Abstract
Background:
Hospitals increasingly start methadone treatment for opioid use disorder (OUD). Rapid methadone titration has been described in hospitals with addiction consult teams; however, most hospitals lack these specialty services. We evaluated whether a pharmacist-led rapid methadone titration protocol (“RAMP-UP”) increased the proportion of hospitalizations that reached a minimally therapeutic dose of methadone (≥60 mg) before discharge.
Methods:
We conducted a retrospective, quasi-experimental, pre-post study comparing outcomes 7 months before versus 7 months after the implementation of RAMP-UP. The study included all adults admitted to a large academic medical center between March 2022 and April 2023 who newly initiated methadone for OUD. We used multivariable regression with generalized estimating equations to evaluate the primary outcome of achieving a total daily methadone dose of at least 60 mg by hospital discharge. Secondary outcomes included days to therapeutic dose, total daily methadone dose, and opioid-related safety events.
Results:
We identified n = 155 admissions, 55 pre-intervention and 100 post-intervention, among 124 unique patients with OUD. After RAMP-UP, a higher proportion of encounters reached ≥60 mg total daily dose (TDD) of methadone, but this difference was not significant in adjusted analyses (45.5% pre-implementation vs 60.0% post-implementation, P = .06). Post-implementation encounters had higher methadone TDD on discharge (62 vs 55 mg; P = .04) and, among those who reached 60 mg TDD, achieved this dose faster (9.0 vs 4.0 days, P < .001). Safety events were uncommon and similar (11.0% vs 12.7%; P = .80).
Conclusions:
Pharmacist-guided rapid titration accelerated attainment of therapeutic methadone doses and did not increase adverse events. Although the adjusted increase in the proportion achieving ≥60 mg TDD by discharge was not statistically significant, these findings demonstrate how inpatient clinical pharmacists can modernize inpatient OUD care.
Highlights
Hospitals are essential sites of care for opioid use disorder.
Pharmacists are uniquely well suited to guide implementation of rapid methadone.
Pharmacist-guided rapid methadone initiation was associated with a higher proportion of patients reaching a minimally therapeutic dose of methadone with no increase in safety events.
Introduction
Hospitals are essential sites of care for opioid use disorder (OUD) amid the ongoing North American overdose crisis.1,2 Nearly 2% of US hospitalizations include an opioid-related diagnosis, 3 and all-cause mortality for patients with OUD 12 months after hospital discharge is ~8%, similar to mortality rates after hospitalizations with myocardial infarction. 4 Alongside buprenorphine, methadone is one of 2 medications for OUD that reduce all-cause mortality by nearly 50%. 5 Per federal regulations, methadone can be administered for OUD treatment by hospital clinicians during medical or surgical hospitalization.6,7
Longstanding guidelines for starting methadone treatment for OUD in outpatient settings recommend 20 to 30 mg on day 1 (with an option for an additional 10 mg) followed by increasing the daily dose 5 to 10 mg after 3 to 7 days.8,9 This dosing approach was developed to account for methadone’s specific pharmacology and risks in unmonitored settings: Methadone accumulates with once-daily dosing due to its half-life of ~24 hours; it can prolong the QT interval; and it induces its own metabolism at varying rates.10-13 The traditional outpatient dosing approach takes more than 2 weeks to achieve a minimally therapeutic maintenance dose of at least 60 mg daily. 8 This approach does not account for the increased potency of fentanyl compared to heroin,14-16 nor for continuous monitoring available in hospital settings.
Over the past decade, fentanyl and other high-potency synthetic opioids have largely replaced heroin in the unregulated opioid supply. 17 This shift has renewed interest in methadone treatment 18 and prompted addiction experts to explore faster methadone titrations for hospitalized patients. 1 Ten single-site, single-arm retrospective cohort studies, 8 of which were completed in the inpatient setting, have found that much faster methadone titrations are feasible and can be completed safely.19-25 A recent systematic review 25 found these approaches achieved mean total daily doses (TDD) doses of about 40 mg on day 1, 70 mg by day 5, and 82 mg by day 7 and were guided by addiction consult teams. However, most hospitals do not have addiction consult services, potentially limiting the generalizability of these findings as generalists without specialty training may not feel comfortable applying novel dosing approaches to a complex medication.
At this study site during the intervention, as in most hospitals without addiction consult teams, 26 the psychiatry consult liaison (CL) service guided methadone initiation for OUD. Psychiatry CL team members varied in their experience and comfort with methadone titration, with most adhering closely to published guidelines. The study site hospital has a high volume of patients with OUD using fentanyl and frequent patient-directed discharges. 27 In this setting, rapid methadone titration was viewed as a potential strategy to improve opioid withdrawal treatment and retention, but psychiatry teams did not feel comfortable implementing this approach without additional support due to concerns about over-sedation and QT prolongation.
In hospitals without addiction consult services, pharmacists are uniquely well-suited to guide implementation of rapid methadone titration based on their expertise in pharmacology and ability to monitor for adverse drug effects. The aim of this study was to assess whether a pharmacist-led methadone initiation approach, Rapid Access to Methadone for hospitalized Patients with OUD at Penn (RAMP-UP), increased the proportion of patients who achieved minimally therapeutic methadone maintenance doses for OUD treatment before hospital discharge in a hospital without an addiction medicine consult service. As secondary and exploratory outcomes, we also assessed safety and hospitalization outcomes for patients starting methadone treatment for OUD before versus after RAMP-UP. We hypothesized that RAMP-UP would increase the proportion of individuals who achieved therapeutic methadone dose prior to discharge without an increase in safety events.
Methods
Study Design and Setting
This was a pre-post study of adult patients admitted to a large, quaternary care academic hospital in Philadelphia from March 2022 to May 2023 who were started on methadone treatment for OUD. This study was approved by the institutional review board with a waiver of informed consent for the use of deidentified data. We followed the Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines for cohort studies. 28
Prior to the RAMP-UP intervention, psychiatry CL services followed a traditional outpatient methadone titration: 30 mg initial dose with increases of 10 mg every 3 to 7 days. During the RAMP-UP intervention, a pharmacist (the author E.R.C.) was available to collaborate with psychiatry teams to implement a rapid methadone titration protocol. The pharmacist had completed 1 year of general residency training and was in the process of completing specialized training in pain management during the RAMP-UP intervention. During the first month of the intervention, the pharmacist supported the psychiatry team through direct consultation, joint visits with the psychiatry providers, and close monitoring for patient sedation and pertinent laboratory values and electrocardiogram (EKG) changes.
The rapid titration protocol targeted a 60 mg TDD of methadone in 3 days: 30 mg once + 10 mg at least 4 hours later on day 1, 40 mg once + 10 mg at least 4 hours later on day 2, and 50 mg once + 10 mg at least 4 hours later day 3. 21 During the first month of RAMP-UP, all hospitalized patients initiating methadone underwent rapid methadone titration. Candidate patients were referred by psychiatry services to the pharmacist, who prospectively collected detailed data on QTc interval and adjuvant dosing of short-acting opioids for safety assessments. After the first month, the pharmacist moved in to support a different service. Psychiatry services could implement rapid methadone titration with or without consulting the same pharmacist based on their comfort and experience. Pharmacist support after the first month was provided through telephone calls or embedded chat feature in the electronic health record.
We assessed methadone dosing, safety, and hospitalization outcomes in the 7 months preceding versus after the implementation of RAMP-UP.
Cohort Definition and Identification
We extracted data from an electronic health record reporting database (Clarity for Epic Systems Corporation). The intervention started in October 2022. The 7-month observation period before the intervention was March to September 2022. The 7-month observation period after the intervention was October 2022 to April 2023.
To identify patients with OUD and exclude patients who received methadone for chronic pain, we included only adult patients admitted during the study period who received a single dose of ≥40 mg of oral methadone (or equivalent in intravenous [IV] or intramuscular [IM] formulations, using a 1:2 conversion for IV/IM:oral). This criterion was chosen based on face validity because methadone for chronic pain is typically dosed as 5 to 20 mg given 3 times daily. 29
Next, to exclude patients continuing outpatient methadone treatment, we excluded patients who received a single dose of ≥60 mg by the end of the second day of receiving methadone. Patients who received at least 60 mg by the end of the second day were assumed to be continuing outpatient methadone treatment while hospitalized, not to be newly initiating methadone treatment. We allowed for up to 2 calendar days because methadone dose verification is sometimes delayed during hospitalization. We validated this identification method of methadone initiation for OUD by reviewing 10 randomly selected patients, all of which were appropriate for inclusion.
Exposure
The exposure was methadone treatment initiation for OUD. To describe the intervention during each period, we report the TDD of methadone by day of methadone titration, with day 1 defined as the first calendar day during which methadone was administered. Since methadone is typically dosed with the largest dose in the morning, we defined day by calendar day, not by 24-hour period.
Outcomes
We compared patient characteristics including age, sex, race, ethnicity, primary insurance, and Charlson comorbidity score before and after RAMP-UP. Encounter characteristics such as primary service, admission diagnosis, hospital length of stay (LOS), and urine drug test (UDT) results were compared between encounter groups.
The primary outcome was the proportion of patients in each arm who achieved at least 60 mg TDD of methadone by the day of discharge. The dose of 60 mg daily was selected because it is considered to be the minimally effective maintenance dose as defined by American Society of Addiction Medicine 8 and the Substance Abuse and Mental Health Services Administration. 30 As secondary dosing outcomes, we assessed the number of calendar days from the first methadone dose until the target TDD of 60 mg was achieved (among those who did achieve this dose level), and the methadone dose for each of the first 5 days of methadone dosing among patients who remained hospitalized on each day.
To assess safety, we compared the rate of opioid-related safety events before versus after implementation of RAMP-UP. A methadone-related safety event was defined as either naloxone administration or sedation (Richmond Agitation Sedation Scale [RASS] 31 <−2 or Pasero Opioid-induced Sedation Scale [POSS] 32 >2) after the first methadone dose administration. 27 Patients were monitored according to the medical and surgical unit standards, including RASS and/or POSS measurements collected as part of routine care. We also assessed the proportion of before medically advised (BMA) 33 discharges, LOS for hospitalizations with and without BMA discharge, and hospital readmissions within 30 days of discharge.
Last, we report additional data on QT interval changes and use of short-acting opioids during the first month of the RAMP-UP intervention, when these data were collected prospectively by the RAMP-UP pharmacist.
Statistical Analyses
To compare patient characteristics between cohorts, we used Pearson chi-squared tests for categorical variables and Kruskal-Wallis nonparametric tests for continuous variables. To compare encounter characteristics, we used generalized estimating equations (GEE) to account for possible correlations induced by some patients contributing more than one encounter. For dosing, safety, and hospitalization outcomes (reported as encounter characteristics), we again used GEE to account for multiple admissions from the same patient. These models controlled for age, gender, race/ethnicity, and primary service as prespecified potential confounders for the outcomes of interest. To compare dosing periods during the first 5 days of an encounter, we used GEE models with period-by-day interactions. For methadone initiations during the first month of RAMP-UP, EKGs were obtained daily, we also provide descriptive statistics for baseline QTc (before methadone), changes in QTc interval, and oral morphine equivalent (OME) of short-acting opioids used during methadone titration. We used SAS (v9.4) for analyses and a 2-sided significance level of P < .05.
Results
Sample Identification and Description
We identified 457 hospital admissions (176 pre-RAMP-UP and 281 post-RAMP-UP) where a single dose of ≥30 oral methadone (or equivalent in IV/IM formulations) was administered during the study period. After limiting the sample to admissions with at least 1 single dose ≥40 mg and without a single dose of ≥60 mg by day 2, we identified 155 total hospital admissions among 124 unique patients where methadone was newly initiated to treat OUD. Of these, 55 admissions were pre-RAMP-UP (31% of the original sample of 176 encounters in this period) and 100 admissions post-RAMP-UP implementation (36% of the original sample of 281 in this period).
Among the 124 unique patients, mean age was 43 years (SD 11.6); 49 (40.0%) were female; 24 (19.4%) self-identified as black or African-American, 87 (70.2%) as white, and 13 (10.5%) other or unknown race; 12 (9.7%) identified as having Hispanic or Latino ethnicity; and 84 (68.3%) were Medicaid insured (Table 1). Of the 155 hospital admissions, 74.8% were to Medicine services and median LOS was 9 days (interquartile range [IQR] 4, 22). Of 110 admissions with UDT performed, 92.7% detected fentanyl and/or norfentanyl and 38.2% detected methadone; however, it is unclear whether UDT samples were obtained after initial methadone administration as UDT collection times were not included in the data.
Patient and Encounter Characteristics by Cohort.
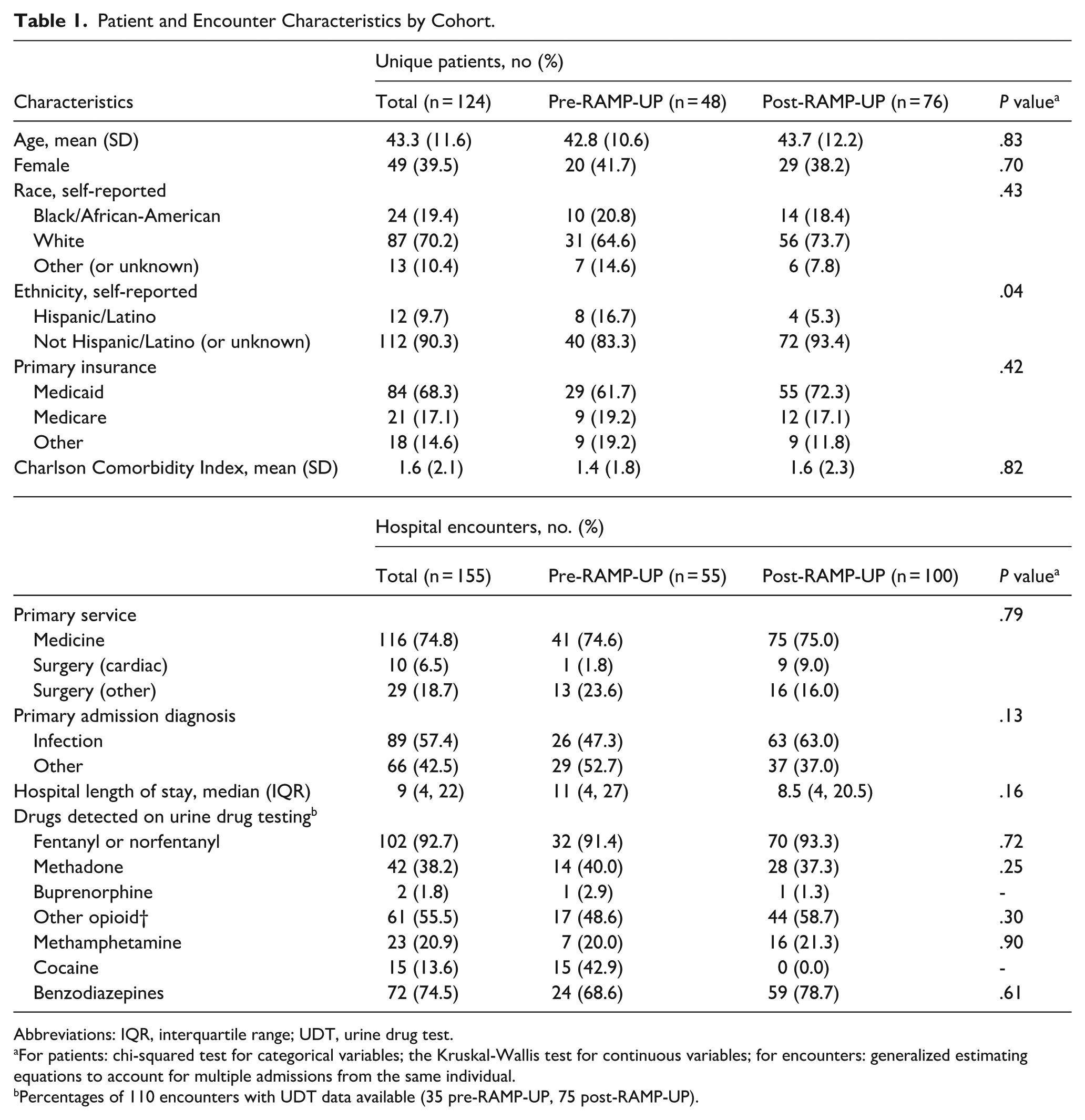
Abbreviations: IQR, interquartile range; UDT, urine drug test.
For patients: chi-squared test for categorical variables; the Kruskal-Wallis test for continuous variables; for encounters: generalized estimating equations to account for multiple admissions from the same individual. b Percentages of 110 encounters with UDT data available (35 pre-RAMP-UP, 75 post-RAMP-UP).
Dosing Characteristics
A higher proportion of encounters reached minimally therapeutic methadone dose (60 mg TDD or higher) during hospitalization after RAMP-UP was implemented compared with before RAMP-UP; however, this difference was not statistically significant in adjusted analyses (45.5% pre-RAMP-UP vs 66.0% post-RAMP-UP, P = .04 in unadjusted analyses, P = .06 in adjusted analyses; Table 2). Among the 91 encounters (25 pre-RAMP-UP, 66 post-RAMP-UP) that did reach 60 mg TDD, encounters after RAMP-UP reached this dose faster (9.0 days [IQR 5.0, 23.0] pre-RAMP-UP vs 4.0 days [IQR 2.0, 90] post-RAMP-UP, P < .001 in unadjusted and adjusted analyses). After the RAMP-UP intervention, the total methadone dose was higher on titration days 2 to 5 (P = .005 unadjusted, P = .007 adjusted) as well as on the day of discharge (P = .04 on unadjusted and adjusted analyses); however, the difference in methadone TDD by day and on discharge was only modest (eg, 54.8 mg TDD pre-RAMP-UP vs 62.2 mg TDD after RAMP-UP on discharge; Table 3).
Methadone Dosing, by Cohort.
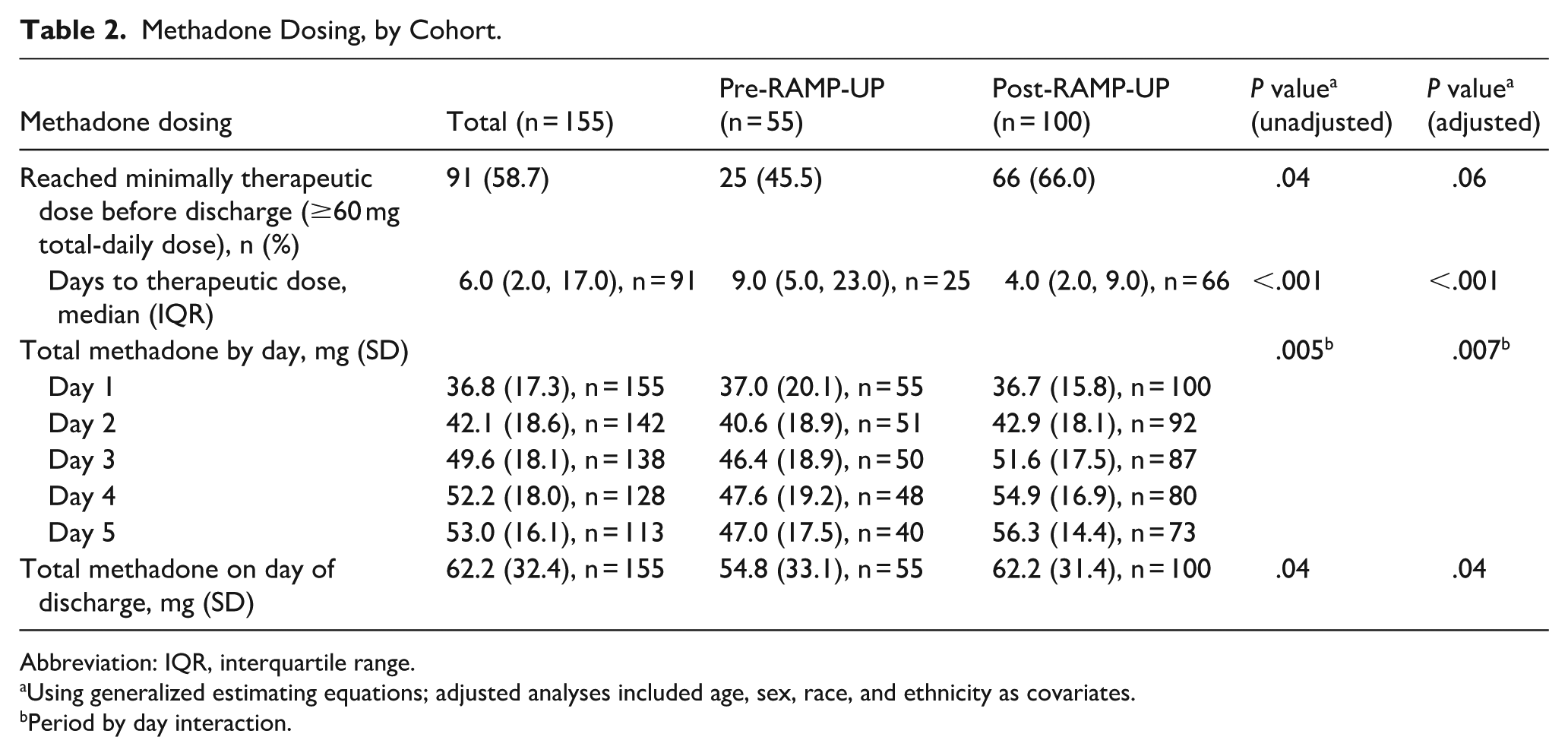
Abbreviation: IQR, interquartile range.
Using generalized estimating equations; adjusted analyses included age, sex, race, and ethnicity as covariates. b Period by day interaction.
Safety and Hospital Disposition Outcomes.
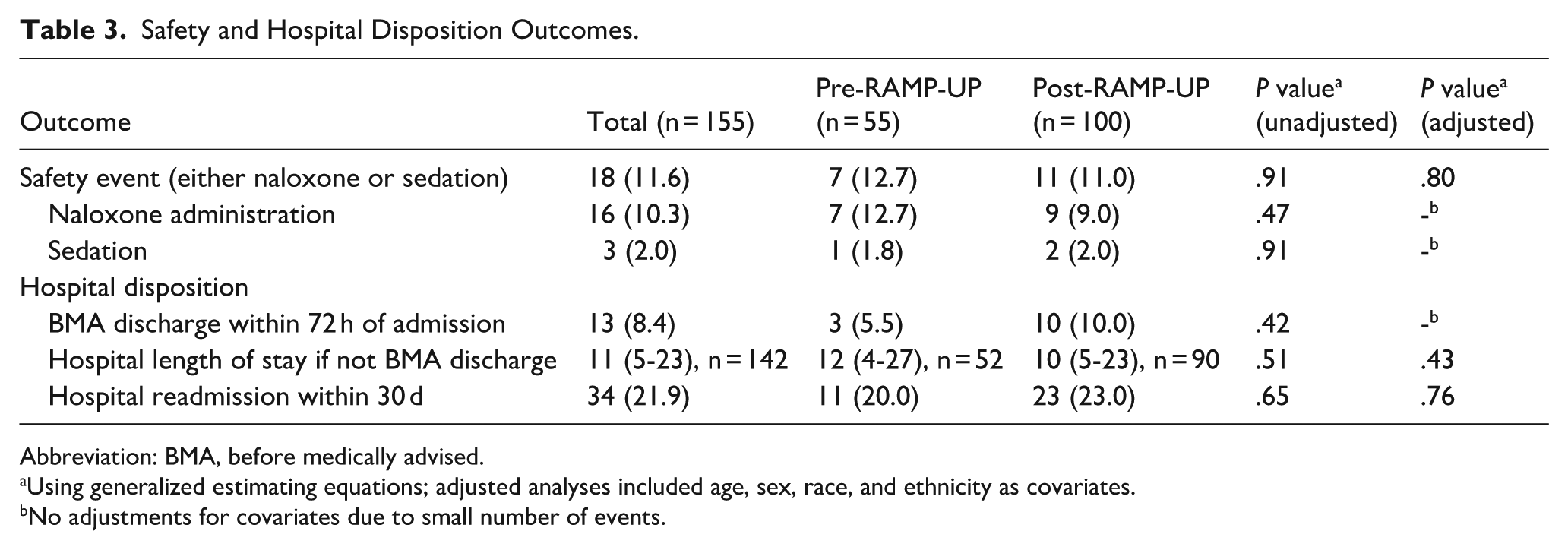
Abbreviation: BMA, before medically advised.
Using generalized estimating equations; adjusted analyses included age, sex, race, and ethnicity as covariates. b No adjustments for covariates due to small number of events.
Safety and Hospital Disposition Outcomes
There were few methadone-related safety events (defined as naloxone administration, RASS <−2, or POSS >2 after first methadone administration) per hospital admission, both before and after RAMP-UP implementation, with no significant differences by cohort (12.7% pre-RAMP-UP, 11.0% post-RAMP-UP, P = .80 in adjusted analyses). The rate of BMA discharges in the first 72 hours of admission was similar in both cohorts (5.5% pre-RAMP-UP and 10.0% post-RAMP-UP, P = .42 adjusted analyses), as was LOS for non-BMA discharges (median 12 days [IQR 4, 27] pre-RAMP-UP vs 10 days [IQR 5, 23] post-RAMP-UP, P = .43 adjusted analyses) and the frequency of hospital readmissions at 30 days (20.0% pre-RAMP-UP vs 23.0% post-RAMP-UP, P = .76 in adjusted analyses).
Additional Safety and Dosing Data From the First Month of RAMP-UP
During the first month of the RAMP-UP, 11 patients started methadone, all of whom underwent rapid methadone initiation. Median methadone dose by day and on discharge for these patients was 40 mg (IQR 40.0, 40.0) on day 1, 50 mg (IQR 50.0, 50.0) on day 2, 60 mg (IQR 20.0, 60.0) on day 3, and 70 mg (IQR 65.0, 72.5) on discharge. Ten patients (90%) achieved a TDD of at least 60 mg prior to discharge. There were no recorded administrations of naloxone and no recorded instances of over-sedation. Ten patients (90%) received concomitant short-acting full-agonist opioids (most often immediate release oxycodone, oral hydromorphone, and/or IV hydromorphone) with median daily (IQR) OME of short-acting opioids of 90 mg (IQR 30, 145) on Day 1, 134 mg (IQR 26, 202) mg on day 2, and 22.5 mg (IQR 0, 112) on day 3. At baseline, prior to methadone initiation, 1 patient had QTc >500 ms. This patient had QTc 504 ms before methadone and received methadone after a risk/benefit assessment and discussion; after 40 mg methadone on day 1, the QTc rose to 527 before falling to <500 ms on subsequent days (all with continued methadone dosing). During methadone titration days 1 to 3, only that same single patient (9.0%) had QTc >500 ms. The mean change in QTc across these 11 patients from days 1 to 3 was +16 ms (SD 18.8).
Discussion
At an academic hospital without an addiction consult service, pharmacist-guided rapid methadone initiation was associated with a higher proportion of patients reaching a minimally therapeutic dose of methadone (≥60 mg daily); however, this was not statistically significant on adjusted analyses. With pharmacist-guided titration, patients reached minimally therapeutic doses about 5 days faster, achieved modestly higher doses prior to discharge, and did not have a higher rate of methadone-related safety events. To our knowledge, this is one of the first studies to assess the impact of pharmacist guidance on hospital-based care for persons with substance use disorder.
Following a previously published taxonomy, 26 this intervention can be understood as an example of hospital-based opioid treatment, where hospital staff with interest and expertise focus on improving a single aspect of OUD treatment – in this case methadone initiation. This study demonstrates opportunities and limitations of this approach. The model is scalable, since pharmacists and psychiatry services are integral parts of almost all hospitals. Partnerships between pharmacists and psychiatry services could serve as a model for improving OUD care by modernizing inpatient methadone dosing in hospitals without addiction consult services.
Adjusted analyses in this study did not demonstrate a significant increase in achieving minimally therapeutic methadone doses before discharge. In this context, it is important to note that, after the 1-month RAMP-UP pilot, the pharmacist was no longer proactively involved in methadone titration. A single month of protocol socialization among psychiatry and admitting services may not have been powerful enough to sustainably change practice. Teams may have reverted to a traditional titration schedule in the absence of the pharmacist. A longer pilot period with proactive pharmacist engagement may have resulted in more robust uptake. Additionally, the null results for the primary outcome may highlight ways in which fully funded interprofessional addiction consult service models can offer more extensive support to hospitalized patients with OUD. 26 These services have been associated with reduced mortality. 34
This study builds on prior work19-22 to provide additional support for the safety of hospital-based rapid methadone titration, even in hospitals without addiction consult services. Although the dosing approach used in this study is faster than traditional outpatient dosing, it is still relatively conservative compared to alternatives described by some addiction consult service and outpatient opioid treatment programs.23,24 Most patients in this study received adjuvant short-acting opioids for ongoing opioid withdrawal, craving, and acute pain. This suggests that higher methadone doses or faster titrations may be safe and appropriate, at least for carefully selected patients, and could limit exposure to short-acting opioids, which are not available as long-term OUD treatment in the United States.
This study has several limitations. First, the retrospective, pre-post design limits causal claims and cannot account for unmeasured confounding, most notably trends in OUD treatment that were independent of this intervention. For example, during the study period, many patients were concerned for buprenorphine-precipitated withdrawal35,36 and xylazine wounds were increasingly common 37 ; in this context, escalating doses of methadone with adjuvant short-acting opioids were often used as initial treatment of opioid withdrawal at the study hospital, 27 even among individuals who never wished to reach 60 mg TDD. These secular trends may have increased the proportion of patients who received methadone for withdrawal, only to be transitioned to buprenorphine with low-dose initiation. These trends may have also contributed to the increase in proportion of BMA discharges. Second, this study relied on safety data collected during routine clinical care, which may be imprecise. Third, although our identification strategy for new methadone treatment of OUD performed well on validation with 10 randomly selected charts, this approach has not been externally or extensively verified. Last, there was a lack of available data to determine secondary outcomes for all patients, particularly readmissions or other utilization outside of our health system.
Conclusions
In a hospital without an addiction consult service, pharmacist-guided implementation of rapid methadone titration did not result in a statistically significant increase in patients who reached minimally therapeutic methadone doses by discharge; however, there was an association with faster methadone titrations and higher methadone doses with no increase in methadone-related safety events. This study demonstrates how pharmacists can serve a critical role in improving and modernizing addiction practice, even in hospitals without the resources to fund addiction experts. The approach to rapid methadone titration described in this study was feasible and safe but may have required more robust support from pharmacy services for a longer period for sustained change. As the complexity of withdrawal management and OUD initiations increases in the era of fentanyl and other high-potency synthetic opioids, there may be additional opportunities to leverage pharmacists to improve the care of hospitalized patients with OUD.
Footnotes
Ethical Considerations
Institutional review board approval was not required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIDA 1K23DA062008-01 (APT).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
