Abstract
Background
Although uncommon, the deep venous thrombosis (DVT) and pulmonary embolism (PE) are serious complications of spine surgery that worsen patient outcomes. Prophylactic measures to prevent the formation of DVT have been explored in spine surgery but given the multitude of potential prophylactic methods and agents, a comprehensive review and understanding of prophylactic agents and their indications is essential.
Objective
Conduct a review of the major DVT prophylactic agents used in spine surgery to provide a cohesive review on the indications, benefits, and drawbacks of commonly used DVT prophylactic agents.
Methods
A literature review on DVT prophylaxis in spine surgery was conducted, with high strength, Levels I and II, and evidence being prioritized, where patient outcomes and complications from prophylactic measures were quantified.
Results
The rates of DVT and PE from spine surgery can vary based on procedure with fusion surgeries, especially multi-level fusion surgeries, anterior approach surgeries, and trauma spine surgeries having greater rates of DVT. Mechanical prophylaxis was found to have reasonable efficacy with minimal drawbacks but may potentially not be strong enough for DVT prophylaxis in higher risk patients. Chemoprophylaxis when indicated is best done with low molecular weight heparin (LMWH) or direct factor X inhibitors (DFXIs). Determination of need for chemoprophylaxis should be done on a patient-by-patient basis. When indicated, DVT chemoprophylaxis should be initiated in the first 48 hours postoperation to maximize effectiveness and safety.
Conclusion
DVT prophylaxis is critical for spine surgery patients to improve patient outcomes. Determining what style of DVT prophylaxis will be most effective for a patient can ensure that patients are able to recover from surgery quicker and safer.
Keywords
Venous Thromboembolism in Spine Surgery: Overview of Incidence, Risk Factors, Pathophysiology, and Clinical Signs
DVT Incidence and Risk Factors in Spine Surgery
The incidence of deep vein thrombosis (DVT) and pulmonary embolism (PE) has increased in recent years secondary to the complexity of spine surgery. Venous thromboembolism (VTE), manifesting as DVT or PE, is considered one of the most frequent complications following a surgical spinal intervention. 1 The rates of DVT and PE following spinal surgery varies based on the procedure type, setting, and approach, with overall measured incidences of PE ranging from 0.06% to 8% and DVT from 0.35% to 15.5%.2-8 Some of the variability in estimates can be explained by study design, as prospective studies are more likely to detect asymptomatic VTE. 2 Regardless of study design, the presentation and risk of DVT and PE vary widely, making it essential to distinguish clinically significant VTE events.
Current literature suggests that thromboembolic risk differs based on the indication for spine surgery—whether elective, trauma-related, or oncologic. A recent meta-analysis found that elective surgeries had an overall incidence of DVT and PE at 3.3% and 0.4%, respectively. 9 Although trauma-related spine surgery is typically considered higher risk, existing data report relatively low rates of VTE, with DVT occurring in 1.7% of cases and PE in 0.9%. 7 However, traumatic injury itself is an independent risk factor for DVT, necessitating increased vigilance, specifically in patients with spinal cord injuries, who have an overall VTE rate of 9.6%. 10 In addition, malignancy is a significant independent risk factor, associated with a 7-fold increased risk of venous thromboembolism. 11 Thus, postoperative DVT and PE rates in patients undergoing surgery for spinal metastases have been reported to be 6.2% and 6%, respectively, with a 1.3% mortality rate secondary to PE. 12 Eskildsen et al. found advanced age (greater than 60), obesity, smoking, congestive heart failure, estrogen-containing contraceptives, pulmonary circulation disorders, family history of VTE, poor postoperative mobilization, and an American Society of Anesthesiologists Classification greater than 3 to be moderate risk factors for VTE in spine surgery. 13
Pathophysiology and Clinical Signs of Venous Thromboembolism Specific to Spine Surgery
The exact mechanisms underlying deep vein thrombosis (DVT) formation are not fully understood; however, they are generally explained through the framework of Virchow’s triad (stasis of blood flow, endothelial dysfunction, and hypercoagulability), as described in the 19th century. It is believed that a lack of shear stress on vein walls from stasis of blood flow may promote the expression of proinflammatory and prothrombotic genes. Subsequent dysfunction of the endothelium results in the loss of the anticoagulatory and fibrinolytic factors, and a promotion of the prothrombotic phenotype. Endothelial dysfunction is more common in the elderly, and it is believed that this may play a role in the observed increased frequency of DVT in the geriatric population. DVTs are composed of large amounts of fibrin created from the overactivation of both the intrinsic and extrinsic coagulation pathways. Factor XIII has been specifically studied for its role in thrombogenesis, with elevated levels associated with increased clot formation. Additionally, platelets are thought to stabilize thrombi and enhance immune cell recruitment, further contributing to the development of the initial fibrin clot. 14
Several factors have been linked to increased risk of postoperative DVT specifically in spine surgical patients. In the perioperative setting, stasis is typically a result of immobilization, either through prolonged operative time or increased bed rest following surgery. 15 Operative times longer than 261 minutes have been linked to increased rates of postoperative DVTs. 16 Although measures to prevent venous stasis, such as sequential compression devices, have been developed, postoperative immobilization of the lower limbs continues to be a significant contributing factor of the development of DVT after surgery. 17
Clinically, DVT is often asymptomatic, making diagnosis based on physical findings alone challenging. When symptoms are present, they may include unilateral or bilateral leg swelling, fatigue, visible superficial veins, and localized pain, tenderness, warmth, or discoloration—typically near the site of the thrombus. 18 Homans Sign is a classic clinical sign of DVT, where a patient experiences pain in the calf during passive dorsiflexion of the ankle. However, the accuracy of Homans Sign has been long disputed with studies finding accuracies from 8% to 56%, and a false positive rate greater than 50%. 19 If the DVT progresses to pulmonary embolism (PE), cardiopulmonary symptoms such as dyspnea, angina, and syncope typically ensue. Massive PEs typically cause systemic hypotension and right-sided heart strain and heart failure when not treated in a timely manner. Smaller PEs may present with cough and hemoptysis. 18
Due to the high occurrence of asymptomatic DVT, and disputed accuracy in established clinical signs, symptoms alone are not reliable predictors of DVT. 20 Clinical examination has shown inconsistent accuracy, with sensitivity ranging from 60% to 96% and specificity ranging from 20% to 72%. 20 Diagnosing recurrent DVT is particularly challenging, as approximately 70–80% of symptomatic patients have symptoms that are more indicative of chronic venous insufficiency or other causes of lower extremity pain. 21
Diagnosis and Monitoring
The Wells criteria is a widely used clinical screening tool for DVT, incorporating risk factors such as active cancer, immobility, and prior DVT to generate a score ranging from 0 to 9. A score below 2 suggests a low probability of DVT, while a score of 2 or higher indicates the need for further evaluation. In a retrospective study, the Wells score demonstrated a sensitivity of 100% and a specificity of 90%.22,23
Diagnostic options include D-dimer testing and extremity venous Doppler ultrasound. While D-dimer is highly sensitive, its low specificity limits reliability in postoperative settings. A negative Wells score and D-dimer may obviate the need for ultrasound where clinical suspicion remains high. Doppler ultrasound is the diagnostic standard for detecting DVT. If clinical suspicion remains high despite a negative Doppler ultrasound, a repeat scan is recommended within 5–7 days.
24
Figure 1 illustrates the Wells criteria and recommended diagnostic workflow. Wells score criteria and flow for usage of Wells score in diagnosis of DVT.
The Wells criteria for PE and Geneva Score both assess PE risk, focusing on factors such as prior DVT, immobilization, current hemoptysis, and active malignancy. 25 The Geneva Score incorporates additional factors, including recent surgery and unilateral lower limb pain or edema. However, Shen et al. found that the Wells score demonstrated higher sensitivity and accuracy when compared to the Geneva score, suggesting it may be a better tool in assessing PE risk and determining the need for further PE workup. 26 If a patient’s Wells or Geneva score indicates a need for further assessment, the diagnostic approach mirrors that for DVT, including D-dimer test, extremity ultrasound and urgent computer tomography (CT) of the chest with angiography.
Implications of DVT on Outcomes in Spine Surgery
Morbidity and Mortality in Spine Surgery Patients
VTE, manifesting as DVT or PE, has been reported to be the third leading cause of hospital-associated death in the United States. 27 While DVT alone poses minimal risk to patient mortality, the progression of DVT to a PE can be life-threatening in surgical patients. In cervical spine surgery patients, the presence of a PE has been associated with a 15- to 48-fold increase in mortality rates, while the presence of a DVT has been linked to a 10- to 30-fold increase. 28 Similar trends have been found in patients undergoing lumbar spine injury, where postoperative DVT and PE have been associated with 12- to 17-fold and 38- to 150-fold increase in mortality, respectively. 29 Beyond mortality risks, the diagnosis of DVT and PE is associated with a significant increase in hospital costs ranging from $30,000 to $50,000. 28 Given that the U.S Department of Health and Human Services identifies cost as a leading barrier to medical care, these financial burdens may subsequently have a negative impact on patient adherence to treatment and recovery. 30
Spine surgery patients diagnosed with either a DVT or PE additionally experience longer hospital stays, extending from an average of 2–7 days to 14–26 days. 28 Approximately 4% of patients diagnosed with a postoperative VTE will be readmitted to the hospital following a secondary VTE event, with over half occurring within 30 days of discharge. 31
Influence on Functional Recovery
The presence of DVT or PE has been associated with delays in postoperative mobilization, subsequently prolonging rehabilitation time. 32 DVT events have been linked to complications such as post-thrombotic syndrome, characterized by chronic pain and swelling, which limits mobility and often leads to an extended recovery time. 33 Early mobilization is essential for reducing complications and hospital stays following spine surgery, highlighting the negative impact of VTE diagnosis in patient’s postoperative rehabilitation. 34 Pulmonary embolism (PE) has also been associated with lasting psychological effects, including post-traumatic stress disorder (PTSD), which can further hinder postoperative recovery. 35
Current Management Recommendations for DVT Prophylaxis in Spine Surgery
Mechanical Prophylaxis
Mechanical prophylaxis methods such as Intermittent Pneumatic Compression (IPC) and Sequential Compression Devices (SCDs) are non-invasive devices that utilize mechanical force to improve blood flow in the lower extremities. Takahashi et al. performed a retrospective cohort study investigating the effectiveness of the mechanical prophylactic measures of foot pumps and elastic stockings in a population of spine surgery patients who had undergone fusion or decompression surgery. They found that the rate of symptomatic PE rates in the non-prophylaxis cohort (n = 541) to be 1.5% compared to 0.2% in the prophylaxis cohort (n = 1434). 5 The most recent North American Spine Society (NASS) guidelines recommend the use of mechanical compression devices during or immediately after spine surgery, continuing until the patient is fully ambulatory, to reduce the risk of venous thromboembolism. 36
While mechanical prophylaxis has a low complication risk profile, patient adherence to mechanical prophylaxis regimens has remained problematic. Cragie et al. performed a meta-analysis of adherence to mechanical prophylaxis, noting adherence rates of around 75% and decreasing to 20% by postoperative day 7. Patients often demonstrated non-adherence due to discomfort and sleep disturbances caused by mechanical devices, suggesting that relying solely on mechanical prophylaxis may be ineffective in reducing VTE risk due to poor patient compliance. 37
Dearborn et al. conducted a retrospective cohort study of 318 spine surgeries where all patients were provided with mechanical prophylaxis (compression stockings and pneumatic compression leggings). They found 7 symptomatic PEs (2.2%) with 6/7 occurring after combined anterior/posterior spinal fusions (6% of studied combined anterior/posterior spinal fusions), while only one occurred after posterior decompression and fusion (0.5%). They concluded that mechanical prophylaxis alone may not confer enough VTE protection for patients undergoing more involved spine procedures, particularly those requiring multiple approaches. 38 Mosenthal et al. conducted a systematic review and meta-analysis of 28 articles comparing mechanical prophylaxis and chemoprophylaxis, noting a nonsignificantly higher rate of DVT and PE in mechanical prophylaxis patients (DVT: 1%, PE: 0.81%) compared to chemoprophylaxis patients (DVT: 0.85%, PE: 0.58%). They additionally noted a greater PE mortality rate in spine surgery patients when compared to other surgical patients, and suggested that chemoprophylaxis may be indicated for high-risk patients in conjunction with mechanical prophylaxis. 39
Chemoprophylaxis
Several pharmacologic agents have been employed for postoperative VTE prophylaxis in spine surgery, including low molecular weight heparin (LMWH), antiplatelet agents, fondaparinux, Factor X inhibitors (apixaban and rivaroxaban), and unfractionated heparin. Surgeons may hesitate to use chemoprophylaxis due to concerns about bleeding complications, particularly the development of epidural hematomas, which can result in severe neurological outcomes. Glotzbecker et al. conducted a systematic review of postoperative spinal hematomas, analyzing 16 studies and finding that the incidence of epidural hematoma with chemoprophylaxis ranged from 0% to 0.7%, suggesting that the actual risk may be lower than commonly perceived. 40 Anaspure et al. conducted a meta-analysis of 14 studies involving 13 754 spinal trauma surgery patients and found that chemoprophylaxis was not significantly associated with an increase in postoperative bleeding compared to controls (RR = 1, P = 0.549). However, they also found no significant difference in VTE prevalence between groups (RR for VTE in the non-chemoprophylaxis group = 1.03, P = 0.119), highlighting the ongoing challenge in determining the necessity of chemoprophylaxis. 41 Furthermore, Rahmani et al. performed a meta-analysis of 220 932 elective spine surgeries and found no statistically significant difference in VTE rates between patients receiving VTE chemoprophylaxis and those who did not receive them, 9 demonstrating the conflicting information on the role of prophylactic measures for spine surgery patients. These findings suggest that chemoprophylaxis may play a role in the management of spine surgery patients and may possess a more favorable safety profile than previously assumed. Despite their widespread use, no global or national consensus currently exists regarding the optimal agent or regimen, leaving most decisions to individual surgeon preference.
Low Molecular Weight Heparin (LMWH)
Low molecular weight heparin (LMWH, -parin suffix) has become a prolific agent for DVT prophylaxis, frequently used in both elective and trauma settings. LMWH agents work by binding to and propagating antithrombin III, a protein that inactivates several enzymes involved in the clotting cascade, mainly factor Xa. Shapiro et al. conducted one of the largest prospective studies (288 patients) evaluating the effectiveness of LMWH (n = 55) compared to mechanical prophylaxis (n = 211) in degenerative cervical and lumbar spine surgery patients identified as high risk for VTE. Their study demonstrated that although patients in the LMWH group had significantly greater rates of delayed wound healing and deep infections (9%) compared to the mechanical prophylaxis group (0.47%), the incidence of epidural hematoma and VTE remained low and comparable. Zeng and Peng conducted a larger retrospective study involving 947 spine surgery patients who received postoperative therapeutic LMWH and found a significantly lower VTE rate (0.21%) compared to the control group, which received no chemoprophylaxis (1.6% among 814 patients). 42 However, the potent antithrombotic effect of LMWH was also reflected in a marginally nonsignificant increase in bleeding events in the LMWH group (1.8%) vs the control group (0.74%). Among the studies conducted to date, the literature largely supports LMWH as an effective agent for VTE prophylaxis in spine surgery and spinal cord injury, demonstrating significant reductions in VTE rates with an acceptable safety profile—though careful consideration of bleeding and wound-related complications remains essential.43,44
Unfractionated Heparin
Unfractionated heparin (UFH) has been in use as a potent antithrombotic agent for almost 100 years. Like LMWH, UFH binds to antithrombin III to inactivate enzymes in the clotting cascade, primarily targeting factor Xa. However, UFH also effectively inhibits factor IIa (thrombin), whereas LMWH predominantly inactivates factor Xa with only moderate inhibition of factor IIa, resulting in UFH exerting broader anticoagulant effects. Macki et al. conducted a matched cohort study of 562 patients comparing LMWH to UFH in spine surgery. Despite finding equivocal incidence of VTE events between both treatment strategies, they observed a trend suggesting that LMWH may be associated with fewer complications than UFH when used for DVT prophylaxis (odds ratio for complications with UFH vs LMWH = 3.5 [0.48-20.35], P = 0.227). 45 A prospective study of 119 spinal cord injury patients conducted by a multicenter group of investigators found that LMWH significantly reduced VTE events during the rehabilitation phase compared to UFH (relative risk reduction = 67%, P = 0.052). In contrast to the matched cohort study, both agents were found to have low rates of complications, including hematomas, during the rehabilitation period. 46
McLynn et al. performed a large retrospective cohort study of 2855 patients undergoing elective cervical, thoracic and lumbar spine surgery. They found that while UFH was not associated with a significant reduction in VTE events (relative risk of VTE with UFH being 0.68, P = 0.424) when compared to mechanical prophylaxis alone, it was linked to a significant increase in postoperative hematomas requiring reoperation (RR = 7.8, P = 0.02). 47 Shiu et al. conducted a large retrospective review over a 13-year period, examining 62 patients who developed VTE following spinal trauma surgery, alongside 174 propensity-matched controls. They found that patients who received UFH had a significantly higher reoperation rate compared to those who received LMWH (31% vs 6.5%, P = 0.02). Furthermore, compared to the control group, UFH was associated with a markedly increased odds of bleeding complications (OR = 43.1, P = 0.01). Among the cohort studies conducted to date, UFH appears to have a weaker safety profile for VTE prophylaxis compared to alternative agents, particularly LMWH. Additionally, the literature suggests that UFH offers equivalent or potentially lower efficacy than LMWH in preventing VTE events. These findings imply that UFH may not be the optimal choice for VTE chemoprophylaxis in spine surgery patients. However, given the limited availability of high-quality evidence specifically evaluating UFH in this context, further research is warranted.
Fondaparinux
In addition to LMWH and unfractionated heparin, fondaparinux has emerged as another chemoprophylactic agent of interest in spine surgery. Fondaparinux works similarly to UFH and LMWH, binding to antithrombin III to exert its antithrombotic effects. However, fondaparinux exclusively binds to antithrombin III, leading to inhibition of factor Xa, with no effect on factor IIa, unlike UFH and LMWH. Fourman et al. conducted a retrospective cohort study evaluating the effectiveness of fondaparinux for VTE prevention in patients undergoing elective lumbar spine surgery. 48 All patients in the treatment group also received 325 mg of aspirin daily for one month after discharge. The study demonstrated a significant reduction in DVT events with fondaparinux use (0% in fondaparinux group compared to 2.9% in control, P = 0.02), and a nonsignificant lower rate of increased wound drainage (18.5% in fondaparinux group compared to 25.5% in control, P = 0.18). Notably, there were no differences in deep infection rates or transfusion requirements between the fondaparinux and control groups, and no postoperative epidural hematomas were observed. The findings of Fourman et al. suggest that fondaparinux may offer a favorable safety profile and effective VTE reduction. However, the current literature on its use for VTE chemoprophylaxis in spine surgery is limited, highlighting the need for further studies—particularly those directly comparing its efficacy to other chemoprophylactic agents.
Antiplatelet Agents (Aspirin and Clopidogrel)
Antiplatelet agents, such as aspirin (ASA) and clopidogrel, offer an alternative approach to anticoagulation by irreversibly inhibiting platelet aggregation, thereby preventing clot formation. Traditionally, their use in orthopedic surgery has been limited due to concerns about their irreversible effects, which complicate the management of bleeding by making reversal more difficult. Fiasconaro et al. performed a retrospective cohort study looking at different chemoprophylactic agents in 83 839 patients who underwent anterior cervical discectomy and fusion and/or posterior lumbar fusion. They found that aspirin (ASA) was associated with higher odds of requiring blood transfusion compared to LMWH (OR = 1.48 [1.17-1.86]), albeit with comparable VTE rates to LMWH. 49
However, in many patients, ASA and clopidogrel may be initiated preoperatively for cardiovascular indications, raising the important question of whether these agents should be discontinued before spine surgery. Zhang et al. performed a meta-analysis of 4 studies assessing the safety of continuing aspirin therapy (especially bleeding risk) in spine surgery patients. They found no increase in risk of blood loss during surgery (95% CI [–111.72 to −0.59] P = 0.05) or rates of postoperative blood transfusion ([0.00–0.27), P = 0.05). 50 A systematic review and meta-analysis by Goes et al. reported similar findings, showing no significant difference in perioperative blood loss between patients who continued ASA and those who discontinued it prior to surgery (553.9 mL [468–639.9] vs 538.7 mL [427.6–649.8], respectively; P = 0.96). 51 Taken together, these findings suggest that while antiplatelet agents like ASA may have a less favorable safety profile compared to LMWH when used as prophylactic agents, their continuation in patients already taking them for other indications—such as cardiovascular disease—does not appear to significantly impact perioperative blood loss.
Factor X Inhibitors (Apixaban and Rivaroxaban)
Factor Xa inhibitors have recently gained attention as chemoprophylactic agents in spine surgery. Although their mechanism resembles that of fondaparinux, a key distinction lies in their mode of action: fondaparinux inhibits factor Xa indirectly by enhancing antithrombin III activity, whereas direct factor Xa inhibitors—such as apixaban and rivaroxaban—bind directly to factor Xa, effectively interrupting the coagulation cascade. Bagheri et al. conducted a prospective cohort study of 130 patients receiving either LMWH (n = 65) or apixaban (n = 65) after posterior decompressive spine surgery. They found no statistically significant differences between the 2 agents in VTE incidence, hematoma formation or complication rate. 52
Shafiei et al. conducted a randomized clinical trial involving 244 spine surgery patients, assigning them to receive either rivaroxaban (n = 123) or LMWH (n = 121). They found that rivaroxaban was associated with a significantly higher postoperative drain volume (P = 0.02), but there were no other significant differences between the 2 groups in terms of complications or surgical outcomes. 53 Du et al. conducted a randomized controlled trial involving 665 patients undergoing lumbar surgery, comparing rivaroxaban (n = 341) to LMWH (n = 324). Consistent with the findings of Shafiei et al., they reported no significant differences in the incidence of VTE or bleeding events between the 2 groups. 54 Based on the available evidence, these findings suggest that direct factor Xa inhibitors, such as apixaban and rivaroxaban, demonstrate comparable efficacy and safety profiles to LMWH in the prevention of VTE in spine surgery patients.
Timing of Chemoprophylaxis
The primary concern regarding the use of chemoprophylaxis in spine surgery is rooted in bleeding complications, particularly spinal epidural hematomas. In addition to selecting the appropriate chemoprophylactic agent, a growing body of evidence suggests that the timing of administration may play an important role. Strom et al. conducted a retrospective single-institution study of 367 patients undergoing laminectomy with or without fusion, noting that delaying LMWH initiation by 24-36 hours after surgery did not cause epidural hematoma in any of the patients. Interestingly, acute VTE was still diagnosed in 14 patients (3.8%) who exhibited independent risk factors of age >60 years, smoking, and obesity. 55 In contrast, Lui et al. noted that early (within 24 hours of spinal cord injury) use of LMWH was not associated with increased risk of hemorrhagic complications in a prospective observational study of 162 patients. 56
In one of the largest retrospective studies to date, Dhillon et al. analyzed 6869 consecutive spine surgeries and reported an average chemoprophylaxis initiation time of 1.46 days postoperatively. Among patients who received chemoprophylaxis (n = 1904) vs those who did not (n = 4965), time-to-VTE was significantly shorter in the non-chemoprophylaxis group (3.6 vs 6.8 days). Importantly, the incidence of postoperative epidural hematoma was low and comparable between groups (0.18% in the control group vs 0.21% in the chemoprophylaxis group). They found that chemoprophylaxis was safe to start in spine surgery patients from one day before surgery, to 3 days postoperation without an increase in epidural hematoma risk.
Kim et al. conducted a large retrospective cohort study involving 1432 trauma patients who underwent surgical fixation for spine fractures. Patients were stratified based on the timing of chemoprophylaxis initiation—either within 48 hours postoperatively (n = 48) or after 48 hours (n = 158). There were no reported cases of bleeding; however, the incidence of VTE was lower in the early prophylaxis group (2.08%) compared to the late group (7.59%), suggesting a potential benefit of earlier initiation. 57 Cunningham et al. conducted a retrospective cohort analysis of 3870 patients undergoing elective spinal procedures, concluding that preoperative DVT chemoprophylaxis did not affect the rates of VTE manifestation when compared to a group without preoperative chemoprophylaxis (OR 0.91 compared with none) and moderately increased rates of spinal epidural hematomas (OR 1.33 compared to control). 58 Cunningham’s study, however, was limited by a minimal VTE patient population (n = 19) and epidural hematoma population (n = 16). These findings suggest that initiating chemoprophylaxis within 2 days of spine surgery may be safe and does not appear to increase the risk of epidural hematoma, but preoperative chemoprophylaxis or chemoprophylaxis initiated after 2 days postoperatively may be less effective and safe.
Determination of Need for DVT Chemoprophylaxis in Spine Surgery
In 2015, Eskildsen et al. introduced an evidence-based algorithm to assess the need for VTE chemoprophylaxis in spine surgery patients. The algorithm evaluates risk across 3 primary domains: patient-specific factors, surgical procedure characteristics, and neurological bleeding risk. Each domain is stratified into low, moderate, or high risk, contributing to a composite score that guides prophylactic decision-making. 13 Patient-related factors include established VTE risk promoters such as advanced age, a body mass index (BMI) over 30, a history of VTE, and active malignancy. Surgical factors consider decompression procedures to carry lower VTE risk, whereas fusion procedures—particularly multi-level fusions—are classified as higher risk. Neurological bleeding risk is determined by both the surgical approach and anatomical location. Non-decompressive procedures are considered to pose minimal bleeding risk, while decompressive surgeries, especially those involving the cervical or thoracic spine, are associated with higher bleeding risk for which chemoprophylaxis may not be ideal.
Based on the cumulative risk assessment, patients categorized as low risk were typically managed with mechanical prophylaxis alone. Those at moderate risk were considered for chemoprophylaxis on a case-by-case basis, while high-risk patients received chemoprophylaxis—typically enoxaparin—initiated 24 to 36 hours postoperatively and continued for approximately 2 weeks.
Shapiro et al. validated the Eskildsen algorithm by administering LMWH to patients identified as high risk for VTE based on the tool. Of the study population, 20.7% (n = 55) were classified as high risk and received chemoprophylaxis. Among these patients, the incidence of postoperative hematoma was 0.75%, which falls within the range reported in the literature (0.3%–3.5%). Delayed wound healing or deep infection occurred in 2.26% of patients, also consistent with reported rates of 2%–3%. Additionally, the VTE rate was 0.38%, which is lower than the published incidence of 1.0%–1.9%. These findings support the algorithm’s utility in identifying appropriate candidates for chemoprophylaxis while maintaining a favorable safety profile. 59 Given that the observed complication rates were within or below published values, these findings suggest that implementing an algorithmic approach to guide prophylactic measures may improve the efficacy and safety of VTE chemoprophylaxis in spine surgery.
Conclusion
Postoperative VTE is a significant concern and remains the third leading cause of hospital-associated death in the United States, with its occurrence worsening patient outcomes. Spine surgeries, particularly fusion procedures and trauma cases, carry a heightened VTE risk—one that increases with the number of levels fused. While mechanical prophylaxis poses minimal risk, chemoprophylaxis offers the greatest reduction in VTE incidence, though it comes with a higher bleeding risk.
Effective prophylaxis requires individualized risk stratification, taking into account factors such as BMI, age, sex, procedure type, and surgical location. Cervical and thoracic spine surgeries carry a higher risk of epidural bleeding, necessitating a careful balance between preventing thrombosis and minimizing bleeding complications. Consequently, decisions regarding chemoprophylaxis are often made on a case-by-case basis. Clinical decision-making can be supported by algorithms—such as the one developed by Eskildsen et al.—which incorporate both VTE and bleeding risk considerations.
Summary of Common VTE Prophylactic Measures, With Indications, Benefits, and Drawbacks.
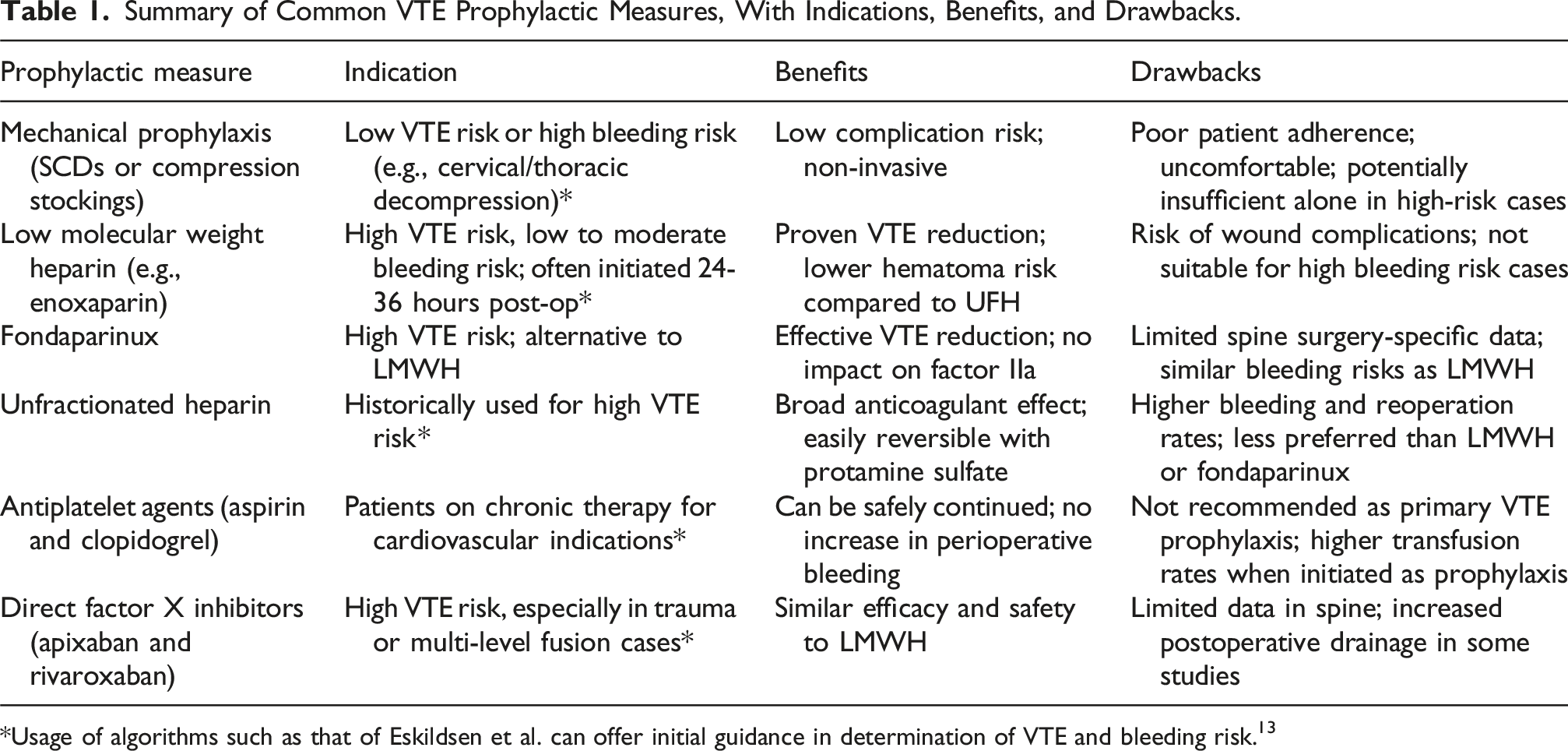
*Usage of algorithms such as that of Eskildsen et al. can offer initial guidance in determination of VTE and bleeding risk. 13
DVT Prophylactic Measure Recommendations.

**Wright et al. described a grading scale for recommendations for orthopedic surgeons based on the strength of evidence supporting the recommendation. An “A” indicates good quality evidence backing, with primarily Level I studies supporting the intervention. A “B” indicates fair quality evidence backing, with primarily Levels II and III studies supporting the intervention. A “C” indicates poor quality evidence backing with primarily Levels IV and V studies supporting the intervention. 60
Footnotes
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
