Abstract
Background
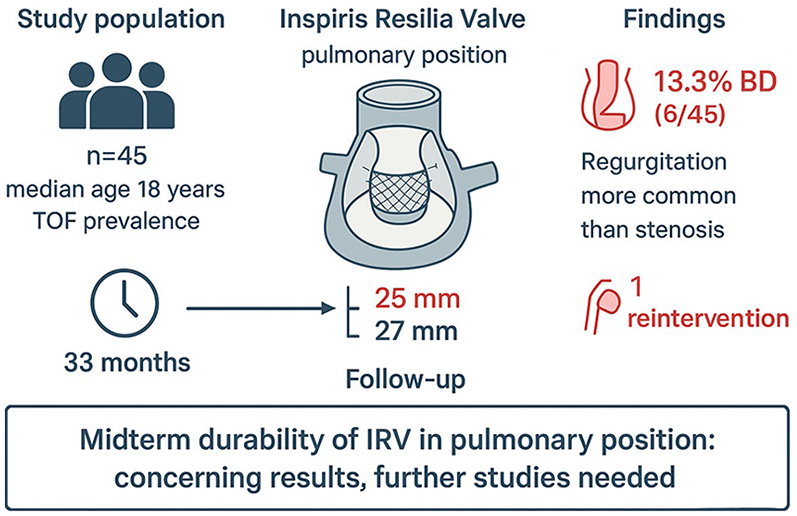
To evaluate hemodynamic performance and midterm durability of the Inspiris Resilia Valve (IRV) in pulmonary position in patients with different cardiac anomalies.
Methods
We retrospectively reviewed the medical records of 45 patients who underwent pulmonary valve implantation with IRV between January 2018 and August 2023. Demographic data included primary diagnosis, age at surgery, intraoperative data, and follow-up (FU). Primary outcomes are bioprosthesis dysfunction (BD) and prosthesis-related reintervention. BD was defined as moderate or severe bio-prosthesis insufficiency or trans-prosthesis peak gradient >50 mmHg.
Results
Most common primary diagnosis was Tetralogy of Fallot (28 patients). Indication for surgery was severe pulmonary regurgitation with significant right ventricle (RV) dilatation, with median RV end-diastolic volume of 164 (151–174) ml/m2. Median age and weight at surgery were 18 years (interquartile range [IQR] 15–30) and 62 kg (IQR 47–72). Most common IRV size implanted was 25 mm (31 patients), followed by 27 mm (11 patients). After a median FU time of 33 months (IQR 18–45), 6 patients (13.3%) developed BD and 1 required trans-catheter reintervention. All patients who developed BD had a 25 mm IRV implanted. Our statistical analysis showed that higher peak gradient and higher RV systolic pressure were associated with increased BD occurrence (p value < 0.001); moreover, bioprosthesis regurgitation was a more significant cause of BD than stenosis (p value < 0.001).
Conclusions
Despite short–medium-term FU, our results suggest that IRV durability in the pulmonary position is concerning. Further discussion and collaboration are needed to evaluate this prosthesis for pulmonary valve replacement on a larger scale and with a longer FU.
Key Point

Background
Implantation of a prosthetic valve is indicated for patients with pulmonary regurgitation who have previously undergone right ventricular outflow tract (RVOT) reconstruction.1–3 Pulmonary valve replacement (PVR) is among the most common congenital procedures performed, and several options currently exist for off-label PVR in patients beyond infancy (bioprosthesis, mechanical valve, or valve conduit). However, there is no consensus on whether it represents the best choice for this procedure. Bioprosthesis valves are likely the most widely used due to their availability and the fact that they do not require permanent anticoagulation therapy, which is particularly advantageous for young patients. Stented bioprosthetic valves have been designed for use in the aortic position but are also extensively implanted in the pulmonary position. The Inspiris Resilia Valve (IRV) (Edwards Lifesciences LLC) is a new bioprosthesis that utilizes innovative tissue preservation technology aimed at enhancing long-term durability. Resilia tissue is bovine pericardial tissue treated with a special integrity preservation technology that selectively eliminates free aldehydes, a key factor in tissue calcification, while also protecting and preserving the tissue. In adult patients, this bioprosthesis, when used in the aortic position, has demonstrated a good safety profile with excellent hemodynamic performance over 5 years of follow-up, showing 100% freedom from structural valve degeneration.4–6 Moreover, IRV features a unique hinge mechanism in valves <27 mm, allowing radial dilation without fracturing the stent frame during future transcatheter valve-in-valve (ViV) procedures. Recently, IRV has begun to be implanted in the pulmonary position, and reports have already shown varying concerns regarding the bioprosthesis’ performance.7–9 Edwards Lifesciences has initiated a prospective, nonrandomized, single-arm, multicenter trial (COMMENCE-P) to study the efficacy of the Inspiris valve in the pulmonary position. 10 This study aims to evaluate the hemodynamic performance and midterm durability of IRV implanted in the pulmonary position in our patients.
Methods
Study Population
We retrospectively reviewed the medical records of all pediatric and adult patients diagnosed with congenital heart disease who underwent PVR with IRV between January 2018 and August 2023 at Bambino Gesù Children's Hospital and Gemelli University Hospital. During the study period, all patients undergoing surgical PVR at our institutions received the IRV with no other types of prostheses or valved conduits used. The Institutional Review Board of both hospitals approved the current study. Collected demographic data included primary diagnosis, age at surgery, previous surgeries, cardiopulmonary bypass (CPB) time, aortic cross-clamping (X-Clamp) time, associated surgical procedures, and follow-up data. Follow-up was completed in all patients (100%), and all data were collected using the clinical and echocardiographic database. Routine transthoracic echocardiograms (TTEs) and cardiac magnetic resonance imaging (cMRI) were performed to determine the timing of interventions, and computed tomography was conducted in selected patients for reoperative planning. Preoperative comprehensive echocardiography is routinely performed and reported in the Cardiac Surgery Echo lab. Chamber quantification, comprehensive assessment of right ventricle (RV) geometry and systolic function, and the severity of valve dysfunction were graded according to a multiparametric approach and were performed following current recommendations. 11 Specifically, indications for PVR were related to symptoms and/or evidence of right ventricular chamber enlargement (right ventricular end-diastolic volume [RVEDV] ≥ 150 mL/m2) determined by cMRI. The size of the bioprosthetic was selected based on RVOT size with the aim of implanting a bioprosthesis large enough for potential transcatheter PVR. After implantation, patients underwent antiplatelet therapy consisting of 3 to 6 months of aspirin 100 mg, after which continuation was left to the discretion of the referring cardiologist.
All patients had a discharge TTE and at least 1 follow-up TTE; no patients were excluded due to unavailable imaging data.
Study End-Points
The primary endpoint of the analysis was freedom from bioprosthetic dysfunction (BD), while the secondary endpoint was freedom from prosthesis-related reintervention. BD was defined based on transthoracic echocardiography performed at the last available clinical follow-up and included evidence of either stenosis or regurgitation. Stenosis was defined as a transvalvular peak gradient >50 mmHg or a peak velocity of approximately >3.5 m/s. Regurgitation was graded as trivial, mild, moderate, or severe, in accordance with current echocardiographic valve assessment guidelines.12,13 Prosthesis-related reintervention was defined as any surgical or transcatheter procedure performed on the pulmonary valve due to dysfunction of the IRV.
Operative Technique
Two approaches were used in our study according to the presence of previous sternotomies. In case of redo-sternotomy, CPB was instituted by central aorto-bicaval cannulation except in 2 instances where, due to strong mediastinal adhesion, we used peripheral cannulation by groin vessel in 1 case and neck vessel in the other patient according to our approach. 14 After the previous transannular patch was longitudinally incised and the native leaflet was completely resected. The posterior sewing ring of the bioprosthesis was secured with a running suture along the native pulmonary annulus. We position the IRV such that there is a slight posterior angulation in relation to the native annulus, thereby achieving a more natural lie within the pulmonary trunk. A large bovine pericardial patch was then used to reconstruct the anterior wall of the pulmonary artery and the remaining anterior prosthesis sewing ring. The remaining part of the patch was finally used to complete the RVOT reconstruction with another running suture. The other technique was used in 5 patients with no previous surgery, where we performed a minimally invasive approach through a J-ministernotomy at the 4th intercostal space with aorto-right atrial cannulation (Figure 1). The native pulmonary artery was incised, and the pulmonary leaflets were resected. The bioprosthesis was then secured to the native pulmonary ring with a running suture similar to the technique previously described.
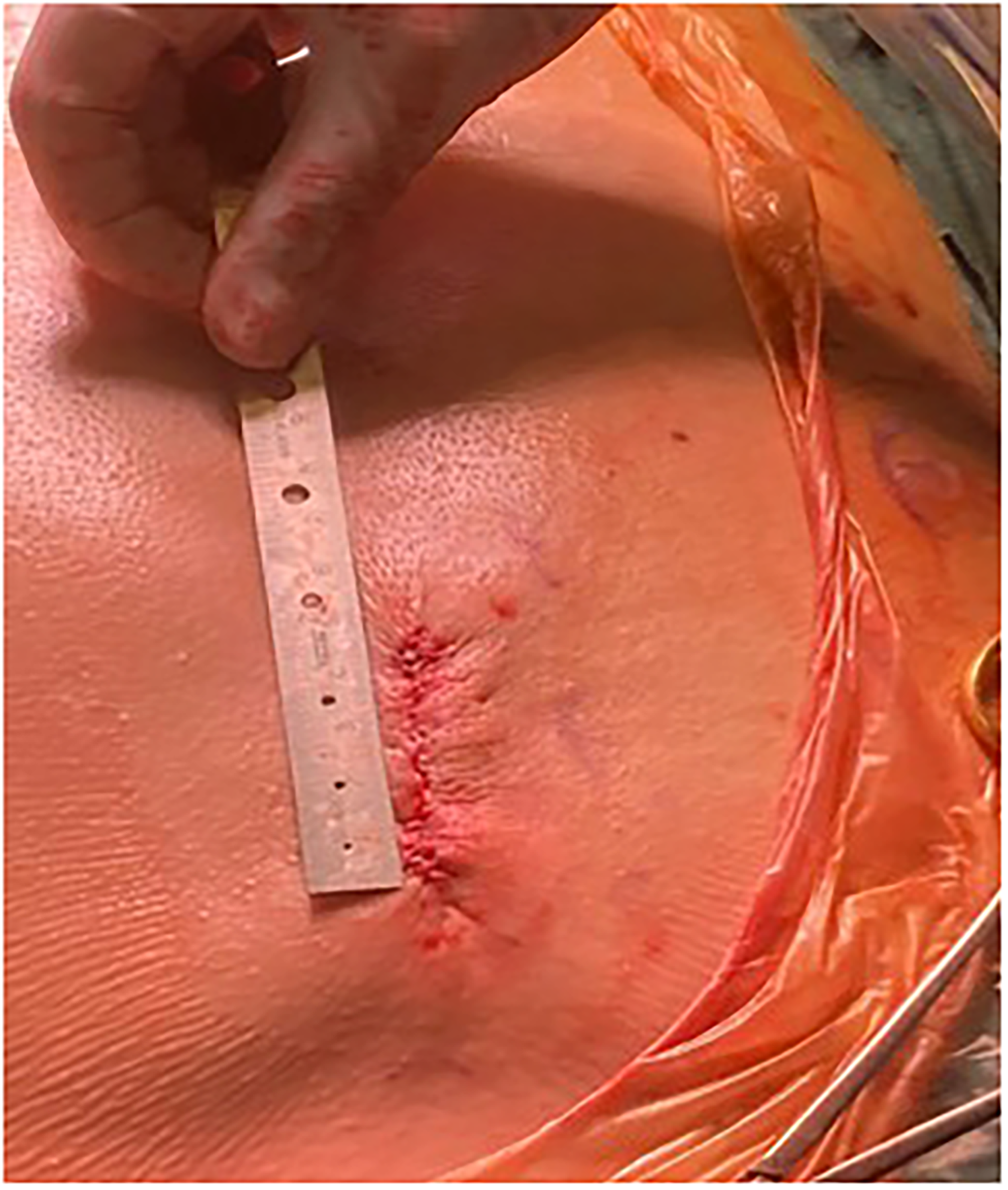
J Ministernotomy Incision at the 4th Intercostal Space for Pulmonary Valve Replacement.
Statistical Analysis
Data were summarized as absolute counts and percentages if related to categorical items and by median, interquartile range (IQR), and range for quantitative variables. The association between the presence of dysfunction (primary endpoint) and categorical factors was assessed through the chi-square test, while the difference in distribution of quantitative variables between patients with or without dysfunction was measured by the nonparametric Mann–Whitney U test. All analyses were performed using IBM-SPSS v.28.0 software.
Results
Our population study included 45 patients: 32 males and 13 females. The most common primary diagnosis was Tetralogy of Fallot (TOF) in 28 patients, followed by valvular/supravalvular pulmonary stenosis (PS) in 10 patients, pulmonary atresia with intact ventricular septum (PAIVS) in 3 patients, and pulmonary atresia with ventricular septal defect (PA/VSD) in another 2 patients; the remaining 2 patients were diagnosed with Fallot canal and transposition of the great arteries, respectively (Table 1). Indication for surgery was severe pulmonary regurgitation with severe RV dilatation in all patients, with a median preoperative RVEDV of 164 (151–174) mL/m2. The median age and weight at surgery were 18 years (IQR 15–30) and 62 kg (IQR 47–72). Associated surgical procedures were performed on 19 patients and included pulmonary artery reconstruction (6 patients), residual atrial septal defect closure (6 patients), tricuspid valve repair (4 patients), pacemaker replacement (1 patient), residual ventricular septal defect closure (1 patient), and innominate artery reimplantation (1 patient). The most common size of the IRV implanted was 25 mm (31 patients, 68.9%), followed by 27 mm (11 patients, 24.4%) and 23 mm (3 patients, 6.7%). In 31 patients, implantation was performed on a beating heart, whereas in 14 patients, the aorta was cross-clamped. The mean CPB and cross-clamp time (when necessary) were 90 min (IQR 63–118) and 35 min (IQR 22–85), respectively. All patients, except for 5, underwent at least 1 previous sternotomy. At the median follow-up time of 33 months (IQR 18–45), no deaths or IRV endocarditis occurred, and 6 patients (13.3%) developed bioprosthesis degeneration (BD). Among these, 1 patient, after 62 months, required a percutaneous ViV procedure for severe bioprosthesis stenosis and bioprosthesis regurgitation (BR); 1 child developed severe BR 15 months postsurgery, and 4 children presented moderate BR at 33, 73, 31, and 19 months following surgery. All patients who developed BD underwent implantation of a 25 mm IRV. No valve thrombosis episodes were detected. All follow-up data are reported in Table 2. Our univariable analysis did not show significant associations between gender and the size of the bioprosthesis or IRV dysfunction. Additionally, age at surgery is not a predictive factor for bioprosthesis degeneration (Table 3). BR, primary diagnosis, and tricuspid regurgitation at follow-up are associated with IRV dysfunction (P < 0.001, P < 0.001, and P = 0.024, respectively). Furthermore, bioprosthesis peak gradient and right ventricular systolic pressure (RVSP) are linked to an increased risk of IRV dysfunction (P < 0.001) (Table 4).
Preoperative and Intraoperative Data.
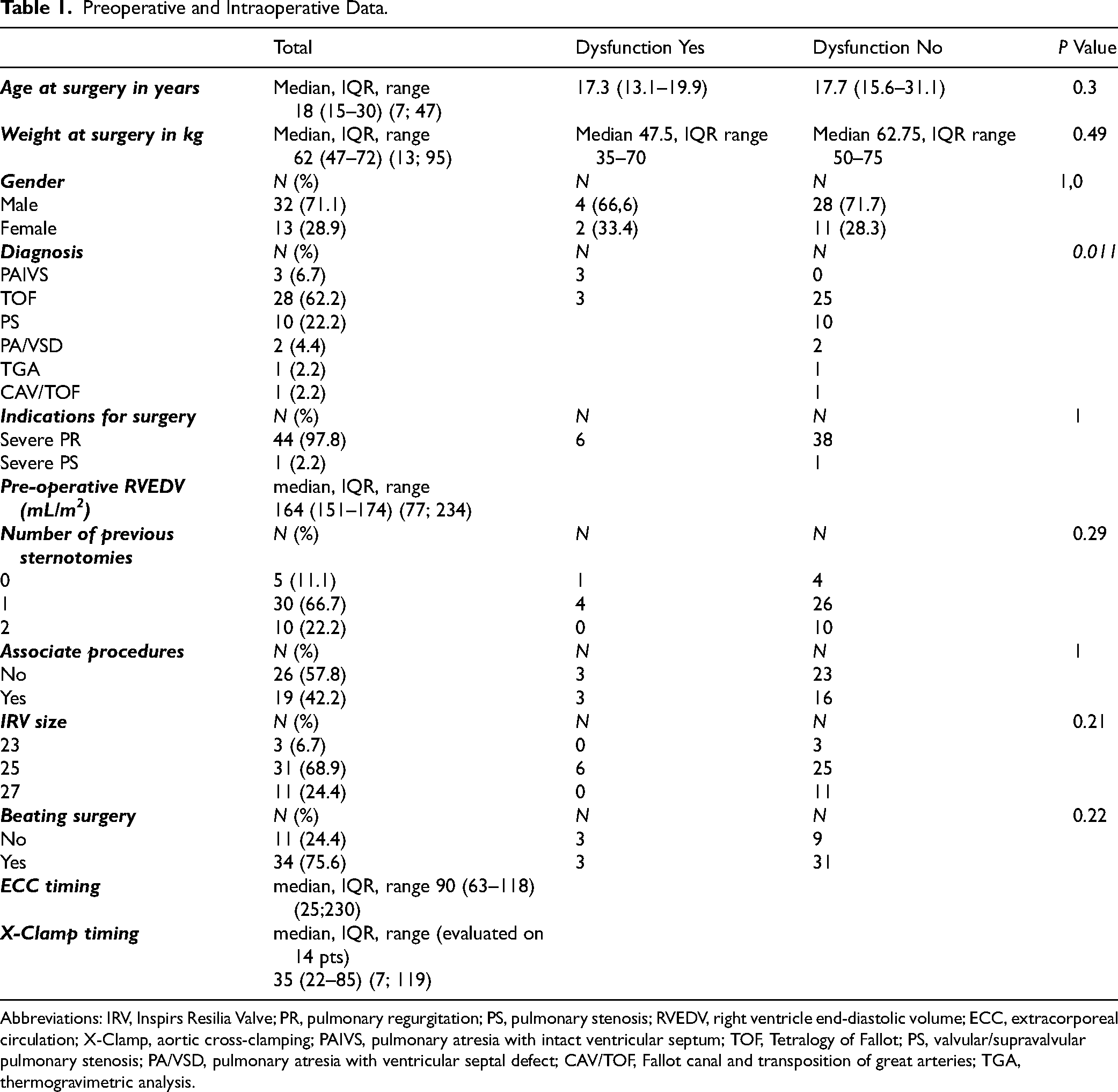
Abbreviations: IRV, Inspirs Resilia Valve; PR, pulmonary regurgitation; PS, pulmonary stenosis; RVEDV, right ventricle end-diastolic volume; ECC, extracorporeal circulation; X-Clamp, aortic cross-clamping; PAIVS, pulmonary atresia with intact ventricular septum; TOF, Tetralogy of Fallot; PS, valvular/supravalvular pulmonary stenosis; PA/VSD, pulmonary atresia with ventricular septal defect; CAV/TOF, Fallot canal and transposition of great arteries; TGA, thermogravimetric analysis.
Follow-up Data.
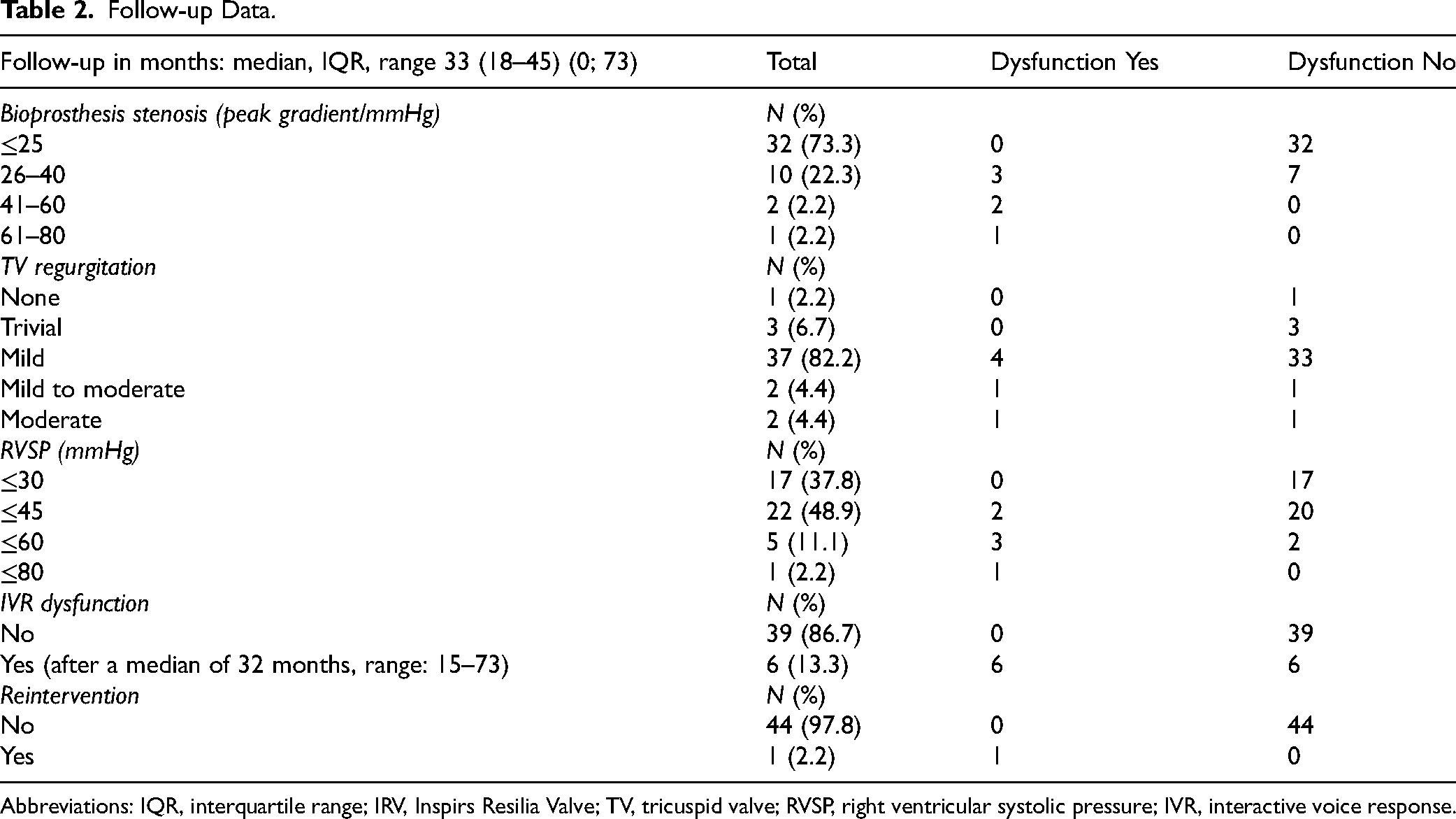
Abbreviations: IQR, interquartile range; IRV, Inspirs Resilia Valve; TV, tricuspid valve; RVSP, right ventricular systolic pressure; IVR, interactive voice response.
Risks Factors for Dysfunction.
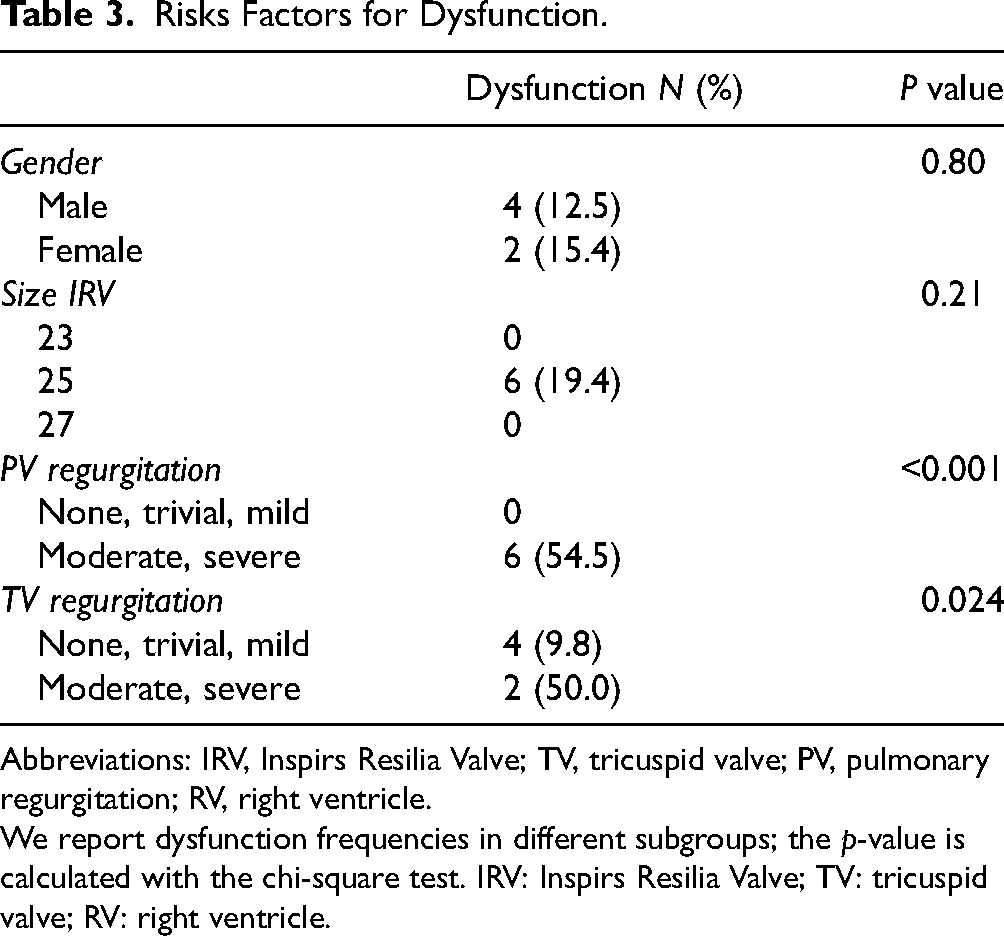
Abbreviations: IRV, Inspirs Resilia Valve; TV, tricuspid valve; PV, pulmonary regurgitation; RV, right ventricle.
We report dysfunction frequencies in different subgroups; the p-value is calculated with the chi-square test. IRV: Inspirs Resilia Valve; TV: tricuspid valve; RV: right ventricle.
Statistical Analysis

Abbreviations: IQR, interquartile range; RVEDV, right ventricular end-diastolic volume; RVSP, right ventricular systolic pressure.
We report median values and interquartile range of 2 groups: dysfunction YES and dysfunction NO; the P value is calculated with the Mann-Whitney test.
Discussion
Currently, valve selection remains dependent on the surgeon or institution. The choice of pulmonary bioprostheses often relies on data from adult aortic valve replacement (AVR) studies. The IRV is a relatively new bovine pericardial prosthesis used for surgical AVR, featuring several innovative modifications designed to prevent oxidation and calcifications, facilitate dry storage, and enhance prosthesis durability. 4 The Inspiris valve is secured with overlapping cobalt chromium alloy bands and a polyester shrink sleeve, allowing the valve frame to expand with balloon valvuloplasty to enable future valve-in-valve replacement without compromising the effective orifice area. 5 This bioprosthesis has recently been adopted for PVR, with several groups reporting their experiences with contradictory results. Some recent studies involving a small number of patients (6–21 patients) indicated good outcomes with the IRV in the pulmonary position, showing no evidence of dysfunction in short-term follow-up.15,16 In contrast, other groups have started to highlight early concerns about outcomes with this device. Said et al 17 found that BR had progressed in 48% of patients at a mean follow-up of 16 ± 8 months Ragheb et al 9 compared outcomes of IRV and mosaic valves and discovered that significant BR was markedly more common after PVR with IRV. In our population study, the rate of patients who developed BD (13.3%) is noteworthy, particularly considering the lack of follow-up time; thus, according to a recent report, analysis of risk factors and potential mechanisms for this development is warranted. Our statistical analysis indicated a correlation between BD and BR more than stenosis. This data is intriguing as it suggests that dysfunction may not be strictly related to leaflet degeneration or calcification, reaffirming the excellent bioprosthesis hemodynamic profile already documented when implanted in the aortic position. Several theories can be proposed to explain the early development of BR. Some authors suggest that the relatively flexible frame of IRV may permit distortion during implantation within the unsupported RVOT,7,17 unlike aortic implantation in the supported left ventricular outflow tract, which can explain the favorable results of IRV in AVR. Indeed, the aortic annulus resides within the fibrous skeleton of the heart, whereas the pulmonary root lacks a fibrous annulus and originates directly from the RV infundibulum. Additionally, the pulmonary root is a highly distensible structure, with its external diameter shown to increase by a mean of 33% under normal RV pressures. 18 Thus, in cases of elevated RV pressure (as seen with RVOT obstruction), the absence of a rigid annulus to support the expandable IRV stent frame may lead to an increase in the internal pulmonary annulus, resulting in leaflet malcoaptation. Our statistical analysis indeed revealed a correlation between BD and high preoperative RV pressure. Therefore, it could be speculated, as previously reported, that the intended benefit of an expandable support structure of IRV may contribute to long-term instability, leading to PVR failure. 19 Expandable VFit technology is available in valve sizes 19 to 25 mm, and all of our patients who experienced dysfunction were implanted with the 25 mm Inspiris Resilia, while no failures have been noted thus far in patients with the IRV 27 size (10 patients). Although these results are not statistically significant, they align with findings from other studies.17,20 According to Said et al, 17 we can speculate that rapid geometric RV remodeling post-PVR led to changes in the prosthesis sewing ring over time, resulting in restricted leaflet mobility and the subsequent development of significant BR. Furthermore, Knox et al 20 suggest that valve angulation during implantation via a minimally invasive approach (left anterior mini-thoracotomy) might cause prosthetic distortion; however, this explanation does not clarify the early failures in cases of full sternotomy implantation, as observed in our series. From a surgical point of view, we implanted IRV only by continuous running suture and, despite potential benefits reported in other studies with interrupted suture, we don’t use this approach. Another proposed failure mechanism relates to the IRV leaflets’ function in pulmonary circulation. 7 These leaflets are thicker than those of other bioprostheses, which may result from the multiple chemical treatments utilized to minimize calcification. While this characteristic might be inconsequential in stable, high-pressure aortic circulation, the lower pressure in pulmonary circulation may render it insufficient to adequately close the prosthetic leaflets, especially following the normalization of right-sided pressures during initial follow-up. According to this idea, all our IRV regurgitation is central and not peripheral. Although not used in our cohort, oral anticoagulation for 3 months postoperatively, as recommended by some guidelines, is used to prevent thrombosis or dysfunction. Indefinite antiplatelet therapy may reduce subclinical thrombosis, and this merits further study. Lastly, despite the well-documented utility of Inspiris valve frame enlargement in AVR, transcatheter PVR is presently associated with limited case reports.7,21 One patient in our series underwent transcatheter valve implantation without frame enlargement but through stent fracture, similar to other bioprostheses. It may be hypothesized that early regurgitation of the IRV results from a combination of factors, including valve design and the anatomic and physiologic characteristics of the pulmonary root. Further discussion and collaboration, along with the use of advanced cross-sectional imaging, are needed to better evaluate the performance of this prosthesis in the pulmonary position on a larger scale and over a longer follow-up period. More data is crucial to develop innovative approaches in terms of tissue engineering and/or identify preventive treatments to mitigate structural bioprosthetic valve degeneration development and progression, and, in turn, significantly improve biological valve durability according to Sallè et al. 22
Limitations
Limitations of this study include its retrospective methodology and short duration of follow-up due to the relatively recent adoption of the Inspiris prosthesis. Our theories about the mechanisms of IRV failure remain theoretical, as we have not yet had any explanted Inspiris valve specimens for pathologic analysis.
Conclusions
Based on our mid-term results, we have currently suspended the use of the IRV for routine PVR. Our preferred choice for surgical PVR now includes the Epic Max prosthesis valve (Abbott, Santa Clara, Calif). Our study included a relatively small number of patients with short- to mid-term follow-up and should therefore be considered preliminary. However, our findings raise concerns regarding the durability of the IRV in the pulmonary position.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Italian Ministry of Health with “Current Research funds.”
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
