Abstract
Background:
Optimal insulin titration is essential in helping people with type 2 diabetes mellitus (T2DM) to achieve adequate glycemic control. Barriers of people with diabetes to implementation of titration include lack of self-efficiency and self-management skills, increased diabetes-related distress, low treatment satisfaction, poor well-being, as well as concerns about hypoglycemia and insulin overdose. My Dose Coach is a digital health tool for optimizing titration of basal insulin that combines a smartphone app for patients with T2DM and a Web portal for healthcare professionals.
Methods/Design:
This is a prospective, open-label, multicenter, randomized controlled parallel study conducted in approximately 50 centers in Germany that are specialized in the treatment of diabetes. Patients in the intervention group will use the titration app and will be registered on the Web portal by their treating physician. Control group patients will continue their current basal insulin titration without using the app. The primary outcome is the mean change in HbA1c levels at the 12-week follow-up. The secondary outcome measures include patient-reported outcomes such as diabetes-related distress, self-management, empowerment, self-efficacy, treatment satisfaction, and psychological well-being as well as fasting blood glucose values.
Conclusion:
This digital health tool has been previously implemented in several independent pilot studies. The findings from this multicenter randomized controlled trial can provide further evidence supporting the effectiveness of this tool in patients with T2DM and serve as a basis for its clinical integration.
Trial Registration:
German Register for Clinical Studies-DRKS-ID: DRKS00024861
Background
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder characterized by persistently elevated blood glucose levels (hyperglycemia) due to progressive failure in insulin production or function. The management of T2DM focuses on controlling glycemic levels and preventing further complications. 1 When lifestyle modifications, such as dietary changes, weight reduction, or physical activity, and oral medication fail to provide adequate glycemic control, the addition of basal insulin is often sufficient to attain adequate glycemic control. 2 After starting the therapy, an essential task of the treating physician and the person with diabetes is to titrate the dose of basal insulin to achieve the agreed individual HbA1c target value. Consequently, the dose of basal insulin must be adapted to the fasting blood glucose value. In the titration phase, the individual fasting glucose target of the participants should be gradually achieved and subsequently maintained. 3
Nevertheless, it has been shown in many cases that the treatment goals are not achieved in everyday clinical practice and titration is either not performed at all or not often enough. In a retrospective longitudinal analysis of six countries, Mauricio et al 4 found that in 2330 patients in Germany who received basal insulin therapy, an average HbA1c upon therapy initiation was 8.5%, with 81.9% of patients having an HbA1c > 7%, and as many as 29.8% an HbA1c > 9.0%, despite the recommendation for titration. 4 In another study involving 11 696 patients from the UK registry evaluating the real-world scenario of T2DM management, only 36.5% had their treatment intensified during the study period and the median time from initiation of basal insulin to treatment intensification was 4.3 years. Among patients clinically eligible for treatment intensification (HbA1c ≥ 7.5%), only 30.9% had their treatment regimen intensified and the median time to intensification was 3.7 years. 5
A common reason for nonachievement of glycemic targets in people with type 2 diabetes who are treated with basal insulin in addition to oral antidiabetic medication is often inadequate titration of the basal insulin dose. The delay in titration can be due to various reasons. Basal insulin titration can be seen as a complex process that requires the following: (1) regular measurement of fasting blood glucose; (2) documentation of the measured values; (3) acknowledgment of the insulin dose adjustment (in case of three consecutive increased fasting blood glucose values); (4) calculation of the mean or median glucose value; (5) availability of the titration chart; (6) understanding and correct application of the titration scheme; and (7) modification of the insulin dose and its injection. Although the treating physician determines the dose of required insulin and creates a titration scheme, the affected person with diabetes is responsible for measuring his or her own blood glucose concentration and injecting the correct insulin dose. If a problem occurs at any stage of the titration process, the basal insulin therapy will fail. In addition, real or perceived risk of hypoglycemia6 -9 is an additional barrier for effective titration of basal insulin doses. Thus, possible problems with the implementation of titration in daily life range from simple forgetfulness and poor arithmetic skills (eg, calculation of mean value) to psychological factors such as lack of self-efficacy or self-management skills, increased diabetes-related distress, low treatment satisfaction, and poor well-being.10 -16
Digital applications such as insulin titration apps may have the potential to overcome these barriers. The large multicenter Take Control trial shows that participants with digitally assisted self-titration had better diabetes control in terms of HbA1c lowering than adults titrated by a physician alone, with similar participant-reported overall outcomes, clinically relevant reductions in emotional distress, and hypoglycemic rates. 17 The AUTOMATIX study compared the glycemic effects of an insulin titration app with routine insulin titration by the physician. No significant advantage of app-assisted titration over routine titration was demonstrated. The percentage of participants who met their titration goals was slightly lower in the app titration group. Similar results were obtained with respect to fasting blood glucose reduction. This study showed noninferiority of app titration compared with routine titration, but not superiority, although this study was conducted as a noninferiority study with a larger sample than required for a superiority study. 18
Bergenstal et al 19 demonstrated that an insulin titration app, “d-Nav,” significantly reduced A1c by 1.0 percentage point, while routine titration reduced A1c by 0.3 percentage point. However, the percentage of low glucose values (>53 mg/dL and <70 mg/dL) was also significantly increased. These results suggest that average glucose levels were reduced by the titration app, but at the expense of a higher exposure to low glucose levels.
In summary, the current evidence for the efficacy of insulin titration apps is limited due to different study designs (noninferiority and superiority studies) and conflicting results regarding their impact on hypoglycemic values.
My Dose Coach is a digital health tool (Web portal for clinicians and app for patients) designed to help people with type 2 diabetes who have just started basal insulin therapy as well as those with type 2 diabetes who are on basal insulin therapy and not meeting their blood glucose targets, to overcome perceived barriers and achieve their individual titration goals. Besides titration of insulin doses, this digital health software also addressed prevention of low glucose values by offering different options to respond to low glucose values during insulin titration.
This health tool has already been implemented in India and Mexico and evaluated in independent pilot studies.20 -22 The results from these studies demonstrated that the use of this digital health tool successfully decreased HbA1c levels, achieved easier target fasting blood glucose levels, and improved overall well-being without any severe hypoglycemic events.20 -22
The aim of this multicenter randomized controlled study is to investigate the effectiveness of using this digital health tool in improving glycemic control as a medical benefit and patient-relevant structural and procedural improvements, as assessed by patient-reported outcomes via the implementation of the physician-determined titration algorithm in patients’ daily lives within the German healthcare system.
Methods
Design of the Study
This is a prospective, open-label, multicenter, randomized controlled parallel study conducted in approximately 50 centers specialized in diabetes. Figure 1 provides a detailed overview of the study design. Financial sponsor of this study is Sanofi Aventis Germany. The responsibility for the operational conduct of the study lies with the Research Institute of the Diabetes Akademie Mergentheim (FIDAM), which is independent of the producer. The study will be conducted in accordance with the recommendations guiding physicians in biomedical research involving human subjects adopted by the 18th World Medical Assembly, Helsinki, Finland, 1964, and later versions. It will also be conducted in accordance with the European Medical Device Regulation, Regulation (EU) 2017/745 of April 5, 2017, and ISO 14155:2020. Ethical approval has been granted by the Ethics Committee of the Medical Association of Baden Württemberg (F-2021-016) and by the 14 competent local Ethics Committees for each study location.
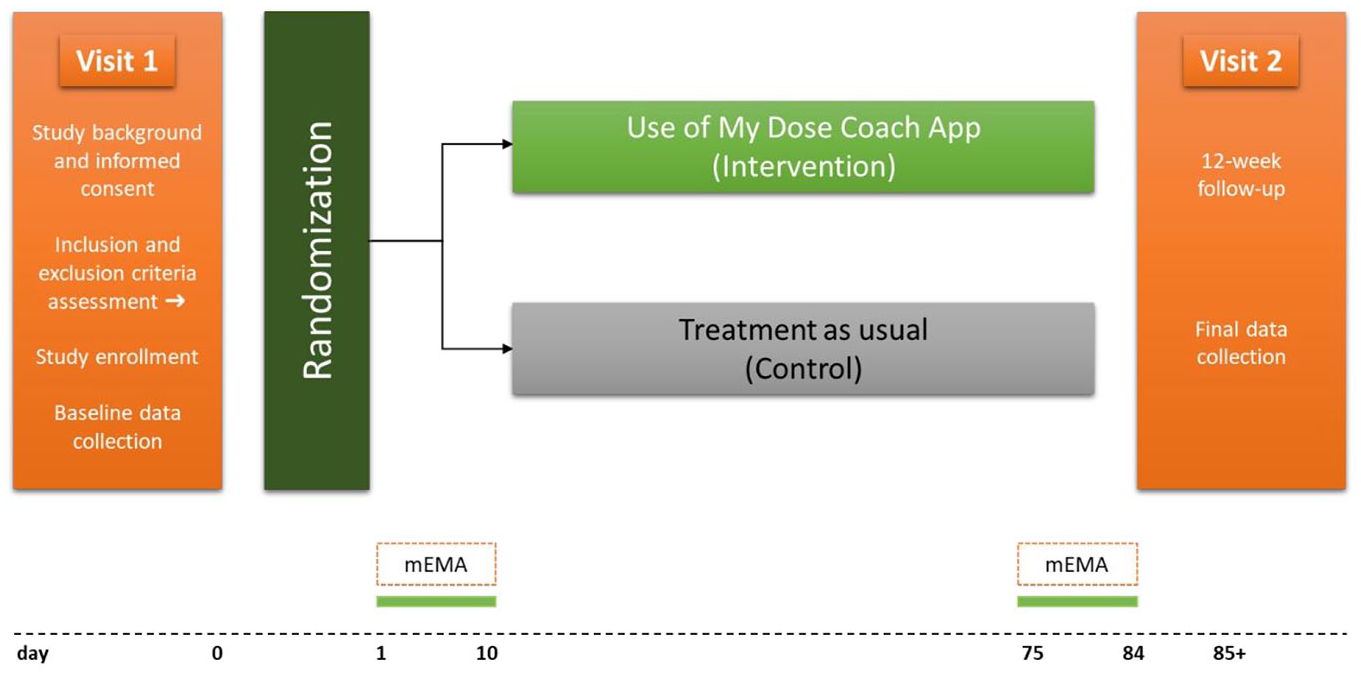
Study design. Abbreviation: mEMA, mobile ecological momentary assessment.
Participant Population
Eligible study participants must meet all of the following inclusion criteria: (1) diagnosis of T2DM; (2) indication for a basal insulin therapy in combination with oral antidiabetic agents; (3) ≥18 years old; (4) HbA1c level >7.5% (58.5 mmol/mol) and <10.0% (85.6 mmol/mol) (last measured HbA1c value from the medical record of the participant); (5) use of a smartphone compatible with the app (iPhone 5 and higher with iOS 10 or higher; Android version 4.4 or higher); (6) willingness to agree to the terms and conditions of the titration app; (7) proficiency in written and spoken German; and (8) presence of written informed consent for study participation.
Patients will be excluded if they meet any of the following criteria: (1) treatment with prandial insulin (meal insulin); (2) body mass index (BMI) < 25 kg/m2; (3) renal insufficiency (glomerular filtration rate < 30 mL/min); (4) presence of an acute mental disorder that may impair the patient’s ability to properly complete the therapy or the study (as assessed by the treating physician); (5) use of another titration app; (6) inability to provide consent due to limited legal capacity or legal guardianship; (7) connection to the sponsor or clinical investigator (eg, employees of the sponsor or participating site and their family members); and (8) participation in another clinical trial during the same period.
Randomization and Blinding
Study participants will be assigned randomly either to the intervention group or to the control group at a 1:1 ratio. Randomization will be performed using a computer-generated algorithm (Research Randomizer; Urbaniak GC, Plous S. Research randomizer [version 4.0] [computer software]. 2013. Research Randomizer [version 4.0] [Computer software]. http://www.randomizer.org/. Accessed June 22, 2022). Randomization will be performed in blocks of six. The block size was concealed against the study centers.
Study centers will receive concealed envelopes with the randomization results for each participant separately. The respective envelope will be opened by the study staff after informed consent was signed by the participant, and inclusion and exclusion criteria were checked at the first study visit. The participant will be allocated either to the intervention group (use of the titration app) or to the control group (titration of insulin doses by a written titration scheme) accordingly. Due to the nature of the intervention, blinding of participants and study centers providing the intervention will not be possible.
Interventions
Participants of the intervention arm will get access to the titration app, and participants of the control group receive treatment as usual.
Intervention group
This digital health tool consists of two components: the Web portal for physicians and the titration app for patients. This digital health tool is expected to support both patients and their treating physicians in optimal insulin titration by facilitating the titration process and reducing the risk of possible errors. Close monitoring can reduce uncertainties about insulin therapy, which is particularly helpful when starting a basal oral therapy, as there is little experience with insulin therapy and titration.
The Web portal
A central component of the digital health tool is Web portal for the physicians, which is used for participant management. After registering the participant in the Web portal, the physician creates an individual dose plan (titration scheme). Physicians have to define the starting dose of basal insulin, the individual fasting target range, the width of the respective correction range, and the corresponding dose increase of the basal insulin according to their clinical judgment of individual insulin sensitivity, multimorbidity, and hypoglycemic risk of the individual participant. Based on this assessment, the physician also has to define a hypoglycemic threshold and the management of hypoglycemic values in daily routine. If a hypoglycemic glucose value is detected, the physician can choose between the three courses of action:
Stop the titration immediately and contact the practice to readjust titration scheme,
Continue titration until the next contact with the practice or readjustment of titration scheme, or
Continue titration, since the algorithm will also reduce basal insulin doses.
The titration recommendations from the Web portal are transferred directly to the patient’s smartphone and displayed in the app. When the participant logs in for the first time, the Web portal sends a confirmation link that the participant must activate to connect and synchronize the app with the Web portal. After that, all entries that the participant makes in the app are automatically transferred to the Web portal. In this way, the physician is able to monitor the patient’s therapy at any time and take any corrective actions, if necessary. When the physician makes any updates or changes to the dosing plan, the updated plan is sent to the patient. The participant will receive a text message with a link to the update, and once he or she taps the link and logs in, the dosing plan is automatically updated.
The titration app
The titration app assists patients with T2DM with optimizing titration of daily basal insulin based on the fasting blood glucose values determined by the treating physician. Participant has to have a profile on the physician’s Web portal and complete a one-time registration by activating the confirmation link.
Participants have to perform at least one fasting blood glucose self-test per day. The results of this test and the applied basal insulin doses are entered in the app. The app calculates the median fasting glucose value and recommends, based on the physicians settings, the corresponding basal insulin dose. The values entered by the patients in the app are automatically transmitted to the physician via the Web portal, which enables the physicians to track the therapy progress and make changes to the therapy plan, if necessary. Table 1 presents all functions of the digital health tool.
Functions of the My Dose Coach App.

Participants in the intervention group will use the titration app to adjust their basal insulin dose during the 12-week intervention period. The treating physician will create a profile for each participant on the portal, enter the individual titration plan, and share it with the patient. During the intervention phase, the participant will use the titration app to adjust their basal insulin dose and to conduct and document their basal insulin therapy. The data will be regularly synchronized via the Web portal so that the treating physician can monitor the patient’s therapy at any time and take any corrective actions, if necessary.
Control arm (treatment as usual)
Participants in the control group should follow their diabetes therapy and titration of basal insulin as agreed with their treating physician. The physician will hand out an individualized titration scheme, which should be followed by the participant. Adjustment of the titration algorithm will be made after consultation with the physician in case of not meeting the titration targets or occurrence of hypoglycemia without the use of another titration app.
Data Collection
Data will be collected at the baseline (visit 1) and at the final assessment (visit 2). Questionnaires will be used to collect patient-reported outcomes. Glucose data (blood glucose levels) will be collected from blood glucose monitoring systems using the DIABASS system (Balingen, Germany). The DIABASS is a glucose management platform with which glucose data can be pseudonymously sent to FIDAM. If downloading data via DIABASS is not possible due to technical issues, the glucose data will be collected with the practice’s own software and transmitted to FIDAM as a pseudonymized file.
No study-related blood sampling is planned for determining the HbA1c value. The HbA1c value of the participants is either taken from the medical record of the participant or determined in the central laboratory as part of a routine blood test. For the upcoming quarterly routine blood tests, an additional tube is filled in the central laboratory for the recording of the HbA1c value.
Visit Schedule
Baseline (visit 1)
Study participants will be randomly assigned to one of the two study groups (intervention or control) in a 1:1 ratio and will be asked to complete the required questionnaires. The treating physician will document the last HbA1c value from the medical record of the participant or schedule the collection of an additional blood sample to determine the HbA1c value in the central laboratory as part of a routine blood test. The glucose data of the last 14 days prior to study inclusion will then be downloaded. Participants in the intervention group will download the titration app and the mobile Ecological Momentary Assessment (mEMA; ilumivu) app on their smartphone and will be provided with the instructions for use.
Intervention
For all study participants (control group and intervention group), the titration of basal insulin will be performed at the discretion of treating physician in accordance with the treatment guidelines of T2DM. The total duration of the intervention and follow-up phase will be 12 weeks (84 days).
Participants in the intervention group will use the basal insulin therapy for the 12-week study period using the titration app. Feedback and, if necessary, an adjustment of the titration algorithm by the physician will be implemented via the Web portal. In addition, the participants in the intervention group will record their experience of using the titration app (“real life experience”) via the mEMA app once a day over a period of 10 days at the beginning and end of the 12-week study period. Participants in the control group will continue their current therapy or as agreed with the treating physician. Monitoring of the titration of basal insulin in the control group will be done according to the practices of the treating facility. Control patients may not use the My Dose Coach app or any other (diabetes) app for titration during the study period.
Final assessment (visit 2)
After the 12-week intervention period, participants in both groups will complete final questionnaires. The treating physician will document the last HbA1c value from the medical record of the participant or schedule the collection of an additional blood sample. All glucose data recorded since the baseline assessment will be downloaded. For participants in the intervention group, the two study apps will be uninstalled. These participants will also complete a separate questionnaire to evaluate the usability of the titration app. No further follow-up is planned in this study.
Outcomes
Primary outcome
The primary objective of this study is to evaluate whether the use of My Dose Coach is associated with a medical benefit as assessed by improved glycemic control and reduced HbA1c levels in patients with T2DM in comparison with patients not using the digital health tool during the 12-week study period. Therefore, we will test the following hypothesis:
Secondary objective
The secondary objective of this study is to examine between-group differences in patient-related structural and procedural improvements of diabetes care after 12 weeks. In particular, diabetes-related distress (coping with disease burden and stress reduction), self-management (adherence to therapy), empowerment (patient sovereignty), self-efficacy (patient sovereignty), treatment satisfaction, and overall psychological well-being (quality of life) will be assessed using various validated questionnaires (see Table 2). In addition, the efficacy of the diabetes therapy will be measured based on the collected glucose data. Safety aspects of the intervention will be specifically analyzed by comparing between-group differences in exposure to hypo- or hyperglycemic glucose values.
Outcome Measures.
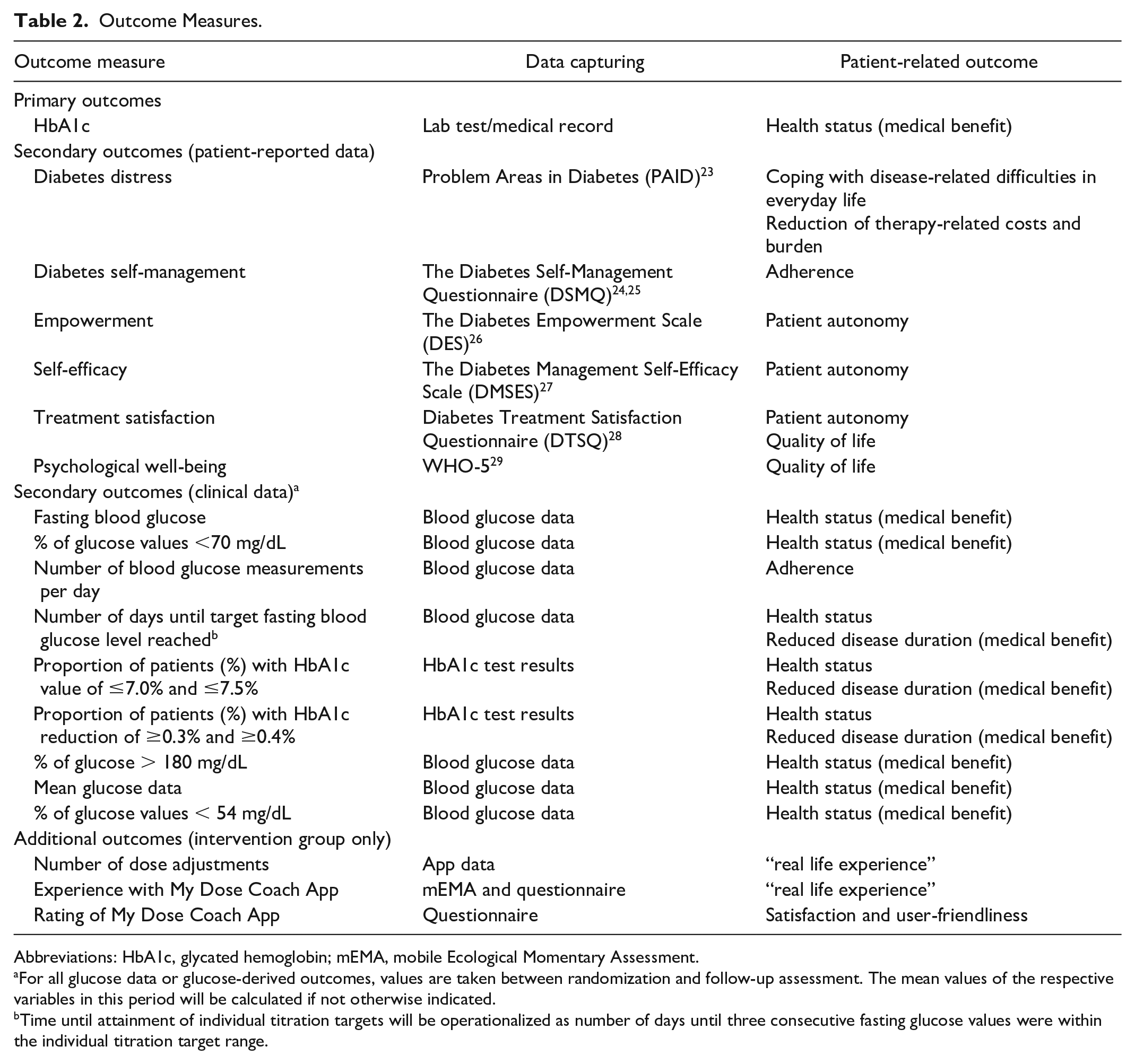
Abbreviations: HbA1c, glycated hemoglobin; mEMA, mobile Ecological Momentary Assessment.
For all glucose data or glucose-derived outcomes, values are taken between randomization and follow-up assessment. The mean values of the respective variables in this period will be calculated if not otherwise indicated.
Time until attainment of individual titration targets will be operationalized as number of days until three consecutive fasting glucose values were within the individual titration target range.
Outcome Measures
The primary outcome measure is HbA1c level at the end of the 12-week period, representing the targeted improvement of health status (medical benefit from treatment). The secondary outcome measures include patient-reported outcomes. Additional measures will be made in the intervention group, including number of dose adjustments and experience with and evaluation of the titration app. Study outcome measures, data capturing, and patient-related outcomes are summarized in Table 2.
Statistical Analysis
Sample size was estimated using an expected mean effect size of
The primary statistical analysis will be performed for the intention-to-treat population. In addition, a sensitivity analysis on the per-protocol population will be performed. Participants included in the per-protocol analysis have provided baseline as well as follow-up data, accessed the randomly allocated intervention, and had a follow-up period longer than 41 days and no longer than 126 days. Continuous variables will be presented as mean ± standard deviation or as median with interquartile range, and categorical variables will be reported as frequencies and percentages.
The analysis of covariance will be used to compare groups for the results on the primary and secondary outcomes. Independent variable is the group allocation (intervention vs control group). The dependent variables are the difference between the baseline and follow-up results of the respective primary or secondary outcome variable with the respective baseline value as a covariate. For all glucose data or glucose data–derived outcomes, all available data between randomization and follow-up assessment will be the dependent variable with the respective baseline values as covariate. In case of deviations from the normal distribution, the data are transformed to van der Waerden scores to achieve normal distribution. If this cannot be achieved, nonparametric methods will compare only the follow-up values, respective the values collected between randomization and follow-up assessment. In case of significant baseline differences in HbA1c between study centers, the study centers will be adjusted for. For nominal data, logistic regression models with adjusted values will be used. Multiple imputation with the jump-to-reference approach will be used for handling missing data. Predictors for missing data imputations will be baseline dose of basal insulin, baseline insulin unit per kg body weight, preexisting insulin therapy versus new initiated insulin therapy, baseline body mass index, and age. Sensitivity analyses are also planned, particularly with regard to relevant subgroups (eg, existing vs new initiation of basal insulin therapy) and in the per-protocol population. All statistical analyses will be performed using IBM SPSS Statistics version 29 (IBM, Armonk, New York). A
Hierarchical Testing of Potential Positive Supply Effects.
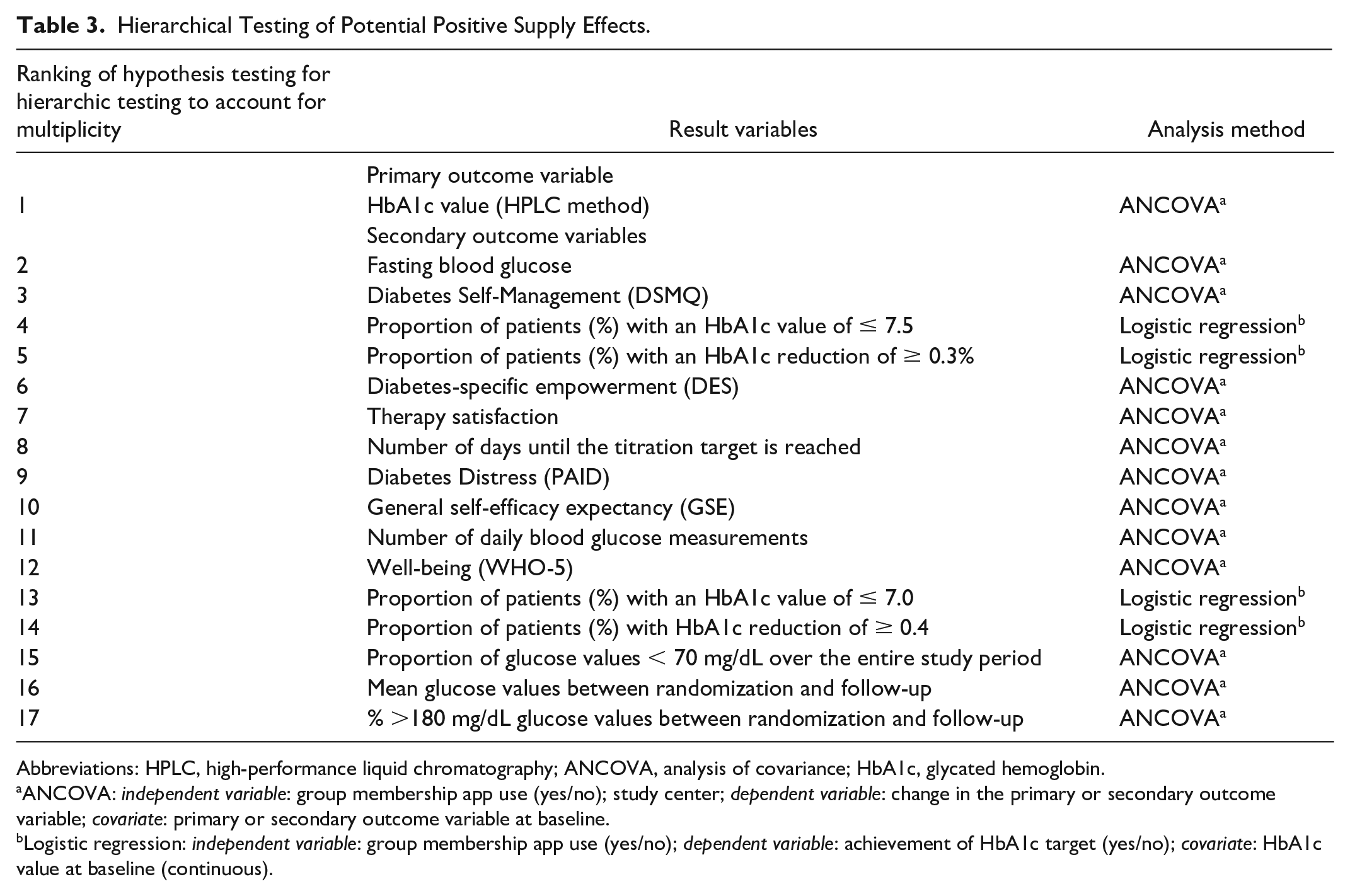
Abbreviations: HPLC, high-performance liquid chromatography; ANCOVA, analysis of covariance; HbA1c, glycated hemoglobin.
ANCOVA:
Logistic regression:
Discussion
Despite a growing body of research on diabetes treatment strategies, most patients with T2DM have poor glycemic control and do not reach their target treatment goals. The widespread use of smartphones and Web communication across all demographic groups and the ability to process and communicate data in real time make these modalities ideal platforms for simple and effective diabetes management programs. The evaluated digital health tool has previously shown successful basal insulin titration and reduction of fasting blood glucose in Indian and Mexican populations of patients with T2DM.20 -22 The current study is expected to confirm these results in a large sample of patients with T2DM under the conditions of the German healthcare system.
In a pilot study of 137 insulin-naive patients with T2DM in India, the digital health tool was used with a standard titration algorithm to achieve a predetermined fasting blood glucose level of 90 to 120 mg/dL during the 12-week study period. 22 Patients were instructed to measure their fasting blood glucose daily, enter the values into the app, and titrate their daily basal insulin dose according to the physician recommendations. After 12 weeks of titration, there was a 2.7% reduction in HbA1c and 59 mg/dL reduction in fasting blood glucose. The mean time from initiation of basal insulin titration and target fasting blood glucose level was 41 days and was associated with an average increase in basal insulin dose of 50%. No events of symptomatic hypoglycemia or hospitalizations were recorded during the study period. 22
In another pilot study, 337 patients with T2DM who registered to use the digital health tool between September and December 2018 and entered at least one fasting blood glucose value were analyzed. 21 The observation period lasted until February 2019 and participants were considered to be on target if three consecutive fasting blood glucose values were within the defined target range. During the observation period, 34% of participants reached their individual fasting blood glucose target within the median time of 25 days. The fasting blood glucose was reduced on average from 165.8 mg/dL to 108.0 mg/dL in this participant group and was within the target range of 100 to 125 mg/dL. The average increase in basal insulin dose was from 16.2 to 19.4 units/day. 21
In a pilot study of 158 patients with T2DM using the digital health tool in Mexico, 58.9% of participants reached their target self-measured plasma glucose level with a mean reduction of 30.9 mg/dL. 20 Target fasting blood glucose level was reached in 55.7% of participants with a mean reduction of 63.4 mg/dL and no severe hypoglycemic events. HbA1c was reduced by 1.78% on average with the last observation carried forward. There was also a mean increase of 2.23 points in World Health Organization Five Well Being Index. 20
If successful in this study, this digital health tool could become an essential part of clinical practice in the management of patients with T2DM and overcome practical barriers to glycemic control in those patients. The software offers several advantages over traditional treatment approaches, including less complex titration process, better adherence, improved timing and greater HbA1c target achievement, and reduced costs and resource utilization, among others. It is important to note, however, that some people with diabetes who are less computer-proficient, such as older patients or those with cognitive impairment, may benefit less from digital health interventions like the evaluated tool, and the results of this study may, therefore, not be generalizable to these patients.
Conclusion
The use of digital tools and telehealth systems in people with diabetes ensures immediate and precise responses to blood glucose levels by providing frequent monitoring and real-time data. They also allow regular communication between people with diabetes and their physician, which helps to optimize the treatment plans of people with diabetes and decrease the risk of long-term complications. My Dose Coach is a modern solution for optimal glycemic control and facilitates insulin titration in people with T2DM, and findings from this multicenter randomized controlled trial can provide further evidence supporting its effectiveness.
Footnotes
Abbreviations
DRKS, deutsches Register für klinische Studien; FIDAM, Research Institute of the Diabetes Akademie Mergentheim; mEMA, mobile ecological momentary assessment; T2DM, type 2 diabetes mellitus.
Author Contributions
N.H., D.E., and B.K. wrote the manuscript. G.F., T.R., and G.F. revised the manuscript and contributed to the discussion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.H. has received fees for Advisory Board Participation from Abbott. He also received fees for lectures from Berlin Chemie, Sanofi, and Novo Nordisk. He also reports research support from Sanofi, Berlin Chemie, Roche, and mySugr. D.E. has received fees for Advisory Board participation from Dexcom, Roche Diabetes Care, mySugr, and Medtronic. He also received fees for lectures from Dexcom and Berlin-Chemie AG. K.F.G has no conflict of interest. T.R has no conflict of interest. G.F. is general manager of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm (IfDT, Ulm, Germany), which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. G.F./IfDT has received grants, speakers’ honoraria, or consulting fees from Abbott, Agamatrix, Ascensia, Berlin-Chemie, Beurer, Boydsens, CRF Health, Dexcom, i-SENS, LifeScan, Lilly, Metronom Health, Medtronic, Menarini, mySugr, Novo Nordisk, PharmaSens, Roche, Sanofi, Sensile, and Ypsomed. B.K. reports grants from AstraZeneca, Berlin-Chemie, Roche Diabetes Care, Abbott Diabetes Care, AstraZeneca, Dexcom, and Ypsomed, and personal fees from Berlin-Chemie, Roche Diabetes Care, Novo Nordisk, Medtronic, Ascensia Diabetes Care, Abbott Diabetes Care, and Eli Lilly.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by Sanofi Aventis Germany.
