Abstract
Background
Nirmatrelvir/Ritonavir (nir/rit) is an antiviral combination, often sold under the brand name Paxlovid, used to treat COVID-19, particularly in high-risk individuals. Numerous COVID-19 treatments exist, but clinical studies have reported conflicting data on their efficacy, particularly in unvaccinated patients.
Objectives
This study aims to compare 28-day mortality, hospitalization, intensive care unit (ICU) admission, observation status, and hypoxia rates among unvaccinated COVID-19 patients treated with nir/rit in the emergency department (ED) versus those who did not receive nir/rit.
Design
This retrospective, propensity-matched cohort study utilized the TriNetX database, which included records of 112 million patients from 64 academic medical centers and healthcare organizations across the U.S. The study period was 2022–2024.
Methods
The treatment cohort consisted of COVID-19-unvaccinated ED patients aged 18 and above who tested positive for COVID-19 via RNA test and were administered nir/rit on the same day. Patients admitted for observation or inpatient care on the same day were excluded. Outcomes assessed within 28 days post-ED diagnosis included mortality, ICU admission, inpatient hospitalization, observation status, and hypoxia (O2 saturation ≤90%). Propensity score matching adjusted for demographics and 21 pre-existing conditions associated with COVID-19 severity and mortality.
Results
A total of 194 181 unvaccinated COVID-19 patients were identified, with 14 799 receiving nir/rit and 179 382 not receiving it. After propensity matching, 14 799 patients were included in each cohort. Paxlovid treatment was associated with lower mortality (risk ratios [RR] = 0.28, P < .001), fewer ICU admissions (RR = 0.53, P = .01), decreased inpatient visits (RR = 0.40, P < .001), reduced observation status (RR = 0.36, P = .004), and less hypoxia (RR = 0.49, P < .001). Trends were similar before propensity matching.
Conclusions
Nir/rit treatment in unvaccinated adults diagnosed with COVID-19 in the ED is associated with significantly lower mortality, ICU admissions, hospitalizations, observation status, and hypoxia, supporting its use in this patient population.
Plain Language Summary Title
Plain Language Summary
Introduction
Since the emergence of COVID-19 in December 2019, there have been a total of 776 million positive cases and over seven million COVID-19-related deaths worldwide as of July 2024. 1 The World Health Organization (WHO) issued a strong recommendation for nirmatrelvir/ritonavir (nir/rit) (eg, Paxlovid) use in high-risk COVID-19 patients in 2022 based on early trial data, while the U.S. Food and Drug Administration (FDA) granted full regulatory approval in 2023 following a complete review of efficacy and safety data.2,3,4 Nir/rit is an oral antiviral treatment composed of nirmatrelvir and ritonavir, designed to treat COVID-19 in individuals at risk of severe outcomes. Nirmatrelvir inhibits the main polyprotein protease (Mpo) of SARS-CoV-2, preventing replication of the virus, while ritonavir, an HIV-1 and HIV-2 protease inhibitor, irreversibly inhibits CYP3A, thereby increasing nirmatrelvir concentrations. 5 While past studies have shown mixed results regarding the efficacy of nir/rit, 5 unvaccinated COVID-19 patients typically face a poorer prognosis.
To date, there are a multitude of treatment options for COVID-19, ranging from supportive care to antiviral medications and monoclonal antibodies. However, there has been conflicting data from clinical studies conducted regarding the efficacy of the various treatment options. One study stated that many treatment options, prior to nir/rit, did not show effective reduction in the risk of progression to severe disease in positive patients, or were too expensive and difficult to treat on a large scale. 6 This same study reported that nir/rit showed a significant decrease in reducing the progression of severity in high-risk patients with an hazard ratios (HR) of 0.54. 6
Several studies have analyzed the efficacy of nir/rit in comparison to other treatment options, such as sotrovimab, in vaccinated populations and noted no substantial difference in the risk of developing severe COVID-19 between the two medications. 7 Recent observational studies have consistently shown that nir/rit is generally more effective than molnupiravir in reducing hospitalization and mortality, particularly among older or high-risk patients, regardless of vaccination status.8,9,10,11 In a recent study that analyzed the safety and efficacy of nir/rit in elderly patients, there was a significant difference in the nucleic acid shedding time of patients on nir/rit (9.32 days) and the control group not on nir/rit (11.11 days). 12
Additionally, given the high acquisition and administrative costs of COVID-19 therapeutics, the sustainability of their widespread use remains an important consideration. Pharmacoeconomic analyses suggest that nir/rit offers comparable efficacy to other antivirals, such as remdesivir, with meaningful cost savings, underscoring the need to interpret clinical outcomes alongside long-term financial impact.
The primary objective of this study is to evaluate the outcomes of unvaccinated COVID-19 patients who were treated with nir/rit in the emergency department (ED), compared to those who did not receive the medication.
Methods
TriNetX is a global federated health research network providing de-identified access to retrospective electronic health records (diagnoses, procedures, medications, lab values, genomic information) from over 112 million patients in 64 large healthcare organizations (HCOs) within the United States. In this study, we used the US Collaborative Network to identify patients who were unvaccinated against COVID-19 and were evaluated in the ED. The two cohorts identified for this study were Cohort 1, unvaccinated patients who received treatment with nir/rit in the ED, and Cohort 2, unvaccinated patients who did not receive treatment with nir/rit in the ED.
Cohort Selection
We identified COVID-19 patients of all ethnicities, races, and genders using TriNetX Code TNX 9088 (SARS coronavirus 2 and related RNA (Presence), Lab Result: Positive). In the database, there were 14 799 unvaccinated patients who received nir/rit, and 179 382 unvaccinated patients who did not receive nir/rit, using RxNORM:2587892 for nirmatrelvir and RxNORM:85762 for ritonavir. The dataset was limited to those patients whose index event occurred on or after January 1, 2022, through March 24, 2024. Two cohorts were generated with these presets. Cohort 1 consisted of unvaccinated adults (SARS-CoV-2 Vaccine, NLM:CVX:213) with a COVID-19 diagnosis (TNX:9088) who were administered Paxlovid (RxNORM:2587892, RxNORM:85762), resulting in 14 799 patients. Cohort 2 consisted of unvaccinated adults with a COVID-19 diagnosis who were not treated with Paxlovid, resulting in 179 382 patients. In addition, patients admitted to observation or inpatient care on the day of the ED visit were excluded from analysis.
Outcomes
We performed the outcome analysis between the two cohorts for five events: death, intensive care unit (ICU) (admission or critical care procedural time), inpatient encounter, observation encounter, and O2 sat ≤90%. This analysis included outcomes that occurred in the time window that started 1 day after the first occurrence of the index event and ended 28 days after the first occurrence of the index event. Patients with outcomes prior to or after the time window were excluded from the analysis.
To control potentially confounding risk factors for the measured outcomes, we performed propensity score matching based on demographics, including age at diagnosis, race, ethnicity, and gender. Additional propensity matching was performed for pre-existing conditions associated with mortality and COVID-19 severity including: COPD (ICD-10 code J44), asthma (J45), malignant neoplasms of lymphoid, hematopoietic and related tissue (C81-96), fibrosis and cirrhosis of the liver (K74), diabetes mellitus (E08-E13), Human immunodeficiency virus (HIV) disease (B20), unspecified dementia (F03), dementia in other diseases classified elsewhere (F02), vascular dementia (F01), schizophrenia (F20), certain disorders involving the immune mechanism (D80-D89), nicotine dependence (F17), pregnancy, childbirth and the puerperium (O00-O9A), cystic fibrosis (E84), hypertensive diseases (I10-I1A), acute kidney failure and chronic kidney disease (N17-N19), ischemic heart diseases (I20-I25), acute myocardial infarction (I21), cerebral infarction (I63), overweight and obesity (E66), and malignant neoplasm of the bronchus and lung (C34). 13 We used the balanced cohort tool in TriNetX for matching.
Statistical Analysis
We performed univariate analysis using the measure-of-association tool in TriNetX, which compares outcomes within the designated time frames for each cohort reported both as risk ratios (RR), odds ratios, 95% confidence intervals (CIs), and P-values. We obtained de-identified patient data from the TriNetX US Collaborative Network database on March 24, 2024, and we performed the data analyses on the same date. The TriNetX platform provides access to aggregated counts and statistical summaries of de-identified patient records. No protected health information or personal data is available to platform users; therefore, this project was exempt from the institutional review board review (www.trinetx.com). This methodology has been previously validated and currently follows STROBE guidelines for observational cohort studies.14,15
Results
We identified 112 million patients in the TriNetX United States Collaborative Network from 63 academic medical centers/ HCOs. A total of 194 181 COVID-19 unvaccinated patients were identified. In Cohort 1 of unvaccinated patients treated with nir/rit in the ED, we identified 14 799 patients. In Cohort 2 of unvaccinated patients not treated with nir/rit in the ED, there were 179 382 patients. Most demographic variables, except for gender (male/female), American Indian or Alaskan Native race, and Unknown race, were significantly different between the two cohorts. Similarly, most pre-existing medical conditions associated with COVID-19 mortality showed statistically significant differences between cohorts (Tables 1 and 2).
Demographics of Cohort 1 and Cohort 2 before and after propensity matching.

SD, standard deviation.
Characteristics of Cohort 1 and Cohort 2 before and after propensity score matching.
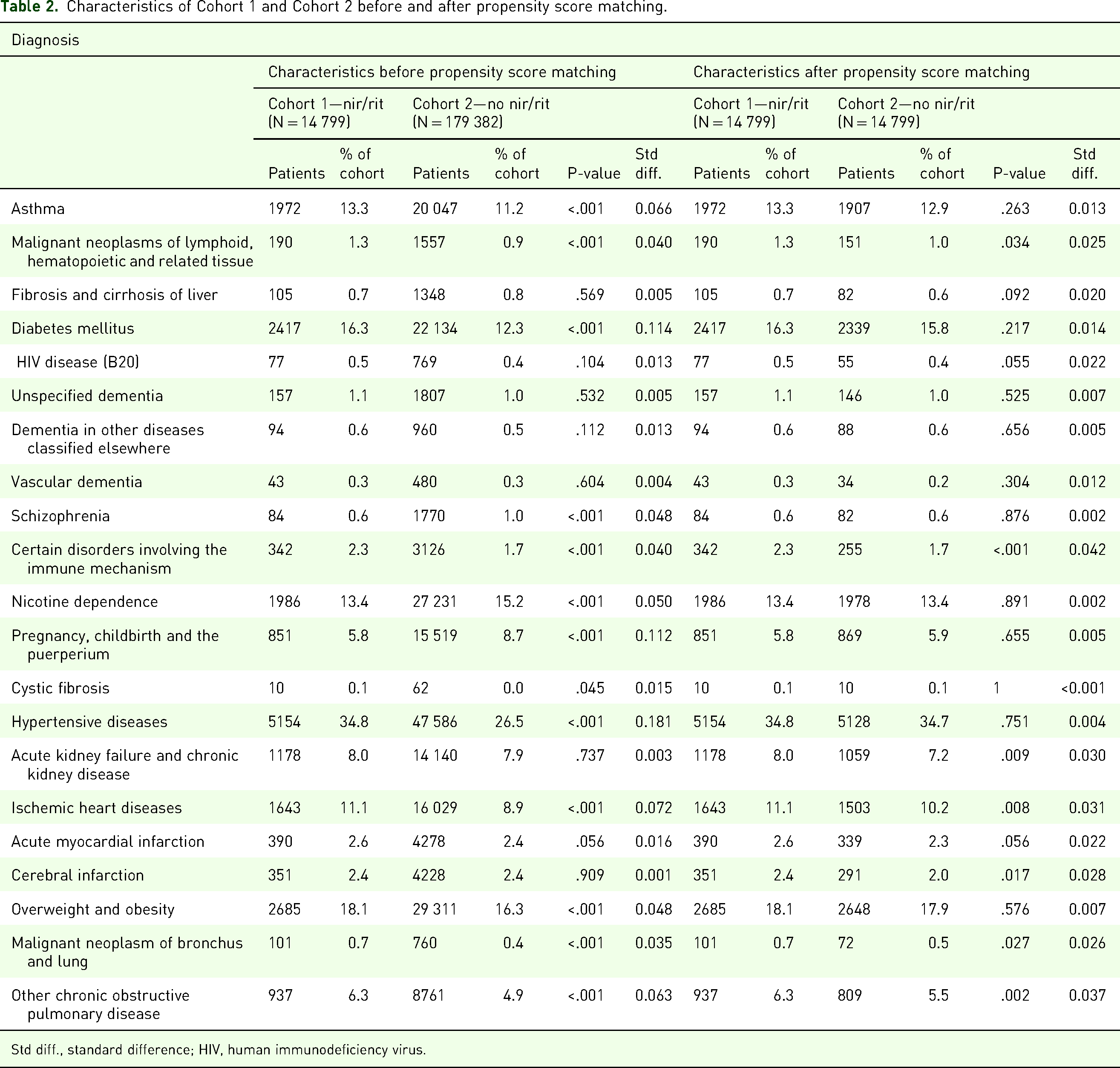
Std diff., standard difference; HIV, human immunodeficiency virus.
After exclusions and propensity score matching, basic demographic information and prior clinical diagnostic groups associated with mortality, there were a total of 14 799 patients evenly matched between the nir/rit and no nir/rit groups. Mean age in Cohorts 1 and 2 before matching was 55.2 and 46.6, respectively. After matching, the mean ages were 55.2 and 55.3, respectively. After matching, the male-to-female ratio was 1:1.51 in Cohort 1 and 1:1.51 in Cohort 2. The frequency of common comorbidities is included in Table 2. After matching, nearly all demographic variables and pre-existing conditions were balanced, with no statistically significant differences remaining between groups (Tables 1 and 2). It should be noted that TriNetX reports infrequent outcomes (≤10 events) as 10; therefore, the observed difference for the cystic fibrosis group may be slightly underestimated.
After propensity matching, unvaccinated patients treated with nir/rit in the ED were found to have reduced mortality (0.1% vs 0.4%, RR = 0.28, CI [0.166, 0.472], P < .001), reduced ICU admissions or critical care billing (0.2% vs 0.3%, RR = 0.53, CI [0.327, 0.873], P = .011), reduced inpatient admissions (0.7% vs 1.8%, RR = 0.40, CI [0.307, 0.514], P < .001), reduced observation admissions (0.1% vs 0.2%, RR = 0.36, CI [0.175, 0.745], P = .004), and reduced risk of O2 saturation being ≤90% (0.3% vs 0.6%, RR = 0.49, CI [0.336, 0.702], P < .001) compared to the unvaccinated patients not treated with nir/rit (Table 3)
Comparison of outcomes in Cohort 1 and Cohort 2 before and after propensity score matching.
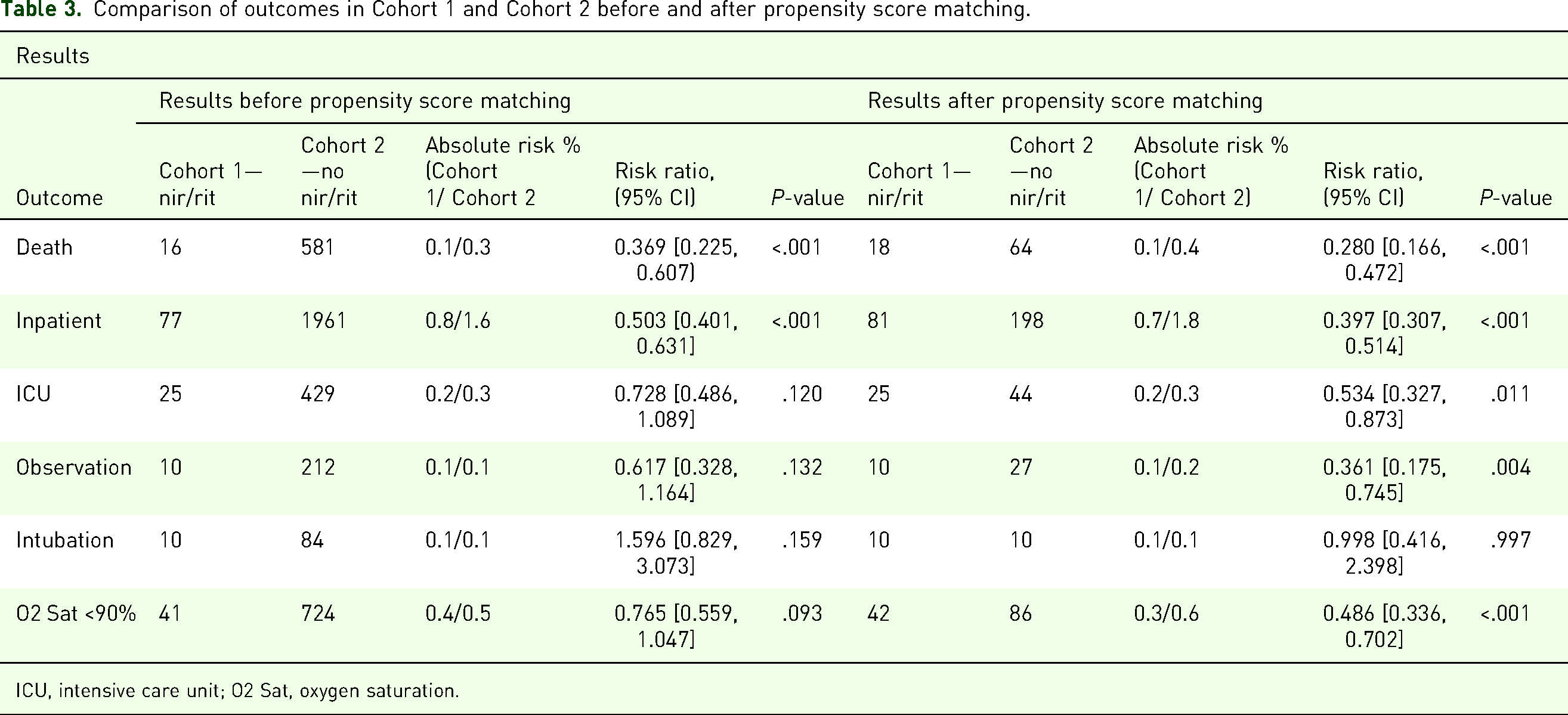
ICU, intensive care unit; O2 Sat, oxygen saturation.
Discussion
This large, multicenter, propensity-matched, retrospective cohort study analyzed the effects of nir/rit on unvaccinated adults with COVID-19. The results demonstrated that nir/rit was associated with significantly reduced mortality, ICU admissions, hospitalizations, observation status, and hypoxia in these patients. The strength of this study comes from its size and propensity matching for age, sex, race/ethnicity, and 21 diseases that the CDC reports are associated with COVID-19 severity and mortality, 13 including ischemic heart disease, cerebrovascular disease, malignant neoplasms of the lung, COPD, and so forth (Table 2).
This study demonstrated that all-cause mortality was significantly lower in unvaccinated adults treated with nir/rit compared to those who did not receive the antiviral. Previous research has consistently shown that Paxlovid reduces mortality in vaccinated individuals10,16 and supports its efficacy in unvaccinated populations as well,17,18 though studies focusing on unvaccinated individuals often involve smaller sample sizes. Additionally, some studies have reported higher mortality rates than observed in our analysis. For instance, a 2023 study by Xie reported a mortality rate of 5.97% among hospitalized COVID-19 patients and included other antiviral therapies. 19 Our findings provide further evidence that nir/rit's mortality benefits extend to unvaccinated individuals. This supports its use in the ED and strengthens clinician confidence in prescribing it for this population.
In addition to mortality, this study also examined ICU admissions and found that nir/rit use in unvaccinated adults led to fewer ICU admissions. A 2023 meta-analysis demonstrated reduced ICU admissions with nir/rit, 20 but it did not differentiate by vaccination status. Our study builds on this by showing that unvaccinated individuals also benefit from reduced ICU admissions. Contradictory literature exists, such as the study by Cai et al, which found no reduction in ICU admissions in patients with acute kidney injury (AKI) treated with nir/rit. 21 However, our study controlled for AKI and chronic kidney disease through propensity matching, enhancing its power to detect true differences. These results suggest that prescribing nir/rit in the ED can reduce ICU admissions.
In addition to ICU admissions, we investigated inpatient admissions and observation status. This study found that both hospitalizations and observation status were reduced in unvaccinated adults prescribed nir/rit. Previous research has supported reduced inpatient admissions,22,23 which our results reinforce. However, some literature, such as the study by Chew et al, contradict our results, suggesting no significant difference in hospitalization 28 days post-diagnosis. 2 This study's larger sample size significantly enhances the power of the results across all-cause mortality, ICU admissions, and inpatient/observation admissions. Compared to the contradictory findings of Chew et al, our cohort was nearly 90 times larger, allowing the opportunity to strengthen or challenge prior studies conducted with limited sample sizes and lower statistical power. 2 Paxlovid may also help prevent long-term post-COVID-19 sequelae and preserve work ability in high-risk patients, as these complications are more likely to develop following severe disease or hospitalization. 24 To our knowledge, no prior research has examined observation status outcomes for nir/rit, making our findings the first to report on this outcome.
This study also investigated hypoxia and found that nir/rit reduced its incidence in unvaccinated adults. Yang et al suggested that mechanical ventilation significantly improved hypoxia in COVID-19 patients and found further improvement with Paxlovid, attributing it to reduced viral load and tissue injury. 25 We found that nir/rit alone improved hypoxia in unvaccinated adults. To our knowledge, no previous research has specifically examined nir/rit's effects on hypoxia. Further research could compare the improvements between nir/rit and mechanical ventilation.
Limitations
This is a retrospective cohort study, and as such, we can only make claims of correlation, not causation. Additionally, due to the privacy policies of TriNetX, we are unable to assess the clustering of data among the various sites from which our analysis is drawn. Another limitation is that our data comes from the HCOs within the US Collaborative Network of the TriNetX database, which does not account for external hospitals or organizations that are not affiliated. This may introduce a selection bias and limit the generalizability of the findings for international populations.
Although we have propensity-matched for basic demographic characteristics and 21 comorbid conditions, there may be other conditions that introduce confounding biases to our data. Those who were not given nir/rit may have been given alternative treatments such as NRICM101, which has primarily been used outside of the US. Lack of stratification in covariates may have also impacted the outcomes, such as in patients with obesity or hypoalbuminemia, in which higher classifications have been shown to be correlated with increasing COVID-19 severity.26,27
While nir/rit has demonstrated significant reductions in mortality and severe outcomes, its long-term sustainability remains a concern due to high costs and access limitations. The high retail cost of medications is often attributed to the expenses of drug development and clinical trials, and nir/rit is no exception, with a 5-day course that remains costly and without a generic alternative. Although co-pay assistance programs are available for eligible individuals, processing delays may cause some patients to miss the drug's effective treatment window. The price of these medications may limit use among self-pay patients; however, assistance programs can substantially reduce costs for those with Medicaid or without insurance. Additionally, some studies have shown that nir/rit was already more cost-effective than other antivirals for COVID-19 and may further help to reduce overall costs through reductions in hospitalization.28,29 Regardless, future efforts should focus on optimizing cost-effectiveness, streamlining access programs, and ensuring equitable distribution to promote sustainable antiviral use.
Conclusions
Nir/rit is a novel therapeutic for COVID-19, and with rising vaccine hesitancy, it is crucial to assess its effects in both vaccinated and unvaccinated individuals. Clinicians have been using their judgment in prescribing nir/rit, and our study complements existing research by focusing on unvaccinated adults. We provide evidence supporting the prescription of nir/rit to high-risk unvaccinated patients, as it results in reduced mortality, ICU admissions, hospitalizations, observation status, and hypoxia.
Supplemental Material
sj-docx-1-cra-10.1177_29768675251413432 - Supplemental material for Effectiveness of Nirmatrelvir/Ritonavir in Unvaccinated COVID-19 Patients Treated in the Emergency Department: A Retrospective Propensity-Matched Cohort Analysis
Supplemental material, sj-docx-1-cra-10.1177_29768675251413432 for Effectiveness of Nirmatrelvir/Ritonavir in Unvaccinated COVID-19 Patients Treated in the Emergency Department: A Retrospective Propensity-Matched Cohort Analysis by Dietrich von Kuenssberg Jehle, Raheed Sunesra, Shania M Williams, Nathaniel J Bilby, Krishna K Paul and Kelcie Hill in Therapeutic Advances in Pulmonary and Critical Care Medicine
Footnotes
Author Contribution(s)
Availability of Data and Materials
Data and materials are available upon request and approval through the TriNetX database.
Ethics Approval and Consent to Participate
Any data displayed on the TriNetX Platform in aggregate form, or any patient-level data provided in a data set generated by the TriNetX Platform, only contains de-identified data as per the de-identification standard defined in Section §164.514(a) of the HIPAA Privacy Rule. The process by which the data is de-identified is attested to through formal determination by a qualified expert as defined in Section §164.514(b)(1) of the HIPAA Privacy Rule. This formal determination by a qualified expert, refreshed in December 2020, supersedes the need for TriNetX's previous waiver from the Western Institutional Review Board (IRB). Additionally, the University of Texas Medical Branch IRB waived the need for ethics approval and patient consent for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this non-interventional study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the UTMB Institute for Translational Sciences, supported in part by a Clinical and Translational Science Award (UM1 TR005443) from the National Center for Advancing Translational Sciences at the National Institutes of Health (NIH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
