Abstract
Background:
The opioid overdose epidemic continues as a serious world problem, with postoperative opioid prescribing recognized as a critical contributor to opioid use disorder (OUD) development. Identifying risk factors associated with OUD in the perioperative setting is essential for developing targeted prevention strategies. This study aimed to evaluate the association of social determinants of health (SDoHs) and mental health conditions in predicting OUD within 1 year following surgery.
Methods:
This retrospective cohort study used de-identified electronic health records from the TriNetX Research Network, evaluating over 295 000 matched patients across individual risk factor cohorts. The index event was defined as the first opioid exposure within 3 days after their first surgery. Patients with and without risk factors were propensity score matched and then odds ratios (ORs) and absolute risks were calculated for postoperative OUD within 1 year.
Results:
Significance at P < .001 was present for all SDoHs assessed with individual ORs ranging from 0.77 (benzodiazepine use, n = 3 026 362) to 8.3 (homelessness, n = 16 997). A cohort with known mixed risk factors showed an additive association with OUD risk. A validation cohort confirmed the methodology, showing no association between levothyroxine use and OUD development (OR 1.0, P = .416, n = 222 471). The definition of OUD in this study was based on ICD-10 coding, which does not perfectly align with DSM-5 diagnostic criteria and may capture patients with persistent opioid use but without formal OUD diagnosis.
Conclusions:
Key SDoHs and mental health conditions significantly increase the risk of postoperative OUD. These findings underscore the need for preoperative risk stratification and targeted interventions, including multimodal analgesia and integrated mental health care to mitigate OUD risk in surgical populations. Addressing these factors is critical for advancing perioperative care and combating the opioid overdose crisis.
Keywords
Introduction
The opioid overdose epidemic remains one of the most pressing public health crises in the United States. Although provisional 2024 data from the National Center for Health Statistics indicate a significant decline in opioid overdose deaths, from 83 264 in 2023 to 79 377 in 2024 (a 4.7% decrease), the overall toll remains high. Concurrently, opioid use disorder (OUD) prevalence continues to be elevated, highlighting the devastating impact of the opioid crisis. 1 While lethal concentrations of synthetic opioids (eg, fentanyl and its derivatives) adulterate 5 in 10 counterfeit prescription pills and are the top contributor of overdose fatalities, prescription opioids (eg, oxycodone) account for the highest percentage of non-fatal overdose emergency room visits in 2022.2-4 This continued public health threat necessitates a multi-faceted approach to mitigating the crisis. Prescription drug monitoring programs (PDMPs) provide support to providers improve opioid prescribing practices, inform clinical decisions, and safeguard individuals at risk. 5 In addition, identifying key demographic groups at heightened risk of developing OUD before their first exposure to opioids may also serve as a proactive approach to mitigate OUD development.
One key area of concern is perioperative opioid prescribing. While opioids play a critical role in managing post-surgical pain, inappropriate prescribing, including lack of individualized approaches that account for OUD risk factors, can inadvertently expose patients to increased risks of developing OUD. This is particularly significant given that many individuals who misuse opioids report initial exposure through prescription medications. 6 Thus, understanding the factors contributing to postoperative OUD risk is crucial for informing prevention strategies. While individuals across all demographics are impacted by the opioid overdose epidemic, certain characteristics may be associated with heightened susceptibility to OUD. 7 Social determinants of health (SDoH), such as socioeconomic status, employment, and housing stability, as well as comorbid mental health conditions, may predispose individuals to developing OUD following perioperative opioid exposure. However, data identifying the most critical predictors of OUD development remain limited, particularly in the perioperative setting. Rather than focus on a specific operative intervention, our findings add to the existing literature by providing a high-level overview of the association of SDoHs and the development of OUD among patients undergoing any surgical procedure for which opioids are prescribed.
To address this gap, we conducted a comprehensive analysis of electronic health records (EHRs) to identify the demographic and clinical predictors most strongly associated with OUD development within 1-year follow-up to surgery. We hypothesized that certain SDoHs, including homelessness, unemployment, and comorbid psychiatric conditions, would emerge as the strongest predictors of postoperative OUD. By identifying these associations, our study provides valuable insights to guide more targeted perioperative opioid prescribing practices and preventive interventions for at-risk populations as well as future studies quantifying the causative impact of these SDoHs on the development of OUD.
Materials and Methods
We used the US Collaborative Network from 68 health care organizations (HCOs) in the TriNetX Research Network. The database was accessed, and analyses were run in October 2024. The available data included information about the demographics, diagnoses coded in the International Classification of Diseases, 10th Revision (ICD-10), procedures coded in the Current Procedural Terminology (CPT), and medications coded in the Veterans Affairs (VA) National Formulary. The network used on the TriNetX database contains de-identified patient records from 118 432 981 patients. Our study contained data from 2011 to 2025 with an average of 2256 discrete facts per patient. Data are aggregated, and only summary statistics are available; individual-level records and specific health care facility identifiers are not accessible. Due to the anonymous nature of the data, informed consent was waived. The TriNetX platform complies with the Health Insurance Portability and Accountability Act and General Data Protection Regulation. The Western Institutional Review Board has granted a waiver of informed consent since the TriNetX platform only aggregates counts and statistical summaries of de-identified information.
A flowchart of the general cohort design is provided in Figure 1. This strategy was applied to each cohort. The only change made to each cohort pair was the inclusion and exclusion criteria for the social determinant being examined for that pair. Using the homelessness cohort pair as an example, the experimental cohort included patients who, before the index event, reported homelessness in their chart. The matching control cohort excluded any patient who had homelessness on their chart at any point before the index event. Specific modifications were made based on the assessed SDoHs and were identified using ICD-10 Z codes. For example, Z62 codes (“Problems related to upbringing”) were used to identify childhood trauma, including a history of abuse, neglect, or removal from the home; Z63 codes (“Other problems related to primary support group”) identified family problems, such as family disruption, marital discord, or absence of family members; Z81 codes (“Family history of mental and behavioral disorders”) captured family mental health history, including depression or substance use in relatives; and Z56 codes (“Problems related to employment and unemployment”) captured unemployment-related issues, such as job loss, occupational stress, or insecure employment. Similar strategies have been successfully used to assess opioid use outcomes using electronic health record (EHR) data.8-11

Overview of cohort design for database queries.
The index event was defined as the first documented opioid exposure within 3 days of the first recorded surgery and for the purposes of this manuscript, these index criteria represent perioperative opioid use. Thus, all patients in the study were both opioid and surgery naive prior to the index event. Additional inclusion criteria for the cohorts were set based on the presence of the SDoH before the index event (Figure 1). A pair of cohorts was created for each SDoH of interest, with an experimental cohort requiring the presence of the risk factor in question and a matching control lacking this risk factor. Any patients who had received opioids before the index event were excluded. Out of an abundance of caution, patients with a history of opioid dependence (ICD-10 F11.2) were excluded. Patients were also required to have been alive for the full 1-year follow-up period. Cohorts were not mutually exclusive, given the ubiquitous nature of many SDoHs. Additionally, to see how the various risk factors affect outcomes in concert, the 3 risk factors with the largest populations (alcohol disorder, nicotine dependence, and depressive episode) were combined into a single mixed cohort and assessed against a matching control. The primary outcome was the development of OUD—as defined by ICD F11 within 1 year of the index event.
The propensity scores for the experimental and control cohorts were matched for each analysis. Each cohort was propensity score matched (PPM) to its respective control group as a means to adjust for the difference in baseline characteristics and generate comparison cohorts with matched characteristics. We incorporated the following covariate demographic factors: age, sex, race, and ethnicity.
Many of the SDOHs and mental health conditions examined in this study are ubiquitous and often interrelated, making it difficult to isolate their independent effects without significantly reducing the sample size, limiting statistical power, and potentially excluded important subpopulations, thereby restricting the generalizability of our findings. The goal of this study was not to establish definitive causal relationships but rather to provide a broad, population-level assessment of postoperative OUD risk within 1 year of surgery. The integrated TriNetX-web portal matching was applied using a 1:1 ratio by a greedy nearest neighbor for each demographic characteristic.
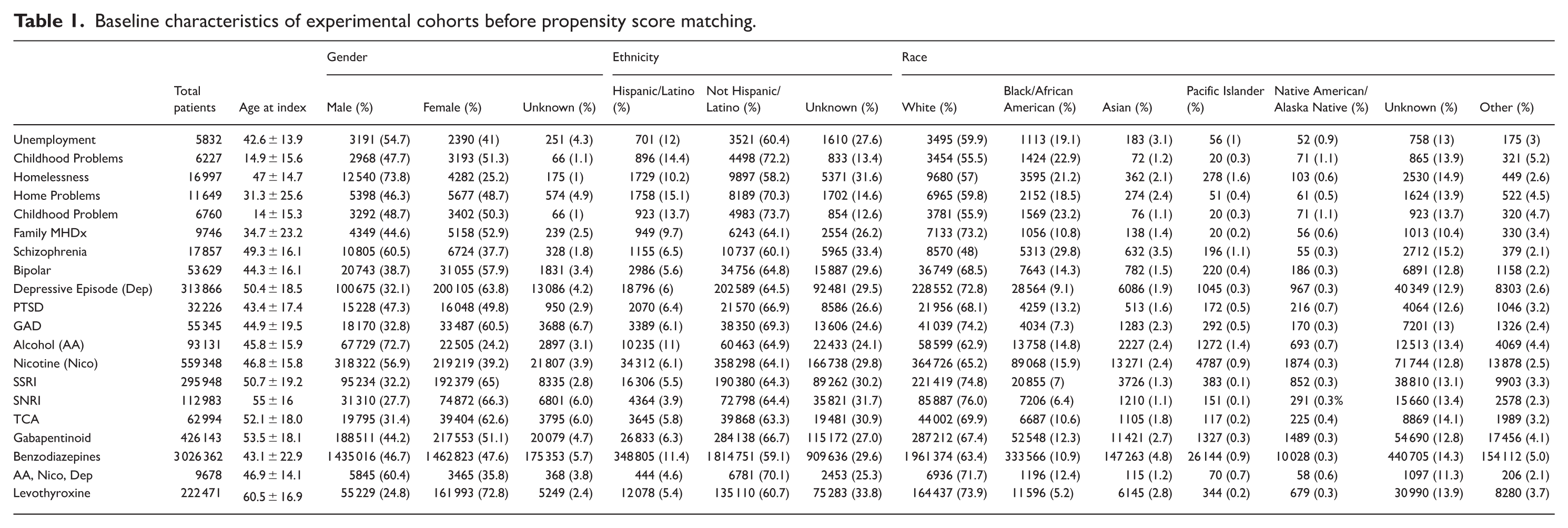
The standardized difference (Std diff) was used to evaluate the baseline characteristics in the propensity score-matched cohorts. Std diff < 0.1 is considered a small difference. 12 All demographic characteristics had a Std diff < 0.001 in all cohort pairs evaluated. Baseline characteristics before propensity score matching are shown in Table 1. Due to the large size of the control cohort, the characteristics of the experimental group remained unchanged after matching; thus, the post-matching characteristics table is identical to the pre-matching table and is not presented separately.
Baseline characteristics of experimental cohorts before propensity score matching.
Incident opioid-related disorders (ICD-10 F11) occurring anytime within 1 year of the index event were assessed. The odds ratio (OR) of developing OUD was calculated for each experimental and control cohort pair. In addition, the absolute risk was calculated for each cohort to give context to the reported OR.
Results
The analysis, which included matched cohorts ranging in size from 5832 to 559 348 patients depending on the risk factor, identified significant associations between several comorbidities and the risk of developing OUD within 1 year following surgery. Control cohorts were matched 1:1 for each comparison (Table 2). Homelessness was the strongest predictor, with an OR of 8.3 and a corresponding risk of 4.336%, highlighting its critical impact on OUD development. Similarly, a history of childhood problems (OR 6.4, risk 1.095%) and unemployment (OR 5.6, risk 3.429%) were strongly linked to increased OUD risk. Other notable predictors included family medical health issues (OR 4.8, risk 1.457%), nicotine dependence (OR 4.0, risk 1.609%), and post-traumatic stress disorder (PTSD; OR 4.3, risk 2.566%). Alcohol use disorder (OR 3.2, risk 1.773%), family problems (OR 3.1, risk 1.082%), and schizophrenia (OR 3.8, risk 2.066%) were also associated with higher OUD risk, although with slightly lower odds ratios. Mental health conditions such as generalized anxiety disorder (GAD; OR 2.9, risk 1.4%), history of depressive episodes (OR 2.9, risk 1.137%), and bipolar disorder (OR 5.6, risk 2.593%) demonstrated significant associations as well. In contrast, selective serotonin reuptake inhibitor (SSRI) use was associated with a comparatively lower magnitude of risk (OR 1.5, risk 0.638%) relative to other risk factors analyzed, though still indicating increased odds of postoperative OUD. Patients in the mixed category, which included alcohol use disorder, nicotine dependence, and depressive episodes, exhibited the highest risk overall (OR 13.9, risk 4.298%). Sample sizes for the cohorts were between 5832 and 3 026 362. Interestingly, benzodiazepine use was found to be associated with a decreased incidence of postoperative OUD (OR 0.77, risk 0.366%). This finding contrasts with the positive associations observed for all other psychiatric and social risk factors, likely reflecting differences in prescribing and monitoring practices rather than a true protective effect.
Effect of comorbidities on 1-year risk of opioid dependency in opioid naïve patients.

Significance at P < .0001.
Significance at P > .05.
To confirm that our cohort design did not inherently bias results toward detecting an association with OUD, a final comparison was created with the same structure as all previous groups and served as a negative control. An additional inclusion criterion was selected for this group, requiring a prescription for levothyroxine. Levothyroxine, one of the most prescribed drugs in the world, was selected as a negative control assessed prior to opioid exposure to validate the cohort structure.13,14 This group was compared to a control group that did not receive levothyroxine. No significant differences in the risk of developing postoperative OUD (OR 1.0, P = .416) were observed in these 2 groups, validating our cohort creation methodology. All findings—except the levothyroxine group—were statistically significant (p < .0001).
Discussion
Our findings emphasize the impact of SDoHs and mental health conditions on OUD development and offer new insights into the effect of perioperative context for these patients. Previous studies have assessed the incidence and risk factors for postoperative OUD across various surgical contexts.15-18 Our study builds on this work by specifically evaluating the impact of social determinants of health across a broad, undifferentiated surgical population. Additionally, our study design limits the effect of confounders by including a wide variety of surgical procedures including elective, emergent, major, and minor procedures.
The association between socioeconomic instability and heightened OUD risk underscores the profound impact of SDoHs on health outcomes. Homelessness and unemployment exacerbate chronic stress, limit access to non-opioid pain management options, and hinder the utilization of mental health services. 19 Similarly, mental health comorbidities such as bipolar disorder and schizophrenia, with their dysregulated stress and reward pathways, may amplify vulnerability to opioid misuse and dependency. 20
Chronic medical conditions, particularly those linked to systemic inflammation—such as diabetes, rheumatoid arthritis, and chronic pain syndromes—can contribute to increased opioid consumption by enhancing pain sensitivity and disrupting stress regulation. 21 Emerging research indicates that inflammatory processes may serve as a key connection between physical and psychological health, further exacerbating the risk of OUD. For example, pro-inflammatory cytokines like interleukin-6 and tumor necrosis-6α are found at elevated levels in individuals with chronic pain and mood disorders, which may heighten opioid sensitivity and increase the likelihood of dependency. 22 A point-of-care method for quantifying pro-inflammatory cytokines could provide valuable predictive biomarkers, offering insight into pain severity, treatment outcomes, and individual vulnerability to OUD. 23 These mechanisms illustrate the complex interplay between socioeconomic, medical, and psychological factors in shaping OUD vulnerability. Targeted interventions that address not only mental health and SDoHs but also medical comorbidities and inflammatory markers could provide a more comprehensive strategy for reducing OUD risk and improving overall outcomes.
Our findings align with and build upon previous research examining the relationship between SDoHs and OUD. For example, a recent study in patients undergoing spine surgery for lumbar degeneration demonstrated that preoperative opioid users were more likely to concomitantly exhibit disadvantageous SDoHs, including unemployment, lower median income and alcohol use which correlated with increased perioperative opioid utilization. 23 While this study was limited to a specific patient population and included individuals with prior opioid use, our research encompassed a broader surgical cohort and highlights that factors like unemployment and alcohol use are significant predictors of postoperative OUD across diverse surgical contexts.
The decreased observed risk associated with OUD development in those using benzodiazepines is likely a reflection of differences in opioid prescription practices in these patients due to the risks associated with the co-prescription of benzodiazepines and opioids. Thus, more conservative opioid prescribing rather than true protective pharmacologic protection is likely the cause of this observed effect. Because the database does not provide prescription counts or refill information, this explanation could not be directly tested, and future prospective research on this topic would offer important insight into this relationship.
Non-modifiable risk factors, such as male sex and advanced age, have been shown to be predictors of chronic opioid use in the postoperative period. 24 Our study complements these findings by emphasizing modifiable risk factors, including nicotine dependence, alcohol use disorder, and unemployment, which are actionable targets for intervention. Several genetic factors also associate with a heightened risk of developing substance use disorders and their common comorbidities.25-27 By being aware of modifiable and non-modifiable risk factors, physicians can be more aware of at-risk patients and offer tailored perioperative strategies to mitigate the risk of postoperative OUD, thereby contributing to more personalized patient care.
These data show a weaker association of OUD among patients utilizing SSRIs. This observation may indicate that effective psychiatric treatment reduces reliance on opioids for managing emotional distress and pain. 28
The potential interaction between SSRIs and opioids adds another layer of complexity. SSRIs are known to inhibit cytochrome P450 enzymes, including CYP2D6, which is responsible for metabolizing certain opioids. 29 This inhibition can impact opioid efficacy by prolonging their half-life, thereby increasing the risk of sedation or overdose, as highlighted in pharmacological studies. 30 These pharmacological interactions underscore the importance of careful monitoring and dose adjustments when SSRIs and opioids are prescribed together.
Furthermore, the relationship between depression, pain, and opioid use is notably complex. Studies examining predictors of pain management in depressed patients suggest that effectively treating depressive symptoms can alter pain perception and reduce the use of opioids for pain. 31 This finding emphasizes the critical role of integrated mental health care, particularly through the optimization of psychiatric treatment with SSRIs, in mitigating OUD risk in perioperative settings. By improving mental health outcomes and addressing chronic pain, SSRIs can significantly influence opioid-related risks.
Our study underscores the necessity of a targeted, multidisciplinary approach to perioperative care to address the elevated OUD risk associated with SDoHs and mental health conditions. Preoperative screening and risk stratification can identify at-risk patients, enabling clinicians to implement tailored interventions. Such tailoring has been previously proposed in the literature with ideas including:
Utilizing multimodal analgesia (eg, over-the-counter medications or regional anesthesia) and nonpharmacologic remedies (eg, physical therapy, acupuncture) to minimize opioid exposure. 32
Providing enhanced patient education and shared decision making to improve pain management and reduce postoperative reliance on opioids. 33
Establishing closer follow-up protocols with surgeons to monitor for signs of misuse and adjust prescriptions as necessary. 34
Coordinating care with mental health professionals to educate patients about opioid misuse. 35
Identifying at-risk patients and then utilizing these strategies has the potential to improve patient outcomes and contribute to broader public health efforts to mitigate the opioid overdose crisis and incidence of OUD. Effectiveness of and adherence to treatment for OUD is affected by SDoHs.36,37 Taking a patient’s specific social context into account in the perioperative period holds the promise to help mitigate the development of OUD in vulnerable populations.
Several limitations of this study warrant consideration and further investigation. Although propensity score matching minimizes confounding, unmeasured variables such as pain severity, provider prescribing behaviors, and cultural attitudes toward opioids may have influenced our results. The observational nature of our study limits causal inferences, and generalizability may be constrained by the specific patient population and healthcare systems included in the TriNetX database. Additionally, the reliance on ICD-10 codes introduces potential misclassification biases, and the dataset does not account for patients lost to follow-up. In addition, social determinants of health do not have specific diagnostic criteria akin to medical conditions. This means that the inclusion or exclusion of these determinants in a patient’s EHR is based on the clinical judgment of the provider. This introduces variability in how SDoHs are documented, which may lead to underreporting or misclassification. These limitations, however, reflect real-world EHR documentation practices and do not systematically bias the observed associations, particularly in such large and diverse cohorts.
Our analysis did not assess the effect of the type of surgical procedure performed. We chose not to stratify by procedure type to focus on the overarching impact of social determinants of health across diverse surgical populations. Future studies may build on this work by examining specific surgical subgroups and evaluating interactions between clinical and social risk factors.
Our exclusion criteria, particularly the omission of patients with prior opioid dependence or those who died within 1 year, may have skewed our sample toward healthier individuals, further limiting generalizability. To address the potential competing risk of death, we excluded patients who died during the follow-up period, but we acknowledge that this exclusion may introduce post-index selection bias, since mortality may be related to opioid exposure or shared risk factors. The TriNetX database only captures deaths reported within participating health systems and thus some deaths may not have been recorded, which remains a limitation of this study. Additionally, because this was a retrospective study utilizing a large national database, no sample size calculation or power analysis was conducted. This may limit the interpretation of the precision of some subgroup comparisons.
The decision was made to include patients whose only exposure to opioids was on the day of surgery even though these patients make up a significant proportion of each cohort pair. Since patients undergoing elective or day surgery would receive their prescription for opioids on the day of their surgery, these patients would have been excluded from the study. Excluding these patients would disproportionately remove patients undergoing same-day surgical cases and bias the cohorts toward more complex inpatient-type procedures. These patients were retained in the study to preserve a representative cross-section of all surgical populations, acknowledging that this approach may include a proportion of patients who received opioids only intraoperatively or in the post-anesthesia care unit and that these patients would potentially have only been exposed to opioids while still under the effect of anesthesia and at no point thereafter.
A key limitation of studies using ICD-10 codes (F11) to identify OUD, is that the lack of direct correspondence to DSM-5 diagnostic criteria. Codes such as F11.90 (“opioid use, unspecified, uncomplicated”) may over capture patients with ongoing opioid use who do not meet the clinical threshold for OUD, potentially leading to misclassification of the outcome. Additionally, all research based on EHR data, such as this study, has the limitation of being only as accurate as the person who is inputting the information. Thanks to the large patient populations and the magnitude of both the effects and significance of the findings, we feel confident in our results.
This study did not account for the specific opioid ingredient (eg, oxycodone), dose, or duration of prescription, as these data are not consistently available within the TriNetX platform. While these variables are important predictors of OUD, our analysis was specifically designed to evaluate the influence of SDoHs on postoperative OUD risk, rather than the effects of specific pharmacologic or surgical factors.
We conducted 2 supplemental sensitivity analyses to assess the robustness of our findings and the ICD-10 codes selected: (1) exclusion of patients with preoperative F11.1x (“opioid abuse”) codes in addition to F11.2x, and (2) exclusion of F11.90 (“opioid use, unspecified, uncomplicated”) as a primary outcome. Findings can be found in Supplemental Tables 1 and 2, respectively. To streamline these analyses, each was performed on 4 representative cohorts—Homelessness, Childhood Problems, Unemployment, and Nicotine. These were selected based on either the largest observed odds ratio increases or cohort size. In all cases, results were directionally consistent with our original analyses, with no significant changes to the primary conclusions.
Future research should address these limitations by incorporating additional variables, evaluating regional differences in prescribing practices, and assessing the impact of newer policies, such as PDMPs. Research into statewide PDMP outcomes has revealed beneficial trends, such as shorter opioid prescription durations post-surgery, yet has also identified unintended drawbacks, including sudden cessation of opioid prescribing in OUD individuals and patients seeking controlled substance prescriptions in neighboring states.38-40 This underscores the complexity of opioid prescribing and the need for a balanced approach that mitigates misuse-related risks while ensuring appropriate pain management. Longitudinal analyses would provide deeper insights into how prescribing patterns evolve in response to policy changes and their effects on patient outcomes.
The large, diverse dataset employed here allowed for a comprehensive analysis of multiple SDoHs and mental health factors, ensuring a broader representation of the general population. Additionally, these data represent real world evidence pulled directly from healthcare organization data. Patients included in these analyses are representative of both insured and uninsured patients in the hospital setting rather than being based on curated questionnaires or insurance-based aggregators. Propensity score matching reduced confounding, while the inclusion of a contemporary cohort reflected current prescribing practices, enhancing the relevance of our findings.
Conclusion
These findings emphasize the strong association of SDoHs with the development of postoperative OUD. Addressing and acknowledging these factors through tailored interventions is essential for improving patient outcomes and advancing public health efforts to combat the opioid overdose epidemic. Clinicians, researchers, and policymakers must collaborate to enhance screening tools, fund research into effective interventions, and develop policies that address underlying contributors to OUD risk and support physicians in navigating the treatment of high-risk patients. By integrating evidence-based strategies into clinical practice and policy frameworks, we can work toward reducing the burden of opioid misuse and its associated harms.
Supplemental Material
sj-docx-1-sat-10.1177_29768357261418824 – Supplemental material for Association of Socioeconomic and Psychiatric Factors with Postoperative Opioid Use Disorder: A Retrospective Cohort Analysis
Supplemental material, sj-docx-1-sat-10.1177_29768357261418824 for Association of Socioeconomic and Psychiatric Factors with Postoperative Opioid Use Disorder: A Retrospective Cohort Analysis by Trevor L. Murphy, Victoria Till, Christina R. Merritt, Kathryn A. Cunningham and George Golovko in Substance Abuse: Research and Treatment
Supplemental Material
sj-docx-2-sat-10.1177_29768357261418824 – Supplemental material for Association of Socioeconomic and Psychiatric Factors with Postoperative Opioid Use Disorder: A Retrospective Cohort Analysis
Supplemental material, sj-docx-2-sat-10.1177_29768357261418824 for Association of Socioeconomic and Psychiatric Factors with Postoperative Opioid Use Disorder: A Retrospective Cohort Analysis by Trevor L. Murphy, Victoria Till, Christina R. Merritt, Kathryn A. Cunningham and George Golovko in Substance Abuse: Research and Treatment
Footnotes
Acknowledgements
We also acknowledge the TriNetX platform for providing access to the large scale de-identified dataset that made this analysis possible.
Author Note
None.
Ethical Considerations
This study utilizes data from the TriNetX network. This platform complies with international and national data privacy regulations, such as the HIPAA in the United States and the GDPR in the European Union. All healthcare organizations contributing to the TriNetX network uphold stringent ethical principles, including securing informed consent for initial data collection and ensuring provisions for its secondary analysis.
Author Contributions
Trevor L. Murphy, BS: Conceptualization, Data curation, Formal analysis, Methodology, Writing. Victoria Till, BS: Investigation, Literature review, Visualization, Writing. Christina R. Merritt, PhD: Validation, Writing, Project administration. Kathryn A. Cunningham, PhD: Supervision, Writing, Funding acquisition. George Golovko, PhD: Supervision, Methodology, Writing, Data interpretation, Corresponding author responsibilities.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support was provided by UG3DA052282, UG3DA050317, Cure Addiction Now Foundation, and the Center for Addiction Sciences and Therapeutics at the John Sealy School of Medicine, University of Texas Medical Branch. This research was also supported by the UTMB Institute for Translational Sciences, supported in part by a Clinical and Translational Science Award (UM1 TR005443) from the National Center for Advancing Translational Sciences at the National Institutes of Health (NIH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data from the private TriNetX network was utilized and thus cannot be shared but it is available through a TriNetX license.
Supplemental Material
Supplemental material for this article is available online.
