Abstract
Background:
Opioid use disorder (OUD) can be associated with cognitive impairment. However, little is known about how patients with OUD self-perceive any cognitive impairments, particularly in attention, executive function, and memory. No studies have addressed this issue in patients with OUD and self-reported adherence to office-based medication-assisted treatment (MAT).
Methods:
This was an observational, multisite, cross-sectional survey study to evaluate self-reported cognitive function in adults with OUD, enrolled at 7 sites in the U.S., who had received ⩾3 months of office-based MAT. A cognitive self-report survey was administered to a clinical sample of adults with OUD (N = 255) adherent to buprenorphine (BUP; N = 139) or extended-release naltrexone (XR-NTX; N = 116). Impairments in attention, executive function, and memory were assessed with relevant items of the Executive Function Index (EFI) and Prospective Retrospective Memory Questionnaire (PRMQ). Factor analysis explored item factor structure; item scores were summed to generate scales, and association was estimated by linear regression.
Results:
Approximately one-third of participants reported head injury and nearly one-half reported psychiatric comorbidities, particularly anxiety and depression. Most participants reported difficulties in ⩾1 cognitive item: 80.8% (206/255) for EFI and 49.0% (125/255) for PMRQ. Factor analysis suggested 3 cognitive scales (attention, executive, memory). Differences between the XR-NTX and the BUP groups on memory, executive function, and attention either were not found or lost significance after adjustment for neurological history. seizure). All scales were associated with a range of demographic and clinical variables.
Conclusions:
Most adults receiving office-based MAT for OUD reported self-perceived cognitive impairment, but neurological comorbidities may also play a role in these perceptions.
Keywords
Introduction
Opioid use disorder (OUD) can be associated with cognitive impairment in the absence of intoxication or withdrawal. 1 A recent meta-analysis used objective neuropsychological functioning tests to identify a pattern of mild general cognitive impairment in patients with OUD, with a large effect on complex psychomotor ability, as well as effects on attention, working memory, memory, visuospatial ability, verbal fluency, and executive functioning. 2 It is not known whether these cognitive deficits precede, or are a consequence of, chronic opioid use. Another important consideration is whether the pharmacotherapies used for treatment of OUD (methadone, buprenorphine [BUP], extended-release naltrexone [XR-NTX]) may affect cognition.
In a randomized trial assessing cognitive function in patients seeking to discontinue BUP and initiate XR-NTX, improvements in several cognitive domains were associated with the transition from BUP to XR-NTX, particularly in participants entering the study on low-dose (<8 mg/day) BUP. 3 Investigators noted that these improvements in cognitive performance may be due to the discontinuation of BUP, the treatment with XR-NTX, or both. 3 However, little is known about how patients with OUD perceive these domains of cognitive impairment, particularly in executive function, attention, and memory. This subjective experience of cognitive function is directly relevant to quality of life as a treatment outcome in OUD, and is important to consider when tailoring treatment for individual patients. We undertook this investigation to understand the implications of objectively measured OUD treatment group-based differences in cognitive performance for patients’ subjective experience of cognitive functioning.
Another aspect of patients’ perception of their own cognitive function is metacognition—that is, the ability to realistically assess one’s own cognitive performance. It is recognized that a lack of insight (eg, into drug dependence severity) is a feature associated with substance use disorders. 4 In a study of metacognitive self-monitoring ability, individuals with opioid dependence undergoing methadone maintenance treatment had defective self-monitoring ability in visual perception compared with a matched healthy control group, despite intact perceptual acuity. However, there was no significant difference in memory metacognitive ability, despite weaker first-order memory performance by individuals with opioid dependence. These results suggest that addiction does not cause pervasive impairment in metacognitive ability; rather, the researchers observed impairment in only 1 specific task (the perceptual task, but not the memory task). 5
Although objective neuropsychological functioning tests are the gold standard to assess cognitive functioning, they have limited ecological validity. For example, because the tasks are assessed in a controlled environment, they may not detect subtle changes that impact daily functioning. 6 In contrast, self-report measures of cognitive functioning specifically ask an individual about tasks of everyday life involved in daily functioning (eg, trouble with multitasking, trying to plan, and thinking about consequences) and assess awareness of a person’s own abilities. This self-perception of cognitive functioning has been shown to precede identification of impairments by objective neuropsychological functioning tests 7 and, ultimately, may be an important quality of life outcome for individuals with OUD. In prior study of cognitive dysfunction in methadone-maintained patients, Copenhaver et al developed the cognitive dysfunction risk score (CDRS) based on demographics, medical and neurological history, psychiatric diagnosis and substance use history, in order to identify patients whose cognitive symptoms were likely to impede treatment.
The assessment of cognitive functioning in individuals with OUD is complicated by their medication-assisted treatments (MATs), also known as medications for opioid use disorder (MOUDs). There are 3 US Food and Drug Administration-approved MATs for opioid dependence: methadone, buprenorphine (BUP), and extended-release naltrexone (XR-NTX).8,9 In small studies with objective standardized neuropsychological functioning tests, methadone and BUP have shown some ability to improve cognitive impairment in patients with OUD, but treated individuals still show impairment relative to healthy controls.10-17 To date, no studies have addressed how patients with OUD who are adherent to office-based MAT perceive their own cognitive functioning. Recent work by Copenhaver et al 18 found that patients receiving methadone in opioid treatment programs (OTPs) exhibited evidence of cognitive challenges likely to interfere with treatment engagement. And on the Brief Inventory of Neuro-cognitive Impairment (BINI), methadone-maintained individuals reported neurocognitive symptoms which fell into 3 categories: minimal, moderate, or significant multi-domain concerns, the last of which may warrant specific accommodation strategies to improve treatment outcomes. 19 To our knowledge, this is the first investigation of self-reported cognitive impairment in patients treated with BUP or XR-NTX.
Therefore, the primary objective of this study was to explore the prevalence and types of self-reported cognitive impairments in attention, overall executive function, and memory in adults with OUD treated with BUP or XR-NTX for at least 3 months. We also report an exploratory objective that investigates factor structure of cognitive items and a self-report cognition instrument for use with adults with OUD receiving MAT, as well as the examined association between the cognitive scales and demographic, clinical, and medication variables. This is an important clinical question for individuals with OUD receiving MAT and is based on a recent analysis of data from the 2012 to 2013 National Epidemiologic Survey on Alcohol and Related Conditions-III (NESARC-III), a nationally representative survey of adults in the US, 20 which found that impairments in attention and executive functioning are positively associated with substance use. These investigators developed and validated 2 cognitive scales based on items from the Executive Function Index (EFI). 21 Our study focuses on adults receiving office-based MAT (BUP or XR-NTX), as methadone-maintained individuals represent a distinct group with supervised dosing and multiple weekly visits often combined with other psychosocial services and were therefore not included.
Methods
Study Design
This was an observational, multisite, cross-sectional survey study to evaluate self-reported cognitive function in adults with OUD enrolled at 7 academic or clinical research sites in the United States (NY, NJ, MD, PA, NC, IL, FL) who had received ⩾3 months of office-based MAT. This study received ethical approval from the Copernicus Group IRB on January 11, 2019. Prior to Screening, patients provided electronic informed consent.
Study Population
Participants meeting eligibility criteria were recruited from the participating sites. Survey participants were male and female adults, 18 to 60 years of age with a Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, diagnosis of OUD, maintained for ⩾3 months on BUP (sublingual or injectable) or XR-NTX, and free of illicit or nonprescribed opioids (by urine drug test). Participants were not experiencing symptoms of intoxication or withdrawal. Participants were recruited with a target of equal representation for BUP and XR-NTX, as well as for the age groups of 18 to 29 years, 30 to 44 years, and 45 to 60 years. Participants were excluded from the survey if they had current alcohol, benzodiazepine, or cannabis dependence (by medical record, subject’s self-report, and/or urine drug test). Participants were compensated between $35 and $50 for their study involvement.
Background Assessment
The following information was collected from participants: demographics; medical and psychiatric history; OUD information; MAT information; use of psychotropic and potentially sedating medications (anxiolytics, antidepressants, neuroleptics, psychostimulants, sleeping medications, and sedative antihistamines) in the last 30 days; health care resource utilization in 4 domains (ie, outpatient treatment, emergency treatment, urgent care/walk-in clinic, and hospitalizations); and recreational/illicit substance use in the past 7 and 30 days, selected from a predefined list of substances.
Survey
The survey was completed at a regularly scheduled office visit. Sites were trained on the use of an electronic tablet to facilitate the subject-reported questionnaires. There were no “interviews” involved, as the questionnaires were self-reported. The web-based survey was electronically administered on a tablet computer and took approximately 50 minutes to complete. For each participant, the survey was administered as a single assessment at a single time point, with no restrictions on the time of day or duration of the survey. Each subject was assigned an anonymous subject ID. Each participant completed the survey directly on the tablet; patients attested that staff had given the tablet to the patient for completion of the questionnaires.
The survey instrument included 81 items in 9 domains covering cognition and health (including sexual function, which is not reported in this manuscript). The survey included cognitive subscales from the EFI 21 and the Prospective and Retrospective Memory Questionnaire (PRMQ) 22 ; bothersomeness scale; Epworth sleepiness scale (ESS) 23 ; the Patient Health Questionnaire-2 (PHQ2) 24 ; the Treatment Effectiveness Assessment (TEA) 25 ; Treatment Satisfaction Questionnaire for Medication (TSQM) 26 ; the Work Productivity and Activity Impairment (WPAI), 27 and the Changes in Sexual Function Questionnaire (CSFQ), 28 which was optional.
This analysis focused on items from cognitive questionnaires that assessed impairments in attention/executive functions and memory (Appendix A). To assess impairments in attention/executive functions, the cognitive subscales of the previously validated EFI questionnaire (ie, those reporting strategic planning, organization, and impulse control) were used. 21 Attention and executive function scales have been validated in a large, nationally representative, general population sample. 20 To assess impairments in memory, the previously validated PRMQ 22 was used, which reported memory failures in everyday life. For both questionnaires, impairment in the items indicated perceived difficulties/failures to perform the task and was defined as endorsing either of the 2 most “extreme” responses.
Sample Size Considerations
The estimated sample size for this study was approximately 270 participants treated with BUP (n = 135) or XR-NTX (n = 135), as determined by the common rule-of-thumb of an n ⩾ 10 for each variable in the factor analysis model 29 to achieve all the primary, secondary, and exploratory objectives listed as study objectives in the protocol. The actual sample size for this study ended up with 255 participants treated with BUP (n = 139) or XR-NTX (n = 116), as the COVID-19 pandemic led to significant delays in enrollment. The n in each medication group was >100; thus the study was considered sufficiently powered to identify differences between groups.
Statistical Analysis
For the background information and primary objective, descriptive data are provided, including the proportion of patients in each treatment group with cognitive impairment on the EFI and PRMQ scales. Descriptive analyses were analyzed using SAS 9.4.
Tablets used to collect data were equipped with design features to alert participants if an item had been missed, before proceeding to the next page of the survey. This method of data collection was expected to minimize any missing data. In descriptive analyses, number and percent missing data were summarized by MAT type (buprenorphine or XR-NTX) and total.
Factor Analysis
Factor analysis (FA) was used to determine the factor structure of self-reported cognitive functioning items and to inform the development of cognitive scales for use in patients with OUD receiving MAT. FA was carried out in an iterative manner, with models tested and then re-configured where fit statistics suggested poor model fit. For both confirmatory FA (CFA) and exploratory FA (EFA), items were considered part of a factor with loadings > 0.40, and good model fit was based on the comparative fit index (CFI ⩾ 0.95), Tucker-Lewis index (TLI ⩾ 0.95), and root mean square error of approximation (RMSEA; <0.06 recommended, <0.10 acceptable).30-33 FA was carried out using Mplus 7.11, 34 using the weighted least squares mean and variance adjusted estimator, as appropriate for categorical items. 35 CFA used delta parameterization, and EFA used geomin oblique rotation and correlation for row standardization.35,36 Additionally, for each factor, to determine if the items were sufficiently correlated to compose a scale, internal reliability was assessed with Cronbach’s α, using SAS 9.4, with acceptable values ⩾.65.37,38
First, CFA determined whether the EFI data from 9 of the 12 items fit the 2-factor model (attention [mixed-up sequence, trouble multitasking, lose track, trouble summing up, and lose interest] and executive [future planning, learn from mistakes, consider consequences, and monitor self]), identified previously in NESARC-III data. 20 Next, EFA determined the structure of the 8 PRMQ memory items. Then, additional EFA models determined if the 3 additional EFI items (use strategies, lose temper, take risks) fit into the attention, executive, or memory domains. This resulted in adding “lose temper” to the attention factor. Cronbach’s α-values for the 6 attention items and 8 memory items were acceptable, but lower for the 4 executive items; information from the additional EFA models was used to suggest a 3-item executive factor. 30 Based on the results of this FA, 3 scales—attention, executive, and memory—were generated by summing relevant items. Higher scale scores indicate better cognitive functioning (ie, fewer perceived difficulties/failures to perform the task). To determine which variables were associated with cognition, we used linear regression to model each scale as a function of the following variables: MAT type (XR-NTX; BUP), age, sex, education, years actively used opioids, years on MAT, use of other medications, medical history (head injury, seizures), drug use, and psychiatric disorder history (depression, anxiety). In addition, the association of MAT type with each cognitive scale was adjusted for sociodemographic and clinical characteristics. The statistical software used for this analysis was Mplus 7.11.
Results
Demographic Characteristics, Medical History, and Substance Use History
The survey was completed by 255 participants treated with BUP (N = 139) or XR-NTX (N = 116). Demographic characteristics were generally reflective of a population with OUD; most (78% [199/255]) were White and the mean age of all participants was 37.6 years (Table 1). There were more females in the BUP group and more Black or African American patients in the XR-NTX group.
Demographic characteristics and medical history.

The percentages are based on participants who have suffered from anxiety or depression in the past 30 days.
Abbreviations. BUP, buprenorphine; MAT, medication-assisted treatment; OUD, opioid use disorder; SD, standard deviation; XR-NTX, extended-release naltrexone.
Approximately one-third (32.9% [84/255]) of participants reported a history of head injury and nearly up to one-half reported psychiatric comorbidities in the past 30 days, particularly anxiety (43.9% [112/255]) and depression (45.5% [116/255]; Table 1). There appeared to be more participants in the BUP group who reported posttraumatic stress disorder (BUP 41.0% [57/139], XR-NTX 24.1% [28/116]), panic disorder (BUP 25.9% [36/139], XR-NTX 9.5% [11/116]), anxiety (BUP 54.7% [76/139], XR-NTX 31.0% [36/116]), and depression (BUP 54.0% [75/139], XR-NTX 35.3% [41/116]) compared with those in the XR-NTX group. Prior history of active opioid use was generally long-term (mean [standard deviation (SD)] 9.2 [7.2] years) and appeared to be similar between groups (BUP 8.2 [6.3] years; XR-NTX 10.5 [8.1] years). Few participants self-reported substance use over the past 7 and 30 days; the most used substances (apart from nicotine and caffeine) were marijuana (20% of adults used in the past 7 days and 23.1% in the past 30 days) and alcohol (7.1% of adults used in the past 7 days and 14.9% in the past 30 days; Supplemental Table 1 in Appendix B). The rate of nicotine use in the past 30 days (76.1%) was higher than in the general US population (14% prevalence for cigarette smoking). 39 No participants reported using methylenedioxymethamphetamine, phencyclidine, hydrocodone, codeine/morphine, or hydromorphone. More participants had used opioids (heroin, oxycodone) in the past 30 days in the BUP group (6.5% [9/139]) than in the XR-NTX group (0.9% [1/116]). Additionally, 1 participant in the BUP group (0.7% [1/139]) and no participants in the XR-NTX group reported using fentanyl in the past 30 days.
Cognitive Impairment
Most participants reported impairment on at least 1 cognitive item: 80.8% (206/255) for EFI and 49.0% (125/255) for PMRQ. Overall, the BUP and XR-NTX treatment groups showed similar prevalence for impairment in at least 1 EFI item (BUP 82.7% [115/139], XR-NTX 78.4% [91/116]) or impairment in at least 1 PMRQ item (BUP 50.4% [70/139], XR-NTX 47.4% [55/116]). However, some numerical differences between groups were found for specific items (Figures 1 and 2).

Percentage of participants reporting impairment in an Executive Function Index (EFI) item. Impairment was indicated by the following responses: a“a lot/very much” or b“not at all/a little.”
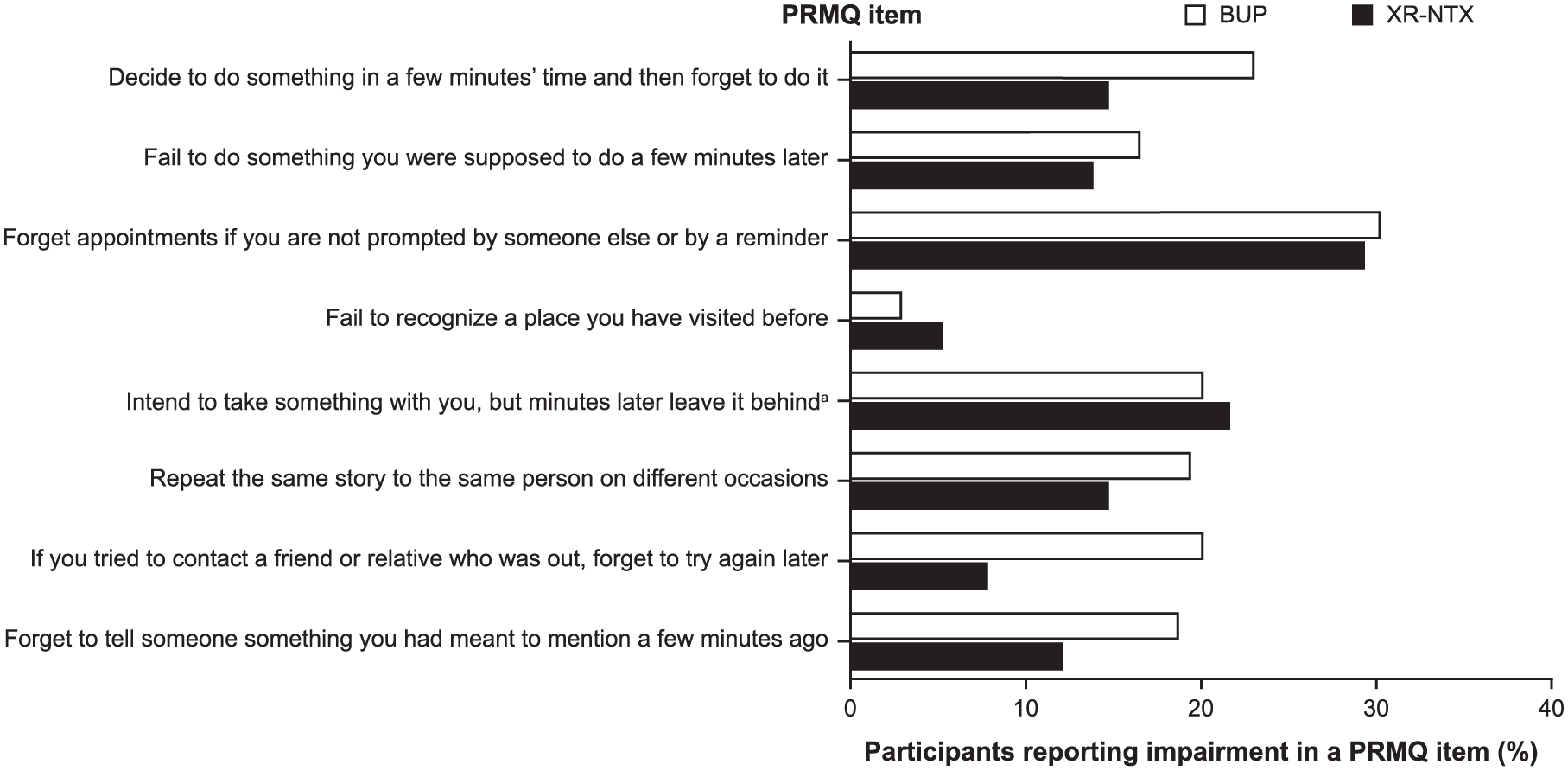
Percentage of participants reporting impairment in a Prospective Retrospective Memory Questionnaire (PRMQ) item. Impairment was indicated by the following responses: “very often”/“quite often.”
Cognitive Scales
Although the data fit the 9-item, 2-factor (attention and executive) model 20 in confirmatory factor analysis (CFI = 0.975; TLI = 0.965; RMSEA = 0.068; Supplemental Table 2 in Appendix B), further exploratory factor analyses were conducted to include other EFI items, leading to the identification of different factors. For the 8 memory items, EFA showed 1 eigenvalue above 1 (first 4: 4.94, 0.69, 0.57, 0.48), suggesting that the items fit a single-factor model. 40 CFI and TLI showed very good model fit (>0.95), while RMSEA was within the bounds of acceptable fit (<0.10; Supplemental Table 3 in Appendix B). The EFA model of the 5-item attention factor with the extra 3 EFI items suggested 2 factors (Supplemental Table 4 in Appendix B), with “lose temper” loading with the 5 attention items (Supplemental Table 5 in Appendix B), leading to a 6-item attention factor with good internal reliability (α = .82). The EFA model of the memory factor with the extra 3 EFI items suggested 2 factors (Supplemental Table 6), with none of the extra items loading on the memory factor, leading to an 8-item memory factor with good internal reliability (α = .89). The EFA model of the 4-item executive factor with 3 extra EFI items suggested 2 factors (Supplemental Table 7 in Appendix B), with no clear distinction between the executive items and extra items. Thus, the 4 executive items were considered for the scale, but internal reliability was below the acceptable level (α = .57); instead, the 3 items that loaded best onto the first factor and fit a 1-factor model (Supplemental Table 8 in Appendix B), and showed good reliability (α = .66), were selected.
The analyses suggested 3 cognitive scales (where higher scores are equivalent to less impairment, ie, better cognitive functioning): attention, executive, and memory. The attention factor included 6 EFI items (lose temper, mix up sequence, trouble multitasking, lose track, trouble summing up, and lose interest), the executive factor included 3 EFI items (use strategies, consider consequences, and monitor self), and the memory factor included all 8 items from the PRMQ. We propose that this set of scales be called the Cognition Scale for Medication-Assisted Treatment (CS-MAT).
Attention scale scores were significantly (P < .05) associated with medication type (Figure 3). Attention scale scores were higher (ie, better self-reported function) for XR-NTX (mean [SD], 17.4 [4.3]) than BUP (15.8 [4.9]; standardized beta—regression coefficient [95% confidence interval]: 0.35 [0.12-0.59]). This association remained significant after adjusting for demographic and clinical control variables, but the effects were attenuated and lost significance (P ⩾ .05) when adjusting for medical history (head injury, seizure; Table 2).

Association of demographic and clinical variables with cognition on the Cognition Scale for Medication-Assisted Treatment (CS-MAT). N = 255. A significant association (P < .05) is indicated by black squares. Variables that were not associated with the scale are indicated by gray squares. Any substance use (past 30 days) included alcohol, nicotine, caffeine, nonmedical benzodiazepines, nonmedical cannabis, cocaine, heroin, and amphetamine or methamphetamine use. There were no significant associations, apart from amphetamine or methamphetamine use (which is included on the figure), including any drug excluding alcohol, nicotine, and caffeine. Upon adjustment for medical history (head injury, seizure), differences in attention scale scores between XR-NTX and BUP were attenuated and lost significance.
Association of OUD MAT (XR-NTX vs BUP) with Attention Scale, Adjusted for Sociodemographic and Clinical Characteristics.

Abbreviations: ADHD, attention deficit hyperactivity disorder; BUP, buprenorphine; CI, confidence interval; MAT, medication-assisted treatment; OUD, opioid use disorder; XT-NTX, extended-release naltrexone.
Note. Entries with P < .05 are shown in italics. Regression coefficients are for the association of medication type with the attention scale, with different covariate controls in each row.
Neither memory scale scores nor executive scale scores were associated with a difference between BUP and XR-NTX (Figure 3).
Attention scale scores were also associated with sex (less self-reported impairment for males compared with females), education (less impairment with higher education), use of medications (benzodiazepines for anxiety, medications for depression, stimulants for attention deficit hyperactivity disorder, medications for sleep), head injury, seizures, anxiety, depression, and amphetamine or methamphetamine use (for all of these factors, less impairment was noted with a “no” response). Executive scale scores were associated with age (less impairment for 18-29 years compared with 45-60 years), MAT treatment duration (less impairment if duration was 13-24 months compared with at least 37 months), sex (less impairment in males), medication for depression, and amphetamine or methamphetamine use (for these factors, less impairment was noted with a “no” response; Figure 3). Memory scale scores were associated with sex (less impairment in males), age (less impairment for those aged 18-29 years compared with 45-60 years), medication for depression, head injury, anxiety, and depression (for these factors, less impairment with a “no” response; Figure 3).
Discussion
To our knowledge, this is the first study to investigate self-reported cognitive impairment in adults with OUD receiving office-based MAT in a real-world treatment environment. We found that among survey participants with OUD receiving office-based MAT and adherent to BUP or XR-NTX, approximately one-third reported head injury; up to nearly one-half reported psychiatric comorbidity, particularly anxiety and depression; and more than one half reported perceived cognitive impairment.
We adapted the self-reported cognitive functioning items to a specific self-report cognition instrument for use in patients with OUD receiving office-based MAT in real world environment, the CS-MAT. Use of the CS-MAT indicated that no significant differences in self-reported cognitive impairment between XR-NTX and buprenorphine groups were appreciated after adjustment for head injury/seizures. There were no differences in memory or executive scale scores between treatment modalities. In general, there was more cognitive impairment in females (compared with males), and in individuals receiving medications for health conditions (compared with those not receiving medications). We believe our findings highlight the prevalence, and potential importance, of cognitive concerns in stable patients receiving office-based MAT. These findings also extend the work of Copenhaver et al., 18 who found a high risk for cognitive dysfunction in patients with OUD engaged in methadone treatment. In addition, our findings will be of interest to researchers evaluating treatment efficacy and to clinicians seeking to be informed of patient-reported outcomes in patients with OUD.
In our study, the prevalence of self-reported cognitive impairments in adults receiving stable MAT for OUD was at least 50%, similar to previous findings of impairment in prospective memory among long-term opioid users. 41 In general, self-perceived impairment was similar between participants receiving BUP or XR-NTX, although differences in attentional functioning between adults receiving BUP and XR-NTX before covariate adjustment may warrant further investigation. Although we did not assess cognition before treatment initiation, other studies have shown that both BUP and XR-NTX can improve objective neuropsychological function. A recent study found that patients with OUD adherent to BUP-naloxone (N = 20) for 6 months had improved learning, memory, and global functioning. 42 And, in a clinical trial of participants with OUD receiving BUP who transitioned to XR-NTX, several cognitive domains improved within the first month on XR-NTX, particularly for patients who transitioned from low-dose BUP. 3
Interestingly, the items identified for the executive scale in this study differed from those that comprised the executive functioning scale from a previous study of the general US population. 20 We also identified an additional item for the attention scale. These findings suggest that some items may not be perceived consistently between populations, which is supportive of development of a specific questionnaire for individuals with OUD receiving MAT, as reported here.
The demographic information collected in our study also provided insights into the population of patients with OUD receiving office-based MAT. For example, although our study found that approximately one-third of adults receiving office-based MAT for OUD reported a head injury (ever, or treated for), it is possible that the number is overestimated given the nonspecific nature of the question. Nevertheless, recent evidence suggests that there is a link between traumatic brain injury and the development of OUD, with the potential for OUD also being a risk factor for future head injury. 43 This may help clinicians identify individuals who are at higher risk for OUD, and further research is needed to explore whether a history of head injury is associated with greater risk of self-reported cognitive impairment. In addition, the rates of psychiatric comorbidities observed in our study population are consistent with the prevalence of co-occurrence of OUD and mental disorders. A survey study of patients with OUD who were starting BUP treatment at a family medicine clinic (N = 100) found that 44% screened positive for anxiety, 31% for depression, and 52% for posttraumatic stress disorder. 44 A recent analysis of data from the 2015 to 2017 National Survey on Drug Use and Health found that 64.3% of individuals with OUD also had a psychiatric illness in the past year. 45 Further, there appears to be a bidirectional relationship between comorbid OUD and anxiety disorders, which can negatively affect prognosis. 46
Given the observational nature of our study, there are associated limitations. First, our results may not apply to the total OUD MAT population, owing to differences in demographics, study entry exclusion criteria, treatment duration, and comorbidities. For context, buprenorphine has demonstrated greater ease of induction than XR-NTX, as the latter can only be initiated after a period of detoxification. 47 The analysis presented here only included those patients who have successfully been inducted onto XR-NTX. In addition, buprenorphine and methadone maintenance treatment have been found to be associated with reduced all-cause and opioid-related mortality. 48 While fewer data are available for XR-naltrexone, the only clinical trial (N=570) directly comparing buprenorphine to XR-naltrexone found these treatments to be comparably effective and safe options. 47 Second, our study did not meet the expected sample size because of limitations imposed by the COVID-19 pandemic. While a larger sample can provide more precise estimates, the sample size was sufficient for factor analysis, based on the rule of thumb of n ⩾ 10 for each item. 29 Third, patients who volunteer to participate in an observational research study may differ from the general MAT population. Fourth, our results may also be influenced by the site investigator, as the study investigator judged whether there was concomitant or past use of alcohol, benzodiazepine, or cannabis dependence based on medical records, subjects’ self-reports, and/or urine drug tests. Fifth, we did not assess cognitive impairments before treatment initiation with BUP or XR-NTX, so we could not evaluate improvement following MAT (although this would be of clinical interest), and our study was not designed to support cross-sectional comparisons. Sixth, the survey included items from the EFI and PRMQ questionnaires, which have not been previously validated in an OUD population. Seventh, the prevalence of self-reported impairment was threshold-dependent, as impairment was defined as endorsing the 2 most “extreme” responses in both questionnaires. Despite these limitations, and given that our measures of self-rated cognitive impairments may by nature be influenced by personal bias and varying levels of self-awareness, our study provides a unique perspective on the daily functioning of adults with OUD in a real-world environment, and the limitations do not detract from the impact we identified on the quality of life of each individual. Future studies should confirm factor structure and assess validation of our proposed scales and comparison with objective neuropsychological functioning tests.
Conclusions
In conclusion, this study found that most adults receiving office-based MAT for OUD reported at least 1 self-perceived cognitive impairment. In general, cognitive impairment was widespread and variable. The potential clinical implications from our study for patients with OUD receiving MAT include consideration of (1) screening for cognitive challenges, including identification of risk factors associated with greater risk of cognitive impairment (eg, females in methadone maintenance, those receiving medications for concurrent psychiatric conditions, amphetamine use), in OTPs or office-based settings where OUD patients are treated; (2) development of a validated screening instrument for assessment of cognitive dysfunction in the OUD population; (3) providing appropriate psychosocial support resources for individuals with comorbid psychiatric conditions, as these may enhance overall patient well-being and thereby promote recovery; (4) educational training to improve executive functions 49 ; and (5) strategies to reduce the negative impact of cognitive difficulties on daily functioning.
Overall, our study identified major clinical conditions (psychiatric comorbidities and cognitive impairment) that should be taken into consideration by clinicians, as impairments in attention and executive functioning may influence the success of MAT for OUD.
A limitation of this study is the absence of a non-OUD comparator group, which would enable assessment of prevalence of self-reported cognitive deficits in the general population. Further research is needed on the effects of selected medications for OUD treatment on patients’ cognitive functions.
Supplemental Material
sj-docx-1-sat-10.1177_29768357251367574 – Supplemental material for Self-Reported Cognitive Impairments in Office-Based Treatment for Opioid Use Disorder with Buprenorphine or Extended-Release Naltrexone
Supplemental material, sj-docx-1-sat-10.1177_29768357251367574 for Self-Reported Cognitive Impairments in Office-Based Treatment for Opioid Use Disorder with Buprenorphine or Extended-Release Naltrexone by Efrat Aharonovich, Dvora Shmulewitz, Abigail Zavod, Elizabeth Keane, Anna Legedza, Deborah Hasin and Maria A. Sullivan in Substance Use: Research and Treatment
Supplemental Material
sj-docx-2-sat-10.1177_29768357251367574 – Supplemental material for Self-Reported Cognitive Impairments in Office-Based Treatment for Opioid Use Disorder with Buprenorphine or Extended-Release Naltrexone
Supplemental material, sj-docx-2-sat-10.1177_29768357251367574 for Self-Reported Cognitive Impairments in Office-Based Treatment for Opioid Use Disorder with Buprenorphine or Extended-Release Naltrexone by Efrat Aharonovich, Dvora Shmulewitz, Abigail Zavod, Elizabeth Keane, Anna Legedza, Deborah Hasin and Maria A. Sullivan in Substance Use: Research and Treatment
Footnotes
Acknowledgements
The authors would like to thank all individuals who completed the survey. Editorial assistance and medical writing support were provided by Rebecca Jarvis, PhD, of Envision Pharma Inc., and funded by Alkermes, Inc. This manuscript was developed in accordance with international guidelines for Good Publication Practice (GPP 2022).
Ethical Considerations
This study received ethical approval from the Copernicus Group IRB on January 11, 2019. Prior to Screening, patients provided electronic informed consent.
Informed Consent/Patient Consent
All participants provided informed consent.
Author Contributions
All authors were involved in the study design and participated in the interpretation of study results and in the drafting, critical revision, and approval of the final version of the manuscript. Additionally, Dr. Anna Legedza conducted the descriptive analyses and Dr. Dvora Shmulewitz conducted the factor analysis and regression analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This analysis was sponsored by Alkermes, Inc., the manufacturer/licensee of XR-NTX (Vivitrol®). Alkermes, Inc. was involved in the study design, data collection, data analysis, and preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Efrat Aharonovich has served as a consultant for Alkermes, Inc., and Dr. Efrat Aharonovich, Dr. Dvora Shmulewitz, and Dr. Deborah Hasin have served as investigators on Alkermes’-funded research. Ms. Elizabeth Keane, Dr. Anna Legedza, Dr. Abigail Zavod, and Dr. Maria A. Sullivan were employees of Alkermes, Inc. during the study and development of the manuscript, and may own stock in the company. Ms. Elizabeth Keane is currently an employee of Mural Oncology. Dr. Anna Legedza is currently an employee of Lyndra Therapeutics. Dr. Abigail Zavod is an employee of Vertext Pharmaceuticals. Dr. Maria A. Sullivan is currently an employee of Eli Lilly and Company.
Data Availability Statement
The data collected in this study are proprietary to Alkermes, Inc. Alkermes, Inc. is committed to public sharing of data in accordance with applicable regulations and laws.
Trial Registration Number/Date
Not applicable.
Grant Number
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
