Abstract
Background:
The United States’ (US) opioid overdose epidemic has evolved into a combined stimulant/opioid epidemic, a pattern driven in part by mitigating opioid overdose risk, variable substance availability, and personal preferences. This study aimed to investigate the association between self-reported substance preference (heroin or methamphetamine) and behavioral/health outcomes among individuals who used both heroin and methamphetamine in the rural US.
Methods:
The Rural Opioid Initiative is a consortium of 8 research cohorts from 10 states and 65 rural counties that recruited individuals reporting past 30-day injection of any substance or opioid substance use by any route from 1/2018 to 3/2020. Analyses were restricted to participants ⩾18 years, who self-reported either heroin or methamphetamine as their preferred substance and past 30-day use of both heroin and methamphetamine. We examined cross-sectional associations between preferred substance (heroin versus methamphetamine) and behavioral and health outcomes using random effects meta-analysis with adjusted regression models.
Results:
Among 1239 participants, 61% (n = 752) reported heroin as their preferred substance. Adjusting for age, sex, and race/ethnicity, methamphetamine preference was associated with lower prevalence ratios for current naloxone possession (adjusted prevalence ratio [aPR] = 0.68; 95% Confidence Interval [95% CI] = 0.59-0.78; P-value ⩽ .001), of ever being told they had the hepatitis C virus (HCV; aPR = 0.72; 95% CI: 0.61-0.85; P-value ⩽ .001) and a personal history of overdose (aPR = 0.81; 95% CI = 0.73-0.90; P-value ⩽ .001).
Conclusion:
In our study analyzing associations between preferred substance and various behavioral and health outcomes amongst people who use both heroin and methamphetamine, a majority of participants preferred heroin. Methamphetamine preference was associated with lower prevalence of naloxone possession, ever being told they had HCV, and prior history of an overdose. This study underscores the need for targeted harm reduction services for people who prefer methamphetamine in rural areas.
Keywords
Introduction
Data suggest the US has entered the fourth wave of the opioid overdose epidemic: a combined stimulant/opioid epidemic,1 -3 with a notable increase in methamphetamine use among people who use opioids in rural communities.4 -9 Polysubstance use, the practice of taking 2 or more classes of substances together or within a short time period, is common among people who use drugs (PWUD) and a driving factor of overdose deaths.10 -13 Motivations for the concurrent or sequential use of stimulants with opioids have been reported by some as a strategy to mitigate opioid overdose risk or to reduce opioid withdrawal symptoms.14 -17 Additional motivations include preferences for the simultaneous effects of both substances and increased availability and affordability of methamphetamine in local communities.7,15,18,19 In addition, some people who use methamphetamine are unintentionally exposed to opioids, and especially fentanyl, putting them at elevated risk for overdose because they are opioid naïve. 20
In the US, there are high rates of opioid/methamphetamine polysubstance use in rural communities.7,9,21,22 Polysubstance use, especially in the form of opioid/stimulant polysubstance use, is an important contributor to negative outcomes such as overdoses among PWUD in rural areas.10,13,23 Some PWUD have previously reported incorporating stimulants with their opioid use as a perceived way to reduce their risk of an overdose; however, evidence shows that the opposite is actually true—use of stimulants and opioids together increases the risk of an overdose. 23 From 2015 to 2021 there was a 7.5-fold increase in overdose deaths in the US.24,25 Nearly half of all fatal overdoses in the US in 2019 involved 2 or more substances and approximately one-third of fatal overdoses involved opioids and stimulants. 26 In the US, PWUD in rural communities experience high rates of fatal overdoses, 26.2 substance overdose fatalities per 100 000 standard population in 2020, and these rates have been steadily increasing.3,27,28 Substance availability and frequency of adulteration/contamination of substances with fentanyl, may leave rural communities at higher risk of overdose.1,29,30
Combined stimulant/opioid use likely drive other poor outcomes among PWUD in rural areas. 31 For example, among people who use opioids in the rural US, methamphetamine use has been negatively associated with past 30-day methadone treatment and retention of methadone treatment. 32 Understanding the unique outcomes of polysubstance use and what factors drive these outcomes, particularly among individuals who use both opioids and methamphetamine in rural communities, is essential for targeting interventions and harm reduction strategies.
PWUD differ in their preferences for opioids and stimulants. Individuals’ preferred substance is associated with various demographic characteristics, but has not been well studied with respect to behavioral and health outcomes. 33 While the adverse outcomes of opioid and stimulant polysubstance use are well documented, little is known on the unique outcomes associated with preferred substance among individuals that use both opioids and stimulants in rural US communities.5,23,34 -36 Understanding an individual’s substance preference may assist health care providers and addiction treatment professionals to understand underlying behaviors and level of risks to appropriately tailor interventions or prioritize efforts when resources are limited. We analyzed high-risk behaviors and health outcomes among PWUD who used both heroin and methamphetamine from rural regions across the U.S. in relation to their stated preferred substance. We hypothesized that risk behaviors and health outcomes vary based on preferred substance (heroin or methamphetamine) among individuals who used both in the past 30 days.
Methods
Study design
This analysis utilized cross-sectional, multi-research cohort data from the Rural Opioid Initiative (ROI). In brief, the ROI is a biphasic, multi-agency funded cooperative agreement with the goal of better understanding and addressing the opioid epidemic in rural settings within the US. The ROI includes 8 research cohorts spanning 10 states (Illinois, Kentucky, New England [Massachusetts, New Hampshire, and Vermont], North Carolina, Ohio, Oregon, West Virginia, and Wisconsin) and 65 US counties. 1 Individuals were recruited into the ROI from January 2018 to March 2020, and were eligible for inclusion if they self-reported past 30-day injection of any substance “to get high” or past 30-day use of opioids (e.g. heroin, fentanyl, pharmaceutical pain medication, etc.) by any route “to get high.”
Study participants
Eligibility criteria included a minimum age (⩾18 for 6 of 8 studies and ⩾15 for 2 studies) and residence in one of the 65 participating rural counties. All studies obtained local institutional review board approval for research activities and data sharing within the ROI. All participants provided written consent prior to questionnaire administration. This analysis was restricted to participants ⩾18 years. Analyses were restricted to participants who (1) reported either heroin or methamphetamine as their preferred substance, and (2) reported using both heroin and methamphetamine in the 30 days prior to enrollment. These restrictions allow us to control for confounding by substance use patterns and is intended to ensure that comparisons are being made among people who have equivalent patterns of substance use. This will allow us to focus on the differences among substance preferences and not substance use consequences as all participants are using the same 2 substances (but express preference for one or the other).
Study procedures
Participants were recruited using respondent-driven sampling (RDS). 37 Each research cohort recruited seeds to initiate peer recruitment from syringe service programs, local health departments, community health centers, and other agencies or community outreach programs. Seeds were provided coupons to recruit peers, and financial incentives were offered for recruitment ($10-$20 per eligible peer) and to participate in the study ($40-$60). Standardized surveys were administered to participants through audio computer-assisted self-interviews (A-CASI), computer-assisted personal interviews (CAPI), or computer-assisted self-interviews (CASI).38 -40 The survey included questions on demographic characteristic, substance use practices and related risks, engagement with harm reduction services, access to and utilization of health care, including treatment for substance use disorder, and sexual behaviors, amongst others.
Measures
Participants were asked “currently, which is your drug of choice for getting high,” and allowed to select one substance on the assessment from the following list of substance options: heroin, street fentanyl or carfentanil powder, opiate painkillers, synthetic opioids, buprenorphine, methadone, prescription anxiety drugs, cocaine or crack, methamphetamine, crystal meth, or amphetamine, gabapentin, clonidine, and other. Heroin and methamphetamine were selected for comparison as they were the most frequently preferred and used substances among ROI participants. 1 The primary exposure of interest in this analysis was preference for heroin versus methamphetamine “for getting high,” among PWUD who reported both heroin and methamphetamine use in the past 30 days.
To understand differences between heroin or methamphetamine as the preferred substance, we evaluated outcomes in 4 domains: (1) past 30-day use of other, additional substances (fentanyl, opioid pain medication, buprenorphine, methadone, benzodiazepines, cocaine/crack, gabapentin, and clonidine “to get high”), simultaneous injection of an opioid and a stimulant, and heavy episodic drinking (defined as having 4 drinks (females)/5 drinks (males) or more in a day); (2) health outcome-related ( possession of naloxone, self-reported previous testing for the hepatitis-C virus [HCV], ever been told they had the HCV, and personal history of overdose); (3) past 30-day injection drug use (IDU) and syringe/paraphernalia behaviors (current IDU, frequency of IDU, use of a syringe or needle previously used by someone else, use of supplies [e.g. cottons, cookers, spoons, or water] previously used by someone else, syringe mediated drug sharing [i.e. injecting substances that somebody else prepared, mixed, or divided with a used syringe], and use of multiple injections per injection episode [MIPIE]); and (4) past 30-day sexual behaviors and risk factors (traded sex for substances, money, housing, or other needs, sex without a condom, and sex with someone who injects substances without a condom).
Statistical analyses
Descriptive statistics were calculated to describe demographic characteristics of participants that used both heroin and methamphetamine in the past 30 days by their preferred substance (heroin or methamphetamine). We also estimated the prevalence ratios for preferred substance and the outcomes of interest by research cohort using multivariable relative risk regression with robust standard errors using the Huber-White estimator. 41 Pooled effects across research cohorts were calculated using traditional random-effects meta-analysis to account for the heterogeneity between populations across research cohorts. 42 Meta-analytic approaches have proven themselves useful as a statistical method to combine and summarize results from multiple independent studies within a larger consortium with similar research aims.43,44 Multivariable models were adjusted for age, sex, race/ethnicity. A sensitivity analysis replicating the methods utilized for the multivariable models in the main analysis was performed to estimate prevalence ratios using multivariable relative-risk regression restricted to participants who reported past 30-day IDU.
Results
Of the original 3048 participants who participated in the ROI consortium, 832 were excluded because they reported a substance other than heroin or methamphetamine as their preferred substance, 975 were excluded because they did not report both heroin and methamphetamine use in the past 30 days, and 2 were excluded because they were less than 18 years old. The final analytical dataset included data from 1239 individuals (Figure 1).

Flow chart depicting study participants’ eligibility in this analysis.
Overall, 41% of participants were female, the average age was 34.4 years (standard deviation [SD]: 9.3; Table 1). A majority of participants were non-Hispanic white (82%) and had experienced homelessness in the past 6 months (58%). A majority of participants reported heroin as their preferred substance (61% (n = 752)). Demographic and social factors, other than geographic region, did not differ by preferred substance.
Demographic characteristics by preferred substance among rural opioid initiative participants with self-reported past 30 days use of heroin and methamphetamine.

Abbreviation: SD, standard deviation.
Race/ethnicity are mutually exclusive categories. Hispanic includes everyone who is Hispanic. White, Black, American Indian, and Other/Unknown race include those who are White, Black, American Indian, or Other/Unknown race and not Hispanic.
Does not add up to 100% because participants could select more than one option.
“Full-time work (40 hour/week),” “Part-time work,” and/or “Retirement check.”
“Public assistance check – like TANF (Temporary Assistance for Needy Families), AFDC (Aid to Families with Dependent Children), etc., ” “Disability check, like SSI (Supplemental Security Income), military, or other,” and/or “Someone supports me.”
“Selling drugs,” “Selling sex,” and/or “Theft, shoplifting, or stealing.”
Defined as living from place-to-place, “couch-surfing,” on the street, in a car, park, abandoned building, squat, or shelter.
Other substances used in the past 30 days was common and differed by participants’ heroin or methamphetamine preference (Table 2). Participants who reported heroin preference had nearly double the prevalence of self-reported past 30-day fentanyl use, compared to participants who reported methamphetamine preference, 60% and 35%, respectively. Individuals reporting heroin preference had a higher percentage of using other sedating drugs, such as gabapentin and clonidine in the past 30 days. Individuals reporting methamphetamine preference, compared to heroin preference, more frequently reported heavy episodic drinking in the past 30 days, (57% and 47%, respectively). Fewer participants who reported methamphetamine preference reported currently possessing naloxone (34% versus 53% in the heroin preference group).
Bivariate associations by preferred substance and outcomes of interest among of Rural Opioid Initiative participants with self-reported past 30 days use of heroin and methamphetamine (N = 1239).
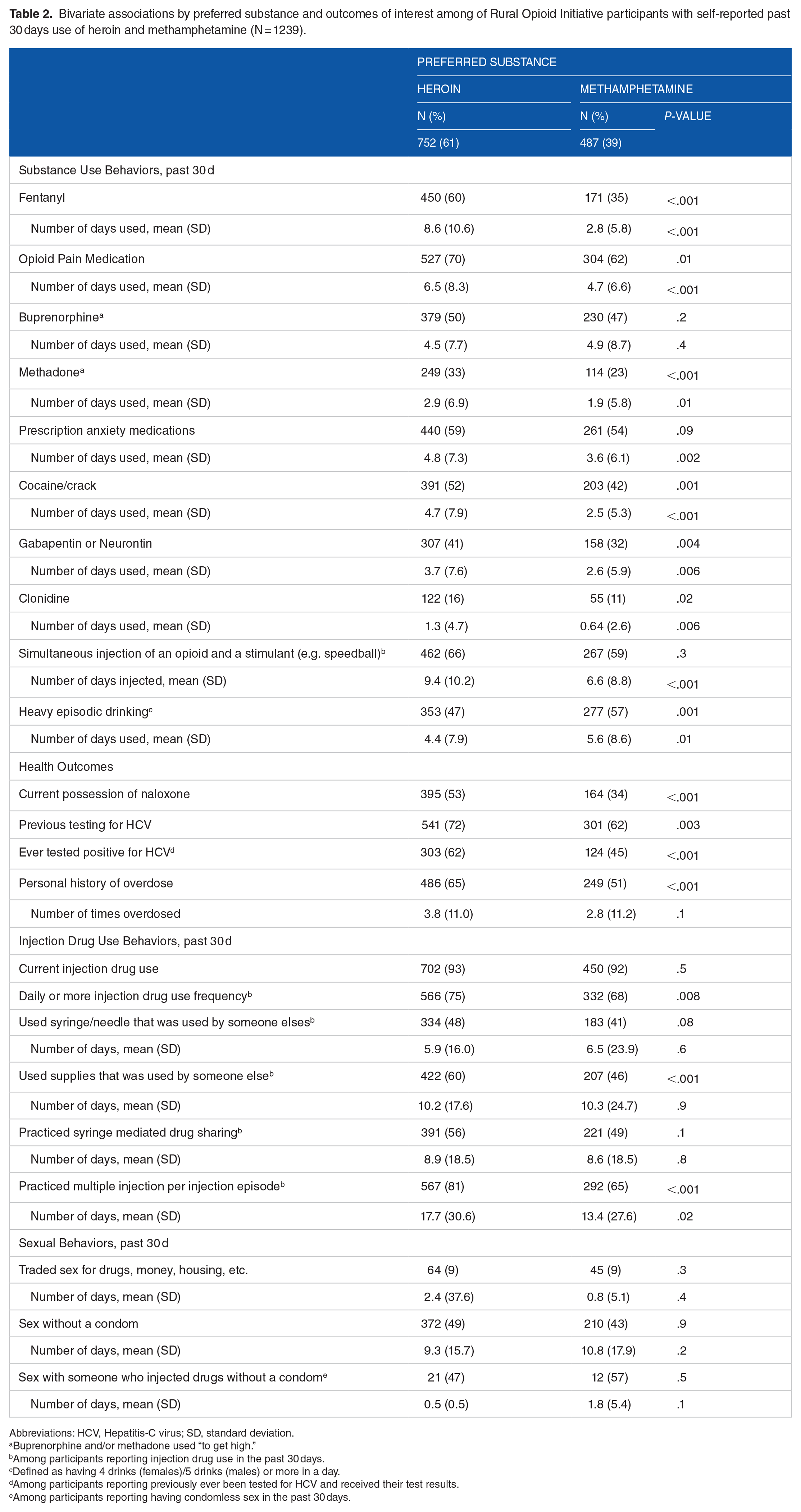
Abbreviations: HCV, Hepatitis-C virus; SD, standard deviation.
Buprenorphine and/or methadone used “to get high.”
Among participants reporting injection drug use in the past 30 days.
Defined as having 4 drinks (females)/5 drinks (males) or more in a day.
Among participants reporting previously ever been tested for HCV and received their test results.
Among participants reporting having condomless sex in the past 30 days.
In adjusted models, methamphetamine preference was associated with a lower prevalence ratio of past 30-day fentanyl use, opioid pain medication use, methadone use, and cocaine/crack use, compared to heroin preference (Table 3). Additionally, methamphetamine preference was associated with a 32% lower prevalence ratio of currently possessing naloxone (adjusted Prevalence ratio (aPR) = 0.68; 95% Confidence Interval [95% CI] = 0.59-0.78; P-value ⩽ .001). Methamphetamine preference was also associated with 28% lower prevalence ratio of reporting ever being told they had the HCV (aPR = 0.72; 95% CI: 0.61-0.85; P-value ⩽ .001) and a 19% lower prevalence ratio for reporting ever experiencing an overdose (aPR = 0.81; 95% CI = 0.73-0.90; P-value ⩽ .001), compared to heroin preference participants. Methamphetamine preference was also independently associated with a lower prevalence ratio for daily or more IDU frequency in the past 30 days (aPR = 0.91; 95% CI = 0.85-0.97; P-value ⩽ .01), past 30-day use of supplies used by someone else (aPR = 0.88; 95% CI = 0.79-0.98; P-value ⩽ .02) and past 30-day practice of multiple injections per injection episode (MIPIE; aPR = 0.87; 95% CI = 0.81-0.93; P-value ⩽ .001), compared to heroin preference. There were no associations between preferred substance and sexual risk behaviors in the past 30 days.
Multivariable prevalence ratio regression associations by preferred substance and outcomes of interest, adjusting for age, sex, and race/ethnicity among participants in the Rural Opioids Initiative who used both heroin and methamphetamine in the past 30 days (N = 1239; Reference: Heroin as preferred substance).
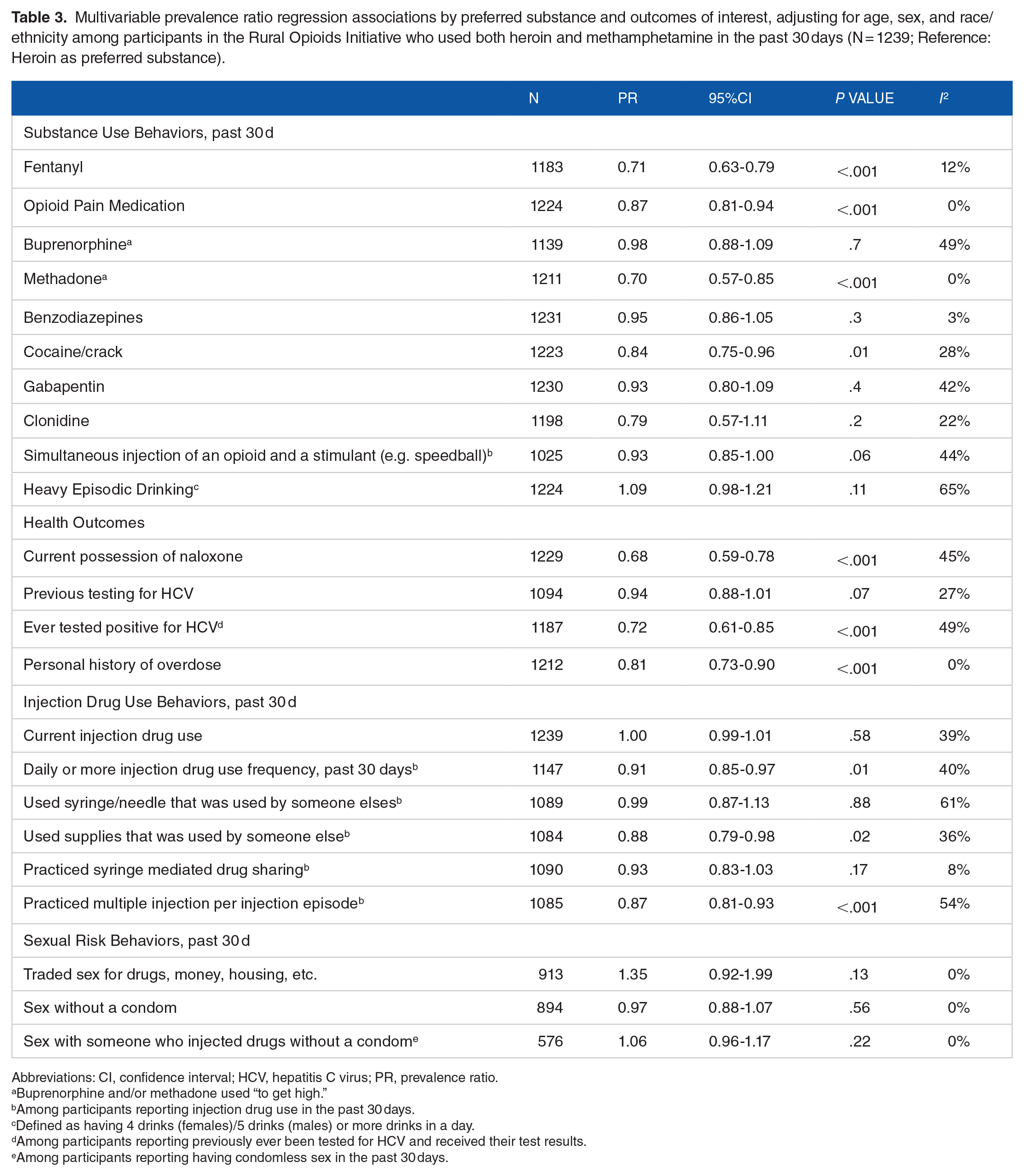
Abbreviations: CI, confidence interval; HCV, hepatitis C virus; PR, prevalence ratio.
Buprenorphine and/or methadone used “to get high.”
Among participants reporting injection drug use in the past 30 days.
Defined as having 4 drinks (females)/5 drinks (males) or more drinks in a day.
Among participants reporting previously ever been tested for HCV and received their test results.
Among participants reporting having condomless sex in the past 30 days.
The sensitivity analysis we performed that was restricted to participants who reported past 30-day IDU demonstrated slightly attenuated associations between preferred substance and outcomes of interest, however results did not significantly change compared to the main analysis (Supplemental Table 1).
Discussion
This study demonstrates how identification of an individual’s preferred substance might be a way to understand health risks and vulnerabilities among people that use both heroin and methamphetamine in rural communities. Our findings suggest methamphetamine preference is associated with various substance use behaviors, health outcomes, and IDU behaviors, including lower possession of naloxone, a lower prevalence of HCV, and a lesser likelihood of a prior overdose.
Methamphetamine preference was associated with lower prevalence ratios of fentanyl, opioid pain medication, and methadone use in the past 30 days. However, methamphetamine preference was associated with a lower prevalence ratio of past 30-day cocaine/crack use. One possible explanation for this finding could be substance supplies in participants’ areas. This result calls for further investigation to understand why within our study population, people who prefer methamphetamine are less likely to have used cocaine/crack in the past 30 days, despite both substances being a stimulant.
Our findings also showed that preferred substance was associated with having ever been told they had HCV. Among health outcomes, methamphetamine preference was associated with a 28% lower prevalence ratio of ever being told they had the HCV and a 19% lower prevalence ratio of a personal history of an overdose. Since, in the adjusted model, there was no significant association between preferred substance and previous testing for HCV, these findings may be a result of substance use severity. In our study, those reporting methamphetamine preference had a lower prevalence of daily or more IDU in the past 30 days, compared to heroin preference (68% versus 75%, respectively). IDU and daily IDU are associated with higher risk of both HCV infection and overdose.45 -48 HCV infection is also highly correlated with injection networks, therefore, the different networks that emerge from substance preference and injection use may be driving the differences in HCV risk. 49
Among our study participants, methamphetamine preference was associated with lower prevalence ratio of currently possessing naloxone, compared to heroin preference. This finding is important because all study participants are at-risk of experiencing an opioid overdose, given the affirmative self-reported past 30-day heroin use, and potential contamination of the methamphetamine supply with fentanyl. Despite use of both heroin and methamphetamine in the past 30 days, participants that reported methamphetamine as their preferred substance had a 32% lower prevalence ratio of possessing naloxone. This finding is consistent with another study noting that people who use methamphetamine reported less naloxone possession, than people who use heroin. 50 The absolute rates in both preferred substance groups show room for improvement via public health interventions and education, however people who prefer methamphetamine and also use heroin have more than two-thirds of participants not reporting possession of a life-saving intervention. The low prevalence of naloxone among all participants is a clear point of need for public health intervention. This is especially pertinent within rural communities where emergency services may take an extended amount of time to reach an individual that is experiencing an overdose. 51
Other studies have found that factors positively associated with naloxone possession are high-risk overdose indicators including: self-reported opioid use, injection as preferred substance administration method, and number of recent overdoses.50,52 People reporting methamphetamine as the preferred substance may not self-identify as being at risk of an opioid overdose in their substance use practice, despite their past 30-day heroin use. This theory is supported by another study that found that people who use cocaine were less likely to know how to recognize an opioid overdose and were less prepared to respond to an overdose, both in possessing naloxone and having received naloxone training. 53 While a vast majority of PWUD have heard of naloxone, there remains a gap of PWUD possessing naloxone, even among those who use opioids. 53
ROI previously reported that the polysubstance use of opioids and methamphetamine was associated with greater nonfatal overdoses compared with opioid use alone in rural people who use drugs. 34 A study using methamphetamine-related death data in West Virginia found that most methamphetamine-related deaths also involved opioids. 54 While participants who preferred methamphetamine in our study had a lower prevalence ratio of having a personal history of an overdose, this group may be more susceptible to the consequences of the substances they use being contaminated with fentanyl since they also had a lower prevalence ratio of possessing naloxone and they are opioid-naïve, such that even minimal exposure to fentanyl can substantially increase opioid-related overdose risks. 55 These results may signal important implications among people who use heroin who, despite reporting methamphetamine as their preferred substance, are more vulnerable to fatal outcomes if they experience opioid-related overdoses, due to their lack of naloxone possession.
Finally, methamphetamine preference was associated with a 9% lower prevalence ratio of daily or more injection drug use frequency, a 12% lower prevalence ratio of past 30-day use of supplies that were used by someone else and a 13% lower prevalence ratio of past 30-day practice of MIPIE. These findings may also be associated with the substance use severity difference between heroin and methamphetamine as preferred substance participants mentioned earlier. MIPIE increases the risk of injection-related complications, including viral transmissions, soft tissue bacterial infections, and acute sepsis.56 -59 Tester shots, which is where a person who injects drugs injects themselves with a small amount of the substance to determine how potent the substance is, is a form of MIPIE that has been a harm reduction technique utilized by people who use opioids to reduce the chance of overdose. 59 This may explain why in our study methamphetamine preference is associated with lower prevalence ratio of practicing MIPIE, compared to heroin preference, since the MIPIE practice of tester shots is predominantly used during opioid administration.
Limitations
The results of this study should be interpreted in the context of several possible limitations. The cross-sectional study design precludes inference for causality and use of a power analysis for sample size selection. Preferred substance and our outcomes of interest are collected simultaneously, despite substance preference predating the outcomes. Additionally, data on routes of administrations, aside from IDU, were not collected in ROI, precluding analyses based on alternative methods of administration, such as smoking. Given the nature of RDS and the limited sub-set of US states represented in this consortium, the ROI cohort is not a national probability sample, this may introduce sampling bias into the cohort due to the non-random nature of RDS. Data were self-reported, which may be subject to recall and social desirability biases. While frequency questions are commonly used in substance use research60 -65 and well understood, the ROI specific questions were adapted as part of a rapid epidemiological assessment, thus not specifically pilot tested. Despite these limitations, there are meaningful associations between preferred substance and behavioral and health outcomes for people who use heroin and methamphetamine.
Conclusion
In conclusion, preferred substance was associated with different patterns of other substance use, IDU behaviors, non-fatal overdose, and HCV infection among people who use both heroin and methamphetamine. Most notably, methamphetamine preference, compared to heroin, was associated with a lower prevalence ratio of ever being told they had the HCV, personal history of an overdose, current IDU, sharing supplies used by someone else, and MIPIE. Methamphetamine preference was also associated with lower prevalence ratio of current possession of naloxone. This study highlights the risk profile that emerges by individual’s substance preference and could be utilized to target harm reduction services. This study also emphasizes the importance of ensuring that naloxone distribution programs and overdose prevention efforts reach all individuals regardless of their preferred substance. Addressing barriers and knowledge gaps to naloxone possession amongst people whose preferred substance is methamphetamine, as well as preventing use of other opioids (such as fentanyl, through intentional fentanyl use, as well as testing substances for fentanyl contamination) and sedating medications (such as gabapentin) among individuals with preference for heroin, will be essential for decreasing overdose rates amongst people who use heroin and methamphetamine.
Supplemental Material
sj-docx-1-sat-10.1177_29768357241272374 – Supplemental material for Behavioral and Health Outcome Differences by Heroin or Methamphetamine Preference Among People in Rural US Communities Who Use Both Substances
Supplemental material, sj-docx-1-sat-10.1177_29768357241272374 for Behavioral and Health Outcome Differences by Heroin or Methamphetamine Preference Among People in Rural US Communities Who Use Both Substances by L Sarah Mixson, Bridget M Whitney, Wiley D Jenkins, Thomas J Stopka, P Todd Korthuis, Lydia N Drumright, Stephanie A Ruderman, Peter D Friedmann, Mai T Pho, April M Young, Ryan P Westergaard, David W Seal, Vivian F Go, William C Miller, William A Zule, Judith Feinberg, Hannah LF Cooper, Judith I Tsui, Heidi M Crane and Joseph A Delaney in Substance Abuse: Research and Treatment
Footnotes
Appendix

| Abbreviation | Meaning |
|---|---|
| A-CASI | Audio computer-assisted self-interviews |
| aPR | Adjusted prevalence ratio |
| CAPI | Computer-assisted personal interviews |
| CASI | Computer-assisted self-interviews |
| CI | Confidence intervals |
| GED | General educational development test |
| HCV | Hepatitis-C virus |
| IDU | Injection drug use |
| MIPIE | Multiple injections per injection episode |
| PR | Prevalence ratio |
| PWUD | People who use drugs |
| RDS | Respondent-driven sampling |
| ROI | Rural opioid initiative |
| SD | Standard deviation |
| SSI | Supplemental security income |
| TANF | Temporary assistance for needy families |
| US | United States |
Acknowledgements
The authors thank the other ROI investigators and their teams, the ROI Executive Steering Committee chair, Dr. Holly Hagan, the NIDA Science Officer, Dr. Richard Jenkins, and particularly, the participants of the individual ROI studies for their valuable contributions. A full list of participating ROI investigators and institutions can be found on the ROI website at ![]() .
.
Authors’ Contributions
LSM, BMW, SAR, LND, JIT, HMC, JAD were involved in the conceptualization of the research goals and aims. LSM, BMW, JAD were involved in the formal analysis. BMW was involved in the data curation. MTP, WJ, RW, DS, VFG, WCM, WZ, PTK, PF, TJS, JF, HC, AY, JIT, HMC were involved in funding acquisition. MTP, WJ, RW, DS, VFG, WCM, WZ, PTK, PF, TJS, JF, HC, AY were involved in conducting research investigation. BMW, JIT, HMC, JAD were involved in supervision of research planning activities. LSM, BMW, JAD were involved in the development of methodology design. LSM was responsible for the original draft of the manuscript. All authors were involved in reviewing and editing the manuscript. All authors gave meaningful input on the manuscript. All authors have approved of the final article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication is based upon data collected and/or methods developed as part of the Rural Opioid Initiative (ROI), a multi-study cohort with a common protocol which was developed collaboratively by investigators at 8 research institutions and at the National Institute of Drug Abuse (NIDA), the Appalachian Regional Commission (ARC), the Centers for Disease Control and Prevention (CDC), and the Substance Abuse and Mental Health Services Administration (SAMHSA). Research presented in this publication is the result of secondary data harmonization and analysis and supported by grant U24DA048538 from NIDA. Primary data collection was supported by grants UG3DA044829/UH3DA044829, UG3DA044798/UH3DA044798, UG3DA044830/UH3DA044830, UG3DA044823/UH3DA044823, UG3DA044822/UH3DA044822, UG3DA044831/UH3DA044831, UG3DA044825, UG3DA044826/UH3DA044826, and U24DA044801 co-funded by NIDA, ARC, CDC, and SAMHSA.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
