Abstract

Prosthesis-patient mismatch (PPM) is classified as a non-structural valve dysfunction according to the Valve Academic Research Consortium 3 standardized definitions, and can be characterized as moderate or severe depending on the ratio of bioprosthetic effective orifice area to patient body surface area (EOA/m2). 1 Although the PPM definition is well established, some studies used different criteria or did not adjust the cut-offs in obese patients, leading to bias and preventing comparison between studies. Even when the standardized definitions are used, there is still a difference in the method used to define PPM: the measured or the predicted method. 2 The predicted method (PPMP) was used in the vast majority of surgical aortic valve replacement (SAVR) cohorts and meta-analyses, 3 while the measured method (PPMM) was used in the largest transcatheter (TAVR) cohorts, 4 leading to conflicting results on the incidence and clinical impact of PPM. These discrepancies may be further exacerbated in population at high risk for PPM (eg small annulus). In the present article, focusing on women with small aortic annulus, Fadel and al. 5 nicely demonstrated that both the method used to define PPM and the cut-off adjustment in obese patients had an impact on its incidence in the TAVR and SAVR cohorts. Therefore, the incidence of moderate-to-severe predicted PPM in both cohorts was divided by two after adjustment for BMI (SAVR: 53% to 25%; TAVR 39% to 19%), and the incidence of moderate-to-severe BMI-adjusted PPM was considerably reduced by applying the predicted method in the SAVR cohort (64% by PPMM vs 25% by PPMP, Figure 1A). Unfortunately, the clinical implications of these findings were not investigated.
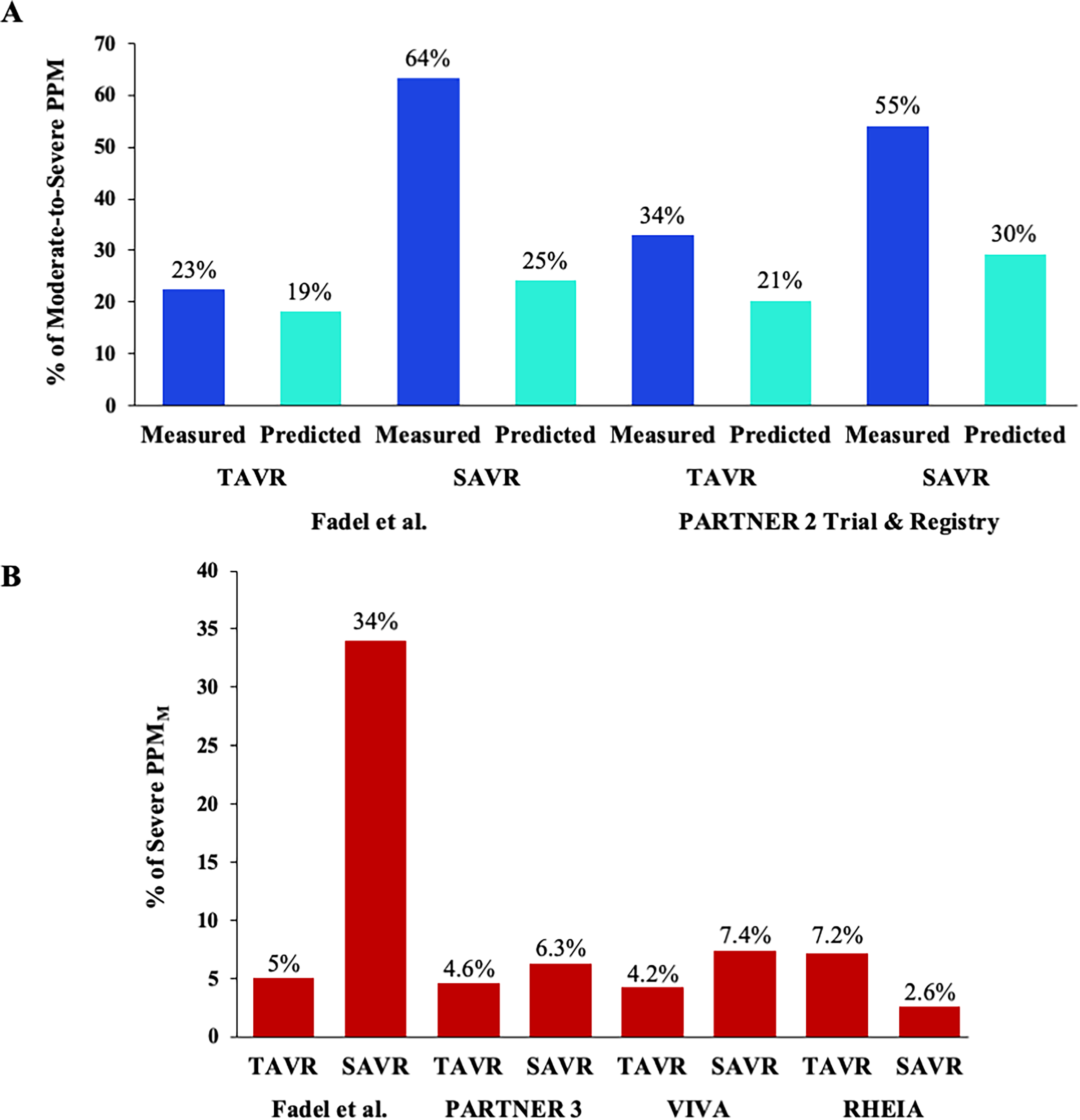
Percentage of prosthesis-patient mismatch in transcatheter versus surgical aortic valve replacement.
Measured or Predicted Method, Does it Matter?
The predicted method uses published reference values for EOA derived from core laboratory analyses of a large number of patients with normally functioning valves, 6 whereas the measured method uses the patient's EOA and left ventricular ejection flow obtained by echocardiography. The predicted method is not influenced by patient flow status or measurement errors, thus avoiding overestimation of severe PPM. The predicted method is thus more specific to the intrinsic hemodynamic performance of the prosthetic valve, and its impact on outcomes does not account for the confounding effect of a low-flow state. In contrast, the measured method captures the prognostic impact of both true PPM and low-flow state. Hence, the measured PPM also account for the presence of low flow (including pseudo-severe PPM) and associated comorbidities. Given that a low flow state can be observed in up to 70% and 90% of patients after TAVR and SAVR respectively, 2 the predicted method should be used as an adjunct to the measured method to better determine the true incidence and impact of PPM. In addition, up to 40% of patients undergoing AVR are obese, which may lead to overestimate the PPM incidence if specific thresholds are not applied. Consistent with our previously published study from the PARTNER 2 Trial and Registry, Fadel et al observed a lower proportion of PPM in SAVR and TAVR patients when PPM was adjusted for obesity, and a higher incidence of BMI-adjusted PPMM than PPMP in SAVR and TAVR patients (Figure 1A).2,5
Which Populations are at High Risk of PPM After AVR?
In contemporary cohorts, the incidence of true severe PPM (ie severe predicted PPM) after AVR in native aortic stenosis (AS) is low (<1% after TAVR and <5% after SAVR) and is only associated with worse outcomes after SAVR (as it is virtually absent after TAVR). 2 However, PPM remains a major concern in high-risk populations, including patients with small aortic annulus or failed bioprosthetic valve, as shown by the studies and randomized controlled trials (RCTs) published in recent years.7–9 Women are also considered to be at high risk for PPM, as up to 75% of them have a small aortic annulus (ie < 430 mm2). The recently presented results of the RHEIA RCT (Randomized researcH in womEn All Comers With Aortic Stenosis, NCT04160130), comparing TAVR and SAVR in women, confirmed the interest in investigating prosthetic valve performance in populations at high risk of non-structural valve dysfunction (ie PPM and paravalvular leak). The study by Fadel et al is therefore up to date, as it only involved women with small annuli. 5
TAVR Versus SAVR in Populations at High Risk of PPM
As the incidence of PPM is higher after SAVR compared with TAVR, the debate about the optimal therapeutic option in populations at high risk for PPM is therefore justified. In the VIVA trial (NCT03383445), 9 the authors compared TAVR versus SAVR in patients with severe AS and small aortic annulus (ie < 23 mm). Interestingly, the incidence of severe PPMM was low in both arms (4.2% after TAVR and 7.4% after SAVR) and no association with clinical outcomes was observed (Figure 1B). In women, the RHEIA RCT also confirmed the excellent hemodynamic valve performance after TAVR and SAVR, consistent with PARTNER 3 results, 10 with a severe PPMM incidence of 7.2% and 2.6%, respectively (Figure 1B). In the present study, Fadel et al reported a higher incidence of severe PPMM after SAVR but not in TAVR (34% vs 5%, Figure 1B). This intriguing result in SAVR arm may be explained by the population selection, which only included women receiving small valves (≤21 mm). Unfortunately, the incidence of severe predicted PPM was not reported and the clinical impact of severe PPM (regardless of method) was not investigated. 5
TAVR in Populations at High Risk of PPM: Balloon- or Self-Expandable Valve?
Recently published studies reported a high prevalence of high residual transvalvular mean gradient and severe PPMM after TAVR in the small native annulus and after valve-in-valve in small failed bioprostheses, especially when using a balloon-expandable valve (BEV).7,8 In the study by Fadel et al, all women received a small BEV (≤23 mm) during the TAVR procedure. Reassuringly, the transvalvular mean gradient (14 mm Hg vs 15.7 mm Hg) and severe PPMM incidence (5% vs 7.1%) were like those reported in the BEV arm of the SMART trial. 5 For completeness, the authors could have provided the mean gradient for each level of PPM severity in SAVR and TAVR cohorts. However, some studies also reported a discrepancy between invasive and echocardiographic assessment of bioprosthetic hemodynamics. Indeed, Doppler-echocardiography overestimated residual gradients and the rate of severe PPM, to a greater extent in small BEV. This discrepancy may therefore explain why the difference in the incidence of PPM between BEV and self-expandable valve was not associated with clinical outcomes.
Populations at High Risk of PPM: The Unknown Future
The presence of a small annulus, severe PPM and a high residual transvalvular gradient induce leaflet shear stress and is therefore associated with accelerated structural valve deterioration (SVD). 11 PPM is also associated with the risk of transcatheter aortic valve thrombosis, 12 which is in turn a risk factor for SVD. Finally, shear stress-induced damage to the leaflets can also lead to infective endocarditis. Given the possible association between small annulus related PPM with structural and non-structural valve dysfunctions, further studies with longer follow-up are needed in these high-risk populations to determine the PPM impact on valve durability and clinical outcomes.
In conclusion, the incidence of severe PPM after AVR is highly dependent of the method applied to determine it. The incidence of true severe PPM is low in contemporary practice, even in high risk population for hemodynamic valve failure such as women with small annulus (<10%). No association between PPM and mid-term outcomes was observed in these high risk populations, but a longer follow-up is necessary to determine its clinical impact.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J. Ternacle is consultant for Edwards Lifesciences, Abbott Structural, General Electric and Philips Healthcare. A. Theron is consultant for Edwards Lifesciences and Medtronic. A. Bernard is consultant for Edwards Lifesciences, Abbott Structural, Medtronic, General Electric and Philips Healthcare.
