Abstract
Background:
Continuous subcutaneous insulin infusion (CSII) therapy aims to enhance diabetes control and management. This study explored the experiences of children/adolescents using CSII therapy and their caregivers.
Methodology:
A mixed-method study was conducted using a basic qualitative descriptive design alongside quantitative methodology. The study involved four children/adolescents with type 1 diabetes mellitus undergoing CSII therapy, as well as four family caregivers. Clinical data, including hemoglobin A1c (HbA1c), time in range (TIR) and diabetic ketoacidosis (DKA), were collected at baseline and 12 months after CSII therapy. Semi-structured interviews were conducted to explore reasons for transitioning to CSII, its impact on diabetes management, its benefits, received support/training, difficulties/challenges in use, and confidence in the technology, with the data analyzed using content analysis.
Results:
HbA1c levels decreased, and TIR improved after 12 months of CSII compared to baseline. All participants had DKA in the year before therapy, but none experienced it during the 12 months of its use. The primary reason for switching to CSII was improved glycemic control, with flexibility and quality of life being secondary factors. Caregivers and children/adolescents viewed CSII therapy positively, citing its effectiveness in improving diabetes management, fostering independence of children/adolescents in managing their diabetes, and enhancing flexibility in outings and meals. Initial challenges with pump operation were temporary, with caregivers appreciating training and support from the manufacturer.
Conclusions:
Results suggest that participants perceived CSII positively, regarding improvements in diabetes management, sleep quality, and lifestyle flexibility.
Keywords
Introduction
The rising prevalence of type 1 diabetes mellitus (T1DM) among adolescents and young adults presents a significant global health care challenge, with the Global Burden of Disease Study (2019) reporting an increase in incidence from 7.78 per 100,000 population in 1990 to 11.07 per 100,000 population in 2019. 1 Saudi Arabia ranks among the highest globally for T1DM incidence, with rates in children and adolescents reaching 109.5 per 100,000, surpassing those of many developed countries.2,3 Complications from diabetes, including macrovascular and microvascular complications, are responsible for much of the disease burden among T1DM patients. 4 To prevent these complications and allow patients to lead as normal a life as possible, managing T1DM requires intensive management aimed at maintaining near-normal blood glucose levels. 5 Multiple daily injections (MDIs) have traditionally been the standard of care for patients with type 1 diabetes, but they are associated with a high risk of severe hypoglycemia and can be particularly challenging for patients needing very small doses, such as children, due to limitations in the accuracy and practicality of current insulin pens. 6 Continuous subcutaneous insulin infusion (CSII) offers a method to achieve intensive insulin therapy without increasing the risk of hypoglycemia by enabling patients to program, adjust, or pause insulin infusion rates and also to deliver precise insulin doses.7,8
The benefits of CSII include a reduced risk of hypoglycemia and severe hypoglycemia, improved glycemic control, and minimized blood glucose fluctuations that contribute to the risk of microvascular and macrovascular complications.7,8 CSII therapy also enhances the quality of life (QoL) for both patients and their families. 9 Many studies conducted in Saudi Arabia have documented the positive effects of CSII therapy on glycemic control.2,3,10 They have also shown increased patient satisfaction with treatment2,3 and improvements in overall QoL. 11 However, there is a lack of data regarding the experiences of children/adolescents using CSII or their parents through qualitative methods. Gaining an understanding of these aspects provides a foundation for a holistic approach to optimizing care by delivering tailored support and guidance in family-centered services. Also, providing information on this topic within the context of Saudi Arabia, with its unique cultural and family structures, will contribute valuable insights to the global body of literature. Therefore, the aim of this study was to explore the experiences of children/adolescents using CSII therapy for the management of T1DM and their family caregivers. Specifically, we sought to understand the reasons behind the transition to CSII therapy, its effect on diabetes management routine, its benefits, the received support/training, the difficulties/challenges associated with its use, and their confidence in the technology.
Methods
Study design
We conducted a mixed-method study that combined qualitative and quantitative methodologies. The qualitative component was a basic descriptive study, which is well-suited for capturing clear and straightforward answers to questions of significant relevance to practitioners and policymakers, without excessive theorization or unnecessary interpretation. 12
Setting and population
In 2021, the Saudi Ministry of Health launched a pilot project to introduce CSII therapy for patients diagnosed with T1DM. The patients were selected for the project based on the eligibility criteria outlined in the Saudi Diabetes Clinical Practice Guidelines by the Saudi Health Council for the use of CSII therapy. As part of the protocol, the factory-calibrated Dexcom G6 continuous glucose monitoring (CGM) system (Dexcom, Inc., San Diego, CA, USA), approved for use in individuals aged ≥2 years, was first applied, and after 14 days, following evaluation of glycemic metrics from the CGM, CSII therapy was initiated. The patients used the insulin pump t:slimx2 with Control-IQ Technology, manufactured by Tandem Diabetes Care, designed for subcutaneous insulin delivery at fixed and variable rates. Using Control-IQ technology, an automated insulin dosing system, the pump automatically adjusts basal insulin delivery based on real-time glucose readings and predictions. This technology works with integrated continuous glucose monitors and alternate controller-enabled pumps. The insulin pumps, along with patient education and monitoring, were provided by VitalAire Arabia, the Saudi branch of the international VitalAire brand. The patients selected for this project consisted of five male and female children/adolescents aged between 12 and 17 years, diagnosed with T1DM. Four of the five children/adolescents who were using CSII therapy for 24 months, along with the family member responsible for their care and supervision of their treatment, consented to participate in this study (Fig. 1).
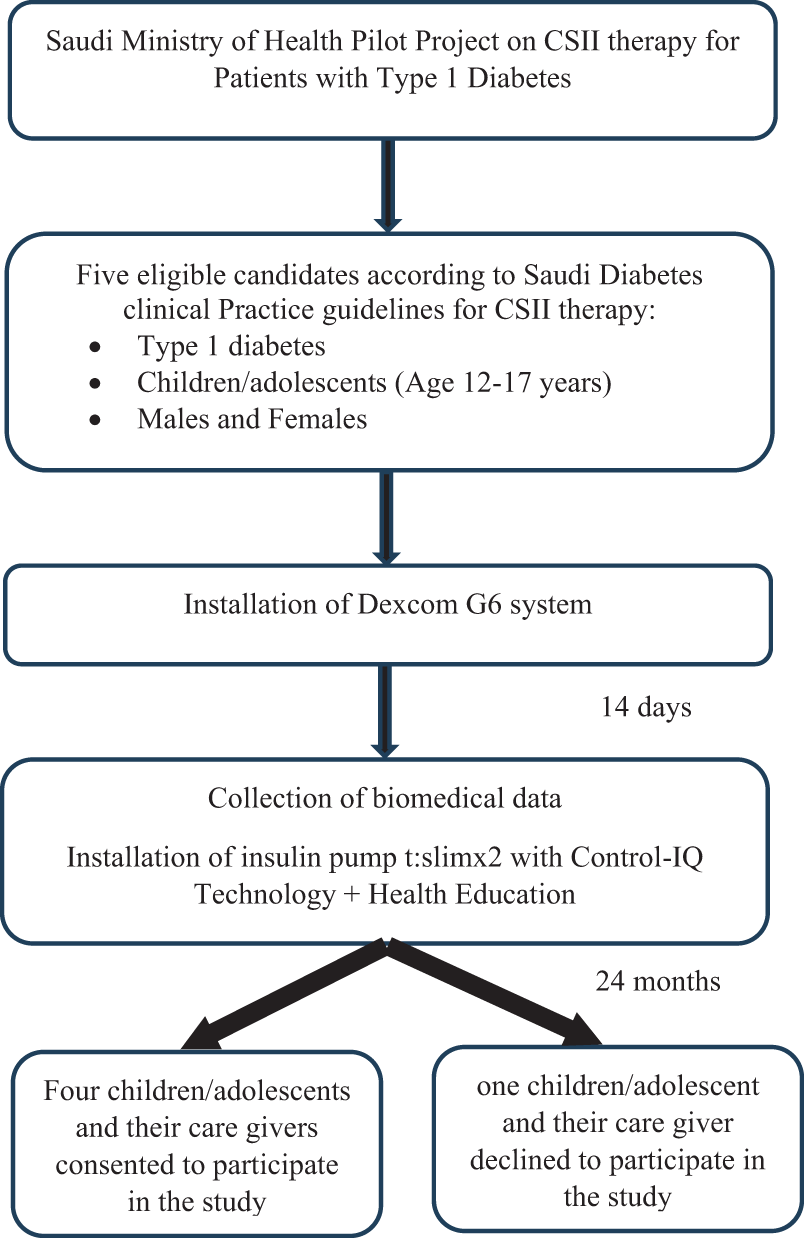
Eligibility criteria and selection of the participants. CSII, continuous subcutaneous insulin infusion.
Data collection
The study employed a mixed-methods approach, incorporating both quantitative and qualitative techniques.
Quantitative sub-study
Clinical data were collected from patient records, including hemoglobin A1c (HbA1c), time in range (TIR), and incidents of diabetic ketoacidosis (DKA). Data collection took place at two time points: before initiating CSII therapy and after 12 months of therapy. TIR was derived from the Dexcom G6 system. Baseline TIR was defined as the percentage of glucose values within the target range of 70–180 mg/dL over the 14 days immediately preceding CSII initiation. All days had data coverage >70 %.
Qualitative sub-study
Children/adolescents and their family caregivers were interviewed using a semi-structured guide that was developed based on themes identified from the literature review. The themes included: reasons for using or transitioning to CSII, effect of CSII therapy on diabetes management routine, benefits of CSII, support and training provided, difficulties associated with CSII therapy use, and confidence in the technology. Prompts were used to encourage participants to share their experiences about CSII. The interviews were conducted in October and November 2024, approximately 24 months after the children/adolescents began using the CSII to allow adequate time for the technology to become integrated into their daily lives. 13 The interviews, lasting 45–60 min, were not audio-recorded but transcribed in full. Informed assent was obtained from the children/adolescents, and informed consent was obtained from their caregivers. The study was approved by the Institutional Review Board at Princess Nourah bint Abdulrahman University (IRB Log Number: 24-0811).
Adolescent assent was obtained following an age-appropriate explanation of the study purpose, procedures, voluntary participation, and the right to withdraw without consequence, in addition to written caregiver consent. Interviews with children/adolescents were conducted privately to promote open expression. All transcripts were anonymized using participant codes, and data were securely stored with access restricted to the research team. Given expressed concerns regarding stigma and disclosure of pump use, particular care was taken to ensure that reported quotations were non-identifiable.
Data analysis
For the quantitative data, we assessed changes in the three clinical indicators at baseline and 12 months after initiating CSII therapy. Due to the small sample (N = 4), the quantitative analyses are descriptive only, with no inferential statistical testing performed. Qualitative analysis was conducted by M.H. and L.A. using qualitative content analysis appropriate for basic descriptive studies. 12 Interviews were documented through detailed field notes, promptly reviewed and cross-checked against the guide and context to ensure accurate interpretation before analysis. The analysis followed three phases: immersion, reduction, and interpretation. 14
During immersion, transcripts were read repeatedly to achieve familiarity with the data. In the reduction phase, coding was performed within a framework informed by literature-derived themes, while allowing inductive identification of subthemes from the data. An iterative coding framework (codebook) was developed, with ongoing refinement of code definitions and category boundaries through constant comparison across transcripts to ensure internal consistency. Coding was conducted manually. The two researchers independently reviewed transcripts and met regularly to compare interpretations; discrepancies were resolved through discussion and consensus, with reference to the original transcripts to maintain data grounding.
Reflexivity was maintained throughout analysis. The researchers critically reflected on how their professional backgrounds and prior knowledge of CSII therapy might shape interpretation and returned to the transcripts to ensure findings reflected participants’ accounts.
In the interpretation phase, codes were organized into subthemes within the predefined thematic framework. Findings were presented as a direct summary of informational content, supported by illustrative participant quotations.
Results
Quantitative sub-study
The four diabetic children/adolescents, aged 12–18 years, included three females and one male. To ensure anonymity, they will be referred to as Child/Adolescent-1, Child/Adolescent-2, Child/Adolescent-3, and Child/Adolescent-4. The clinical data of the children/adolescents before and 12 months after starting CSII therapy are summarized in Table 1. All showed a reduction in HbA1c levels after 12 months of CSII therapy compared to baseline values. Additionally, there was an increase in the percentage of TIR during the 12-month period of CSII therapy compared to the 15- to 30-day period prior to starting therapy for all children/adolescents. Furthermore, while all participants experienced DKA during the 12 months before using the CSII therapy, none experienced DKA during the 12 months of CSII therapy use. Due to the small sample size (N = 4), results are presented descriptively and are not statistically interpretable in terms of inference
Background Information and Clinical Data of the Children/Adolescents at Baseline and 12 Months After Continuous Subcutaneous Insulin Infusion Therapy (n = 4)

HbA1c, hemoglobin A1c; DKA, diabetic ketoacidosis; TIR, time in range.
Qualitative sub-study
Eight respondents, including four children/adolescents and their four caretakers, were interviewed. For three of the children/adolescents, the caregiver responding for the interview were their mothers, while for the fourth, the respondent was a sister. Caregivers will also be referred to as Caregiver-1, Caregiver-2, Caregiver-3, and Caregiver-4. The subthemes identified under the previously identified themes are described below.
Theme-1. Reasons for switching to CSII therapy
Subtheme-1.1. Improving diabetes control
Reasons cited by caregivers for switching from MDI to CSII therapy included persistent irregular blood sugar levels, poor diabetes control reflected by high A1C, and the ease of insulin administration with the pump compared to injections.
The sugar level was not regular and we could not control it. (Caregiver-1)
It is easier to give the insulin by the pump than with the injection. (Caregiver-4)
Theme-2. Effect of CSII therapy on diabetes management routine
Subtheme-2.1. Easier management routine
Caregivers described the impact of CSII therapy on the diabetes management routine as positive. They reported that the overall process of managing diabetes became much easier, with CSII therapy streamlining insulin delivery and management. It also provided greater convenience, allowing caregivers to adjust insulin delivery according to varying situations, such as meals, sleep, or physical activity.
(the pump had) a very positive effect, thank God, … … and controlling the blood sugar level became much better and much easier. (Caregiver-3)
The children/adolescents also described diabetes management as becoming easier with CSII therapy, as it simplified blood sugar level adjustments and control. Specifically, CSII therapy was described as providing convenience in various settings, such as school, home, and social outings, eliminating the need to seek private spaces for injections.
I no longer need to go to the bathroom or an empty place to take an injection, and this is very useful, at school during break, I don’t need to go to the administration and go to the bathroom and waste my time. (Child/Adolescent-3)
Subtheme-2.2. Independence in diabetes management
Caregivers highlighted the independence promoted by CSII therapy, enabling them to manage their diabetes and insulin levels with minimal intervention. This empowerment allowed children/adolescents to take greater control of their condition, reducing the need for constant caregiver involvement.
(the pump facilitated) ease of taking insulin (by him) sometimes without our intervention if the sugar rises, and managing it if he is at school or outside the home. (Caregiver-4)
Children/adolescents also reported feeling more independent in managing their diabetes, requiring less assistance with insulin dose adjustments and blood sugar monitoring compared to using injectable insulin.
(before the pump) I needed help to adjust the insulin pen doses and check my blood sugar. (Child/Adolescent-4)
Theme-3. Benefits of CSII therapy
Subtheme-3.1. Improved diabetes control
The caregivers described CSII therapy as providing excellent control of blood sugar levels, resulting in more stable and predictable readings, leading to improved overall diabetes control.
(After the pump) there is excellent control in the sugar level, and the glycated haemoglobin improved a lot. (Caregiver-1)
The children/adolescents also described the CSII therapy as having a positive impact on their lives, enhancing diabetes control. It was noted for providing better regulation of blood sugar levels, reducing fluctuations, and maintaining readings within target limits.
(before the pump) there were always many highs and lows and readings were always outside the range. (after the pump) even at bedtime my blood sugar is in the range, (there are) no strong highs and lows that cause drowsiness and tiredness all the time. (Child/Adolescent-2)
Subtheme-3.2. Flexible lifestyle
The caregivers highlighted the impact of CSII therapy on their children’s lifestyle, noting it provided greater flexibility and freedom, enabling children/adolescents to go out without preparing insulin pens and accessories, thus making social activities easier to participate in.
(after the pump she can) go out at any time and quickly without having to prepare the insulin pen and the rest of its accessories. (Caregiver-3)
It (the pump) helped and made his life easier, thank God, because before (the pump) we had to help him take insulin and measure sugar, now he goes out with his friends and takes insulin, and measurements are available continuously—his life has become easy, thank God. (Caregiver-4)
The children/adolescents also highlighted the positive impact of CSII therapy on their lifestyle, particularly in terms of flexibility with exercise and food choices. They highlighted features like the exercise mode, which made it easier to engage in physical activities, and the extended insulin delivery system, which allowed for greater dietary flexibility, enabling them to enjoy a variety of food types without worry.
(after the pump) I walk a lot or do simple sports and activate the exercise mode. (Child/Adolescent-4)
… for foods like pizza, baked goods or toast, the extended system helped me eat them without fear and I knew how many carbs I was taking in. (Child/Adolescent-1)
Subtheme-3.3. Sleep quality
Improved sleep quality for the child/adolescent was highlighted as a benefit of CSII therapy, as caregivers no longer needed to wake the child to monitor blood glucose levels.
My daughter sleeps deeply even in times of high (sugar), I can adjust the corrective doses without having to wake her. (Caregiver-1)
The improvement in the sleep quality was also mentioned by children/adolescents due to the assurance that the closed-loop will maintain stable glucose levels.
I can sleep well …. since I do the sleep position. (Child/adolescent-4)
Theme-4. Support and advice for CSII therapy
Subtheme-4.1. Delays in response
While the training and support provided by the manufacturer were generally appreciated, there were issues with delays in responses and supply deliveries, which sometimes required the use of traditional methods like regular blood testing and insulin injections.
It (the support) is somewhat good, but sometimes there is a delay in responding or a delay in sending supplies, so we have to go back to measuring blood and taking insulin with needles for many days and sometimes it reaches a week, and this is very difficult for a child, whether at school or at home. (Caregiver-4)
Subtheme-4.2. Family support
In addition to the support from the manufacturer, the children/adolescents also valued family support, which included reminders, assistance with the pump, and emotional encouragement. Family involvement extended to training and education, helping adolescents learn tasks such as carbohydrate counting and effective pump use.
My sister helps me with it, and brought me a book about counting carbs which I benefited from a lot. (Child/Adolescent-4)
Theme-5. Difficulties/challenges of using CSII therapy
Subtheme-5.1. Transient pump functionality issues
Understanding the pump’s functionality and its effective use for consistent diabetes management was the primary difficulty/challenge faced by caregivers of children/adolescents, particularly during the initial stages of CSII therapy. This included determining appropriate insulin adjustments for different activities, mastering various settings, and developing the skill of calculating carbohydrates. However, they described these challenges as temporary, occurring primarily during the initial phase of CSII use.
(the difficulty was) knowing the correct amount of the exchange and the way of using the different situations like sleep and exercise. Now (I) just use the sleep mode and usual basal mode. (Caregiver-3)
Subtheme-5.2. Overcoming difficulties
Caregivers reported overcoming the difficulties/challenges associated with the pump use through continuous communication with the manufacturer’s support team and the use of educational resources, such as instructional videos, which improved their understanding of the device. Over time, with practice, they learned to adjust insulin levels appropriately, considering factors like illness, the menstrual cycle, or fasting, and became more skilled at accurately calculating carbohydrate intake and adjusting insulin doses.
With practice, I learned the correct amount, and how to increase and decrease according to the changes in the body like in illness and the monthly period and fasting. (Caregiver-3)
Subtheme-5.3. Easy technology
Children/adolescents, on the other hand, cited no difficulties, described the CSII therapy as very easy and comfortable to use, especially after completing proper training.
….On the contrary, using the pump is very easy and comfortable. (Child/Adolescent-1)
(the pump is) very easy, I don’t need to go over the sensor. (Child/Adolescent-3)
Subtheme-5.4. Reluctance to disclose CSII
Some of the children/adolescents preferred to keep their use of the insulin pump private, disclosing it only to close friends while avoiding letting other peers or colleagues know.
None of my friends know about the pump except those close to me because it is a personal matter. (Child/Adolescent-2)
it’s nice that it is Bluetooth, ….(so that) at school, I don’t feel embarrassed in front of my colleagues. (Child/Adolescent-3)
Theme-6. Confidence in the automatic insulin delivery adjustments
Subtheme-6.1. Confidence in technology
Caregivers expressed confidence in the automatic insulin delivery adjustments made by the pump, with no significant issues reported. However, there were occasional instances when manual adjustments were deemed necessary for more precise control.
I feel comfortable, (but) sometimes I need to adjust the doses of the insulin pump by myself. (Caregiver-3)
The children/adolescents also found the automatic adjustments made by the insulin pump reliable, which strengthened their trust in the technology. They felt reassured and confident in the pump’s ability to make necessary corrections, especially during sleep, easing their anxiety about blood sugar control.
I became more reassured that I can sleep well because the pump corrects it. (Child/Adolescent-4)
Discussion
The aim of this study was to describe the experiences and opinions of children/adolescents with T1DM and their caregivers regarding the CSII therapy. The findings suggested that both caregivers and children/adolescents generally viewed CSII positively, regarding diabetes management and daily life.
Reasons for switching to CSII therapy
The primary reason for switching from MDI to CSII therapy, as reported by caregivers, was medical—specifically, the pursuit of improved glycemic control. This finding aligns with previous studies.15,16 HbA1c levels peak among adolescents aged 13–17 years, 17 corresponding to our study population and likely motivating families to adopt advanced technologies to optimize diabetes management. Although improved QoL has been identified as a key driver of CSII adoption in other studies,16,18 it was less influential among caregivers in this cohort.
Effect of CSII therapy on diabetes management routine
In this study, caregivers’ and children/adolescents’ reports suggest that CSII simplified diabetes management. This is particularly relevant during periods of rapid growth and puberty, when insulin requirements fluctuate markedly. 19 Improved management routines and support beyond parental capacity have been widely reported as key advantages of CSII.20,21 Participants in this study also described greater child independence in diabetes care, consistent with previous research showing increased involvement in daily management after CSII initiation.22,23 Adolescents viewed greater responsibility as supportive parental behavior, a factor associated with effective CSII implementation and improved glycemic control. 24
Benefits of CSII therapy
In this study, respondents reported multiple benefits of CSII, particularly improved glycemic stability and diabetes control. These perceptions were consistent with descriptive biomedical indices observed in this cohort, although these findings should be interpreted cautiously. Similar improvements in diabetes control have been reported in other qualitative studies of CSII use.20,22,23
In addition to improved glycemic control, participants in this study reported lifestyle-related benefits of CSII, including greater flexibility in eating and social activities. Consistent with previous studies, this flexibility involved the ability to consume a wider variety of foods,21,25 freedom from fixed meal schedules,18,21,23 reduced injection burden, and fewer supplies to carry. 25 Improved sleep quality was also described, likely reflecting the closed-loop system’s capacity to maintain stable overnight glucose levels.20,21,26 These advantages have been associated with enhanced QoL in other studies. 22 A previous study in Saudi Arabia similarly reported better health-related QoL among children using CSII compared with MDI. 11
Although caregivers in this study recognized the positive impact of CSII on QoL, their focus was primarily on their children rather than themselves. In contrast, other studies report parental benefits, including improved sleep, increased free time, and greater social engagement.20,23–25 Cultural norms may have shaped caregivers’ emphasis on child-centered outcomes over personal gains. Further research is needed to explore the influence of cultural factors on caregivers’ experiences with CSII.
Support and advice for CSII therapy
In this study, despite some concerns about delays in response in some instances, both caregivers and children/adolescents appreciated the training and support provided by the manufacturers. Similar feedback was noted in other studies, where the manufacturers were recognized for offering training and providing technical support. 15 However, there should be a responsive, user-focused support system to address urgent concerns, reduce frustration, and ensure the effective and sustained use of the technology.
Difficulties/challenges of using CSII therapy
Caregivers in this study reported initial technical challenges with CSII use, including pump operation, carbohydrate counting, dose calculation, and basal adjustments, consistent with previous studies. 23 These difficulties were temporary and were reported to improve with practice, training, and ongoing support.23,25,27
Another challenge is that some children/adolescents in this study were hesitant to disclose their insulin pump use, sharing it only with close friends. This reluctance aligns with previous findings, where adolescents feared the pump’s visibility might signal abnormality or provoke stigma.13,18 Such concerns sometimes led to device disconnection or avoiding the handset to prevent public alarms, 13 and in some cases, even discontinuation of CSII use. 28 Strategies are therefore needed, including manufacturer-led interventions, to improve social acceptance and understanding of CSII therapy.17,29
Confidence in the automatic insulin delivery adjustments
Despite initial challenges, caregivers and children/adolescents in this study expressed strong confidence in the insulin pump’s ability to regulate blood glucose. Consistent with previous studies, this trust enabled parents to feel assured their child could sleep safely and participate in unsupervised activities,13,24,27,30 while adolescents could engage socially without fear of hyperglycemia or embarrassment. 13 The sustained positive perception of CSII after 2 years likely reflects direct experience transitioning from MDI, aligning with studies showing higher treatment satisfaction and clear preference for CSII over MDI.18,28
A major limitation of this study is the small sample size, which was determined pragmatically due to the limited availability of eligible participants. Such practical considerations have been cited as a justification for small sample sizes in qualitative research. 31 Qualitative experts note that sample size is often a pragmatic decision shaped by the resources and specific populations available to the researcher. 32 Furthermore, although data saturation was not reached, the adequacy of this study should be evaluated based on its “information power” and the richness of individual accounts, rather than solely on numerical counts or the achievement of saturation.31,32 Another limitation was that participants were drawn from a pilot Ministry of Health program requiring caregiver consent, which may have introduced selection bias and limited generalizability to less engaged families or other socioeconomic groups.
Conclusion
Among our respondents, the primary reason for switching to CSII therapy was improved glycemic control, while flexibility and QoL were less significant factors for caregivers. Caregivers’ and children/adolescents’ reports suggest a positive view of CSII, particularly regarding its effectiveness in stabilizing blood sugar levels, improving diabetes management routine, improving sleeping quality, and enhancing the lifestyle by providing flexibility in outings and eating for children/adolescents. Although caregivers initially encountered challenges in operating the CSII pump, these difficulties were temporary, and they valued the training and support provided by the manufacturer. Some children and adolescents continued to experience feelings of embarrassment and hesitancy to reveal the use of the pump to others.
Authors’ Contributions
All authors contributed to the concept and design of the data. L.A. and M.H. performed the analysis and interpretation of results and drafted the article. All the authors reviewed the article before submission and take full accountability for all aspects of the work.
Footnotes
Author Disclosure Statement
Two of the authors, F.M. and D.F., are employed by VitalAire Arabia, the company that supplies the insulin pumps and provides education to patients and their families on how to use them. The remaining authors declare no conflicts of interest.
Funding Information
No funding was received for this article.
