Abstract
Background:
Ovarian cancer (OC) persists as a major cause of death in gynecological malignancies, primarily due to inherent resistance to chemotherapy and the recurrence of metastatic disease.
Aim:
This investigation aimed to elucidate a previously uncharacterized mechanism of action for the anthelmintic agent mebendazole (MBZ) in OC cells. Specifically, we aimed to determine if MBZ could simultaneously target mitochondrial function and X-linked inhibitor of apoptosis protein (XIAP), thereby triggering an intrinsic apoptotic pathway.
Methods:
In vitro studies were conducted using SKOV3 (adenocarcinoma) and PA1 (teratocarcinoma) OC cell lines. Cytotoxicity was assessed via IC50 determination. Mitochondrial membrane potential (ΔΨm) was evaluated using flow cytometry with rhodamine-123 and confocal microscopy with JC-1 staining. Apoptosis was quantified by annexin V/PI staining. Western blot analysis was used to examine the expression of XIAP, cytochrome c, Bax, and cleaved caspase-3. Molecular docking and density functional theory (DFT) calculations were utilized to investigate MBZ’s interaction with XIAP.
Results:
MBZ exhibited dose-dependent cytotoxicity in both cell lines (IC50: 1.25 µM in SKOV3, 0.312 µM in PA1), with PA1 showing greater sensitivity. Significant ΔΨm depolarization was observed (40% in SKOV3, 33% in PA1), accompanied by increased JC-1 monomer/aggregate ratios (5.7-9.9-fold). MBZ induced apoptosis without significant necrosis (3.2-4.5-fold increase in Annexin V+ cells). Mechanistically, MBZ reduced XIAP expression (2.5-fold) and upregulated cytochrome c, Bax, and cleaved caspase-3. Molecular docking revealed high-affinity binding of MBZ to the XIAP BIR3 domain, comparable with A4E (ASTX660), with unique interactions at LYS299. DFT calculations confirmed MBZ’s comparable electronic properties, including a lower Highest Occupied Molecular Orbital-Lowest Unoccupied Molecular Orbital energy gap relative to A4E.
Conclusion:
This study identifies a novel dual mode of action for MBZ in OC cells, concurrently disrupting mitochondrial function and directly inhibiting XIAP, leading to caspase-dependent apoptosis. This unique mechanism of action suggests MBZ as a promising repurposed therapeutic strategy to potentially overcome chemoresistance in OC.
Introduction
Ovarian cancer (OC) remains a significant health challenge, representing the fifth leading cause of cancer-related deaths among women worldwide. Characterized by its often-asymptomatic progression, OC frequently remains undetected until advanced stages, leading to poor prognosis and limited treatment options. 1 The standard treatment modalities, primarily involving surgery and chemotherapy, face significant hurdles due to the intrinsic resistance exhibited by OC cells. This resistance is driven by various mechanisms such as drug efflux, DNA repair alterations, and apoptosis evasion, making it a critical barrier to effective therapy. This resistance not only complicates treatment regimens but also underscores the urgent need to develop novel therapeutic strategies that can overcome or bypass these mechanisms.2,3 The development of novel therapeutic agents is essential to improve survival rates and quality of life for OC patients. Traditional chemotherapy often leads to significant side effects and does not guarantee long-term remission. New drugs that target specific molecular pathways or exploit vulnerabilities in cancer cells could enhance treatment efficacy and minimize adverse effects. This has led researchers to explore alternative therapeutic avenues, including the repurposing of existing drugs. However, the process of discovering and approving new drugs is both time-consuming and expensive, often taking years of rigorous testing before reaching clinical application. 4 In this context, drug repurposing has emerged as a promising and cost-effective alternative. Drug repurposing involves identifying new therapeutic applications for existing, already-approved drugs. This approach not only significantly reduces the time and cost associated with drug development but also leverages the known safety profiles of these drugs, accelerating their potential clinical use. 5 Among the various classes of drugs being investigated, antihelminthics have garnered attention due to their potential anticancer properties. These drugs, originally developed to treat parasitic infections, have shown potential in targeting various cancer hallmarks, including proliferation, metastasis, and survival. Among these, MBZ stands out as a particularly potent candidate due to its broad spectrum of activity and low toxicity. 6
MBZ, a widely used and well-tolerated antihelminthic drug, has shown potential as an anticancer agent in various cancer types, including lung, breast, and colorectal cancer. 7 Unlike other antihelminthics, MBZ has demonstrated a unique ability to inhibit cancer by microtubule disruption, leading to cell cycle arrest, cell proliferation, inducing apoptosis, and preventing metastasis. Its mechanism of action involves disrupting microtubule dynamics, which is critical for cell division and survival. 8 This disruption not only affects the cancer cells directly but also impacts their ability to migrate and invade surrounding tissues. Furthermore, clinical trials have demonstrated its safety and efficacy across various cancers. 9 For instance, when combined with temozolomide in trial NCT01729260, MBZ improved survival outcomes in high-grade gliomas. 10 Another study (NCT03628079) showed its potential in treating advanced gastrointestinal cancers. 11 Pediatric trials (NCT02644291) further revealed MBZ’s ability to cross the blood–brain barrier, inhibit tumor growth, and maintain a favorable safety profile in brain cancers. 12 Despite these promising findings, there are limited reports investigating its effects on OC. This gap in the literature underscores the need for focused research to elucidate MBZ’s potential as a therapeutic agent for this deadly disease.
One of the critical mechanisms by which MBZ exerts its anticancer effects is through the induction of apoptosis. 13 Apoptosis, or programed cell death, is a tightly regulated process that is often dysregulated in cancer cells, allowing them to survive and proliferate uncontrollably. It can be triggered by various cellular stressors, including mitochondrial dysfunction. The mitochondrial membrane potential (Δψm) plays a crucial role in maintaining cellular homeostasis and energy production. 14 Disruption of Δψm can lead to the release of pro-apoptotic factors, such as cytochrome c, into the cytosol, which activate downstream caspases and ultimately trigger apoptotic pathways. 15 Furthermore, the disruption of Δψm has been linked to the downregulation of X-linked inhibitor of apoptosis protein (XIAP), which is a critical regulator of apoptosis that can inhibit caspase activation. 14 XIAP is often overexpressed in cancer and contributes to chemoresistance by inhibiting caspase activity. 16 By targeting Δψm and XIAP, MBZ may overcome one of the key resistance mechanisms in OC by effectively promoting apoptosis, thereby reducing tumor growth and progression, making it a compelling candidate for further investigation.
Given these promising mechanisms, this study aims to provide a comprehensive evaluation of MBZ’s anticancer effects specifically in OC cells. In our recently published work, MBZ was shown to inhibit OVCAR3 and OAW42 cell progression through suppression of the Girdin–AKT/IKKα/β–NF-κB signaling pathway, with mitochondrial depolarization and apoptosis noted only as downstream consequences. 17 Building on that foundation, the present investigation employs two distinct OC cell lines, SKOV3 (adenocarcinoma) and PA1 (teratocarcinoma), to uncover a mechanistically different route in which MBZ directly disrupts Δψm and suppresses XIAP, thereby triggering caspase-mediated apoptosis. Importantly, unlike our earlier work, this study also integrates in silico docking and molecular dynamics to validate XIAP as a binding target, adding structural and translational depth. To the best of our knowledge, this is the first study to explicitly identify XIAP downregulation as a central mechanism of MBZ-induced apoptosis in OC, supported by both experimental and computational evidence. By elucidating this complementary pathway across different OC subtypes, we aim to provide valuable insights into MBZ’s dual anticancer potential and its suitability as a repurposed therapeutic for improving clinical outcomes.
Materials and Methods
In vitro cell culture for experimental use
The human OC cell lines SKOV3 and PA1 were procured from the National Center for Cell Science (Pune, India). Cells were cultured in RPMI-1640 medium (Thermo Fisher Scientific, Cat# 11875093) supplemented with 10% fetal bovine serum (Thermo Fisher Scientific, Cat# 26140079) and 1% penicillin-streptomycin (Thermo Fisher Scientific, Cat# 15140122) at 37°C in a 5% CO2 humidified incubator (HERAcell 204i, Thermo Fisher Scientific). Cells were routinely passaged at 80–90% confluency using 0.25% trypsin-EDTA (Thermo Fisher Scientific, Cat# 25200056).
Drug preparation and treatment
Mebendazole (MBZ, Sigma-Aldrich, Cat# M1625) was dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich, Cat# D8418) to prepare 10 mM stock solutions, which were further diluted in culture medium to achieve working concentrations. The final DMSO concentration in all experiments was maintained at 0.1% (v/v), which served as vehicle control (VC).
Morphological analysis using phase contrast microscope
SKOV3 and PA1 cells were seeded in 6-well plates at a density of 5 × 106 cells/well and allowed to adhere for 48 hours. Cells were treated with varying concentrations of MBZ (0.156–2.5 µM) or 0.1% DMSO (Sigma-Aldrich, Cat# D8418, USA) as VC for 48 hours. Morphological changes were observed under an Olympus phase-contrast microscope (Olympus, Japan) (magnification: 10×, scale bar = 100 µm) and captured using Olympus Camedia software (E-20P 5.0, Chicago, MI, USA). Images were processed using Adobe Photoshop 2021 v22.5.1.441 (San Jose, CA).
Cell proliferation (viability) assay
Cell viability was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Thermo Fisher Scientific, Cat# M6494) assay as previously described. 18 Briefly, 1 × 104 cells/well were seeded in 96-well plates and treated with MBZ at concentrations ranging from 0.156 to 2.5 µM for 48 hours. After treatment, 10 µL of MTT solution (5 mg/mL in PBS) was added to each well and incubated for 3 hours at 37°C. The formazan crystals were solubilized in 100 µL DMSO, and absorbance was measured at 570 nm using a Multiskan FC microplate reader (Thermo Fisher Scientific). The half-maximal inhibitory concentration (IC50) was calculated using GraphPad Prism 5.0 (GraphPad Software, San Diego, CA, USA).
Flow cytometric profiling of mitochondrial membrane potential in MBZ-treated cells
Flow cytometry was used to evaluate ΔΨm, according to the previously discussed protocol with minor alterations. 19 SKOV3 and PA1 cells were treated with IC50 concentrations of MBZ or DMSO. After 48 hours, cells were detached using Trypsin-EDTA Solution (Thermo Fisher Scientific, Cat# 25200056, USA), washed with PBS (Thermo Fisher Scientific, Cat# 10010023, USA), and incubated with 5 µM rhodamine-123 (Thermo Fisher Scientific, Cat# R302, USA) for 30 minutes. Cells were washed, resuspended in flow cytometry buffer, and analyzed using a BD FACSLyric™ flow cytometer. Changes in rhodamine 123 fluorescence, reflecting altered ΔΨm, were recorded and analyzed using BD FACSLyric™ flow cytometry system software (BD Biosciences, USA). Data were compared with the DMSO control and plotted using GraphPad Prism 5.
Confocal microscopy analysis of JC-1 aggregation and mitochondrial polarization
Confocal microscopy was employed to analyze ΔΨm. 20 SKOV3 and PA1 cells, cultured on coverslips, were treated with MBZ at IC50 concentrations or DMSO control for 48 hours. Cells were washed with PBS (Thermo Fisher Scientific, Cat# 10010023, USA) and stained with 2 µM JC-1 dye (Sigma-Aldrich, Cat# T4069, USA). After incubation and washes, coverslips were mounted using ProLong Gold Antifade Reagent (Invitrogen, Cat# P36930, USA). Images were acquired using an Olympus FV3000 confocal microscope (Olympus, Japan). JC-1 aggregates (red fluorescence, high ΔΨm) and monomers (green fluorescence, low ΔΨm) were analyzed. The green-to-red fluorescence ratio, indicative of mitochondrial depolarization, was quantified using ImageJ software (Version 1.53c, NIH, Bethesda, MD, USA), normalized to the DMSO control, and plotted using GraphPad Prism 5.
Apoptosis quantification: Flow cytometric methods
To assess MBZ’s impact on apoptosis, SKOV3 and PA1 cells were treated with MBZ at IC50 concentrations or a DMSO control. Following a 48-hour incubation, cells were harvested, washed with PBS (Thermo Fisher Scientific, Cat# 10010023, USA), and stained using the Annexin V-FITC/PI Apoptosis Detection Kit (BioLegend, Cat# 640914, USA), following the manufacturer’s instructions. The stained cells were then analyzed using a BD FACSLyric™ flow cytometer (BD Biosciences, USA). Fluorescence emission was measured at 530 nm (Annexin V-FITC) and 617 nm (PI). Cell populations were categorized as live, early apoptotic, late apoptotic, and necrotic, based on Annexin V-FITC and PI staining patterns using BD FACSLyric™ flow cytometry system software (BD Biosciences, USA). Quantitative analysis determined the percentage of cells in each phase. Statistical significance was determined using GraphPad Prism 5 software. Experiments were repeated thrice to ensure reproducibility, providing robust evidence of MBZ-induced apoptosis in OC cells.
Investigation of protein expression using immunoblotting
Protein expression was analyzed as previously described with slight modification. 21 Cells were lysed in RIPA buffer (Cell Signaling Technology, Cat# 9806) containing protease inhibitors (Sigma-Aldrich, Cat# P8340, USA). Protein concentration was determined using the Bradford method with Bradford Reagent (Bio-Rad, Cat# 5000006, USA). Total protein (50 µg/lane) was separated by using SDS-PAGE and transferred to PVDF membranes (Merck, Cat# IPVH00010). Membranes were blocked with 5% BSA (Sigma-Aldrich, Cat# A3059, USA) in TBST and probed with primary antibodies against Bax (1:1000, CST Cat# 2772), Bcl-2 (1:1000, CST Cat# 2872), cleaved-caspase-3 (1:1000, CST Cat# 9661), cytochrome C (1:1000, CST Cat# 11940), XIAP (1:1000, CST Cat# 2045), and GAPDH (1:500, Santa Cruz Biotechnology, Cat# sc-47724) overnight at 4°C. After incubation with HRP-conjugated secondary antibodies (1:5000, CST Cat# 7074/7076), bands were visualized using Immobilon Western Chemiluminescent HRP Substrate (Merck, Cat# WBKLS0500) on a Bio-Rad ChemiDoc MP (Bio-Rad, USA) Imaging System. Densitometric analysis was performed using ImageJ software (Version 1.53c, National Institutes of Health (NIH), Bethesda, MD, USA), and the quantitative graphs were plotted by GraphPad Prism software (V5.0, GraphPad Software, San Diego, CA, USA).
Protein retrieval, preparation, and refinement (protein model preparation)
The crystal structure of XIAP (PDB ID: 5OQW) was obtained from the Protein Data Bank (RCSB). This structure was selected based on its X-ray diffraction data, resolution below 2.5 Å, and high scores in validation metrics. Using PyMOL (version 2.5), the protein was prepared by removing native inhibitors, heteroatoms, and water molecules. Hydrogen atoms were added to optimize protonation states for docking. 22 The resulting macromolecule was further refined, verified, and energy-minimized using Swiss-PDB Viewer to ensure structural integrity and stability. 23 These steps prepared the protein for accurate molecular docking simulations.
Ligand model building and optimization
MBZ and A4E, a co-crystal ligand of XIAP (5OQW), were chosen as ligands, designated MBZ_Dock and A4E_Redock, respectively. Their 3 D structures were retrieved from the PubChem database in .sdf format. Ligand preparation was performed using Open Babel, 24 a component of the PyRx software suite. This step ensures the ligands are in the appropriate format for subsequent molecular docking simulations.
Molecular docking: Energetic and structural analysis of ligand–protein complexes
Molecular docking, facilitated by PyRx software (version 0.8), was employed to investigate ligand-target interactions, focusing on binding orientations, conformations, and affinities within the 5OQW active site. Prior to docking, ligands underwent conversion to the PDBQT format using Open Babel, ensuring compatibility with AutoDock Vina. A grid box, encompassing the A4E co-crystal ligand, was generated to constrain the search space, promoting GRID-based docking. Visualization and analysis of noncovalent interactions in the resulting protein-ligand complexes were conducted using PyMOL. The ligand conformation exhibiting the lowest binding energy (kcal/mol) was identified as the most energetically favorable binding mode. 25 This approach enables a detailed assessment of ligand–protein interactions, offering insights into potential binding mechanisms and compound efficacy.
Computational profiling of ligand interactions: Structural interaction fingerprinting analysis
Structural interaction fingerprinting (SIFt) was employed to model and evaluate protein–ligand interactions in three dimensions. This method generates a binary fingerprint that encodes the structural binding characteristics of the complex, representing its interaction pattern. This fingerprint facilitates efficient organization, analysis, and presentation of large datasets for ligand-receptor complexes, enabling database mining. The SIFt panel within Schrödinger Suite 2020-3 was used to generate interaction fingerprints for selected docking complexes, using receptor grids and ligands as inputs. Results are visualized, highlighting key residues and interaction types (hydrophobic, hydrogen bond donor/acceptor, etc.). Further details on the SIFt model generation can be found in prior research. 26
Density functional theory: Quantum chemical analysis of molecular properties of ligands
Density functional theory (DFT) was employed to investigate the electronic properties of the studied molecules. Calculations were performed using the Gaussian 06 package (Rev.E.01) with the B3LYP functional and SVP basis set. This computational approach facilitated the determination of optimized molecular geometries and the generation of molecular electrostatic potential (MESP) maps. In addition, frontier molecular orbitals (FMOs) were analyzed to derive global and local reactivity descriptors. GaussView 6 was utilized for visualization and validation of the computed parameters. 27
Statistical analysis
Data were presented as the mean ± standard error of the mean (SEM). Statistical significance was determined using either a one-way ANOVA followed by Tukey’s test, a two-way ANOVA followed by Bonferroni’s test, or a student’s t-test. All analyses were conducted using GraphPad Prism software (version 5.0, San Diego, CA, USA). Significance was defined as *p < 0.05, **p < 0.01, and ***p < 0.001.
Results
MBZ reduces concentration-dependent viability and alters morphology in OC cell lines
The SKOV3 and PA1 OC cell lines were subjected to treatment with varying MBZ concentrations (0.156 µM to 2.5 µM) for 48 hours, followed by analysis using phase contrast microscopy to observe any morphological alterations. Phase contrast microscopic analysis revealed dose-dependent morphological changes in both cell lines. In the DMSO (VC)-treated cells, both SKOV3 and PA1 cells exhibited normal morphology with typical epithelial-like appearance, growing in a monolayer with distinct cell boundaries, and high confluency visible under phase contrast. However, exposure to increasing concentrations of MBZ, we observed progressive reduction in cell density (proliferation), accompanied by morphological alterations including cell rounding, detachment, cell shrinkage, and membrane blebbing, and the formation of apoptotic bodies, potentially indicative of cell cycle arrest or apoptosis. Cellular debris has also been observed in the treated samples, hinting at cell death, particularly at higher concentrations (1.25 µM and 2.5 µM) as shown in Figure 1A, B. These visual cues provide preliminary insights into the mechanism of action of MBZ exerts a cytotoxic and antiproliferative effect on both the SKOV3 and PA1 cell lines.

Impact of MBZ on ovarian cancer cells: morphological and viability assessments.
To quantitatively assess the effect of MBZ on the proliferation (viability) of SKOV3 and PA1 cells, an MTT assay was performed. The cells were treated with MBZ at concentrations of 0.156 µM, 0.312 µM, 0.625 µM, 1.25 µM, and 2.5 µM for 48 hours. In SKOV3 cells, MBZ demonstrated a concentration-dependent reduction in cell viability from 93% in the control group to 86%, 78%, 65%, 50%, and 31% at 0.156 µM, 0.312 µM, 0.625 µM, 1.25 µM (IC50), and 2.5 µM concentrations, respectively. The half-maximal inhibitory concentration (IC50) for SKOV3 cells was determined to be 1.25 µM. PA1 cells exhibited greater sensitivity to MBZ treatment compared with SKOV3 cells. Cell viability decreased from 94% in the control group to 73%, 51%, 38%, 27%, and 18% at 0.156 µM, 0.312 µM (IC50), 0.625 µM, 1.25 µM, and 2.5 µM concentrations, respectively. The IC50 for PA1 cells was 0.312 µM, approximately four times lower than that of SKOV3 cells, indicating significantly higher sensitivity of PA1 cells to MBZ treatment (Figure 1C, D). Therefore, the MTT assay provides quantitative data on the extent to which MBZ inhibits the proliferation (or reduces the viability) of SKOV3 and PA1 cells in a concentration-dependent manner, with PA1 cells showing greater sensitivity compared with SKOV3 cells and further confirms the microscopic observations.
Molecular disruption of ΔΨm by MBZ
The effect of 48-hour MBZ treatment on ΔΨm in OC cell lines SKOV3 and PA1 was assessed using flow cytometry with rhodamine-123 staining. Flow cytometry analysis revealed a significant reduction in ΔΨm in both cell lines following MBZ treatment at their respective IC50 concentrations compared with the VC (DMSO). In SKOV3 cells (Figure 2A), MBZ treatment at 1.25 µM (IC50), a substantial shift in the fluorescence histogram toward lower intensity on the x-axis was observed, indicating a reduction in rhodamine-123 uptake. Furthermore, the untreated control group exhibited a mean fluorescence intensity (MFI) of approximately 1350 arbitrary units while the MFI in the treated group decreased to approximately 580 arbitrary units, representing a substantial reduction in ΔΨm in SKOV3 cells (Figure 2B). This change also manifested as a notable increase in the percentage of cells populating the lower fluorescence intensity range, with an overall reduction in the MFI of approximately 57% (p < 0.001), indicating reduced mitochondrial membrane polarization. Similarly, in the PA1 cell line (Figure 2C), the untreated control group reported an MFI of approximately 1,650 arbitrary units. Upon treatment with MBZ at a concentration of 0.312 µM (IC50) for 48 hours, a distinct leftward shift in the fluorescence histogram on the x-axis was observed, indicating a marked loss of ΔΨm. The MFI in the treated PA1 cells decreased to approximately 550 arbitrary units, representing a reduction of approximately 67% (p < 0.001) (Figure 2D). These findings demonstrate a significant reduction in mitochondrial uptake of rhodamine-123, a hallmark of ΔΨm disruption, in both the cell lines following MBZ treatment.

MBZ disrupts mitochondrial membrane potential (ΔΨm) in ovarian cancer cells.
To validate the flow cytometry observations, confocal microscopy analysis using JC-1 staining was performed. In the SKOV3 cell line (Figure 3A), the untreated control group showed cells with mitochondria exhibiting predominantly punctate red fluorescence due to JC-1 aggregates, indicative of high ΔΨm. However, following a 48-hour treatment with MBZ (1.25 µM, IC50), a noticeable alteration in this fluorescence pattern was observed. There was a clear decrease in the intensity of red fluorescence and a corresponding increase in the green fluorescence (increase in JC-1 monomers), indicating a reduction in the red/green fluorescence ratio (JC-1 aggregate/monomer), while there was an increase in the green/red fluorescence ratio (JC-1 monomer/aggregate). Quantitative analysis of the green/red ratio (JC-1 monomer/aggregate) revealed a 9.9-fold change (p < 0.001) in the MBZ-treated SKOV3 cells compared with the control, confirming significant mitochondrial depolarization (Figure 3B). This substantial shift from red to green fluorescence provides strong evidence that MBZ significantly disrupts the Δψm in SKOV3 cells. Similarly, in the PA1 cell line (Figure 3C), the untreated control group showed predominantly red fluorescence within the mitochondria. Upon treatment with MBZ (0.312 µM, IC50) for 48 hours, a similar trend toward decreased red fluorescence and increased green fluorescence was observed. Quantitative analysis demonstrated a 5.7-fold change (p < 0.001) in the green/red ratio (JC-1 monomer/aggregate) in the MBZ-treated PA1 cells compared with the control, indicating mitochondrial depolarization (Figure 3D). Therefore, these results corroborate the mitochondrial dysfunction induced by MBZ, consistent with its known mechanism of action involving apoptosis induction through mitochondrial pathways.
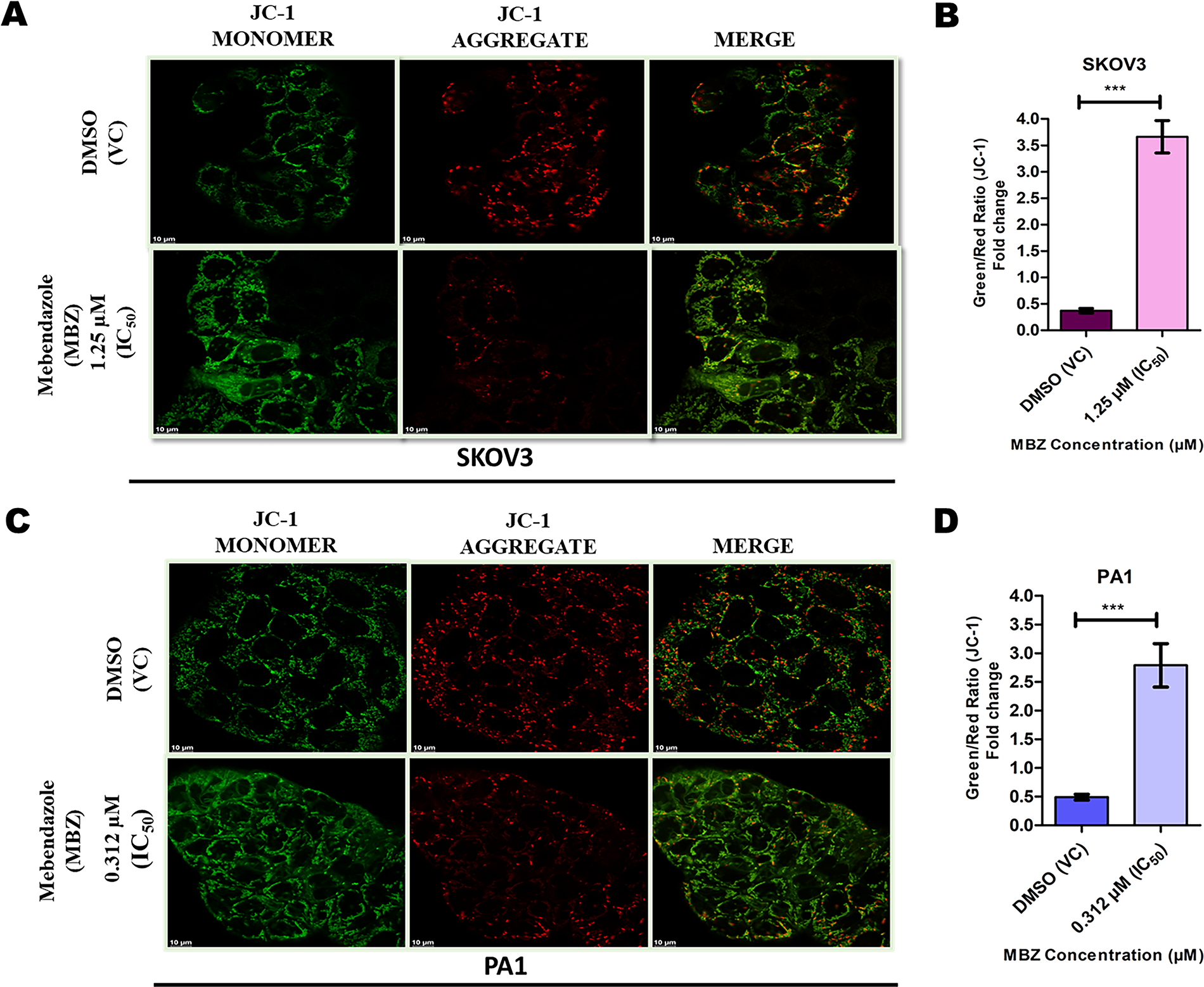
MBZ-induced mitochondrial depolarization in ovarian cancer cells assessed by JC-1 staining.
MBZ-mediated apoptosis in OC: Quantitative assessment of cell death mechanisms
Previous studies exhibited that the alteration of Δψm aligns with the understanding that a decrease in Δψm is a well-recognized early event in the apoptotic cascade. This study aimed to assess the in vitro effect of MBZ on the induction of apoptosis in two human OC cell lines, SKOV3 and PA1, using flow cytometry. The methodology involved treating these cell lines with MBZ at their respective IC50 concentrations for 48 hours, followed by the quantification of apoptosis using annexin-V-FITC/PI conjugated stain and flow cytometry analysis. The analysis revealed a significant increase in the percentage of apoptotic cells in both cell lines when treated with MBZ at their respective IC50 concentrations compared with the VC (DMSO) (Table 1).
MBZ-mediated induction of apoptosis in human ovarian cancer cell lines SKOV3 and PA1
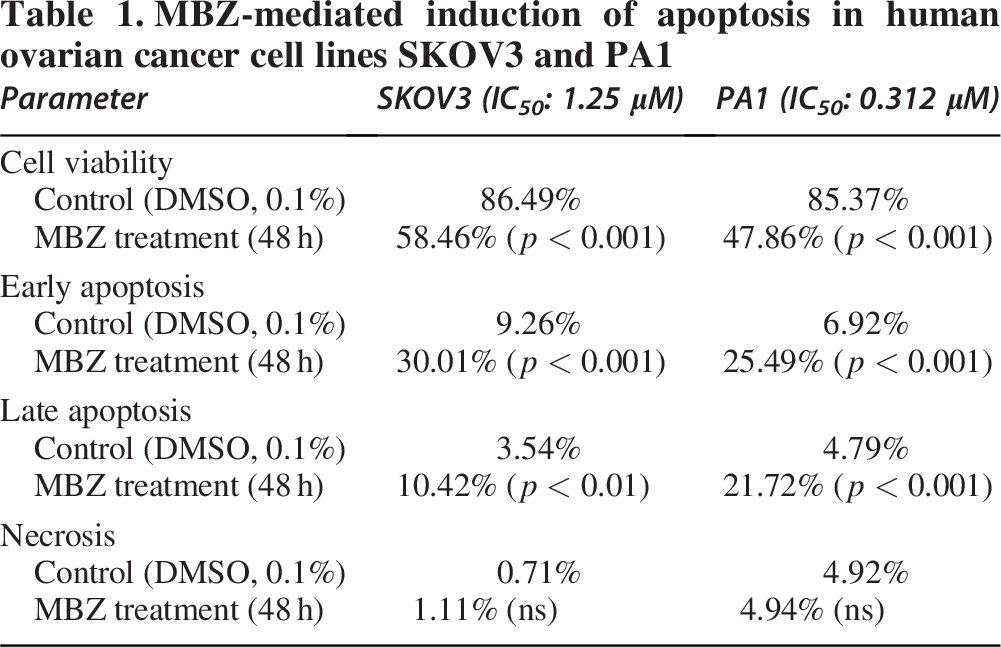
In the untreated control group of SKOV3 cells, which received DMSO as a vehicle, the flow cytometry analysis revealed a baseline distribution of cell populations. The majority of cells, 86.49%, were classified as live (FITC-/PE-), while 9.26% were in early apoptosis (FITC+/PE-), 3.54% in late apoptosis (FITC+/PE+), and 0.71% were necrotic (FITC-/PE+). Following a 48-hour treatment with 1.25 µM MBZ, which represents the IC50 for SKOV3 cells, a notable shift in this distribution was observed. The percentage of live cells decreased to 58.46% (p < 0.001), indicating a substantial reduction in viable cells. Concurrently, the proportion of cells undergoing early apoptosis significantly increased to 30.01% (p < 0.001), and the late apoptotic population also rose to 10.42% (p < 0.01). The percentage of necrotic cells remained relatively low, that is not changed statistically at 1.11% (ns) (Figure 4A). Similarly, PA1 cells treated with 0.312 µM MBZ (IC50) resulted in a significant increase in the percentage of early apoptotic cells from 6.92% in the control group to 25.49% (p < 0.001). Similarly, the percentage of late apoptotic cells increased significantly from 4.79% to 21.72% (p < 0.001). The percentage of live cells decreased significantly from 85.37% to 47.86% (p < 0.001), while the slight increase in necrotic cells to 4.94% was noted, although this change was not statistically significant (ns) (Figure 4C). Moreover, quantitative analysis, further supported by a bar graph, corroborated these observations (Figure 4B, D). These results demonstrate that MBZ induces apoptosis in both SKOV3 and PA1 OC cell lines, leading to a statistically significant shift from live to apoptotic cell populations, with no significant increase in necrosis. Hence, reinforcing the conclusion that MBZ primarily induces programed cell death in SKOV3 and PA1 cells.

MBZ induces apoptosis in SKOV3 and PA1 ovarian cancer cells.
MBZ triggers apoptosis via XIAP-mediated intrinsic mitochondrial pathway
The treatment of SKOV3 and PA1 OC cell lines with MBZ significantly altered the expression of proteins involved in Δψm, leading to apoptosis. To elucidate the molecular mechanism underlying the anticancer effects of MBZ in OC, we investigated the expression of key proteins involved in apoptosis in SKOV3 and PA1 cell lines following 48-hour treatment with MBZ at their respective IC50 concentrations. Western blot analysis revealed significant alterations in the fold change expression of XIAP, cytochrome C, Bcl-2, Bax, and cleaved-caspase-3. In SKOV3 cells treated with 1.25 µM MBZ (IC50), a significant reduction in antiapoptotic proteins was observed, with XIAP expression decreasing by approximately 0.38-fold (p < 0.001) compared with the DMSO control. Concomitantly, there was a marked increase in pro-apoptotic proteins, cytochrome C levels were elevated 2.4-fold (p < 0.001), indicating mitochondrial release into the cytosol, Bax showed a 2.5-fold increase (p < 0.001), while cleaved-Caspase-3, an executioner caspase, demonstrated a 2.2-fold upregulation (p < 0.001) as compared with the DMSO control. Furthermore, the expression of Bcl-2, an antiapoptotic protein, showed a significant decrease in expression to approximately 0.45-fold (Figure 5A, B). Similar trends were observed in PA1 cells treated with 0.312 µM MBZ (IC50). Antiapoptotic XIAP and Bcl-2 levels were significantly reduced by approximately 0.35-fold (p < 0.001) and 0.38-fold (p < 0.001), respectively, as compared with the control. Conversely, the level of pro-apoptotic protein expression was markedly elevated, with cytochrome C levels increased to 1.8-fold (p < 0.001), Bax expression was upregulated to 2.0-fold (p < 0.001), and cleaved-caspase-3 levels elevated to 2.0-fold (p < 0.001) compared with the VCs (Figure 5C, D). The observed protein expression changes were consistent across both cell lines, though PA1 cells exhibited a slightly more pronounced reduction in antiapoptotic proteins and a marginally lower induction of pro-apoptotic factors compared with SKOV3 cells. These results indicate that MBZ treatment activates the mitochondrial-mediated apoptotic pathway (intrinsic apoptotic pathway) in both OC cell lines, characterized by downregulation of antiapoptotic XIAP and Bcl-2, release of cytochrome C, upregulation of pro-apoptotic Bax, and activation of cleaved-caspase-3. Furthermore, the MBZ-induced downregulation of XIAP could potentially contribute to the observed alterations in Δψm, an early event in apoptosis, further amplifying the apoptotic signaling cascade. The consistent downregulation of XIAP in both cell lines, coupled with the robust induction of apoptosis, strongly suggests that targeting XIAP is a key mechanism underlying the anticancer activity of MBZ in OC.

MBZ induces apoptosis via XIAP-mediated mitochondrial pathway in ovarian cancer cells.
In silico investigation: MBZ’s interaction with XIAP and comparison with co-crystallized inhibitor ASTX660 (A4E)
XIAP represents one of the most potent endogenous caspase inhibitors, playing a crucial role in regulating Δψm and programed cell death pathways. Our experimental findings with OC cell lines (SKOV3 and PA1) demonstrated that MBZ treatment disrupts Δψm, leading to decreased XIAP protein expression and subsequent activation of the intrinsic apoptotic pathway. To further understand the molecular basis of these observations and elucidate whether MBZ directly interacts with XIAP, we conducted comprehensive in silico molecular docking analyses to compare the binding affinity of MBZ with the co-crystallized XIAP inhibitor. The XIAP protein structure (Figure 6A) was retrieved from the Protein Data Bank (PDB ID: 5OQW), and its Chain A was utilized for docking studies (Figure 6B). The active site was defined based on the position of the co-crystallized ligand A4E (ASTX660), and a grid box was generated around the binding pocket to ensure precise docking (Figure 6C). The molecular docking procedure was conducted with high precision by employing a GRID-based approach and the docked ligands were visualized to analyze their binding poses and interactions within the active site. As illustrated in Figure 6D, both the ligand (named as A4E_Redock and MBZ_Dock) successfully maneuvered into the binding pocket of XIAP. Quantitative assessment of binding energies yielded docking scores of -8.1 kcal/mol for A4E_Redock and -7.6 kcal/mol for MBZ_Dock. This relatively small difference in binding energy (0.5 kcal/mol) suggests that MBZ has comparable binding affinity to the purpose-designed XIAP inhibitor. Furthermore, the detailed 3 D and 2 D pocket interaction analyses (Figure 6E) revealed that the co-crystallized inhibitor A4E_Redock establishes an extensive network of noncovalent interactions within the XIAP binding pocket. Specifically, conventional hydrogen bonds were formed with THR308 and ASP309, which serve as anchor points stabilizing the ligand-protein complex. These hydrogen bonds are particularly important as they provide directional interactions that confer specificity to the binding. Carbon-hydrogen bonds were formed with GLN319, LEU307, GLU314, and TRP323, although weaker than conventional hydrogen bonds, but contributing incrementally to the overall binding affinity. In addition, salt bridge interactions were observed between A4E_Redock and GLY306 and TYR324. These electrostatic interactions provide substantial binding energy contributions and are often critical for high-affinity binding. The π-cation interactions formed with GLU314 enhance binding stability through favorable electrostatic interactions between the positively charged lysine residue and the electron-rich π systems of the inhibitor. A4E_Redock also formed π-alkyl interactions with TRP323, LYS297, and VAL298. Conversely, MBZ_Dock exhibited a remarkably similar binding pattern with some notable interactions with the critical residues of XIAP (Figure 6F). It formed conventional hydrogen bonds with THR308, GLU314, GLN319, TRP323, and LYS322, which are consistent with the key residues identified in the binding pocket. The formation of hydrogen bonds with THR308 is particularly significant, as this residue has been identified as crucial for XIAP’s interaction. MBZ also formed a salt bridge with GLY306, TYR324, and carbon–hydrogen bond interactions with LEU307. The benzimidazole moiety of MBZ formed π-sigma and π-alkyl interactions with TRP323. In addition, MBZ formed unique conventional hydrogen interactions with residues LYS322, which may contribute to its distinct mechanism of action. The overall interaction profile of both the ligand (A4E_Redock and MBZ_Dock) demonstrates a binding mode that effectively occupies the BIR3 domain binding pocket, with multiple interaction types ensuring both specificity and stability of binding (Table 2). Such strong binding affinity indicates a thermodynamically favorable interaction that would likely be physiologically relevant at therapeutic concentrations of MBZ, supporting our hypothesis that direct XIAP inhibition contributes to MBZ’s anticancer effects in OC cell lines.

In silico molecular docking of mebendazole (MBZ_Dock) and co-crystallized inhibitor (A4E_Redock) with XIAP (PDB: 5OQW).
Molecular docking results of mebendazole (MBZ_Dock) and ASTX660 (A4E_Redock) with XIAP (PDB ID: 5OQW)

SIFt analysis and docking validation: Enhanced structural insights and comparative interactions study
To thoroughly investigate the potential of MBZ as an XIAP inhibitor, we performed detailed molecular docking studies and binding interaction analyses. This visual comparison is fundamental to evaluating whether MBZ occupies a similar binding space as a known inhibitor and to assess the reliability of our docking protocol by examining the accuracy of the redocking process. Figure 7A illustrates the superimposed docking complex of the co-crystal ligand (A4E_CCL), MBZ (MBZ_Dock), and the redocked ligand (A4E_Redock) within the binding pocket of the XIAP protein (PDB ID: 5OQW-Chain A). This structural overlay reveals remarkable spatial concordance between MBZ and the known XIAP inhibitors, suggesting a shared binding mechanism despite their distinct chemical scaffolds, and suggests that MBZ can effectively mimic the interaction pattern of purpose-designed XIAP inhibitors. Furthermore, understanding which residues are involved in the interaction is crucial for elucidating the binding mode and potential mechanism of inhibition. Figure 7B visually identifies the amino acid residues within the XIAP BIR3 domain that form the interaction surface for the co-crystal ligand, MBZ, and the redocked ligand. These residues are: LEU292, LYS297, VAL298, LYS299, GLY306, LEU307, THR308, ASP309, TRP310, GLU314, GLN319, LYS322, TRP323, and TYR324. These residues align precisely with those identified in the 3 D and 2 D pocket interaction analyses of Figure 1E, F, which form hydrogen bonds, hydrophobic interactions, and π–π stacking with the ligands to ensure stable binding.

Structural interaction fingerprinting and docking validation of MBZ (MBZ_Dock) and A4E_Redock with XIAP.
Furthermore, SIFt analysis in Figure 7C highlights the interaction frequencies of these residues as a heat map and provides a comprehensive comparison of the interaction profiles of A4E_CCL, MBZ_Dock, and A4E_Redock with individual amino acid residues in the binding pocket. This detailed fingerprinting reveals both similarities and subtle differences in the binding modes of these compounds. All three compounds showed strong interactions with the critical residues THR308, GLN319, and TYR324, which have been established as essential for the binding of Smac-mimetic XIAP inhibitors. Notably, MBZ (MBZ_Dock) demonstrated interaction patterns remarkably like those of A4E_Redock, particularly with respect to GLY306, LEU307, THR308, GLU314, GLU319, TRP323, and TYR324. This similarity in interaction fingerprinting provides molecular-level evidence, supporting our hypothesis that MBZ can function as an effective XIAP inhibitor. In addition, MBZ exhibited distinctive interaction patterns with LYS299. These unique interactions further feature that MBZs could potentially contribute to specific inhibitory effects on XIAP function and subsequent activation of apoptosis in OC cells.
To validate the reliability of our docking protocol, we conducted a root-mean-square deviation (RMSD) analysis using the Biovia Discovery Studio tool, as shown in Figure 7D. The RMSD value of 0.321 Å between the co-crystal position of A4E (A4E_CCL) and its redocked position (A4E_Redock) demonstrates the high accuracy of our docking procedure. According to established protocols in computational drug discovery, RMSD values below 2.0 Å are considered reliable for docking simulations, with values below 1.0 Å indicating excellent predictive power. Our remarkably low RMSD of 0.321 Å therefore confirms the exceptional reliability of our docking protocol and significantly strengthens the validity of our findings regarding MBZ’s interaction with XIAP. The high accuracy in redocking the known ligand provides strong evidence that the docking protocol is capable of accurately predicting the binding mode of other ligands, including MBZ, to the same protein target.
Electronic insights into the binding potential of the ligands through MESP, Mulliken charges, and FMOs: DFT studies
The MESP mappings were systematically analyzed for A4E (A4E_Redock) and MBZ (MBZ_Dock) ligands to comprehensively evaluate their electron density distribution profiles within the molecules and serve as a crucial tool for predicting molecular interactions. As depicted in Figure 8A, B, the MESP maps utilize a color-coded gradient system to visualize the electrostatic potential variations. For A4E, the potential ranges from −0.185e0 (deep red) to 0.185e0 (deep blue), while MBZ exhibits a range from −0.132e0 to 0.132e0. The deep red regions in these mappings are particularly significant as they represent areas with the highest electronegative potential, making them prime sites for electrophilic attack and potential hydrogen bond acceptors in ligand-protein interactions.

DFT analysis: Electronic properties of A4E (A4E_Redock) and MBZ (MBZ_Dock).
To further validate the MESP analysis, we performed the Mulliken charge population analysis revealed specific charge distributions across the A4E (A4E_Redock) and MBZ (MBZ_Dock) molecules, identifying atoms with the most significant negative and positive charges. For A4E_Redock, the oxygen atoms O18 (−0.536), O12 (−0.459), and the nitrogen atoms N19 (−0.622), N27 (−0.594), N9 (−0.451), and N7 (−0.443) exhibited the highest negative charges. These electronegative atoms are likely to serve as hydrogen bond acceptors during interactions with a protein target. Conversely, the carbon atoms C17 (+0.459742) displayed the highest positive charges, suggesting potential sites for weak hydrogen bond donation (if bonded to hydrogen atoms) or for interaction with electron-rich regions of a protein. In the case of MBZ, the oxygen atoms O9 (−0.454), O22 (−0.427), O20 (−0.417), and the nitrogen atoms N15 (−0.477), N17 (−0.287), N18 (−0.264) carried the highest negative charges, again indicating their potential as hydrogen bond acceptors. The carbon atoms C19 (+0.627060), C16 (+0.605), and C21 (+0.321) exhibited the highest positive charges, suggesting they could interact with electron-rich regions of a target protein. The presence of multiple oxygen and nitrogen atoms with significant negative charges in MBZ highlights its potential for forming multiple hydrogen bonds like the A4E (Supplementary Table S1 and S2).
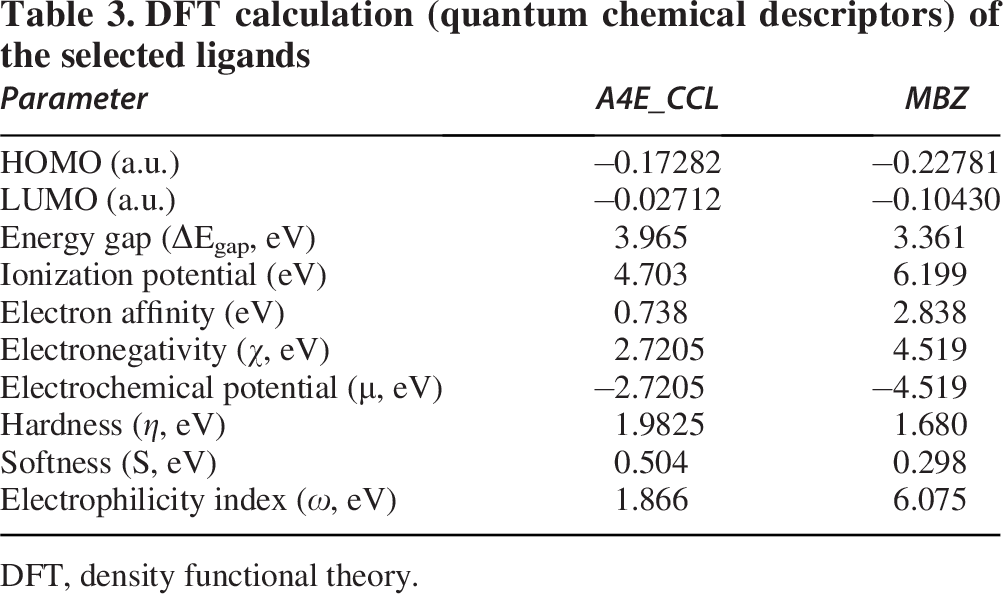
The HOMO-LUMO analysis offers crucial insights into the electron transition properties and chemical reactivity of A4E (A4E_Redock) and MBZ (MBZ_Dock). The Highest Occupied Molecular Orbital (HOMO) represents the molecule’s ability to donate electrons, while the Lowest Unoccupied Molecular Orbital (LUMO) indicates its ability to accept electrons. The energy difference between these orbitals, known as the HOMO-LUMO gap (
Further analysis of quantum chemical descriptors, shown in Table 3, provides additional insights into the reactivity and stability of the molecules. Electronegativity (
DFT calculation (quantum chemical descriptors) of the selected ligands
DFT, density functional theory.
Discussion
Ovarian carcinoma persists as a significant oncological challenge, primarily attributable to its frequently asymptomatic progression to advanced stages and the development of resistance mechanisms against conventional therapeutic modalities. 28 The present study investigates the potential of MBZ, an antihelminthic drug, as a novel therapeutic agent for OC by targeting ΔΨm disruption and apoptosis induction via the XIAP-mediated pathway. The phase contrast microscopy and the MTT assay provide complementary evidence regarding the effect of MBZ on SKOV3 and PA1 OC cell lines. Our results demonstrate that MBZ significantly inhibits the proliferation of both SKOV3 and PA1 OC cell lines in a dose-dependent manner. The observed reduction in cell density and the presence of morphologically altered cells in the MBZ-treated samples, as seen under the microscope, align with the quantitative results from the MTT assay, which demonstrate a significant decrease in cell viability in both cell lines. This concordance between qualitative and quantitative data correlates with the measured reduction in cell viability, suggesting a reliable effect of MBZ on the cancer cells and providing stronger evidence for the validity of the findings. Moreover, the IC50 values of 1.25 µM for SKOV3 and 0.312 µM for PA1 cells indicate that PA1 cells are more sensitive to MBZ compared with SKOV3 cells. This differential sensitivity could be attributed to variations in genetic and molecular profiles between the two cell lines. Our findings are consistent with previous studies that have highlighted the anticancer properties of MBZ. Morphological analysis revealed characteristic features of apoptosis, including cell rounding, shrinkage, membrane blebbing, and the formation of apoptotic bodies, which are consistent with previous studies on MBZ’s effects in other cancer types. For instance, Bai et al. reported similar morphological changes in glioblastoma cells treated with MBZ, further supporting its broad-spectrum anticancer potential. 29 Similarly, Doudican et al. demonstrated that MBZ induced apoptosis in melanoma cell lines with IC50 values ranging from 0.1 to 0.8 µM, comparable with our results with PA1 cells, and Pinto et al. reported that MBZ inhibited the growth of gastric cancer cells with IC50 values of 0.39–0.89 µM, further supporting our observations.30,31 In addition, Sung et al. highlighted MBZ’s ability to induce apoptosis in endothelial cancer cells, suggesting that MBZ targets common pathways across different cancer types. 32 Furthermore, the cytotoxic effects of MBZ observed in this study align with findings from Liang et al., who demonstrated that MBZ induces apoptosis and inhibits migration via the reactive oxygen species-mediated STAT3 signaling downregulation in nonsmall cell lung cancer. 33 Recent studies by Huang et al. specifically reported that MBZ inhibited SKOV3 cell growth with an IC50 of approximately 1 µM after 48 hours of treatment, which closely matches our finding of 1.25 µM. 34 These results collectively underscore the importance of investigating MBZ as a potential therapeutic agent for OC.
The significant disruption of Δψm observed in both SKOV3 and PA1 OC cell lines indicates that mitochondrial dysfunction is a key mechanism in MBZ-induced cytotoxicity. The findings from both the rhodamine-123 flow cytometry and JC-1 confocal microscopy assays consistently demonstrate that a 48-hour treatment with MBZ leads to a significant decrease in the ΔΨm of both SKOV3 and PA1 OC cell lines. This overall consistency provides robust evidence for the mitochondrial depolarizing effect of MBZ. The quantitative flow cytometry data revealed a substantial reduction in the MFI of rhodamine-123 in both cell lines following MBZ treatment, with approximately a 40% decrease observed in SKOV3 cells and a 33% decrease in PA1 cells. These results were further corroborated by the qualitative and quantitative analysis using JC-1 staining and confocal microscopy, which showed a clear shift from predominantly red fluorescence (representing JC-1 aggregates in healthy mitochondria with high ΔΨm) to increased green fluorescence (representing JC-1 monomers in cells with lower ΔΨm) in both cell lines after MBZ exposure. Quantitative analysis demonstrated a 9.9-fold increase in the green/red fluorescence ratio in SKOV3 cells and a 5.7-fold increase in PA1 cells, confirming substantial mitochondrial depolarization. Furthermore, these findings align with previous studies that have reported MBZ-induced mitochondrial damage in various cancer types. Doudican et al. demonstrated that MBZ causes mitochondrial dysfunction in lung cancer cells through oxidative stress. 30 Similarly, Li et al. showed that ivermectin disrupts ΔΨm in glioblastoma cells, leading to cytochrome c release and caspase activation. 35 Similarly, Emmings et al. reported that MBZ triggers mitochondrial depolarization and subsequent apoptosis in colon cancer cells. 36 In addition, Biswas et al. demonstrated that the combination of sulforaphene and cisplatin induced mitochondrial depolarization in SKOV3 cells, as assessed by rhodamine-123 flow cytometry. 37 Previously, direct experimental confirmation of MBZ-induced alterations in ΔΨm within the PA1 cell line has been lacking. However, our present investigation furnishes compelling evidence demonstrating significant MBZ-mediated ΔΨm disruption in PA1 cells. These findings parallel prior observations of MBZ-induced ΔΨm perturbation reported in the SKOV3 cell line. This novel finding, demonstrating MBZ’s efficacy across OC cell lines of differing origins, further broadens the understanding of its cellular mechanism of action and highlights its potential as a therapeutic agent in diverse OC subtypes. Moreover, our study provides direct evidence of this mitochondrial dysfunction by quantifying and visualizing the changes in ΔΨm using established fluorescent probes and analytical techniques. Furthermore, the dual methodological approach employed in this study that is flow cytometry with rhodamine-123 and confocal microscopy with JC-1, provides robust evidence for MBZ-induced mitochondrial dysfunction. The consistency between these complementary techniques strengthens the reliability of our findings and provides spatial insights into the mitochondrial changes that occur following MBZ treatment. This adds to the growing body of evidence supporting the potential repurposing of MBZ as a therapeutic agent for OC.
Altered Δψm after MBZ treatment aligns with the understanding that a decrease in Δψm is a hallmark of an early event in the apoptotic cascade. 14 The activation of this cascade triggers a series of biochemical events, including the translocation of phosphatidylserine from the inner to the outer leaflet of the plasma membrane. This exposed phosphatidylserine acts as an “eat me” signal for phagocytes and is readily bound by annexin-V, a calcium-dependent phospholipid-binding protein, which forms the basis of the annexin-V-FITC staining used in this study to detect apoptotic cells. 38 Annexin V-FITC/PI staining and flow cytometric analysis revealed a significant increase in early and late apoptotic populations in both SKOV3 and PA1 cells following MBZ treatment. In SKOV3 cells, MBZ treatment resulted in a 3.2-fold increase in early apoptotic cells (from 9.26% to 30.01%) and a 2.9-fold increase in late apoptotic cells (from 3.54% to 10.42%) compared with controls. In PA1 cells, MBZ treatment led to a 3.7-fold increase in early apoptotic cells (from 6.92% to 25.49%) and a 4.5-fold increase in late apoptotic cells (from 4.79% to 21.72%). Notably, there was no significant increase in necrotic populations, reinforcing the conclusion that MBZ primarily induces programed cell death rather than necrosis. The absence of a significant increase in necrotic cell populations suggests that MBZ induces a controlled apoptotic process rather than a necrotic one. Apoptosis is generally considered a more desirable mechanism of cell death in cancer therapy, as it is less likely to induce inflammation and promote tumor growth compared with necrosis. The observed significant induction of apoptosis in SKOV3 cells following 48-hour MBZ treatment aligns with findings from previous research. 28 Likewise, Huang et al. demonstrated that MBZ inhibited the growth of various OC cell lines, including SKOV3, and induced intrinsic apoptosis. Even in cisplatin-resistant SKOV3CR cells, MBZ has been shown to induce apoptosis, suggesting a robust mechanism of action that can bypass certain drug resistance mechanisms. 34 The substantial increase in both early and late apoptotic populations in SKOV3 cells observed in this study at the IC50 concentration further strengthens this evidence. Regarding the PA1 OC cell line, while the provided research snippets offer less direct evidence on MBZ-induced apoptosis specifically in PA1 cells, the findings of this study clearly demonstrate a significant increase in apoptosis at its IC50 concentration of 0.312 µM. This suggests that PA1 cells are also highly susceptible to the pro-apoptotic effects of MBZ. While studies have shown MBZ’s anticancer activity in other OC cell lines like OVCAR8, 34 the significant apoptosis observed in PA1 cells in this study contributes novel data to this area. PA1 cells, derived from a human ovarian teratocarcinoma, exhibit different characteristics compared with SKOV3 cells, which originate from an adenocarcinoma.39,40 The significant increase in both early and late apoptotic cells in PA1 cells at its IC50 concentration highlights the broad potential of MBZ as an anticancer agent across different histological subtypes of OC. The significant apoptotic effect of MBZ observed in both SKOV3 and PA1 OC cell lines underscores its potential as a therapeutic agent for this challenging disease. Given the ability of MBZ to overcome cisplatin resistance in OC cells, it holds promise for the treatment of both primary and chemoresistant ovarian tumors. The well-established safety profile and low cost of MBZ, as an anthelmintic drug used for decades, further enhance its attractiveness as a candidate for drug repurposing in the context of OC therapy.
In our previous experiments, we found that MBZ treatment significantly altered Δψm in both SKOV3 and PA1 OC cell lines in a dose-dependent way. This ΔΨm dysfunction impairs ATP generation, induces oxidative stress, and finally triggers the apoptotic cascade. 35 Western blot analysis revealed that MBZ significantly modulates the intrinsic apoptotic molecular signaling axis. The observed downregulation of XIAP, coupled with the upregulation of cytochrome C, Bax, and cleaved caspase-3, further elucidates the molecular mechanisms underlying this mitochondrial-led apoptosis, particularly highlighting the role of XIAP in this process. Our findings expand upon previous work by Nygren et al., who demonstrated that microtubule-targeting agents induce apoptosis through the intrinsic pathway in OC cells. 41 The present study specifically identifies XIAP as a key mediator in MBZ-induced apoptosis in OC, which has not been previously reported. This XIAP-mediated mechanism may explain the broad spectrum of anticancer activity of MBZ observed across different cancer types, as XIAP overexpression is common in many malignancies and associated with chemoresistance. 16 The observed significant reduction in XIAP expression following MBZ treatment represents a critical step in the initiation of apoptosis. XIAP is known to directly inhibit caspases 3, 7, and 9, thereby preventing apoptosis execution. 42 In addition, XIAP plays a crucial role in maintaining mitochondrial membrane integrity through its interaction with the mitochondrial protein SMAC/DIABLO. 43 Our findings of XIAP downregulation are consistent with those reported by Doudican et al., who demonstrated that MBZ induces apoptosis in melanoma cells through suppression of IAP family proteins. The reduction in XIAP levels likely contributes to the previously observed ΔΨm disruption through multiple mechanisms. First, diminished XIAP allows for the activation of pro-apoptotic factors that target mitochondrial membrane integrity. Second, as demonstrated, XIAP directly regulates mitochondrial physiology and can protect against mitochondrial dysfunction. 30 The significant decrease in XIAP expression observed in both cell lines (2.5-fold) may therefore represent an initiating event in MBZ-induced apoptosis. Concurrent with XIAP reduction, we observed decreased expression of the antiapoptotic protein Bcl-2, which is critical for maintaining mitochondrial membrane integrity. The Bcl-2 reduction likely contributes to mitochondrial outer membrane permeabilization, facilitating the release of cytochrome c, as evidenced by the 1.8-2.4-fold increase in cytosolic cytochrome c levels. This finding aligns with the work by Martarelli et al., who reported that MBZ induces cytochrome c release in adrenocortical carcinoma cells. The increased expression of pro-apoptotic Bax further supports the activation of the intrinsic apoptotic pathway. 44 This shift promotes mitochondrial membrane permeabilization and subsequent cytochrome c release, as demonstrated by Chen et al., in cancer cells treated with anticancer-targeting agents. 45 The final activation of caspase-3, as indicated by elevated cleaved caspase-3 levels, confirms the execution phase of apoptosis. This caspase activation is likely a consequence of both reduced XIAP inhibition and cytochrome c-mediated formation of the apoptosome. Similar caspase-3 activation following MBZ treatment has been reported by Pantziarka et al. in various cancer types. 46 The substantial inhibition of antiapoptotic proteins and induction of pro-apoptotic factors at clinically achievable concentrations (IC50 values of 1.25 µM for SKOV3 and 0.312 µM for PA1) support the potential repositioning of MBZ as an anticancer agent for OC.
The in vitro results demonstrate a clear sequence of events initiated by MBZ treatment: The disruption of Δψm in OC cell lines (SKOV3 and PA1), followed by a reduction in the expression levels of the antiapoptotic protein XIAP, and the subsequent activation of the intrinsic apoptosis pathway. This proposed mechanism highlights a novel aspect of MBZ’s action in OC, suggesting that its cytotoxic effects may be mediated, at least in part, through the perturbation of mitochondrial function and the downregulation of XIAP. The central role of XIAP in this process is underscored by its position as a key inhibitor of apoptosis, acting downstream of mitochondrial stress in this context. The novelty of identifying XIAP downregulation by MBZ as a key contributing factor in this specific context warrants further investigation into the precise molecular interactions between MBZ and XIAP. The in silico docking analysis further supports the possibility of a direct interaction between MBZ and the XIAP protein. Molecular docking studies revealed that MBZ exhibits comparable binding affinity (−7.6 kcal/mol) with the co-crystallized XIAP inhibitor ASTX660 (A4E_Redock, −8.1 kcal/mol). The comparable binding energies suggest that MBZ binds to XIAP with an affinity like that of the known XIAP inhibitor, supporting our hypothesis that MBZ may directly interact with XIAP to inhibit its antiapoptotic function. Detailed analysis of the binding interactions revealed that both ligands bind to the same pocket of XIAP. Both ligands formed similar interactions with key residues in the XIAP binding pocket, including hydrogen bonds with THR308, GLU314, GLN319, TRP323, and LYS322, as well as salt bridges and π-alkyl interactions. SIFt analysis highlighted the similarity in interaction patterns between MBZ_Dock and A4E_Redock, particularly with respect to critical residues such as GLY306, LEU307, THR308, GLU314, GLU319, TRP323, and TYR324. In addition, MBZ formed unique interactions with LYS299, suggesting its potential to exert distinct inhibitory effects on XIAP function. These interactions are particularly significant as they involve residues known to be crucial for the inhibition of XIAP’s antiapoptotic function. Our findings are consistent with previous studies on XIAP inhibitors. For instance, Ye et al. reported that ASTX660 inhibitors targeting the BIR3 domain of XIAP interact with critical residues, including GLY306, LEU307, THR308, and GLU314, which are the same residues we identified in our docking analysis for MBZ. 47 Similarly, Wu et al. demonstrated that effective XIAP inhibitors form hydrogen bonds with THR308 and hydrophobic interactions with LEU307, which were also observed in our MBZ docking model. 48 Furthermore, the binding mode of MBZ shows interesting similarities with both peptidic and nonpeptidic XIAP inhibitors described in the literature.49,50 Furthermore, MBZ interacts with the critical residues in the BIR3 domain hydrophobic groove, like the Smac-derived peptides, as reported previously.51,52 However, its smaller size and nonpeptidic nature may confer advantages in terms of bioavailability and resistance to proteolytic degradation. Compared with other small molecule XIAP inhibitors such as those reported earlier,16,53 MBZ exhibits a comparable interaction profile but with a distinct chemical scaffold. This structural novelty could prove valuable for overcoming resistance mechanisms that might develop against other XIAP inhibitors and provides a new starting point for medicinal chemistry optimization efforts. Considering the existing literature on MBZ’s anticancer effects, the predicted interaction with XIAP could offer a novel perspective on its mechanism of action.
The DFT analysis provides insights into the electronic properties of A4E and MBZ ligands through MESP, Mulliken charge distribution, and FMO analysis. MESP mapping revealed significant electronegative regions in both ligands, indicating potential hydrogen bonding sites, consistent with prior studies on quinazoline derivatives. 54 Mulliken charge analysis identified oxygen and nitrogen atoms in A4E and MBZ with high negative charges, suggesting a strong hydrogen bond acceptor potential. These findings align with previous research on bis-thiazolidine amide derivatives, where charge distributions were key determinants of binding interactions. 55 The HOMO-LUMO analysis indicated that MBZ, with a lower energy gap (3.361 eV), showed similar or more reactivity than A4E (3.965 eV). The distribution of HOMO and LUMO suggests MBZ’s higher electrophilicity, making it more prone to intermolecular interactions. These observations align with reports that lower energy gaps enhance molecular reactivity. 56 In addition, quantum descriptors confirmed MBZ’s superior reactivity, with a higher electrophilicity index (6.075 eV) and electronegativity (4.519 eV), reinforcing its potential for biological interactions. Previous studies on flavonoid glycosides have shown similar trends where higher electrophilicity correlated with stronger ligand-protein binding.54,56 Furthermore, this comparative analysis is the first to report FMO and MESP of MBZ and A4E (co-crystal ligand of XIAP). Overall, the electronic properties of MBZ suggest a comparable binding potential and reactivity with A4E for XIAP, which is supported by the observed DFT parameters. These results support DFT as a predictive tool in ligand-protein interaction studies.
Our present work also provides important distinctions from our earlier study. That report, conducted in OVCAR3 and OAW42 OC cells, primarily emphasized MBZ’s inhibition of the Girdin–AKT/IKKα/β–NF-κB signaling cascade, with mitochondrial depolarization and apoptosis observed as downstream consequences. 17 In contrast, the current investigation employs SKOV3 (adenocarcinoma) and PA1 (teratocarcinoma) cells, thereby broadening the spectrum of OC models studied. Here, we identify the mitochondrial–apoptotic axis as the primary mechanistic target, establishing that MBZ directly disrupts ΔΨm and suppresses XIAP, leading to caspase-dependent apoptosis. Importantly, unlike our previous work, this study also integrates an in silico validation framework, including molecular docking, dynamics, and quantum chemical analyses, to corroborate MBZ–XIAP interactions. This methodological innovation strengthens translational significance by providing structural evidence in support of experimental findings. Together, these complementary but distinct lines of evidence show that MBZ exerts nonredundant, dual mechanisms across different OC subtypes-suppressing invasive signaling pathways on one hand and dismantling intrinsic apoptosis resistance on the other. This mechanistic diversity underscores the novelty of the present work and highlights MBZ’s potential as a broad-spectrum, repurposed therapeutic for OC.
Conclusion
Our study shows that MBZ suppresses the proliferation of SKOV3 and PA1 OC cells at clinically achievable concentrations (IC50: 1.25 µM and 0.312 µM, respectively). We identify Δψm disruption and XIAP downregulation as key mechanisms by which MBZ promotes apoptosis, representing a previously unreported pathway in OC. This mitochondrial dysfunction triggers the intrinsic apoptotic cascade, marked by caspase activation and loss of XIAP activity. Supporting this, in silico docking indicates that MBZ binds directly to XIAP with an affinity comparable to established inhibitors, engaging critical residues within the BIR3 domain. Complementary DFT analyses further confirm MBZ’s favorable electronic features for protein interaction. Notably, this work highlights XIAP inhibition and mitochondrial collapse as a central mechanistic route in SKOV3 and PA1 cells. The incorporation of computational docking and quantum analyses, absent in the earlier study, provides structural validation and strengthens translational significance. Together, these findings lay a strong mechanistic basis for MBZ’s anticancer potential in OC. Future studies should extend to in vivo models, explore combination regimens, and advance toward clinical testing, particularly in resistant disease settings. Given its well-established safety record and low cost, MBZ remains a compelling candidate for therapeutic repurposing.
Authors’ Contributions
R.G.: Conceptualization, data curation, formal analysis, investigation, methodology, software, validation, visualization, writing—original draft, review and editing.
Footnotes
Acknowledgments
The author is grateful for the resources, facilities, and support provided by the institution and acknowledges the UGC and GoI for the NET Fellowship. The author gratefully acknowledges Netaji Subhas Chandra Bose Cancer Hospital, Kolkata, for granting access to the Flow Cytometry Instrument facility.
Author Disclosure Statement
The author declare no conflicts of interest.
Funding Information
This research received no external funding.
Data Availability Statement
All data presented in the article are available with the author and can be given upon reasonable request.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
