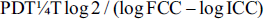Abstract
Background
Mesenchymal stem cells (MSCs) are multipotent cells with self-renewal and differentiation capabilities, making them promising candidates for regenerative medicine. Iliac crest stem cells are a widely used source of MSCs valued for their accessibility, estrogenic potential, and established role in bone regeneration and repair studies. The human mandibular bone marrow (hMBM) is a readily accessible and potentially rich source of MSCs, particularly valuable in orofacial and craniofacial reconstruction.
Purpose
This study aimed to isolate and characterize MSCs from hMBM. The isolation of hMBM-derived MSCs serves multiple purposes, including fundamental research in craniofacial biology and pathology, regenerative medicine, disease modeling, and personalized cell-based therapies.
Methods
While bone marrow-derived stem cells from the iliac crest are widely used for autologous transplantation and bone repair, MSCs from the human mandible have not been extensively explored for regenerative applications. Given the availability of mandibular bone marrow (MBM) during oral and maxillofacial procedures, this study investigated the osteogenic and chondrogenic potential of human mandibular mesenchymal stem cells (hMMSCs) in vitro.
Results
The isolated hMMSCs expressed characteristic MSC surface markers (CD90, CD105, and CD73), exhibited colony-forming ability, and demonstrated tri-lineage differentiation potential. Notably, hMMSCs showed a strong capacity to differentiate into osteoblasts, which surpassed their chondrogenic potential.
Conclusion
hMBM is a viable source of MSCs with significant osteogenic potential. These findings support the potential application of hMMSCs in bone regeneration and mandibular repair strategies. Our data thus exhibits for the first time that hMMSCs are a reliable cell source for mandibular tissue engineering.
Introduction
Regenerative medicine holds immense promise for addressing tissue loss and organ dysfunction by leveraging the body’s intrinsic healing capabilities, often augmented by exogenous cellular therapies. At the heart of this field are stem cells—undifferentiated cells with the capacity to self-renew and differentiate into various specialized cell types. Adult stem cells, particularly mesenchymal stem cells (MSCs), have garnered significant attention due to their multipotency, immunomodulatory properties, and relative ease of accessibility from various tissues.1, 2
Traditionally, the iliac crest has been the primary source of bone marrow-derived MSCs (BMMSCs) for regenerative applications in oral and maxillofacial surgery. 3 However, the associated donor site morbidity, pain, and the need for general anesthesia make it less ideal for certain clinical scenarios, especially in localized craniofacial defects. This has spurred research into alternative, more accessible, and potentially regionally specific stem cell sources.
The human mandible, a dynamic bone involved in mastication, speech, and facial aesthetics, contains cancellous bone filled with hematopoietic and mesenchymal marrow. The close proximity to oral and maxillofacial pathologies, coupled with their accessibility during routine dental and oral surgical procedures (e.g., wisdom tooth extraction, implant site preparation, orthognathic surgery), positions mandibular bone marrow (MBM) as an exceptionally compelling and under-explored source of autologous stem cells. This article aims to consolidate current knowledge regarding the stem cell population within MBM, characterizing their biological attributes, and evaluating their potential clinical impact on craniofacial regenerative strategies.
MSCs have proven to be a prominent cell source for regenerative medicinal purposes and bone tissue engineering.4, 5 The primary cell source for MSCs is are femur, tibia, and pelvis. 6 However, to harvest bone marrow is a technique-sensitive process associated with prolonged operating time, complications at the donor site, and extended post-operative pain management. 7 MSCs can also be obtained from various oral tissues, which include periodontal ligament, 8 apical papilla, 9 dental pulp 10 and human exfoliated deciduous teeth.11, 12 Numerous studies have been reported on the osteogenic potential of oral MSCs as a substitute source for osteogenic regeneration. 10 A comparison is made in various studies between oral sources of stem cells and their MSC counterparts derived from sources other than oral tissues.13, 14
Specifically, the mandible, a part of the lower jaw, is an attractive source of stem cells due to its role in facial development and the potential for craniofacial tissue regeneration. This article aims to explore the stem cell population within human mandibular bone marrow (hMBM), focusing on their characterization, potential, and implications. MSCs have been explored extensively for various therapeutic purposes, including bone regeneration, in recent years.15, 16
The first and foremost described source for MSCs was derived from bone marrow and is considered the most reliable source pertaining to bone regeneration.17, 18 Tibia, femur, and iliac crest are conventionally used sources to acquire BMMSCs for osteogenic regeneration for ages. 19 But it remains uncertain whether human mandibular mesenchymal stem cells (hMMSCs) can be more potent for this purpose.
The mandible callus is composed of cells originating from the neural crest, whereas the tibial bone is entirely of mesodermal origin. 20 Mandibular bone grafts rendered a better outcome compared to the graft procured from the iliac crest with respect to alveolar cleft repair, concluding that there might be less resorption from the graft site. 21
hMBM offers several advantages as a source of MSCs, particularly in the context of orofacial and craniofacial reconstruction. Its proximity to the defect site facilitates localized delivery, minimizing systemic distribution and potential off-target effects. Moreover, hMBM is relatively accessible, making it a practical source for clinical applications.
In this study, we looked at the MBM-derived MSCs and examined their potential toward osteogenic and chondrogenic lineage. The study provides a detailed methodology employed for isolating MSCs from hMBM, discusses their advantages and limitations, and explores their potential applications in the realm of regenerative medicine.
Materials and Methods
Isolation and Culture of Cells
Mandibular bone was collected at the time of the implant procedure (n = 5). Written informed consent was received from all subjects involved in the present study. Mandibular bone was obtained from healthy subjects coming for implant placement within the age range of 20–25 years at the institutional implant center.
The primary cultures for hMMSCs were obtained by enzymatic digestion. Cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) (Gibco) enhanced with 10% fetal bovine serum (FBS) (Gibco) and 1% penicillin/streptomycin (all from Invitrogen, India) and sustained in a humidified cell culture incubator with a temperature of 37 °C, along with the presence of 5% CO2. Cell passage was done with the use of 0.25% trypsin. Experiments were performed with the cells between passage numbers two and five.
Population Doubling Time and Cell Growth Curve
For determining the proliferative potential of hMMSCs, 1 × 104 cells at passage four were seeded into culture plates (12-well), and the cell count was taken every other day for 15 days. Population doubling time (PDT) was calculated after 48 hours based on the following equations:
Where ICC means the initial cell count, FCC means the final cell count, and T is the incubation time (in hours). The growth curve for 9 days was plotted using cell numbers only.
Colony Forming Unit Efficiency
A colony-forming unit-fibroblast (CFU-F) assay was performed to assess the capability of cells to form colonies. Cells were seeded (P4) into 60 mm petri plates at an initial density of 5 × 102 cells per well in culture media (DMEM with 10% FBS and 1% AA). After 10 days of incubation, cells were washed with 1X phosphate-buffered saline (PBS) and stained with 0.3% crystal violet.
Characterization Using Flow Cytometry
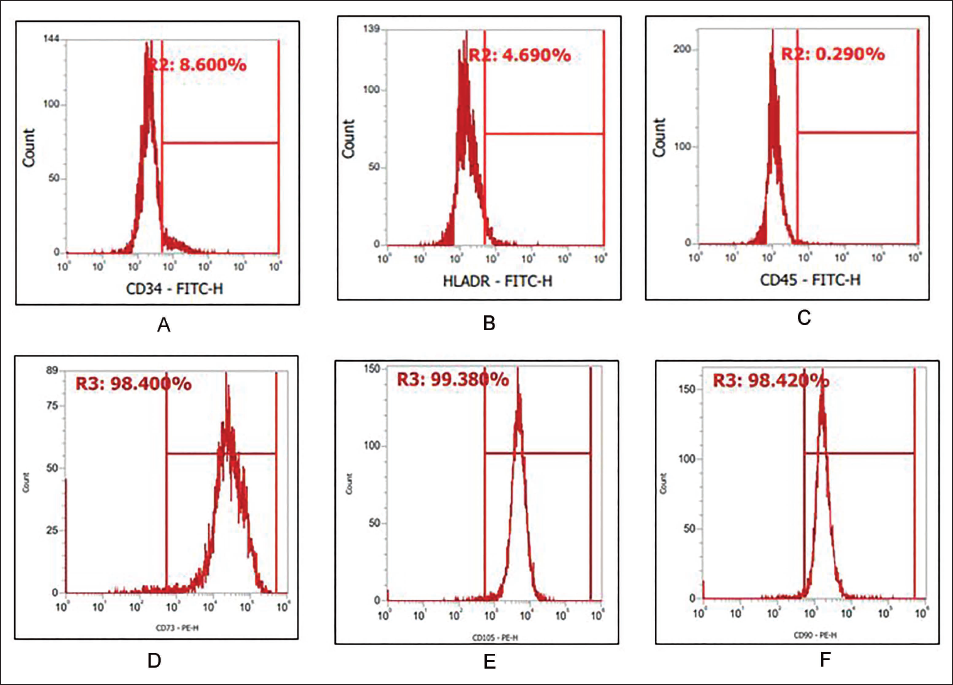
For flow cytometric analysis, passage 4 (P4) hMMSCs were obtained, washed with 1X PBS and incubated for 30 min at 4 ℃ with surface marker antibody, CD34 (8.6%), CD45 (0.29%), human leukocyte antigen (HLA)-DR (4.6%), CD73 (98.4%), CD90 (99.38%), CD105 (98.42%) (All from eBiosciences, USA) antibodies. After washing the cells with 1X PBS, the cells were acquired (10,000 events were acquired per sample) on Attune NxT Flow Cytometer (Thermo Scientific, USA). In comparison to the unstained, the degree of positive staining was computed.
Osteogenic Differentiation Assay
To assess the osteogenic differentiation potential, hMMSCs were seeded at a density of 1 × 104 cells in a 24-well cell culture plate. Once cells are 80% confluent, the culture medium is replaced with osteogenic induction medium (1 mM dexamethasone, 1 mM beta-glycerol phosphate, and ascorbic acid). Osteogenic induction medium was replaced every 3 days. At the end of 21 days, differentiated hMMSCs were stained with Alizarin Red S stain for accumulation of calcium and matrix mineralization as described previously. Stained cells were observed under an inverted phase-contrast microscope to evaluate the formation of mineralized nodules.
Chondrogenic Differentiation
To investigate the chondrogenic differentiation potential, hMMSCs were seeded as described in the section on osteogenic differentiation until 80% confluence was obtained. Additionally, a chondrogenic induction medium containing high-glucose DMEM supplemented with 1X-ITS, 1 mM sodium pyruvate, 100 nM dexamethasone, 50 mg/mL ascorbate-2-phosphate, 40 mg/mL L-proline, and 10 ng/mL TGF-β3 was used in place of the culture media. The media was replaced every 2–3 days while the cultures were cultured for 4 weeks at 37 °C in a 5% CO2 environment. After 21 days, the cells were fixed with 4% paraformaldehyde and stained for proteoglycans and glycosaminoglycans using 0.1% Safranin O and Alcian blue, respectively, to show the chondrogenic differentiation.
Statistical Analysis
The experiments were performed thrice, and the sample analysis was performed (n = 3). All the data values are represented as mean ± standard deviation.
All experiments were performed in triplicate.
Results
Human Mandible MSCs Showed Fibroblast-like Cells
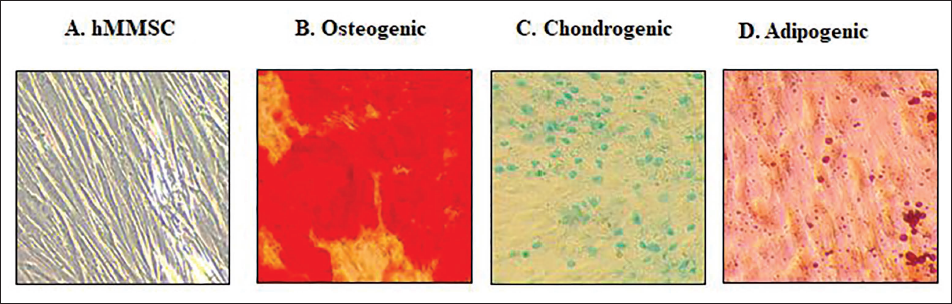
A successful isolation of hMBM will yield a population of adherent, fibroblast-like cells that can be expanded in vitro. Expanded M-MSCs will exhibit a characteristic spindle-shaped, fibroblast-like morphology (Figure 3A).
Colony Forming Potential of hMMSCs
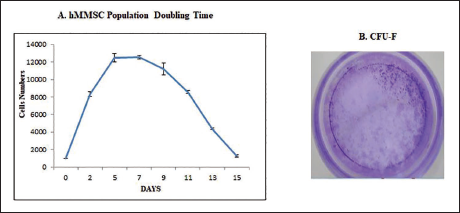
The growth curve for 9 days was plotted using cell numbers only (Figure 1A). To assess the colony-forming potential, CFU assays were performed using 100 cells of hMMSCs. After 14 days of culture in the growth medium, hMMSCs generated a significantly higher number of colonies: 22 ± 3 in single-cell CFU assays (Figure 1B).
A. PDT Shown by hMMSCs at Passage Three, 1,000 Cells/Well were Seeded in 24 Well Plates and Harvesting was Carried Out Every Alternate Day. (B) hMMSCs CFU-F.
M-MSCs Express Stem Cells Characterization
At passage five, hMMSCs expressed MSC-specific surface markers, including CD105, CD90, and CD73, being negative for hematopoietic markers CD45, CD34, and HLA-DR. Flow cytometric analysis confirmed high expression levels of CD105 (%), CD73 (%), and CD90 (%), with negligible expression of CD45 and CD34 (Figure 2). Additionally, hMMSCs demonstrated positive differentiation into both osteogenic and chondrogenic lineages, forming mineralized nodules during osteogenesis and producing glycosaminoglycans during chondrogenesis (Figure 2).
(A–D) hMMSCs Showed Homogenous Mesenchymal Population Stained Negative for CD34 and CD45, Positive for CD73, CD90, and CD105 Surface Marker. (E) PDT Shown by hMMSCs at Passage Three, 1,000 Cells/Well were Seeded in 24 Well Plates and Harvesting was Carried Out Every Alternate Day. (F) hMMSCs CFU-F.
Osteogenic Differentiation Potential of hMMSCs
Cells at a density of 2500/cm2 were seeded in a 24-well culture plate with complete growth medium. Twenty-four hours later, the cells were cultured in induction medium (DMEM supplemented with 10% FBS, 1% AA, 0.1 mM dexamethasone, 50 mM ascorbate 2-phosphate, and 10 mM b-glycerophosphate). The differentiation induction medium was changed twice a week. Control cells were those taken from the basal medium. After 21 days, the cells were fixed with formalin and then stained with 2% Alizarin Red S to demonstrate the osteogenic differentiation and related mineralization (Figure 3B).
hMMSCs Differentiation into Osteogenic, Chondrogenic, and Adipogenic Differentiation. hMMSCs. The Panels Display Representative Photomicrographs of: (A) hMMSCs; (B) Osteoblasts Detected by Alizarin Red S Staining–Mineralized Matrix; (C) Chondrocytes Detected by Alcian Blue Staining–GAGs. (D) Adipogenic Cells Detected by Oil Red Staining.
Chondrogenic Differentiation Potential of hMMSCs
2.5 × 105 cells from each set of the triplicates were spun in a 15-mL polypropylene tube at 1000 rpm for 5 minutes, followed by a DMEM wash, to evaluate the chondrogenic differentiation. High-glucose DMEM supplemented with 1X-ITS, 1 mM sodium pyruvate, 100 nM dexamethasone, 50 mg/mL ascorbate-2-phosphate, 40 mg/mL L-proline, and 10 ng/mL TGF-β3 was used to cultivate the cells. Cell cultures were incubated with 5% CO2 in the atmosphere at 37 °C. The differentiation induction medium was changed every 2–3 days. After 21 days of induction, the cells were fixed with 4% paraformaldehyde and further stained for the presence of glycosaminoglycans using 0.1% safranin O and Alcian Blue for chondrogenic differentiation (Figure 3C).
Discussion
This study investigated the phenotypic and functional properties of stem cells derived from hMBM, with a focus on their MSC characteristics, differentiation potential, and immunophenotypic profile. Our findings support the hypothesis that MBM is a rich and viable source of stem cells with properties comparable to those derived from iliac crest bone marrow, which is conventionally considered the gold standard. 22
Osteoblastic differentiation of bone marrow cells from jaw and long bones exhibits different characteristics. Similarly, site-specific differences were evident in the bone marrow stem cells from the human mandible and iliac crest.23, 24
Phenotypically, the hMMSCs expressed classical MSC surface markers such as CD73, CD90, and CD105, while lacking hematopoietic markers including CD34 and CD45 (Figure 2). This immunophenotypic profile aligns with the criteria established by the International Society for Cellular Therapy for defining human MSCs, confirming the mesenchymal origin of the isolated cells. 22
Furthermore, the CFUs directly influence the difference in the amount of bone marrow.25, 26 Therefore, a CFU assay was performed to evaluate the colony-forming potential of the hMMSCs, which suggested that the human mandible contains a highly dependable and invariable source of MSCs (Figure 1). The success of bone regeneration is highly dependent on the source of stem cells and their availability, which makes the mandible an exemplary source for MSCs from the orofacial region.
In proliferation assays, hMMSCs displayed a higher population doubling rate, suggesting enhanced proliferative capacity. 27 This is a promising feature for tissue engineering and regenerative medicine applications, particularly in the context of craniofacial reconstruction, where cell expansion is often necessary before implantation.
In our study, we examined the feasibility of MSCs isolated from human mandible bone to serve as a cell source for bone regeneration. Differentiation of isolated MSCs into osteogenic and chondrogenic lineages was investigated in the present study. 10 We ascertained very negligible adipogenic differentiation from these cells, indicating their propensity toward hard tissue. The overall osteogenic potential of hMMSCs was found to be remarkable (Figure 3).
Our results unequivocally demonstrated that mandibular bone is a promising source of MSCs and possesses significant osteogenic and chondrogenic differentiation potential. This study is limited by its small sample size and in vitro design, which may not accurately reflect the in vivo behavior and functional potential of MBM stem cells in clinical settings. Additional research required on the osteogenic capacity of human MSCs in a bone scaffolding environment is a viable option for repairing bone abnormalities.
Footnotes
Acknowledgments
The authors thank the Implant Center, Dr. D. Y. Patil Dental College and Hospital for their assistance in obtaining a human mandibular graft. The authors would like to acknowledge Dr. D. Y. Patil Vidyapeeth for providing the facility to carry out the present work.
Authors’ Contribution
Avinash Kharat–Methodology and data collection, Supriya Kheur–Analysis, Avinash Sanap–Manuscript writing, Ramesh Bhonde–Supervision.
Data Availability Statement
Data is available on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Institutional Statement
Ethical approval was obtained from the ethical approval committee of Dr. D. Y. Patil Dental College and Hospital, Pimpri, Pune. The institutions and participants were informed about the objectives, risks, and benefits of the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Signed informed consent was obtained from all participants upon their agreement to participate in the present study.
Statements and Declarations
I hereby declare that the information provided in this resume is true and accurate to the best of my knowledge.