Abstract
Background
[No revision is required.]Pulmonary cryptococcosis (PC) is common in immunocompetent patients, and its early diagnosis is challenging. Multiple studies have addressed the detection of cryptococcal glucuronoxylomannan (GXM) antigen in serum, but few have investigated its detection in bronchoalveolar lavage fluid (BALF).
Objectives
To evaluate the diagnostic value of the Cryptococcus GXM antigen test in BALF for PC in immunocompetent patients.
Design
We collect the clinical data of 138 patients from October 2018 to March 2023 and divide them into a PC group (n = 48) and a non-PC group (n = 90). Perform serum and BALF GXM antigen test on all cases that meet the inclusion criteria.
Methods
To analyze and compare the clinical characteristics of the PC group and non-PC group. We calculate the positive detection rate (PDR) and negative predictive value of BALF and serum GXM antigen tests, as well as their combination, for the PC group.
Results
Most patients in the PC group are male (59.6%), with an average age of 49.4 years. Most of the patients have no underlying disease (51.9%). Diabetes and hypertension are the most common comorbidities. Most patients (53.8%) are asymptomatic. Cough and sputum are the most frequent clinical symptoms, followed by fever and chest distress. Nodules and exudative consolidation are the most common imaging findings, and the culture positivity rate is 25%. Forty patients are positive according to the BALF GXM test, for a positivity rate of 83.3% and a negative predictive value of 91.8%. Moreover, 39 patients are positive according to the serum GXM test, for a positivity rate of 81.3% and a negative predictive value of 90.9%. The positivity rate for serum plus BALF is 95.8%. There is no significant difference (P > 0.05) in diagnostic value between BALF and serum GXM tests for PC. GXM positivity rates differ significantly among paired BALF, serum, and BALF plus serum groups (P < 0.05).
Conclusion
The BALF GXM test is useful for the early diagnosis of PC in immunocompetent patients. Simultaneous BALF and serum GXM testing would increase the rate of early PC diagnosis in immunocompetent patients.
Introduction
Pulmonary cryptococcosis (PC) is mainly caused by Cryptococcus neoformans and Cryptococcus gattii, which invade the lungs from the respiratory tract. 1 The diagnosis of PC is usually based on a combination of clinical and radiological suspicion and laboratory confirmation. The methods used to confirm the infection are culture, direct microscopic, histopathology, serology, and molecular detection. 2 Unless there is invasive cryptococcal infection, the positive rate of sputum or bronchoalveolar lavage fluid (BALF) or blood culture is low, and lung puncture biopsy has a certain degree of trauma, making it difficult to obtain sufficient lung tissue samples. Glucuronoxylomannan (GXM), a type 2T-independent antigen, is the major component of the capsular polysaccharide (Cn CAP) of Cryptococcus neoformans. 3 Cryptococcal capsule-antigen test is a fast, sensitive, and non-invasive detection method for cryptococcal infection, widely used in serum and cerebrospinal fluid, but there is little data on its application in BALF. We perform a statistical analysis of the positivity rate and negative predictive value of blood and BALF detection of Cryptococcus GXM antigen in immunocompetent patients from October 2018 to March 2023, to explore its utility for the early diagnosis of PC in immunocompetent patients.
Methods
The reporting of this study conforms to the Studies of diagnostic accuracy (STARD) statement. 4
Study Design
We enrolled human immunodeficiency virus (HIV)-negative patients with PC from October 2018 to March 2023 and excluded patients with immunosuppression (solid-organ transplant recipients, hematological malignancies, solid malignancies, rheumatic diseases, liver cirrhosis, chronic diseases, corticosteroids, and other immunosuppressive therapies).
5
The diagnostic criteria for pulmonary cryptococcus are as follows: (A). If biopsy shows Cryptococcus, this was considered a proven PC case, whatever the radiological findings. (B). If direct microscopy or cytological exam revealed a yeast typical of Cryptococcus from any cavity, pleural fluid or biopsy (but not bronchoscopy samples), this was also considered a proven PC case, whatever the radiological findings. (C). In those cases with a positive respiratory sample culture of Cryptococcus, without histopathology or microscopic evidence, the patient had also to have new nodules, masses, or consolidation on chest computed tomography (CT) scan to be considered a probable case of PC. (D). In patients without histopathology or microscopic evidence, and serum or chest fluid or respiratory specimens tested positive for Cryptococcus capsule-antigen or the molecular detection
Participants
The inclusion and exclusion criteria for the research subjects in the project proposal are as follows:
Inclusion criteria for research subjects:
Patients over 14 years old who have lung infections or have lung nodules or masses found during physical examinations, excluded patients with immunosuppression, Cases diagnosed with PC with complete clinical data, who underwent serum cryptococcosis capsule-antigen test and agreed to undergo BALF cryptococcosis capsule-antigen test. Informed consent for this study, willing to sign an informed consent form. Patients aged 14-17 who meet the inclusion criteria have their guardians (parents) sign an informed consent form. Meets the diagnostic criteria for PC. Exclusion criteria for research subjects:
Excluding patients with immunosuppression (solid-organ transplant recipients, hematological malignancies, solid malignancies, rheumatic diseases, liver cirrhosis, chronic diseases, corticosteroids, and other immunosuppressive therapies). The patient or family members do not agree to participate in the research project. Those who have contraindications for bronchoscopy or do not agree to undergo bronchoscopy Control group setup: Patients who were admitted to the hospital simultaneously and underwent serum and BALF Cryptococcus Capsular Antigen tests to exclude cases of PC with complete data.
Test Methods
We used the lateral flow assay test strip of Immuno-Mycologics (Norman, OK, USA). Whole-blood samples were extracted using a conventional method and centrifuged at 3000 rpm for 10 min to separate serum. After routine bronchoscopy, 60 mL of 0.9% sterile normal saline was injected into the segmental or subsegmental bronchus of the diseased lung for lavage. Next, 10-15 mL of lavage fluid was recovered and centrifuged; the supernatant was retained. We placed one drop of sample diluent into a test tube, and added 40 µL of BALF supernatant or serum using a micro-sampler. Then, we dipped the colloidal gold test strip into the suspension and incubated it at room temperature for 10 min. The result was considered negative if only the quality control line was present, and positive if both the quality control and detection lines were present.
Statistical Analyses
The chi-squared McNemar test was performed using SPSS v20.0 (IBM Corp., Armonk, NY, USA). Count data are expressed as percentages. Statistical significance was evaluated at P < 0.05. We compared the GXM positivity rate between BALF and BALF plus serum groups. CT images were interpreted by professional radiologists, each with 15 years of experience.
Follow-up Studies
Patients in the PC group were followed up for 3-12 months, with complete absorption or loss of contact ending the follow-up. Patients in the non-PC group were followed up for 1 year, with a clear diagnosis, loss of contact, or death ending the follow-up.
Results
A total of 183 HIV-negative patients with pulmonary lesions of unknown etiology underwent blood and BALF Cryptococcus GXM antigen testing. Among these, 78 patients were diagnosed with PC, 20 immunocompromised patients were excluded, and 10 patients withdrew from the study. Forty-eight patients were included in the PC group. One hundred five patients were diagnosed with non-cryptococcal infections, and 15 patients with infections of unknown etiology were excluded. Ninety patients were included in the non-PC group (Chart 1).

Case and study flow chart of PC group and non PC group.
Participants
There were 48 patients in the PC group (30 male, 18 female), 17-87 years (mean, 49.4 years) of age. Of the patients, 27 and 21 did not and did have comorbidities, respectively; the most common were diabetes and patients were asymptomatic, and 20 had clinical symptoms (19 cough and expectoration, 5 fever, 2 hemoptysis, 1 chest pain, and 5 chest distress). Most patients had normal lung examinations. Pulmonary lesions affected one or both lungs; the left lung was most frequently affected. Imaging findings included nodules (Figure 1), mixed type (Figure 2), and exudative consolidation. Halo signs were common, lobulated signs were visible, and cavities were rare (Figure 3). BALF Cryptococcus GXM testing yielded positive results in 40 patients, serum Cryptococcus GXM testing in 39 patients, and BALF plus serum Cryptococcus GXM testing yielded positive results in 33 patients. Two of the PC patients had negative serum and BALF GXM antigen tests, which confirmed the diagnosis of cryptococcal infection through metagenomic next-generation sequencing (mNGS) (Table 1). Three patients had mixed tuberculosis infections; all patients were treated with fluconazole capsules (antifungal) for 4-6 months, and were followed up for 3-12 months. Chest CT showed various degrees of absorption or complete absorption (Table 2). There were 90 patients in the non-PC group (53 male, 37 female), 18-87 years (mean, 55 years) of age. Among these, 34 patients had bacterial pneumonia, 22 had pulmonary tuberculosis, 14 had lung cancer, 6 had non-tuberculous mycobacillosis, 4 had Aspergillus infection, and 10 patients had other diseases such as interstitial lung disease and chronic granulomatous lesions (Table 2).

Male patient, 42 years of age, no comorbidity. Small nodules were found in the middle lobe of the right lung during a physical examination (left). During follow-up, the number of nodules increased, serum and bronchoalveolar lavage fluid (BALF) capsule-antigen tests were negative, and mNGS detected Cryptococcus in BALF. After 3 months of oral fluconazole capsules (400 mg), chest computed tomography (CT) showed complete absorption of the infective focus (right). mNGS, metagenomic next-generation sequencing.
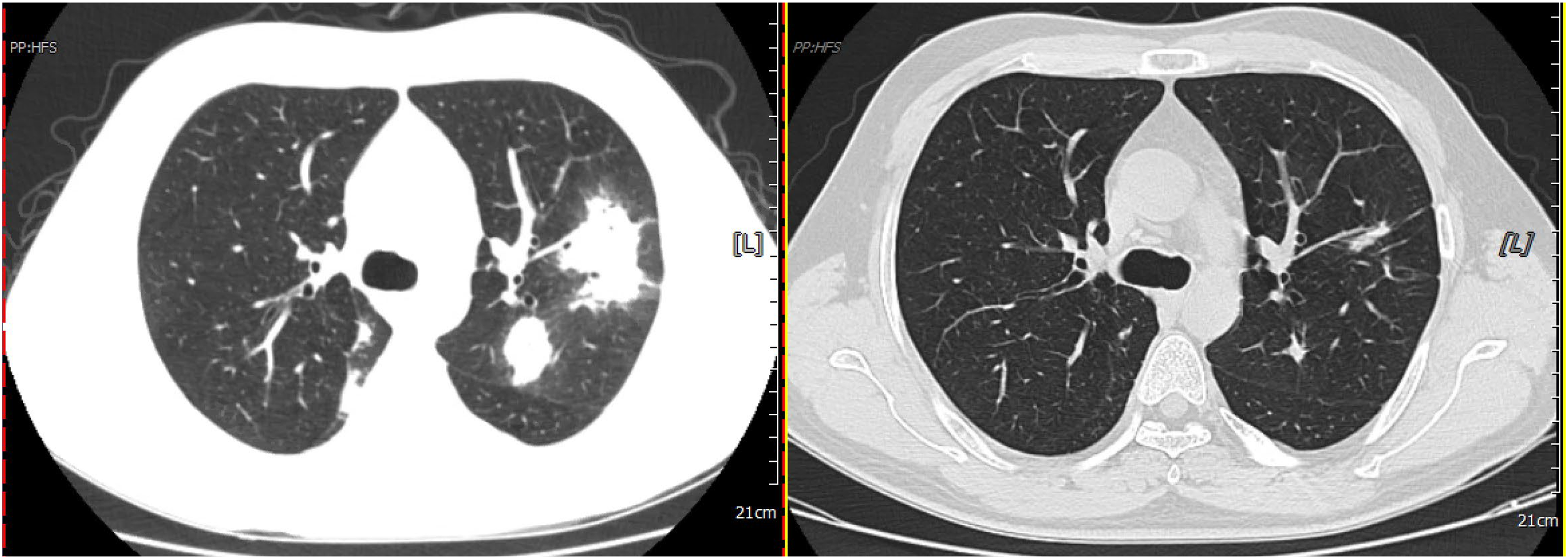
Male patient, 36 years of age, no comorbidity. The patient coughed and expectorated for 3 days; chest CT showed infection in both lungs (Figure 3, left), and a Blood/BALF cryptococcal capsule-antigen test was positive. Cryptococcus neoformans was cultured from BALF. The patient received oral fluconazole capsules (400 mg qd) for 6 months, and the left lung infection was again evaluated (right). BALF, bronchoalveolar lavage fluid; CT, computed tomography.

Male patient, 49 years of age. During antiviral treatment for hepatitis B, He occasionally coughed with blood streaks in the sputum, and a chest CT showed nodules with cavity formation in the left lower lung dorsal segment (left). A blood/BALF cryptococcal capsule-antigen test was positive, and the patient was administered fluconazole capsules (400 mg orally qd). After 6 months of treatment, chest CT showed that the left lower lung nodules had shrunk and the cavity had closed. BALF, bronchoalveolar lavage fluid; CT, computed tomography.
Clinical characteristics of 48 cases PC group.

PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid; mNGS, metagenomic next-generation sequencing.
The characteristics of patients in non-PC group.

PC, pulmonary cryptococcosis.
Test Results
The positivity rates of BALF and serum Cryptococcus GXM testing for PC in immunocompetent patients were 83.3% (40 of 48) and 81.3% (39 of 48), respectively; the difference was not significant (Table 3). The positivity rate of BALF plus serum Cryptococcus GXM testing was 95.8%, which was compared with the positivity rate of single-sample Cryptococcus GXM in pairs (Tables 4 and 5) (P < 0.05). The negative predictive values of BALF and serum Cryptococcus GXM antigen testing were 91.8% and 90.9%, respectively (P > 0.05) (Table 6). We analyzed subgroups and found that in the asymptomatic group, there were 28 cases with a higher positive rate of BALF than serum (92.7% vs 75.0%), while in the symptomatic group, there were 24 cases with a lower positive rate of BALF than serum (75.0% vs 91.6%). But there was no significant statistical significance in either subgroup (P > 0.05) (Tables 7 and 8).
Comparison of BALF and Serum GXM tests in PC group.

Calculate using the chi square 2X2 table, P > 0.05, reject H0, accept H1, and there is no significant statistical difference.
PR, positive rate; PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid.
Comparison of positive detection rates of BALF and BALF + serum combined GXM test in PC group.

Calculate using the chi square 2X2 table, P < 0.05. Accept H0 and reject H1, and there is a statistical difference.
PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid.
Comparison of positive detection rates of serum and BALF + serum combined GXM test in PC group.

Calculate using the chi square 2X2 table, P < 0.05. Accept H0 and reject H1, and there is a statistical difference.
PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid.
Negative predictive values of BALF and serum GXM tests.

Calculate using the chi square 2X2 table, P > 0.05, reject H0, accept H1, and there is no significant statistical difference.
NPV, negative predictive values; FN, false-negative; TN, true negative; PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid.
Comparison of BALF and serum GXM tests in asymptomatic PC group.

Calculate using the chi square 2X2 table, P > 0.05, reject H0, accept H1, and there is no significant statistical difference.
PR, positive rate; PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid.
Comparison of BALF and serum GXM tests in symptomatic PC group.

Calculate using the chi square 2X2 table, P > 0.05, reject H0, accept H1, and there is no significant statistical difference.
PR, positive rate; PC, pulmonary cryptococcosis; GXM, glucuronoxylomannan; BALF, bronchoalveolar lavage fluid.
Follow-up
In the PC group, 3 cases were lost to follow-up, and 45 cases showed absorption and improvement in chest CT after treatment, with good outcomes. After 1 year of follow-up, there was no recurrence or death.
Discussion
Cryptococcus sp. are basidiomycete yeasts and are distributed worldwide in soil, pigeon manure, water, and some foods. Cryptococcus is weakly pathogenic, mainly affecting the immunosuppressed but occasionally causing disease in immunocompetent individuals. 9 Most cryptococcal infections are caused by the inhalation of cryptococcal spores via the respiratory tract. Cryptococci in lung tissues are either immediately removed or encapsulated by a granuloma as latent infection or disease, potentially resulting in systemic spread, depending on the host immune response, and the number and the virulence of the pathogen. The incidence and mortality rates of cryptococcosis are increasing worldwide. PC caused by C. neoformans is an important public health issue. Cryptococcus gattii is more likely than C. neoformans to infect immunocompetent individuals. A cohort study in Australia and New Zealand showed that 44% of immunocompetent patients exhibited C. gattii infection, of which 61% had lung damage. 10 PC accounted for 75% of 218 patients with cryptococcal infection, 62% of whom were immunocompetent. 11 A 10-year multicenter retrospective study in China showed that cryptococcal infection was the third (15.6%) most common pulmonary fungal infection, following Aspergillus (37.9%) and Candida (34.2%). 12 Therefore, PC is an important fungal infection of the human respiratory system.
In immunocompetent patients, the clinical manifestations of PC vary greatly, with most patients showing no symptoms or only mild respiratory symptoms, and a few immunosuppressed patients show respiratory failure caused by severe pneumonia. 13 In this study, 53.8% of the patients were asymptomatic. Cough and expectoration were the most common respiratory symptoms (40.4%), followed by fever and chest tightness (13.5%); hemoptysis and chest pain were rare. Imaging shows diverse, non-specific changes according to immune status, most of which are single or multiple nodular shadows that are easily misdiagnosed as lung cancer or tuberculosis.2,14 In this study, nodules accounted for 41.7%, exudative consolidation for 16.7%, mixed type for 33.3%, and space occupying/mass shadow for 8.3% of all imaging findings. One case was accompanied by pleural effusion, which the pleural fluid GXM test was positive. In all cases, lesions involving one lung and both lungs accounted for 58.9% and 41.1%, respectively. Multiple lesions were common, and some patients exhibited the halo (75.0%), visible cavity (13.5%), and lobulation (20.8%), and spiculation was rare. However, these signs have poor specificity, as they are also indicative of pulmonary Aspergillus and Pneumocystis disease. Most lesions had a broad basal shape close to the pleura around the lower lung. Typically, 18F fluorodeoxyglucose (FDG) positron emission tomography (PET) of PC lesions shows an abnormal concentration of radioactivity, which can lead to misdiagnosis as lung cancer.2,15 In a retrospective study, 88% (37 of 42) of patients with PC showed high FDG uptake, and the maximum standard intake value of OC was 1.4-13.0 (mean 4.9). 15 No patients underwent a PET-CT examination.
Among the methods for clinical diagnosis of PC, lung tissue biopsy has several difficulties. It takes at least 1 week to obtain culture results from specimens from sterile sites, sputum, lavage fluid, and blood, compared with 2 h for the Cryptococcus GXM antigen test, and the positivity rate is low.16–19 Indeed, the serum Cryptococcus GXM antigen test is the most frequently used non-invasive method for detecting cryptococcal infection; however, its sensitivity varies, with previous studies reporting sensitivity rates of 93.3%, 20 and 80.0% and 96.4% in immunocompetent and immunosuppressed HIV-negative patients with PC, respectively. 21 However, in HIV-negative patients with PC, the sensitivity of serum Cryptococcus GXM antigen decreased to 25-56%.22,23 Because the colloidal gold method for detecting Cryptococcus is simple and does not require specialist equipment, it can be conducted in most laboratories. The results of the colloidal gold method show good agreement with the latex agglutination test and enzyme-linked immunosorbent assay, and it was able to detect C. gattii. 24 Our findings showed that in immunocompetent patients, the colloidal gold Cryptococcus GXM detection method had sensitivity and specificity rates of 81.3% and 100% (serum) and 83.3% and 100% (BALF), respectively. These values are significantly higher than that for culture (25.0%).
Few studies have addressed the detection of Cryptococcus GXM antigen in BALF. A Cryptococcus GXM antigen titer in BALF of 1:8 showed 100% sensitivity and specificity in a previous study. 25 In BALF of HIV-negative patients with PC, the sensitivity for detecting GXM antigen was higher than that for serum (82.6% and 73.9%, respectively). 26 In this study, the PC detection rate of BALF Cryptococcus GXM testing was 83.3%, which was non-significantly higher than that of serum Cryptococcus GXM testing (81.3%); by contrast, the rate for BALF plus serum was significantly higher, at 95.8%. BALF and serum Cryptococcus GXM tests had negative predictive values of 91.8% and 90.9%, respectively, a non-significant difference. Clinically, it is highly suspected that PC patients have negative BALF or serum GXM, and there is still a certain rate of missed diagnosis for both. However, in this study, 92 cases had negative BALF and serum GXM, among which 2 cases were diagnosed as PC by NGS method and were false-negative cases with a negative predictive value of 97.8%. Compared to a single specimen, the combined negative predictive value of the two is greater. We analyzed the subgroups and found that the symptomatic group had a higher serum GXM positivity rate, while the asymptomatic group had a higher BALF GXM positivity rate. Patients with asymptomatic suspicion of PC can be screened by BALF GXM test, while those with symptomatic suspicion of PC can be screened by serum GXM test in the early stage. Suspected PC patients, if both specimens are sent for GXM testing, can further improve the positive diagnosis rate, and reduce the missed diagnosis rate.
The mNGS enables unbiased detection of pathogens by high-throughput or large-scale parallel sequencing, thereby facilitating the diagnosis of infections by rare pathogens. 27 The BALF mNGS positivity rates in immunocompetent and immunocompromised patients with PC are 40% and 87.5%, respectively. 20 In this study, PC was confirmed by mNGS in two patients, in whom BALF and serum Cryptococcus GXM tests yielded negative results. The blood and BALF of another patient were positive according to the Cryptococcus GXM antigen test; however, Cryptococcus was not detected by BALF mNGS. The diagnostic utility of BALF mNGS for PC requires further investigation.
This study has certain limitations. We did not conduct a sample size calculation, and the limited sample size in this study may affect the statistical significance of the results. In addition, the exclusion of 15 non-PC cases with unknown etiology cannot truly rule out the possibility of PC, which may have a certain impact on the negative predictive value results.
Conclusion
Cryptococcus GXM testing of BALF samples is useful for the early diagnosis of PC in immunocompetent patients (sensitivity, 83.3%). BALF plus serum Cryptococcus GXM testing show superior performance for the early diagnosis of PC in immunocompetent patients compared with single-sample Cryptococcus GXM testing (positivity rate, 95.8%). Therefore, immunocompetent patients with suspected PC should undergo simultaneous BALF and serum Cryptococcus GXM testing to increase the early diagnosis rate and reduce that of missed diagnosis.
Supplemental Material
sj-docx-1-cra-10.1177_29768675251363301 - Supplemental material for Diagnostic Value of Cryptococcal Glucuronoxylomannan Antigen Testing of Bronchoalveolar Lavage Fluid for Pulmonary Cryptococcosis in Immunocompetent Patients: A Clinical Study
Supplemental material, sj-docx-1-cra-10.1177_29768675251363301 for Diagnostic Value of Cryptococcal Glucuronoxylomannan Antigen Testing of Bronchoalveolar Lavage Fluid for Pulmonary Cryptococcosis in Immunocompetent Patients: A Clinical Study by Shuangxia Dong, Xinjian Dai, Linyan Pang and Yang Huang in Therapeutic Advances in Pulmonary and Critical Care Medicine
Footnotes
Acknowledgements
Appreciate all patients participating in the trial and all our hospital's respiratory doctors.
Author Contribution(s)
Availability of Data and Materials
Allowed free use for learning, prohibited use for commercial purposes.
Ethics Approval and Consent to Participate
This study was approved by the Ethic Committee of Wenzhou Central Hospital (No,:L2020-02-0265). The authors have no ethical conflicts to disclose.
The patients/participants provided their written informed consent to participate in this study. For participants under 18 years old, informed consent was obtained from their guardians (parents) in the form of a signed informed consent form.
Consent for Publication
Consent for publication was obtained from participants in the form of a signed informed consent form.
For participants under 18 years old, informed consent was obtained from their guardians (parents) in the form of a signed informed consent form.
Declaration of Conflicting Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fund Project: Wenzhou Municipal Science and Technology Bureau (y20180605).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
