Abstract
Aims and Objectives
This in-vitro study aims to evaluate and compare the effects of remineralizing mouthwashes based on inorganic fluoride, organic fluoride, or zinc hydroxyapatite on the mechanical properties (yield strength (YS) and modulus of elasticity (ME)) of NiTi and CuNiTi archwires under loading and unloading.
Materials and Methods
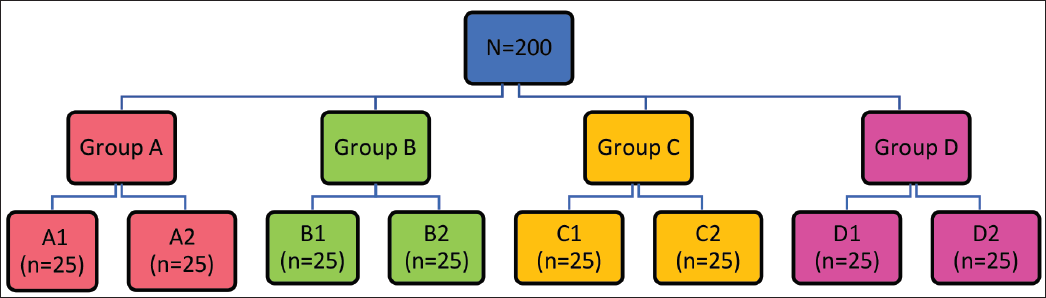
A total of 200 samples of 0.016 × 0.022 NiTi and CuNiTi archwires were divided into four groups (n = 50 per group) and immersed in 5 mL of different remineralization agents. The samples were incubated at 37°C for 1.5 h, then rinsed and tested using a universal testing machine. A three-point bending test was conducted to determine the YS and ME of the wires.
Results
Tukey’s post hoc test and one-way ANOVA were used to analyze differences between groups. The results showed that for NiTi wires, only the inorganic fluoridated mouthwash (Phos-Flur) significantly affected mechanical properties during loading. During unloading, both inorganic (Phos-Flur) and organic fluoridated (Amflor) mouthwashes had a significant effect. For CuNiTi wires, only the inorganic fluoridated mouthwash (Phos-Flur) showed a significant effect during unloading. The non-fluoridated zinc hydroxyapatite mouthwash did not affect either NiTi or CuNiTi wires during loading or unloading.
Conclusion
Non-fluoridated remineralizing zinc hydroxyapatite mouthwash did not affect the loading and unloading mechanical properties of both NiTi and CuNiTi wires when compared with fluoridated mouthwashes. Zinc hydroxyapatite-based mouthwashes may serve as a safer alternative without compromising remineralization effectiveness.
Introduction
One significant and common iatrogenic side effect of orthodontic therapy is enamel surface decalcification near fixed orthodontic appliances.1, 2 Because enamel translucency is directly related to the degree of mineralization, initial enamel demineralization typically manifests clinically as a white spot lesion (WSL).2, 3 Orthodontic appliances increase plaque accumulation, making oral hygiene challenging and raising the risk of enamel decalcification due to low pH near brackets.1, 2 Studies show that orthodontic patients, especially teenagers, are more prone to decalcification. 4 Remineralizing agents are recommended to restore the deeper layers of WSLs and improve aesthetics. 5
Typical mouthwashes recommended for remineralization during orthodontic treatment include those containing inorganic fluoride, organic fluoride, and other non-fluoridated agents.
As a result of the nanotechnology revolution, mouthwashes and dentifrices containing hydroxyapatite nanocrystals were developed. It was discovered to form a layer that shields the enamel structure underneath. The fluoride-free mouthrinse Biorepair (Coswell SpA, Bologna, Italy) contains zinc hydroxyapatite (Zn-HAP) microparticles. The Zn-HAP microparticles (MicroRepair®) are biomimetic to the mineral that forms enamel. The addition of nanocrystals to the mouthrinse provided long-term anti-inflammatory, anti-plaque, and anti-gingival properties. 6 According to studies 7 comparing different fluoridated and non-fluoridated remineralizing mouthwashes, zinc hydroxyapatite toothpastes and mouthwashes offer the highest amount of remineralization capability.
In orthodontic therapy, titanium-based archwires are utilized to provide light, continuous force with high activation levels over prolonged periods. As a result, they are extremely effective as beginning or transitional wires between the initial alignment and the end of treatment. 8 Studies8–12 have shown changes in the mechanical properties of NiTi and CuNiTi wires in response to the use of fluoride-based remineralization agents.
The impact of remineralizing agents based on zinc hydroxyapatite or organic fluoride on the mechanical characteristics of titanium-based wires has not been investigated. It is essential to conduct a study to assess the impact of organic fluoride and zinc hydroxyapatite-based remineralizing agents on the mechanical properties of titanium-based wires, since research has demonstrated that these agents have superior remineralizing potential when compared to fluoride-based agents.
Materials and Methods
The study will be divided into four groups (n = 50 for each group) depending on the type of remineralization agent used. Group A (control group): Deionized water (Figure 1), Group B (inorganic fluoride): Phos-Flur group (Figure 2), Group C (organic fluoride): Amflor group (Figure 3), and Group D (zinc hydroxyapatite): Biorepair group (Figure 4).
Phos-Flur Mouthwash.

Amflor Mouthwash.
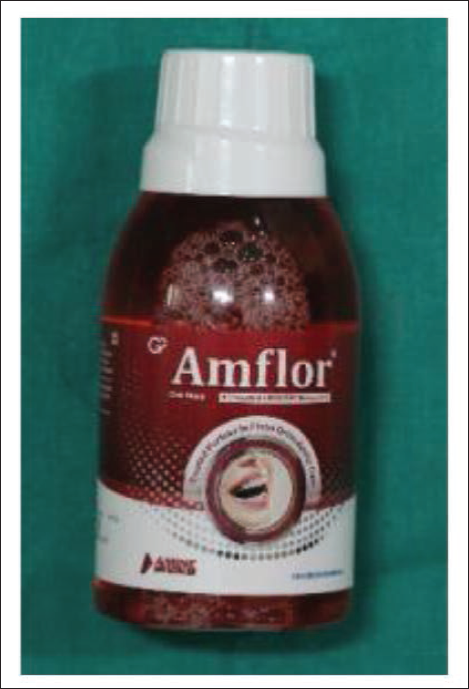
Biorepair Mouthwash.

Deionized Water.

A total of 100 archwires were used in this study, which comprises 50 preformed 3M 0.016 × 0.022 rectangular NiTi archwires (Figure 5) and 50 preformed 3M 0.016 × 0.022 rectangular CuNiTi archwires (Figure 6). From the straight portion of each preformed archwire, two wire specimens of length 25 mm will be cut. We yield a total of 100, 0.016 × 0.022 rectangular NiTi and 100, 0.016 × 0.022 rectangular CuNiTi wire specimens.
Preformed NiTi Archwires (3M)-50.
Preformed CuNiTi Archwires (3M)-50.

All the cut wire specimens will be stored in polypropylene test tubes containing 5 mL of various remineralization agents. Each group will be further divided into two subgroups (n = 25 for each subgroup) based on the type of wire specimen used.
All the samples will be incubated in a water bath at 37°C for 1.5 h (Figure 7). After the predetermined time, all the wire specimens have been removed from the remineralizing solutions, rinsed properly with distilled water, and sent for testing.
Heated Controlled Water Bath.

Specimen Preparation
For the mechanical bending test, a custom-made acrylic jig was designed (Figure 8). Standard edgewise brackets (0.22″ slot; American Orthodontics) with no tip and torque were bonded with cyanoacrylate at an inter-bracket distance of 12 mm. 8
Custom-made Jig.

Mechanical testing followed ANSI/ADA Specification No. 32 using a three-point bending test on a universal testing machine. Wire specimens were mounted on a custom 12 mm span jig, with 25 mm wire segments secured to brackets using a standardized steel ligature method. 13 To simulate oral conditions, the setup was immersed in a 37°C distilled water bath, with temperature controlled by a heater and a thermocouple thermometer (Figure 9). The centers of each wire were deflected at a crosshead speed of 1 mm/min. Each specimen was loaded until a deflection of 3.1 mm was produced (Figure 10); thereafter, they were deloaded (deactivated) at the same crosshead speed until the load became zero.8, 14
Mechanical Testing of Sample in Instron.

Loading of Specimen Till 3.1 mm Deflection.

Comparison of Modulus of Elasticity and Yield Strength of NiTi Archwires. (a) Comparison of ME Among the Four Study Groups During Loading of NiTi Wires. (b) Comparison of ME Among the Four Study Groups During Unloading of NiTi Wires. (c) Comparison of YS Among the Four Study Groups During Loading of NiTi Wires. (d) Comparison of YS Among the Four Study Groups During Unloading of NiTi Wires.
Comparison of Modulus of Elasticity and Yield Strength of CuNiTi Archwires. (a) Comparison of ME Among the Four Study Groups During Loading of CuNiTi Wires. (b) Comparison of ME Among the Four Study Groups During Unloading of CuNiTi Wires. (c) Comparison of YS Among the Four Study Groups During Loading of CuNiTi Wires. (d) Comparison of YS Among the Four Study Groups During Unloading of CuNiTi Wires.
For each wire specimen, the ME and YS were calculated automatically by a software program, Shimadzu Autograph AGS-X, which is linked to the Universal Testing Machine (Instron Matrix 5KN).
Statistical Analysis
The Statistical Package for the Social Sciences (SPSS), version 21, was used for all statistical computations. The data are summarized using the groups’ mean and standard deviation (SD) values. Using the Kolmogorov–Smirnov test, the recorded data in the four study groups were examined for normality. Given that the data were determined to be normally distributed, Tukey’s post hoc test was employed for intergroup comparison, and a parametric test (one-way ANOVA) was performed to determine the overall significance between the groups.
Results
The mean and SD values of ME and YS of NiTi wires while loading and unloading for Group A1 (control group): Deionized water, Group B1 (inorganic fluoride): Phos-Flur, Group C1 (organic fluoride): Amflor, and Group D1 (zinc hydroxyapatite): Biorepair are listed in Table 1.
Comparison of Modulus of Elasticity and Yield Strength Among the Four Study Groups During Loading and Unloading of NiTi Wires Using One-way ANOVA.
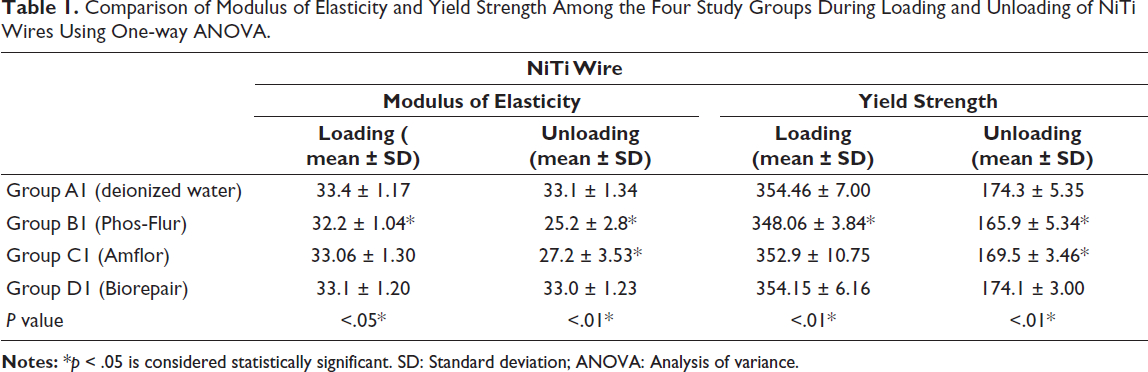
This demonstrates that, in comparison to the control group (deionized water), the mechanical properties (ME and YS) of NiTi wires are significantly impacted by the use of fluoridated remineralizing mouthwashes (Phos-Flur and Amflor). The mechanical characteristics of NiTi wires are not significantly affected by the non-fluoridated remineralizing mouthwash containing zinc hydroxyapatite (Biorepair), Group D1 used in this study, as compared to Group A1 (control group) during loading and unloading (Graph 1).
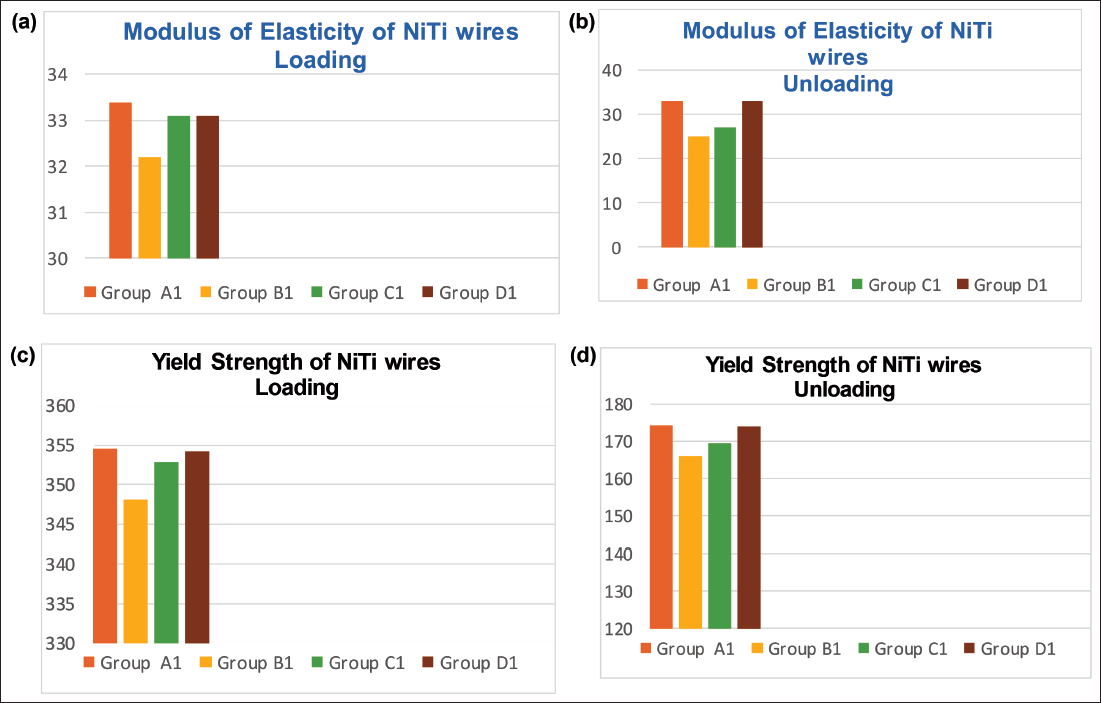
The mean and SD values of ME and YS of CuNiTi wires while loading and unloading for Group A2 (control group): Deionized water, Group B2 (inorganic fluoride): Phos-Flur, Group C2 (organic fluoride): Amflor, and Group D2 (zinc hydroxyapatite): Biorepair are listed in Table 2.
Comparison of Modulus of Elasticity and Yield Strength Among the Four Study Groups During Loading and Unloading of CuNiTi Wires Using One-way ANOVA.
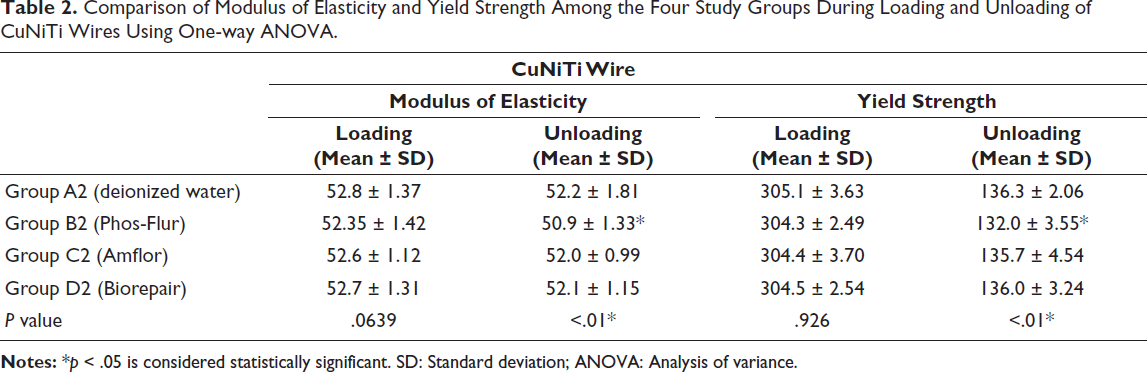
This study demonstrates that none of the test groups’ mechanical properties is significantly impacted when CuNiTi wires are employed during loading. When compared to the control group (deionized water), only inorganic fluoride mouthwashes, such as Phos-Flur (Group B2), significantly affect the mechanical parameters (ME and YS) during unloading (Graph 2).
Discussion
According to Todd et al., WSLs in patients following orthodontic treatment range from 15% to 91%. 15 According to Ogaard et al., 16 the period needed for this WSL manifestation is significantly less than the interval between two orthodontic visit sessions. The progression or reversal of WSLs depends on the balance between demineralization and remineralization. 17 This balance is influenced by factors such as saliva pH, fluoride availability, and calcium and phosphate levels. Enamel demineralization occurs when pH drops below the critical level, leading to hydroxyapatite breakdown. Modern dentistry emphasizes non-invasive treatment to manage WSLs and restore oral health. 18
Mouthwashes that contain inorganic fluoride, organic fluoride, and other non-fluoridated ingredients are commonly recommended for use during orthodontic treatment to promote remineralization.
This study examines the effects of different remineralizing mouthwashes on nickel–titanium (NiTi) orthodontic wires. Colgate Phos-Flur contains 0.04% sodium fluoride (pH 5.1), 19 is alcohol-free, and is widely used among orthodontic patients for preventing enamel demineralization. However, it may weaken orthodontic wires and cause side effects like discoloration or hypersensitivity. Amflor contains amine fluoride (pH 6.8), an organic fluoride with superior and sustained remineralizing capacity than inorganic fluorides.20, 21 It also shows similar drawbacks to inorganic fluoride, but has shown better enamel protection. Biorepair, with Zn-HAP nanoparticles (pH 7.6), represents a newer generation of remineralizing agents with strong anti-caries, anti-plaque, and remineralizing properties.6, 7
While fluoride agents have been shown to degrade the mechanical properties of NiTi and CuNiTi wires, the effects of amine fluoride and zinc hydroxyapatite on wire strength have not been studied. Therefore, this study aims to investigate their impact on the mechanical characteristics of NiTi orthodontic wires, considering their higher remineralizing potential.
Pairwise comparisons of NiTi wires during loading showed statistically significant differences in ME and YS between Group B1 (Phos-Flur) and Group A1 (deionized water), C1 (Amflor), and D1 (Biorepair). No significant differences were found between A1, C1, and D1. During unloading, statistically significant differences were observed between most groups, including A1 versus B1 and C1, B1 versus C1 and D1, and C1 versus D1. However, the difference between A1 (deionized water) and D1 (Biorepair) was not statistically significant.
Fluoridated remineralizing mouthwashes (Phos-Flur and Amflor) significantly affect the mechanical properties (ME and YS) of NiTi wires compared to the control (deionized water). In contrast, the non-fluoridated mouthwash containing zinc hydroxyapatite (Biorepair) does not significantly impact these properties during loading or unloading.
In this study, inorganic fluoridated mouthwash with a pH of 5.1 (Phos-Flur) affected the mechanical properties of NiTi wires during both loading and unloading. Similar results were reported in studies done by Gupta et al. 11 and Koushik et al., 22 though others reported effects only during unloading.
Organic fluoridated mouthwash (Amflor, pH 6.8) influenced NiTi wires only during unloading, aligning with Huang et al., 23 Walker et al., 8 Suryawanshi et al., 24 and Nakagawa et al. 25
Non-fluoridated remineralizing mouthwashes like zinc hydroxyapatite (Biorepair) used in this study, pH 7.6, have no significant effect on the mechanical properties of NiTi wires, both while loading and unloading.
This suggests that the acidic pH of fluoride agents contributes to the breakdown of the protective titanium oxide layer, leading to fluoride-induced hydrogen embrittlement and altered mechanical properties of titanium-based alloys. Inorganic fluoridated mouthwash (pH 5.1) caused greater degradation during both loading and unloading compared to organic fluoridated mouthwash (pH 6.8), which affected properties only during unloading. These findings highlight the role of pH and are supported by similar studies. 26
For CuNiTi wires during loading, pairwise comparisons showed no statistically significant differences in ME and YS between any groups. However, during unloading, significant differences were found between Group B2 (Phos-Flur) and Group A2 (deionized water), C2 (Amflor), and D2 (Biorepair). No significant differences were observed between A2, C2, and D2 during unloading.
The study found that CuNiTi wires are not significantly affected by any test group during loading. However, during unloading, only the inorganic fluoridated mouthwash (Phos-Flur, pH 5.1) significantly impacted mechanical properties compared to the control. Similar 12 and conflicting8, 27 results have been reported in previous studies. Organic fluoridated (Amflor) and non-fluoridated (Biorepair) mouthwashes showed no significant effect during either phase.
The addition of copper to NiTi wires enhances their thermal properties and helps protect against fluoride-induced degradation. Copper acts as an inhibitor against reducing acids like hydrofluoric acid (HF), which typically dissolves protective titanium oxide layers. This copper effect reduces hydrogen penetration and formation of titanium hydride, thereby preserving the wire’s mechanical properties.8, 28
The study highlights that fluoride-remineralizing mouthwashes can weaken the mechanical properties of NiTi wires, and to a lesser extent, CuNiTi wires. Though the observed changes occurred after just 1.5 h of exposure, equivalent to three months of daily one-minute treatments, real-life exposure is likely longer due to post-treatment guidelines and extended use of archwires, suggesting the clinical effects may be more significant over time.
To prevent the overall outcome of the therapy from being hindered and delayed by changes, clinicians should consider the pH of preventive products. Non-fluoridated mouthwashes, like zinc hydroxyapatite, are a safer alternative for preventing WSLs, as they do not significantly alter the mechanical properties of NiTi or CuNiTi wires.
Limitations of the Study
The study has limitations, including not evaluating the effects of saliva or directly testing for hydrogen embrittlement. As an in-vitro study, its results may not fully reflect clinical conditions. Real-life fluoride exposure occurs in shorter, repeated doses rather than continuous exposure. Future research should explore whether cumulative, frequent fluoride applications have similar impacts on NiTi and CuNiTi wire properties.
Conclusion
Inorganic fluoridated mouthwashes had a significant effect on both the loading and unloading mechanical properties of NiTi wires. Organic mouthwashes had a significant effect on only the unloading mechanical properties of NiTi wires. None of the remineralizing mouthwashes compared in this study had a significant effect on the loading mechanical properties of CuNiTi wires. Only inorganic mouthwashes had a significant effect on the unloading mechanical properties of CuNiTi wires. Non-fluoridated remineralizing zinc hydroxyapatite mouthwash did not affect the loading and unloading mechanical properties of both NiTi and CuNiTi wires. Zinc hydroxyapatite-based mouthwashes may serve as a safer alternative without compromising remineralization effectiveness.
Footnotes
Acknowledgements
The authors express their sincere gratitude to all those who contributed to the successful completion of this study. We are especially thankful to the Department of Orthodontics for providing the necessary facilities and continuous support throughout the research process. We extend our appreciation to the laboratory staff for their technical assistance and guidance during the experimental procedures. We are also grateful to our mentors and colleagues for their valuable insights, encouragement, and constructive feedback. Finally, we acknowledge the support of our institution for fostering an environment conducive to research and academic growth.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Institutional Statement
This study was approved by the Institutional Ethics Committee (IEC) under approval number ANIDS/IEC/2023010.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
