Demonstrate a Progressive Decline in Negative Pressure as More and More Fluid Accumulates in the Syringe.
Therefore, we favour using a cannula connected to a suction machine, which ensures consistent and controlled low negative pressure throughout the aspiration process. Recent studies have found no significant difference between high (700 mmHg) and low (200 mmHg) negative pressures when it comes to the issue of adipocyte viability.9-11 Despite this, we favour using lower negative pressures (250-300 mmHg), as we have anecdotally observed a reduction in fascia, fibrous debris, and tissue contamination in the aspirate, which may enhance graft purity. We also believe that lower negative pressures cause less bleeding and fascia disruption, thus minimising donor-site morbidity.
For fat collection, we utilised an ICD jar, which functions as a single-use, sterile, closed system and serves as a highly efficient and economical alternative to expensive commercial systems such as Revolve™ (LifeCell Corp., Bridgewater, New Jersey) and Puregraft™ (Cytori Therapeutics, Inc., San Diego, California).12,13 These collection systems cost between $500-$900. Our setup provides all the advantages of these commercial systems at only a fraction of the cost.
Although centrifugation has long been regarded as the gold standard, several studies have demonstrated that filtration and washing are equally effective.3,4,14,15 Metal strainers may cause increased inflammation and greater oil retention, both of which can adversely affect graft survival. 16 Usage of nylon as a suture or mesh in the form of a biocompatible material does not need to be proven, as it is already an established surgical practice.
Our technique has certain limitations as sieve-based filtration methods may result in some loss of Stromal Vascular Fraction (SVF), which is rich in regenerative cells and plays a critical role in graft survival and tissue integration. 17
Clinically, we have achieved excellent fat retention rates based on subjective clinical evaluation, with no reported cases of fat cysts or necrosis in 208 cases over the past 8 years. No study was undertaken to objectively demonstrate actual fat retention, as this was not the subject of this study. The aim was to simplify an established practice of fat harvesting, collection, and filtration and ensure frugality, safety, and effectiveness.
We hope that our innovative technique for fat harvesting using an ICD jar and fat filtration using a sterilised household strainer will find global acceptance by the surgical fraternity due to its ease and safety.
Footnotes
Author Contribution
Original Idea and Editing of manuscript: Sunil Choudhary. Review of literature and writing of manuscript: Pavan Venkateswar. Writing of manuscript: Soumya Khanna, Madhur Khullar, Raghav Mantri, Prateek Arora.
Consent to Participate
Not applicable.
Data Availability Statement
This study did not generate or analyse any datasets.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The developed technique is an established protocol for which the authors are using frugal, sterile biocompatible substitutes in order to make it cost effective. The authors’ institute does not require ethical approval for reporting individual cases or case series.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
