Abstract
Background:
To expand access to buprenorphine treatment for opioid use disorder (OUD), the X-waiver requirement was removed in January 2023. This study examined changes in OUD diagnosis and buprenorphine prescribing before and after waiver elimination.
Methods:
We conducted a retrospective analysis using TriNetX electronic health record data (EHR) on adults with OUD during the waiver-required (2021-2022) and elimination (2023-2024) periods. Logistic regression estimated the likelihood of new OUD diagnoses and buprenorphine initiation, adjusting for demographic and clinical characteristics.
Results:
Among 1 041 754 adults with an OUD diagnosis (516 508 in the waiver-required and 525 246 in the waiver-elimination periods), 295 675 participants (146 904 in the waiver-required and 148 771 in the waiver-elimination periods) had a new OUD diagnosis recorded. Among those with a new OUD diagnosis, buprenorphine was prescribed to 27 665 (18.8%) participants in the waiver-required and 29 223 (19.6%) participants in the waiver-elimination periods. Those in the waiver-elimination period, compared to those in the waiver-required period, were 2% less likely to receive a new OUD diagnosis (adjusted odds ratio [aOR] = 0.98, 95% confidence interval [CI]: 0.97, 0.98) and 14% more likely to have buprenorphine initiated (aOR = 1.14, 95% CI: 1.12, 1.16).
Conclusion:
In this retrospective analysis of a large EHR dataset, we found that following the elimination of the X-waiver, the incidence of new OUD diagnoses declined by 2%, while subsequent buprenorphine initiation increased by 14%. These findings highlight the impact of regulatory changes on buprenorphine prescribing and underscore the need for continued efforts to reduce barriers to this life-saving treatment for OUD.
Highlights
Since the elimination of the X-waiver in 2023, the electronic health record entry of new opioid use disorder (OUD) diagnoses declined by 2%, and the rate of subsequent buprenorphine initiation increased by 14%.
Individuals with chronic or mental health conditions were more likely to receive buprenorphine treatment.
Racial minority groups were more likely to be diagnosed with OUD, but less likely to receive buprenorphine treatment, suggesting persistent gaps in treatment access.
Introduction
Opioid misuse, use disorder, and related overdose have had a devastating impact on public health, straining healthcare resources, criminal justice system, and societal productivity. Medications for opioid use disorder (MOUD), including buprenorphine, have demonstrated effectiveness in improving outcomes for individuals with opioid use disorder (OUD) and in reducing the risk of fatal opioid-involved overdose. Yet, their use remains underutilized due to persistent barriers such as stigma, inadequate access to care, and policy restrictions.1,2 Overcoming these challenges is critical to expanding access to MOUD, curbing the opioid crisis, and improving public health outcomes.
Buprenorphine is the only opioid-agonist MOUD option that can be prescribed outside of specialized opioid treatment programs (OTPs). 3 As a partial agonist at the mu-opioid receptor and an antagonist at the kappa-opioid receptor, buprenorphine is associated with a lower risk of respiratory depression and overdose compared to full opioid agonists like methadone. 4 It is classified as a Schedule III medication under the Controlled Substances Act. From 2000 through the end of 2022, clinicians were required to complete a specialized training and obtain a waiver (ie, X-waiver) to be able to prescribe buprenorphine for MOUD outside of OTPs. As of January 2023, the Consolidated Appropriations Act of 2023 5 eliminated the X-waiver requirements, enabling any clinician whose DEA license allows for prescribing Schedule III substances to prescribe buprenorphine for MOUD without completing the previously mandated training and X-waiver-related procedures. 6
This regulatory change has the potential to reduce prescribing barriers for clinicians and increase patient access to buprenorphine-based MOUD. A recent study examining pharmacy dispensing data showed an increase in the number of clinicians prescribing buprenorphine between January 2022 (when the X-waiver was still required) and November 2023 (following its removal in January 2023) 7 ; however, the study did not differentiate buprenorphine prescribed for OUD versus pain management, nor did it account for the volume of prescriptions issued. Additionally, changes in the overall number of patients treated with buprenorphine were also unclear due to the short observation period. These limitations highlight the need for further research to evaluate the impact of the new buprenorphine prescribing policy on clinician engagement in and patient utilization of buprenorphine-based MOUD.
This study aimed to address these gaps by leveraging large, real-world electronic health record (EHR) data to assess changes in buprenorphine prescribing among adults diagnosed with OUD before and after the X-waiver elimination.
Methods
Study Design
This retrospective observational study spanned a 4-year period (2021-2024) and compared buprenorphine prescribing-related outcomes between 2 cohorts. The waiver-required cohort included individuals with any OUD diagnosis recorded during the period when X-waiver requirements were in effect (January 1, 2021-December 31, 2022), while the waiver-elimination cohort included individuals with any OUD diagnoses recorded after the waiver requirement was removed (January 1, 2023-December 31, 2024). The study addressed 2 primary questions: (1) whether the incidence of new OUD diagnoses differed following the policy change; and (2) whether the incidence of buprenorphine prescribing after a new OUD diagnosis changed between the waiver-required and waiver-elimination periods.
The study utilized deidentified EHR data from the TriNetX research database (Cambridge, MA, USA), a global health data network containing deidentified EHR data of more than 130 million patients from participating healthcare organizations predominately from the US Data in the TriNetX database have undergone extensive curation and mapping to common clinical entities and terminologies to ensure high usability as well as consistency with the Reporting of studies Conducted using Observational Routinely collected Data (RECORD) guidelines. 8 Because the study used secondary de-identified EHR data, it was determined to be exempt from the Institutional Review Board.
Study Population
To evaluate potential changes in the incidence of new OUD diagnoses between the 2 policy periods, the study included adults (age ≥18) with OUD diagnoses (based on the presence of the ICD-10 diagnostic codes F11.1 or F11.2) recorded in their EHR during the study period. Individuals with a pre-existing OUD diagnosis prior to the study period were included to capture the full population with OUD.
Those with OUD diagnosis recorded for the first time during the study period (ie, OUD diagnosis was not present in the patient’s EHR prior to the study period) were classified as having a new OUD diagnosis; the new OUD diagnosis sample served as a basis for estimating the incidence of new OUD cases. To ensure that the sample included individuals actively engaged in healthcare, eligible participants were also required to have at least 2 outpatient visits during their cohort-specific timeframe. Persons with diagnoses of cancer or receiving care in a skilled nursing/palliative care facility during the study timeframe were excluded because opioid therapy in these populations is guided by a different set of clinical practice guidelines, such as for cancer-related pain or palliative care.
To assess the incidence of initiating buprenorphine following a new OUD diagnosis within the cohort-specific study period, participants with a new OUD diagnosis were categorized into 2 subgroups: (1) those prescribed buprenorphine; and (2) those not prescribed buprenorphine. Buprenorphine treatment was considered “present” (Yes) if participants had at least 2 prescriptions for preparations approved for OUD treatment (and not exclusively for pain), including buccal/sublingual tablets or films or subcutaneous injections identified through the Prescription for Electronic Drug Information Exchange (RxNorm) code sets. 9 Detailed information on diagnostic and medication codes is provided in Table S1.
Outcome Measures and Risk Factors
The incidence of individuals with a new OUD diagnosis and the incidence of buprenorphine therapy initiation after the new OUD diagnosis during each policy period served as the main outcome measures.
To characterize the population and assess for confounding factors that may influence OUD diagnosis and buprenorphine therapy initiation, additional participant data were extracted from TriNetX at baseline for those with a new OUD diagnosis, defined as the date of the new OUD diagnosis. These characteristics included demographic information (sex: male/female; and race/ethnicity: Hispanic, non-Hispanic White, non-Hispanic Black, Other); age (years) when the OUD was first diagnosed; presence (Yes/No) of comorbidities associated with OUD (diagnoses of obesity/overweight, chronic pain, mental health disorders [depression, anxiety, bipolar], substance use disorders [alcohol, tobacco/nicotine, other non-opioid drugs]); presence (Yes/No) of any prescriptions for benzodiazepines, stimulants or gabapentin/pregabalin medications within 6 months before the first OUD diagnosis. Detailed information on the extracted baseline characteristics is provided in Table S1.
Data Analysis
Descriptive statistics (mean ± standard deviation or percentage) were utilized to summarize the characteristics of the study population. Baseline characteristics were compared between the waiver-required and waiver-elimination periods using the Mann-Whitney test for continuous variables and Chi-square test for categorical variables. The Chi-square test was also applied to compare the unadjusted incidences of new OUD diagnoses and those prescribed buprenorphine between the 2 policy periods.
Multiple logistic regression was conducted to assess the likelihood of a new OUD diagnosis (first model) and initiation of buprenorphine treatment after the new OUD diagnosis (second model) during the waiver-elimination period compared to waiver-required period, when controlled for baseline characteristics known to contribute to the risk of OUD. The logistic regression analysis was computed using the Maximum Likelihood Estimation method, which provided regression coefficients, standard deviations (SDs), and P values for each of the model variables. The adjusted odds ratios (aORs) and 95% confidence intervals (CIs) for each baseline variable were calculated to assess the likelihood of each outcome measure. The significance level was set at a 2-tailed P < .05. All statistical analyses were performed using PROC LOGISTIC procedure of the SAS software (Version 9.4, SAS Institute Inc., Carey, NC, USA).
Results
Study Population
The analysis identified 1 041 754 adults with an OUD diagnosis, with 516 508 in the waiver-required and 525 246 in the waiver-elimination cohort.
Among them, new OUD diagnoses were entered for 295 675 adults: 146 904 (28.4%) in the waiver-required and 148 771 (28.3%) in the waiver-elimination cohort (P = .03). Among those with a new OUD diagnosis (Table 1), females comprised close to one-half of the sample, the majority (65.5%) identified as White non-Hispanic, and their first OUD diagnosis was recorded at age 49.1 (SD = 16.1) years. One-fifth of them had diagnoses of overweight/obesity and approximately one-third had diagnoses of chronic pain, anxiety, and/or depression. Among co-occurring substance use disorders (other than those involving opioids), nicotine/tobacco use disorder was the most common (39.5%), followed by drug (22.9%) and alcohol (13.3%) use disorders. Co-prescribing patterns revealed that 57.8% of participants received benzodiazepines, 15.5% received gabapentin, and 2% received stimulants in conjunction with buprenorphine. Both demographic and clinical characteristics were significantly different between the waiver-required and waiver-elimination cohorts, with co-morbidities affecting a larger proportion of individuals in the waiver-elimination cohort (Table 1).
Baseline Characteristics of the Study Population With New OUD Diagnosis.
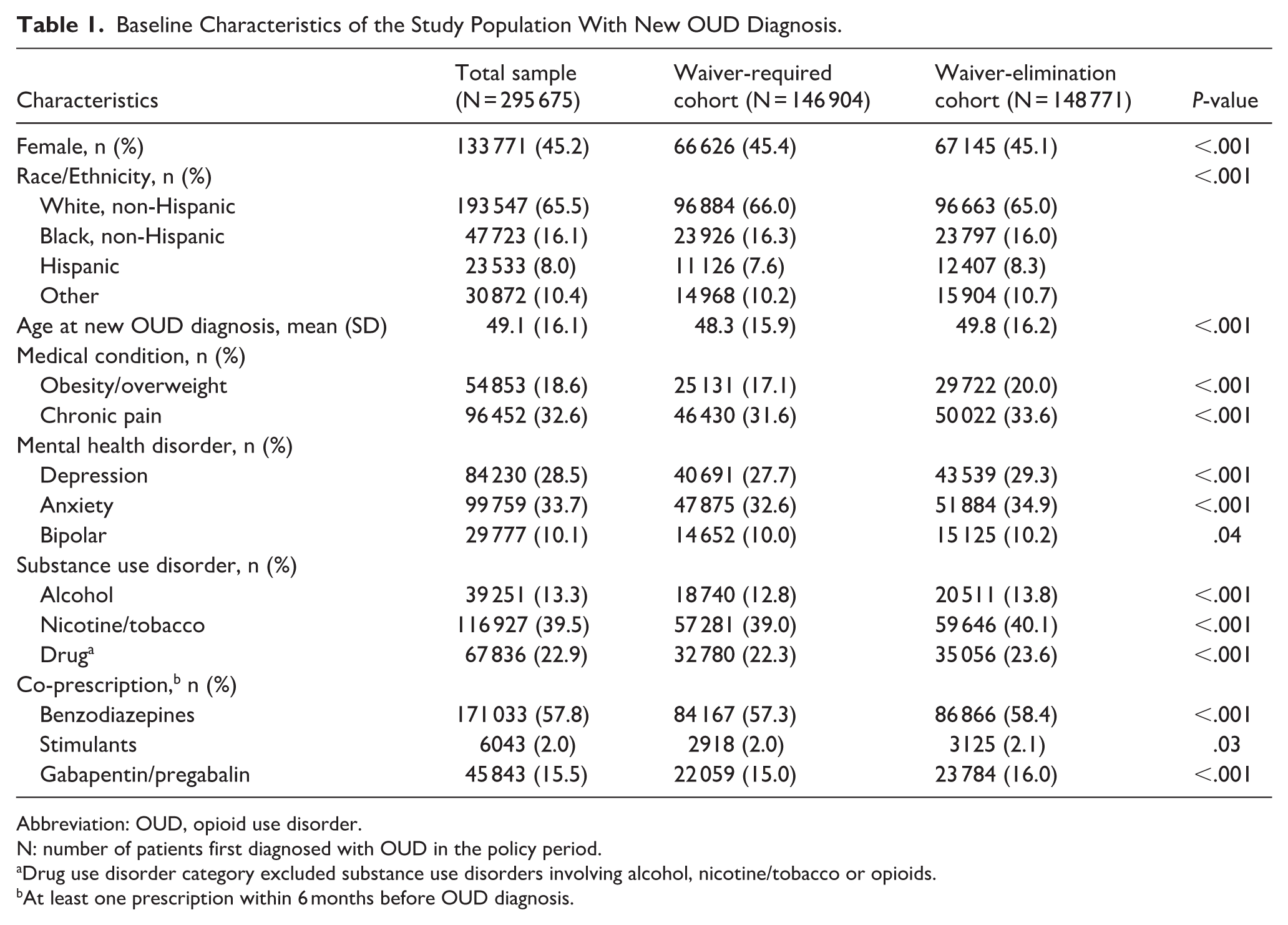
Abbreviation: OUD, opioid use disorder.
N: number of patients first diagnosed with OUD in the policy period.
Drug use disorder category excluded substance use disorders involving alcohol, nicotine/tobacco or opioids. b At least one prescription within 6 months before OUD diagnosis.
Among participants with a new OUD diagnosis, a smaller proportion were prescribed buprenorphine in the waiver-required cohort than in the waiver-elimination cohort (18.8% [27 665/148 771] vs 19.6% [29 223/146 904], P < .001). The average time from a new OUD diagnosis to the first buprenorphine prescription was longer during the waiver-required than in the waiver-elimination period (56.3 ± 176.3 vs 46.0 ± 115.9 days, P < .001).
Changes in New OUD Diagnoses
Logistic regression analysis, controlled for demographic and comorbid confounders, indicated the likelihood of a new OUD diagnosis was 2% lower during the waiver-elimination period compared to the waiver-required period (aOR = 0.98, 95% CI: 0.97, 0.98) (Figure 1a).
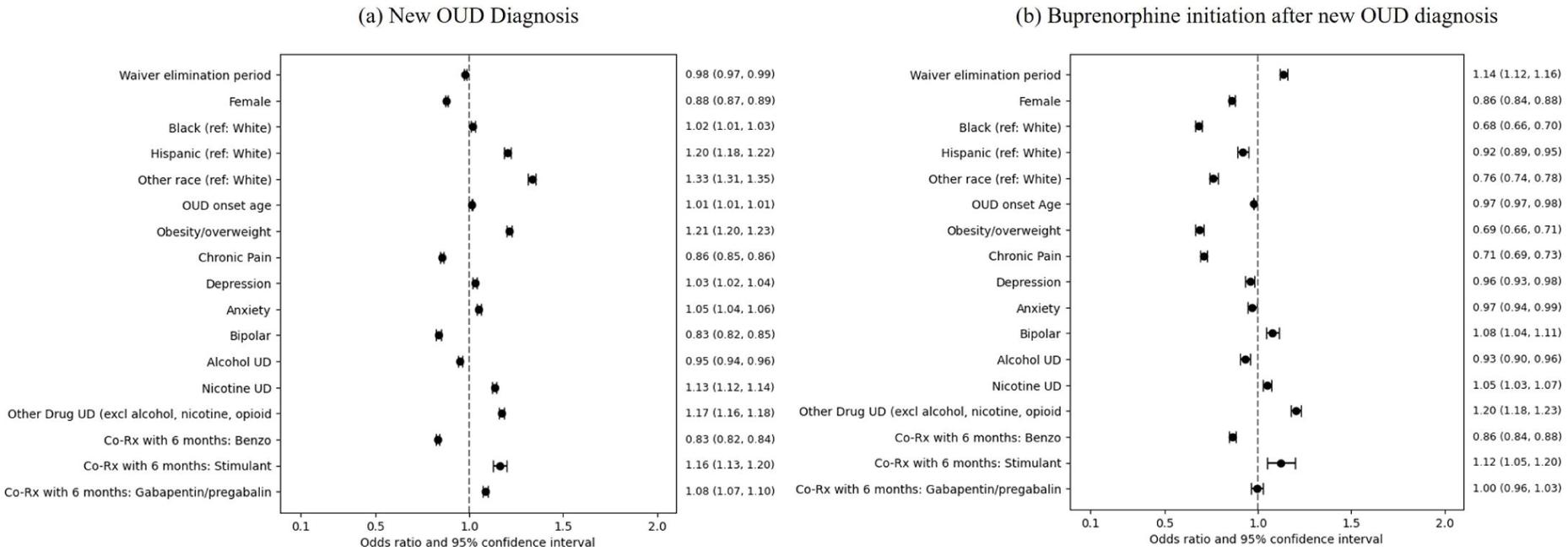
Likelihood of a new OUD diagnosis and buprenorphine initiation after a new OUD diagnosis. (a) New OUD diagnosis. (b) Buprenorphine initiation after new OUD diagnosis.
This analysis also found that females and individuals with chronic pain, or bipolar or alcohol use disorders, and those co-prescribed benzodiazepines were less likely to have a new OUD diagnosis recorded (Figure 1, Table S2). In contrast, individuals from racial minority groups were more likely to be diagnosed with OUD compared to their White counterparts. Increased likelihood of a new OUD diagnosis was also observed among those with co-occurring obesity/overweight status, depression, anxiety, nicotine or drug use disorders, and those prescribed stimulants or gabapentin/pregabalin (Table S2).
Changes in Buprenorphine Initiation After New OUD Diagnosis
Logistic regression analysis included 131 330 adults newly-diagnosed with OUD during the waiver-required period and 126 122 during the waiver-elimination period. Individuals in the waiver-elimination cohort were 14% more likely to be prescribed buprenorphine after their new OUD diagnosis than those in the waiver-required period (aOR = 1.14, 95% CI: 1.12, 1.16) (Figure 1b).
Females, adults who were diagnosed with new OUD at a younger age, those identifying as belonging to racial minorities, those with co-occurring medical conditions, and those with anxiety and depression or alcohol use disorder diagnoses were less likely to have buprenorphine initiated following their new OUD diagnosis. Adults with bipolar disorder and those with nicotine/tobacco or other drug use disorders were more likely to have buprenorphine prescribed after their new OUD diagnosis (Table S2).
Discussion
Buprenorphine is among the first-line treatments for OUD. In 2023, the changes in regulations under the Consolidated Appropriations Act of 2023 allowed prescribers of controlled substances to prescribe buprenorphine for OUD without the previously required X-waiver to increase access to and acceptability of this evidenced-based MOUD. 5 This study leveraged real-world EHR data from multiple healthcare organizations to assess changes in OUD diagnosis and buprenorphine MOUD prescribing patterns following the X-waiver elimination. Although a larger number of adults had an OUD diagnosis recorded in their EHR, our results indicated a 2% decline in the incidence of new OUD diagnoses in the post-waiver elimination, compared to the waiver-required, period. The study also found a 14% increase in the initiation of buprenorphine MOUD among those with a new OUD diagnosis after the waiver elimination.
This study revealed that the incidence of new OUD diagnosis decreased by 2% from the waiver-required to the waiver-elimination period. The decline could be attributed to multiple factors. Many individuals with OUD may have been identified and diagnosed during or shortly after the COVID-19 pandemic, when opioid use and overdose awareness peaked, prompting intensified local and national OUD prevention and treatment efforts and leading to the current reductions in opioid-related overdose fatalities. This could have “reduced” the pool of adults with existing, yet undiagnosed OUD, contributing to the decline in the incidence of new OUD diagnoses in the post-waiver period. In addition, our post-waiver study period (2023-2024) corresponded to the post-pandemic years, when healthcare systems started returning to more routine operations, and intensified targeted OUD screening efforts may have diminished, resulting slight reduction in newly identified OUD cases. Prior initiatives aimed at increasing access to MOUD could have also led to an increased number of individuals who had been diagnosed with OUD earlier; this hypothesis is aligned with our findings of a larger overall number of adults with the EHR-recorded OUD diagnosis after the X-waiver was removed.
Our analysis also showed an increased likelihood of prescribing buprenorphine MOUD to individuals with a newly-diagnosed OUD after the elimination of the X-waiver requirement. This regulatory change may have allowed more clinicians to prescribe buprenorphine, thereby expanding access to MOUD. The elimination of the X-waiver may also lead to a more integrated approach to OUD treatment, with buprenorphine becoming a more routine part of care in primary care and other healthcare settings, outside of addiction medicine-specialized ones, with more patients receiving this treatment.
Yet, the increase in prescribing buprenorphine observed in our study was modest. This pattern may reflect other persistent systemic barriers, including clinician discomfort with, or lack of confidence in, managing OUD due to limited experience or training. Moreover, because the regulatory changes were relatively recent, many clinicians may have experienced uncertainty regarding the new requirements and expectations when treating OUD with buprenorphine, 10 potentially delaying a broader adoption of buprenorphine MOUD. Other barriers to OUD care, including buprenorphine MOUD, such as insurance coverage limitations, insufficient community resources for behavioral treatment or patient-level transportation problems to clinical visits could hinder patient access to buprenorphine MOUD.11,12
Our results indicated a higher prevalence of medical and mental health comorbidities among individuals who were newly-diagnosed with OUD after the X-waiver removal. This finding may suggest clinicians’ increased comfort with buprenorphine therapy, thus extending prescribing buprenorphine to more medically-complex patients, such as those with multi-morbidity (eg, pain and OUD) whose OUD might have previously been undiagnosed and/or untreated. Moreover, our study found that racial minority groups had greater likelihood of being diagnosed with OUD, yet were less likely to receive buprenorphine treatment, highlighting persistent disparities in treatment access and suggesting the ongoing presence of systemic and socioeconomic barriers to evidence-based OUD care.13,14
Lastly, our analysis also identified several risk factors with odds ratios close to 1, suggesting that, although statistically significant, these associations may have limited clinical relevance and likely reflect small effect sizes with modest practical impact. Moreover, because the timing and process of policy implementation varied across states, Future research should examine how the timing and process of implementation of regulatory changes impact diagnosis and treatment of OUD. Such analyses could, for example, help determine whether the uptake of buprenorphine prescribing for newly-diagnosed OUD was affected by different state-level timing of X-waiver elimination or by variations in clinician awareness of these changes or health system implementation capacity.
Limitations
Several limitations may affect the generalizability and conclusiveness of our findings. The study utilized EHR data from health systems participating in the TriNetX research network. Diagnoses and medications from clinics or pharmacies outside this network would not be captured in the analysis, potentially underestimating the prevalence of OUD diagnoses and buprenorphine prescriptions. The existing OUD-related diagnostic codes include “opioid physical dependence” (F11.2). While opioid physical dependence is expected in persons with active OUD, it is also common among those without OUD who are treated with daily long-term opioids. 15 This may lead to erroneous use of F11 codes for individuals with opioid-treated chronic pain who do not have OUD. In addition, buprenorphine therapy for chronic pain was less common in the past; the removal of X-waiver, combined with the new treatment guidelines for chronic pain, 16 could have increased clinician comfort with prescribing buprenorphine to manage chronic pain, outside of the OUD settings, impacting our results. Socioeconomic contexts (eg, health insurance, education and income levels, and neighborhood factors) are not available in the TriNetX database and, therefore, unavailable to be included as confounders in our analysis. Because changes in prescription patterns may take longer to manifest, the existing data may not have fully captured the X-waiver elimination’s long-term impact. Nonetheless, by leveraging a large, real-world EHR dataset, this study provides evidence on beneficial changes in buprenorphine prescribing for adults with newly-diagnosed OUD following the X-waiver removal. Additional research, with longer follow-up periods, is needed to assess the sustained impact of the X-waver elimination on OUD care and OUD-related overdose, mortality and other harms.
Conclusion
Results of this electronic health record-based retrospective cohort study suggest the elimination of X-waiver was associated with increased buprenorphine treatment initiation among adults with newly-diagnosed OUD. While the X-waiver regulatory change has likely improved access to MOUD, other factors, such as provider hesitancy, lack of training, and disparities in treatment access, may continue to hinder adoption of buprenorphine MOUD and limit its impact. Addressing these barriers through provider education, patient outreach, and system-level support could help increase buprenorphine MOUD initiation, with associated benefits. Further research is needed to assess the longer-term impact of X-waiver elimination on buprenorphine MOUD initiation and ways for increasing patient access to this treatment.
Supplemental Material
sj-docx-1-saj-10.1177_29767342251414541 – Supplemental material for Assessing the Initial Impact of X-Waiver Elimination on Buprenorphine Prescribing for Opioid Use Disorder
Supplemental material, sj-docx-1-saj-10.1177_29767342251414541 for Assessing the Initial Impact of X-Waiver Elimination on Buprenorphine Prescribing for Opioid Use Disorder by Wen-Jan Tuan, Sujeong Park, Shazib Altaf and Aleksandra E. Zgierska in Substance Use & Addiction Journal
Footnotes
Ethical Considerations
The study was determined to be exempt from the Institutional Review Board oversight by the Pennsylvania State University’s Human Research Protection Program.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Penn State Inter-Campus Health and Medicine Research Program, which supports interdisciplinary research collaborations across the University’s campuses. Penn State Clinical and Translational Science Institute provides access to the TriNetX network and is supported by a National Center for Advancing Translational Sciences and its Clinical and Translational Science Award (Grant: UL1 TR002014).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest: AEZ was a Board of Directors member and Vice President of the American Society of Addiction Medicine.
Statement of Pre-Print Policy
The submitted manuscript is an original contribution that has not been previously published and is not under consideration for publication elsewhere.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
