Abstract
Nirmatrelvir/ritonavir is a novel drug combination that is authorized by the Food and Drug Administration for the treatment of coronavirus disease 2019 (COVID-19). Ritonavir is a cytochrome P450 3A inhibitor and a P-glycoprotein inhibitor that increases the plasma concentration of tacrolimus and other medications. We describe the cases of two patients treated with nirmatrelvir/ritonavir: a patient who had undergone kidney transplantation and another with a history of hematopoietic stem cell transplantation. Toxic concentrations of tacrolimus were induced in both. This case series highlights the risk associated with the concomitant administration of tacrolimus and nirmatrelvir/ritonavir.
Keywords
Introduction
Coronavirus disease 2019 (COVID-19) was first detected in China in December 2019 and was declared to be a pandemic by the World Health Organization (WHO) on 11 March 2020. 1 The combination of nirmatrelvir and ritonavir (NIM/RTV; Paxlovid®, Pfizer, New York, NY, USA) was authorized by the Food and Drug Administration (FDA) for emergency use in December 2021. 2 The FDA-approved indication for NIM/RTV is the treatment of mild-to-moderate COVID-19 infection in adults and children (over 12 years of age, body mass ≥40 kg) who have a high risk of progression to severe disease. Solid organ transplant recipients (SOTRs) who are taking immunosuppressants are at risk of severe COVID-19 disease. 3 In addition, recipients of hematopoietic stem cell transplants are at high risk of COVID-19-associated morbidity and mortality. Therefore, the early treatment of symptomatic COVID-19 infection is an important means of reducing the risks of severe disease and death in such patients, 4 and they are ideal candidates for treatment using NIM/RTV.
Ritonavir is a cytochrome P450 3A inhibitor and a P-glycoprotein inhibitor and may increase the plasma concentration of tacrolimus and other medications. 2 Indeed, the metabolism of tacrolimus via the cytochrome P450 3A pathway is associated with the potential for multiple drug–drug interactions. 5 Thus, patients who have undergone transplantation and are taking such inhibitors should be monitored in case of NIM/RTV drug–drug interactions. However, there have been few studies of patients who have undergone hematopoietic stem cell transplantation to date. Here, we describe the clinical course and management of two patients who underwent concomitant treatment with tacrolimus and NIM/RTV. The findings highlight the potential for interactions between tacrolimus and NIM/RTV.
We have followed the CARE guidelines 6 in the reporting of these cases and obtained the consent of the patients for treatment.
Case presentation
Case 1
A 32-year-old man presented to our emergency department with complaints of respiratory distress and fever because of a symptomatic COVID-19 infection. The patient had undergone kidney transplantation 7 years previously. His immunosuppression regimen comprised oral prednisone 5 mg/day, oral tacrolimus 4 mg twice daily, and oral mycophenolate 1080 mg twice daily. He was also taking benzbromarone 50 mg/day and petrepril terbutamine 4 mg daily. Two weeks before he was admitted, he had a fever of 38.5°C. On the day after his admission, oral NIM/RTV was prescribed as nirmatrelvir 150 mg and ritonavir 100 mg twice a day. This was a relatively low dose, because he had an estimated glomerular filtration rate (eGFR) <60 mL/minute/1.73 m2 and his creatinine clearance (CrCl) was 59.04 mL/minute. After admission, his immunosuppressants were stopped and NIM/RTV was administered in the form of 10 divided doses. The patient’s blood tacrolimus concentration was measured using a chemiluminescent microparticle immunoassay for the quantitative determination of tacrolimus in human whole blood (Cobas e601, Roche Diagnostics, Basel, Switzerland).
The last dose of tacrolimus was administered 12 hours prior to the start of NIM/RTV therapy. However, despite the subsequent withdrawal of tacrolimus, the trough blood concentration increased to 72.8 ng/mL on the second day following the first NIM/RTV dose. At this time, the patient felt fatigued. His tacrolimus concentration subsequently decreased from 59.44 ng/mL on day 3 to 33.94 ng/mL on day 4, then gradually decreased further, from 15.65 ng/mL on day 5 to 4.06 ng/mL on day 8 (Figure 1). The administration of a lower dose of 0.5 mg twice daily was restarted when the patient was discharged, 2 days after the end of NIM/RTV therapy (Table 2), and the dose was gradually increased to the original level. The blood tacrolimus concentration returned to the normal range 3 days later.
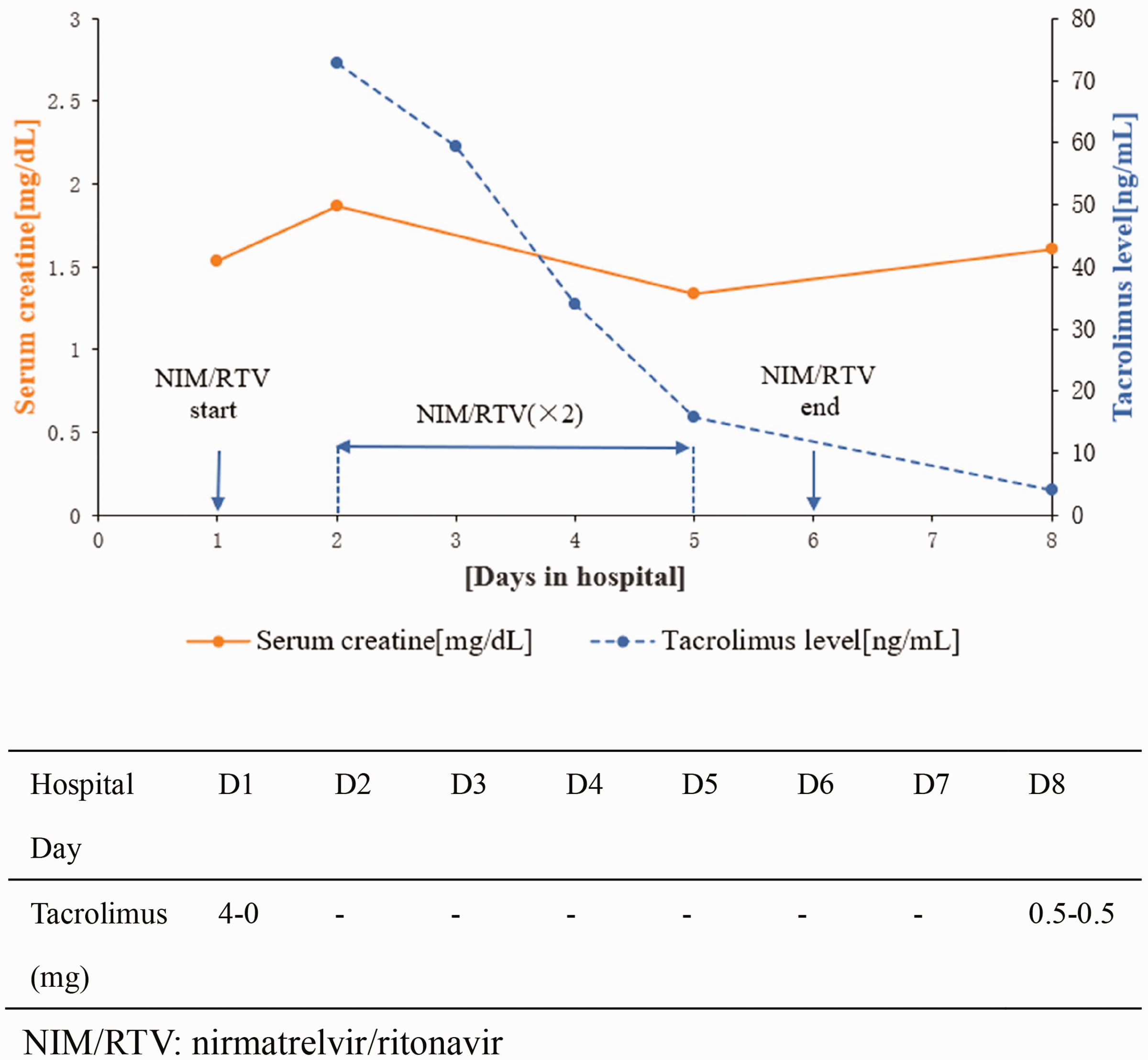
Serum creatinine and blood tacrolimus concentrations of patient 1 during his hospital stay.
Characteristics of the patients.
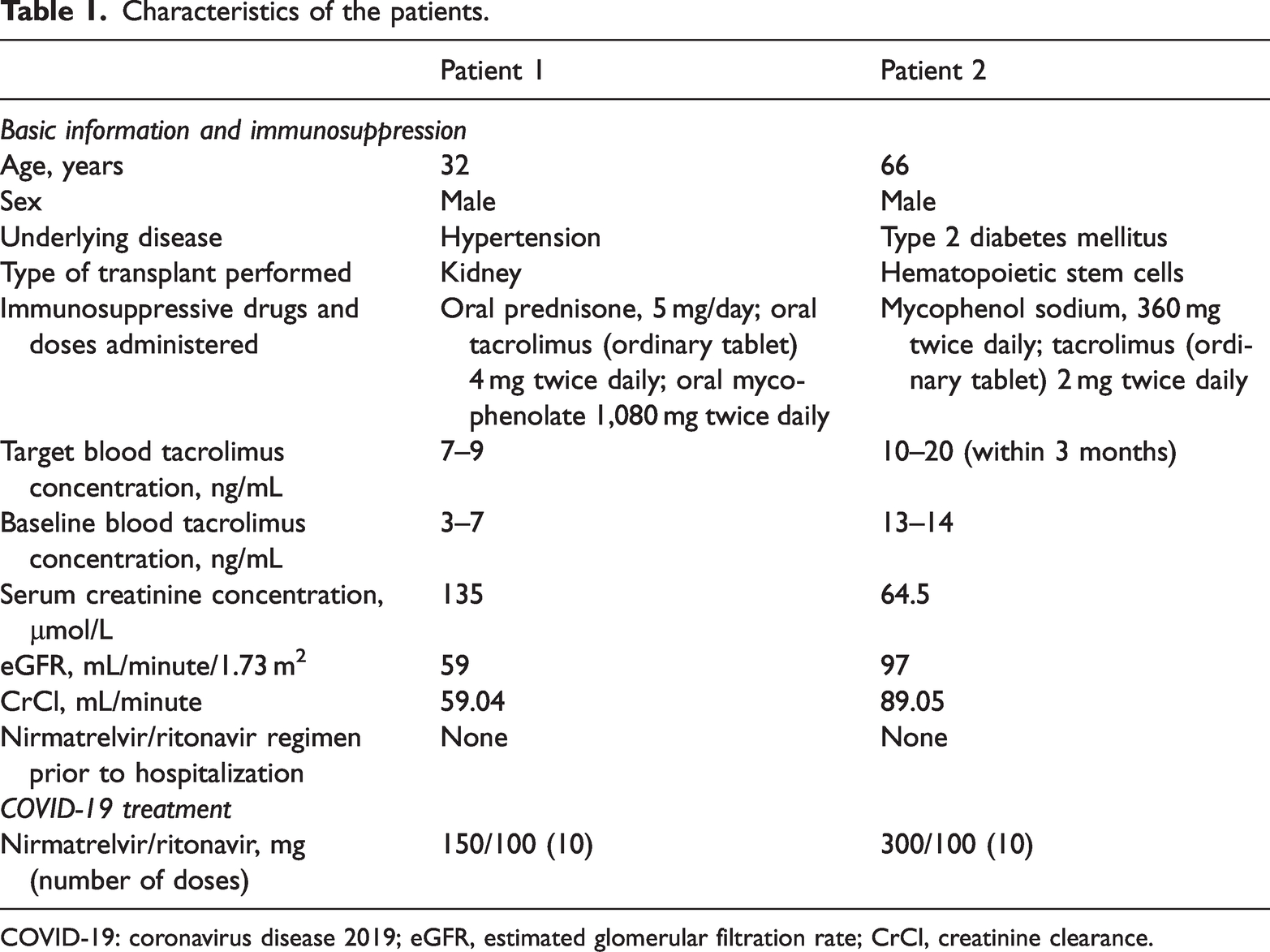
COVID-19: coronavirus disease 2019; eGFR, estimated glomerular filtration rate; CrCl, creatinine clearance.
Daily doses of tacrolimus and nirmatrelvir/ritonavir administered to patient 1.

Tac: tacrolimus; D: day; uid: once daily; bid: twice daily.
The patient’s circulating creatinine concentration increased from 135 μmol/L on day 1 to 165 μmol/L on day 2 (Figure 1), but his kidney function gradually returned to baseline after the NIM/RTV was stopped. The patient’s clinical condition improved and he was discharged from hospital on the eighth day following the start of NIM/RTV treatment. On week after his discharge, the patient’s blood tacrolimus concentration had returned to within the target range.
Case 2
A 66-year-old man developed COVID-19 after having undergone hematopoietic stem cell transplantation. He was taking tacrolimus 2 mg and mycophenol sodium 0.36 g twice daily, and isavuconazole sulfate capsules were administered orally to prevent fungal infection following the transplantation (Table 3), during which period his tacrolimus concentration was stable. NIM/RTV administration (300 mg/100 mg, twice a day) was started on day 1, and given the risk of drug–drug interaction, his tacrolimus dose was reduced to 1.5 mg twice daily at this time (Table 3).
Daily doses of tacrolimus and creatinine concentrations of patient 2 before and after nirmatrelvir/ritonavir administration.

Tac: tacrolimus; D: day; bid: twice daily; uid: once daily.
The patient’s tacrolimus concentration 24 hours after commencing NIM/RTV administration was >40 ng/mL and the trough concentration had increased to >40 ng/mL 72 hours after starting NIM/RTV (Figure 2). At this time, he felt fatigued and nauseous. Given his high tacrolimus concentration, his dose of isavuconazole sulfate was reduced from 200 mg to 100 mg four times daily for days 5 to 8. His tacrolimus concentration had decreased to 20.96 ng/mL 24 hours after the NIM/RTV administration was stopped. Tacrolimus was withheld between days 3 and 6.

Serum creatinine and blood tacrolimus concentrations of patient 2 during his hospital stay.
The patient started administering a lower dose of tacrolimus (0.5 mg twice daily) 2 days after the end of NIM/RTV therapy, then this was increased to 1 mg twice daily for days 8 to 10, and his tacrolimus concentration returned to 10.07 ng/mL on day 8. His dose of isavuconazole sulfate was restored to 200 mg four times daily on day 9. His circulating creatinine concentration increased from 64.5 μmol/L at baseline to 94.5 μmol/L on day 2 (Table 3). His kidney function returned to the baseline level after the NIM/RTV administration was stopped.
Discussion
Here, we have described two patients who experienced a drug–drug interaction between tacrolimus and NIM/RTV, one of whom had a history of kidney transplantation, and another who had undergone hematopoietic stem cell transplantation. In the former, the final dose of tacrolimus was administered 12 hours prior to the commencement of NIM/RTV administration, but because of a lack of relevant clinical experience, the same modification was not made for the latter patient. Given the possibility of drug–drug interactions, the dose of tacrolimus being administered should have been reduced when NIM/RTV administration commenced. However, in the second patient, the final dose of tacrolimus was administered 36 hours following the start of NIM/RTV administration. A lower dose of tacrolimus, of 0.5 mg twice daily, was restarted 2 days after the end of NIM/RTV treatment.
There was another difference between the two patients, because the second patient was concurrently administering isavuconazole to prevent fungal infection, whereas the first was not. However, his tacrolimus concentration was stable while he was taking isavuconazole, before NIM/RTV administration commenced. Given that isavuconazole is an inhibitor of CYP3A4, an interaction with tacrolimus, resulting in an increase in tacrolimus concentration, might have been anticipated.
Tacrolimus is the key component of most immunosuppression protocols that are currently in use worldwide for the optimization of transplant outcomes and to reduce the incidence of rejection. Underdosing can increase the risk of graft rejection and donor-specific antibody formation, whereas overdosing can be associated with adverse effects, such as nephrotoxicity, neurotoxicity, and gastrointestinal defects. 7
A multicenter retrospective study 8 recorded acute kidney injury secondary to high tacrolimus trough concentrations (mean 28.5 μg/L) in 10 of 14 patients after the dose of tacrolimus had been reduced or it had been discontinued during NIM/RTV administration.
The French recommendations for the management of antiviral therapy of COVID-19 using NIM/RTV in patients taking drugs with the potential for interactions, 9 classify tacrolimus as a red-label drug, owing to the significant risk of permanent adverse consequences, implying that it should not be administered alongside NIM/RTV. According to another set of published recommendations with respect to drug–drug interactions involving NIM/RTV, 9 tacrolimus should be discontinued during the first 5 days of NIM/RTV treatment and the tacrolimus trough concentration should be monitored on days 1 to 3. Furthermore, after the reintroduction of tacrolimus, the circulating concentration should be measured every 2 weeks.
The study by Belden et al. 10 showed that restarting standard tacrolimus tablets on the 6th day and tacrolimus sustained-release tablets on the 7th day following the start of NIM/RTV administration caused an increase in the blood concentration of tacrolimus, and therefore they recommended that these two groups of patients should instead restart tacrolimus administration on the 8th and 10th days. Giguère et al. also studied the optimal timing for tacrolimus administration to be restarted following NIM/RTV therapy. 11 An algorithm was proposed based on a model that recommends the administration of a single dose of tacrolimus, equivalent to 1/8th of the total initial daily dose, on day 1 of nirmatrelvir/ritonavir therapy, after which tacrolimus is withheld during the remaining 4 days of treatment. Tacrolimus administration should then be restarted at 50% of the initial dose at the end of day following COVID treatment, with 75% of the initial dose being administered the following day and the full dose being administered on the third day.
It has been documented on numerous occasions that patients who are diagnosed with COVID-19 in primary care and self-medicate at home often require treatment in the emergency department and hospital admission because they develop nausea, vomiting, abdominal pain, fatigue, and other symptoms related to the toxic effects of tacrolimus. 7 However, the administration of some immunosuppressants alongside tacrolimus may be associated with more frequent side effects; therefore, it is important for clinicians to be aware of potential interactions.
With respect to the coadministration of NIM/RTV and tacrolimus, NIM/RTV has a significant effect on the circulating concentration of tacrolimus, with the resulting increase varying according to the doses administered. In a previous study, we found that patients who were taking NIM/RTV frequently showed larger increases in tacrolimus concentration, and indeed one who was administering eight daily doses 12 showed a 35-fold increase in their blood concentration of tacrolimus. Ritonavir is a potent CYP3A4 inhibitor, and when its administration is stopped, its inhibitory effect decreases by 46% to 61% during the first 24 hours, and it is then 70% to 90% lower on the 2nd to 5th days, but it may take 3 weeks for enzyme function to fully recover. 13
Of the published cases of interactions, two patients14,15 were administering 150 mg/100 mg NIM/RTV and the remaining eight were administering 300 mg/100 mg. Patients with severe kidney injury (eGFR <30 mL/minute/1.73 m2) should not administer this combination, including those with end-stage renal disease who are on hemodialysis.
One published case report records an interaction between NIM/RTV and tacrolimus in a patient with myasthenia gravis. 16 The patient’s tacrolimus concentration was >30 ng/mL because she was unaware of the need to stop tacrolimus administration during treatment, but after obtaining her blood test results, her tacrolimus was immediately discontinued.
Regarding the time at which tacrolimus administration should be restarted, the fastest recorded return of the tacrolimus concentration to the baseline range took 4 days, 17 while the longest took 10 days, 15 after which the administration of tacrolimus 2 mg twice daily could be resumed.
The management of such interactions is challenging and requires the discontinuation or tapering of the doses of immunosuppressants, followed by the monitoring of the drug concentrations to guide the timing of their reintroduction. The timing of the reintroduction of tacrolimus can be determined according to the patient’s condition and their blood concentration. In addition, clinicians should consider whether a rescue plan is needed.
In conclusion, the two cases reported herein highlight the importance of the interaction between tacrolimus and NIM/RTV. We have described the course of a patient who had previously undergone kidney transplantation and that of a second who had undergone hematopoietic stem cell transplantation, which places patients at high risk of morbidity and mortality. When infected with COVID-19, such patients are at a high risk of progression to severe disease. Our experience with the second of these patients leads us to conclude that tacrolimus should be withheld while NIM/RTV is administered and be restarted when the blood tacrolimus concentration returns to within the therapeutic range.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241247705 - Supplemental material for Nirmatrelvir/ritonavir treatment of patients with COVID-19 taking tacrolimus: case series describing the results of drug–drug interactions
Supplemental material, sj-pdf-1-imr-10.1177_03000605241247705 for Nirmatrelvir/ritonavir treatment of patients with COVID-19 taking tacrolimus: case series describing the results of drug–drug interactions by Dan Shen, Yinhua Gong, Yulan Qian, Jianguo Zhu and Jie Gao in Journal of International Medical Research
Footnotes
Acknowledgements
The authors are grateful to Mr. Deng Xifan for his guidance regarding the creation of the figures and Jiang Ye from Lianyungang Maternal and Child Health Hospital for performing a literature search.
Author contributions
DS: drafting the manuscript and pharmacokinetic analysis; YG: drafting the manuscript, data analysis; YQ: figure design and revision of the manuscript; JG: case management during hospitalization; JZ: guidance regarding the manuscript. All the authors read and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics approval
Because of its retrospective nature, we did not seek ethics approval for the study described or the informed consent of the patients involved. However, we have de-identified the information regarding the patients that is included in the article.
Funding
This work was supported by the Jiangsu Provincial Medical Key Discipline (grant no. ZDXK202201) and a Jiangsu Pharmaceutical Association Hospital Pharmacy Research Project (grant no. H202312).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
