Abstract
Background:
Patients with opioid use disorder need prompt buprenorphine/naloxone (BUP/NX) and naloxone nasal spray (NNS) access from pharmacies, but audit studies report substantial availability deficits. No specific intervention has increased BUP/NX stocking, and little is known about the impact of transitioning specific NNS formulations to over-the-counter. The purpose of this study was to assess the impact of mailed academic detailing on BUP/NX availability and the ability to obtain NNS without a prescription.
Methods:
A prospective interventional study was conducted in a sample of 1000 Texas community pharmacies assigned 1:1 to intervention and control groups. The intervention group received academic detailing handouts with links to continuing education addressing BUP/NX and NNS. Pre- and post-intervention audits were conducted using a telephonic secret-shopper approach. Outcomes for BUP/NX included stocking, willingness to order if not stocked, and a composite outcome of stocking and willingness to order. NNS outcomes included availability without a prescription. McNemar and Wilcoxon signed-rank tests, and generalized estimating equations (GEE) were used for analyses.
Results:
Data from 757 pharmacies were included in analyses (374 intervention, 383 control). Baseline BUP/NX stocking was low (36.7%) with no statistically significant pre-to-post changes for either group. Most pharmacies were willing to order (60.5%) with no significant pre-to-post changes for either group. GEE analysis found no significant difference in odds of stocking between groups (aOR = 1.08, P = .73), but the intervention group had a significantly higher likelihood to order (aOR = 2.25, P = .02) and of achieving the composite outcome (aOR = 2.17, P = .01). NNS availability increased in both groups but did not differ significantly between groups (aOR = 1.00, P = .99).
Conclusions:
A mailed academic detailing intervention did not change NNS availability or BUP/NX stocking but did modestly increase willingness to order BUP/NX. This intervention is easily replicable and scalable, but modifications to increase the magnitude of its effect are needed.
Highlights
Audits identified gaps in buprenorphine and naloxone access in Texas pharmacies.
Mailed educational handouts did not increase stocking of these products.
Willingness to order buprenorphine products increased modestly.
More intensive educational and policy interventions are needed.
Introduction
The opioid crisis is characterized by a drastic increase in opioid overdose deaths. 1 Access to naloxone, a short-acting, mu-opioid antagonist that can reverse opioid overdose, is imperative for people with opioid use disorder (OUD). 2 However, in 2021, only 60% of surveyed Texas community pharmacies had naloxone nasal spray (NNS) stocked. 3 A recent systematic review indicated similar naloxone availability (62.8%) in community pharmacies among 30 US studies. 4 In March 2023, 4 mg NNS was approved to be sold over-the-counter (OTC), though availability and placement (behind-the-counter vs in the aisle) within pharmacies may vary. 5
Buprenorphine is a Schedule III controlled substance and partial opioid agonist approved for OUD treatment. 6 It is commonly formulated with naloxone (BUP/NX) to ensure intended use via sublingual administration. Its partial agonist mechanism alleviates opioid withdrawal symptoms and reduces cravings, supporting stabilization and treatment engagement. It is associated with a 37% decrease in all-cause mortality in patients who have previously experienced a nonfatal overdose. 7 Despite its effectiveness, it is not widely available in community pharmacies.3,8 Across a sample of Texas community pharmacies, only 42% stocked 8/2 mg sublingual BUP/NX films, a common strength and formulation. 8 These disparities were more pronounced in independent pharmacies versus chains, with 21.5% versus 52.4%, respectively, stocking a 1-week supply. Similar trends were noted in a combined analysis of 11 US states. 8 Pharmacists are essential in OUD treatment by dispensing BUP/NX and NNS and ensuring adequate supply to meet local demand. Targeted education can enhance their preparedness to implement evidence-based OUD care, potentially increasing buprenorphine dispensing. 9
Academic detailing is an outreach-based educational approach that addresses knowledge gaps, enhances evidence-based care, and improves patient outcomes. 10 In-person, one-on-one interactions between a researcher and a learner can increase medication access in community pharmacies.11,12 However, time, geographical distance, and personnel costs limit in-person detailing on a larger scale. A more feasible, scalable intervention to expand BUP/NX access in community pharmacies would be valuable. This study explored whether a mailed academic detailing intervention, with brief academic detailing and links to online continuing education (CE), could increase BUP/NX and NNS availability in Texas community pharmacies.
Methods
A prospective interventional study was conducted to assess the impact of an academic detailing intervention on the availability of 8/2 mg BUP/NX sublingual films and 4 mg NNS without a prescription among Texas community pharmacies. Pre- and post-intervention audits were completed using a secret-shopper approach similar to previous studies conducted by our research team to determine BUP/NX and NNS availability.3,8,13 This study was determined “not human research” by The University of Texas at Austin Institutional Review Board (IRB ID: STUDY00004812). The intervention was conducted in collaboration with Texas Health and Human Services as a part of the Texas Targeted Opioid Response.
The pre-intervention audit was conducted from October 31, 2023 to November 22, 2023 to determine baseline BUP/NX and NNS availability. Academic detailing materials were mailed to pharmacies in the intervention group in early January 2024 and received by pharmacies as early as January 12, 2024. The post-intervention audit was conducted from February 10, 2024 to March 10, 2024.
The study population was obtained from a list of Texas community pharmacies with an active license from the Texas State Board of Pharmacy (TSBP) website. After excluding those not licensed to dispense opioids, 4427 remained and were used to generate a randomized sample of 1000 pharmacies, including 600 chain and 400 independent pharmacies, which is representative of the proportion of these pharmacy types in Texas. Pharmacies were classified as independent if they had fewer than 5 stores under the same name/owner and classified as a chain if they had 5 or more. Pharmacies were initially randomized 1:1 between the intervention and control groups (see Figure 1).
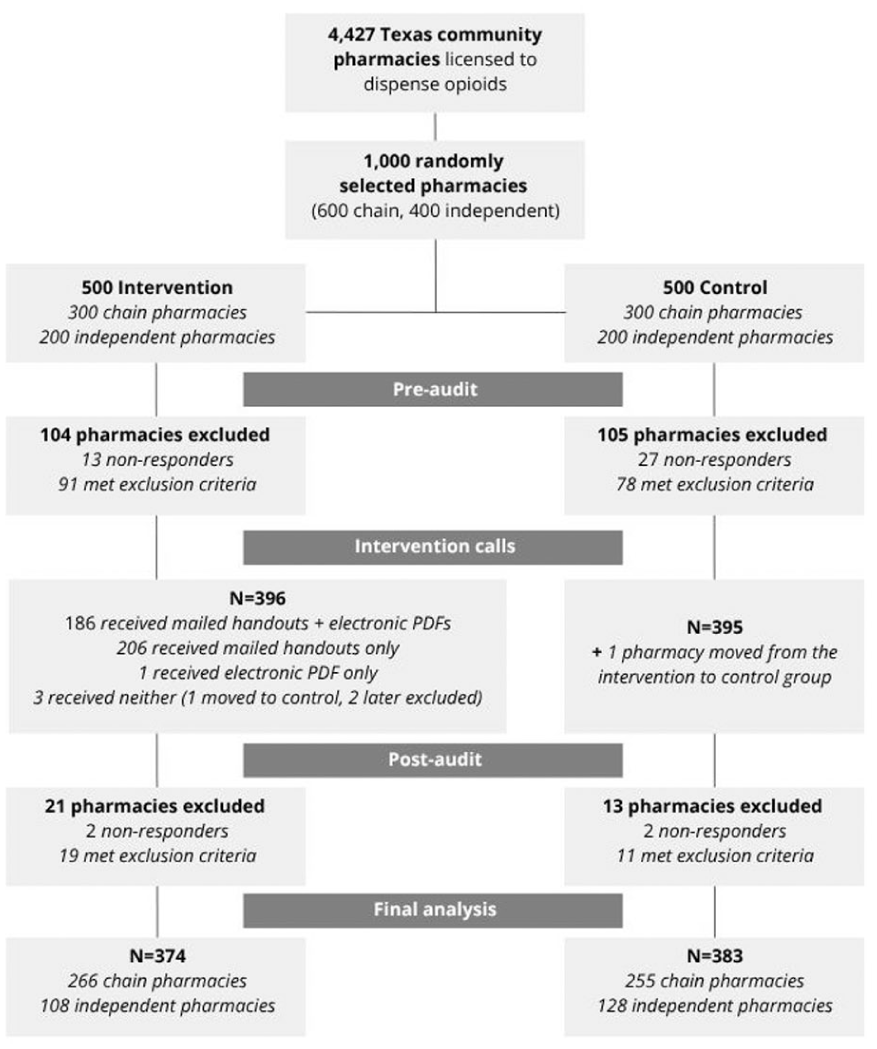
Pharmacy flowchart.
While substantial prior research has documented buprenorphine availability and associated barriers across community pharmacies, few have evaluated interventions designed to improve availability, and none have included actual stocking as an outcome. Therefore, the sample size estimation drew upon related evidence from a naloxone-focused academic detailing intervention completed by members of our research team. 11 Given an estimated 50% baseline buprenorphine availability and 15% difference between the intervention and control groups following the intervention, an alpha of 0.05, and power of 0.8, an estimated 167 pharmacies were needed in each group to demonstrate a statistically significant difference in BUP/NX availability between groups and provide sufficient power for overall and subgroup analyses by pharmacy-type (JMP® Pro 17, SAS Institute Inc., Cary, NC, USA). A total of 1000 pharmacies were randomized to account for excluded pharmacies and nonresponders.
During the pre-intervention audit, pharmacies found to be out of business, not open to the public, specialty (eg, compounding, dermatology, and oncology), or not a community pharmacy (eg, hospital and long-term care) were excluded. Pharmacies were also excluded if they were unreachable, defined as 3 failed attempts to contact them (eg, no answer, sent to voicemail, busy signal, and excessive hold), and were listed as nonresponders if they refused to disclose BUP/NX availability or provide an estimated order time if the medication was not in stock.
The intervention consisted of 2 physical handouts, one for buprenorphine and naloxone (Supplemental Appendix A), and an online CE course (Supplemental Appendix B). Handouts served as a pharmacist’s guide to stocking, dispensing, and patient education and included a link to the freely available online CE. They were printed on 8.5 × 11-inch cardstock, laminated, and mailed to each pharmacy in the intervention group not previously excluded or listed as a nonresponder. Once mailed, callers used a script to contact the intervention group by phone, inform them of the mailed handouts, highlight access to the free online CE, and offer to send electronic versions of the materials for convenience. To ensure only the intervention group interacted with the online CE, it was only available using the QR code on the handouts. The online education was a 1-hour course that met TSBP opioid CE requirements and was available to both pharmacists and pharmacy technicians. The objectives of the CE course were to (1) recall OUD epidemiology and treatment modalities, (2) summarize recent legal and regulatory changes impacting OUD care, (3) analyze the impact of limited buprenorphine and naloxone availability in community pharmacies, and (4) identify action items for pharmacists to integrate evidence-based OUD care into their practice.
Pre- and post-intervention audits were conducted by trained auditors using an identical protocol across intervention and control pharmacies. All auditors completed a 1-hour training and used a standardized script (Supplemental Appendix C) to conduct secret-shopper telephone calls to collect information on the primary and secondary outcomes. Data was entered in a data collection sheet using Microsoft Excel®. Although auditors were aware of pharmacy group assignment, the use of a standardized secret-shopper script minimized potential bias.
The primary outcome was BUP/NX stocking, while secondary outcomes included (1) willingness to order BUP/NX if not stocked, (2) estimated BUP/NX ordering time, (3) ability to attain naloxone without a prescription, and (4) whether naloxone can be obtained behind the pharmacy counter or in store aisles. Primary and secondary BUP/NX outcomes were analyzed by pharmacy group (intervention vs control). Analyses were conducted using Microsoft Excel® and JMP® Pro 17 (SAS Institute Inc., Cary, NC, USA). Paired, nominal data examining differences between the pre- and post-intervention were compared using McNemar’s chi-square statistical test. Non-normally distributed, paired, continuous data examining differences between pre- and post-intervention were analyzed using the Wilcoxon signed-rank statistical test. Subgroup analyses using the same statistical tests were used to compare outcomes by pharmacy group (intervention vs control). Generalized estimating equations (GEEs) with a binomial distribution and logit link were used to compare the change in outcomes between the intervention and control groups. An unstructured working correlation structure was used as the most flexible option to allow for different correlations between pairs of observations. A separate GEE analysis was conducted with each outcome at post-intervention (BUP/NX stocking, willingness to order if not stocked, and stocking/willingness to order as a composite outcome) modeled as a dependent variable and study group (intervention vs control) as the primary predictor, while other covariates included their corresponding pre-intervention measures as within-subject factors and the interactions of study group and their corresponding pre-intervention measures. Adjusted odds ratios (aORs) from the GEE models are reported and interpreted in the Results. A P-value of <.05 signified statistically significant changes between the pre- and post-intervention audits.
Results
Data from 757 pharmacies (521 chain and 236 independent) were included in the final analyses (Figure 1). Of the original 1000 pharmacies, 243 were excluded (125 intervention, 118 control) for meeting exclusion criteria (n = 196) or nonresponse (n = 47). Within the intervention group, 4 of 396 pharmacies did not receive mailed handouts. Of these, 1 was sent an electronic PDF, 2 were later excluded because they were permanently closed, and 1 pharmacy did not receive either mailed or electronic materials and was analyzed with the control group. This pharmacy remained in the analysis because outcomes could still be assessed, despite not receiving the intervention. Almost half of the intervention group (n = 207) provided emails to receive electronic handouts (187 were successfully sent due to firewall or nonworking emails), while 206 pharmacies received only the mailed handout. Among the 393 pharmacies that received the intervention (mailed and/or electronic), 8 engaged with the online CE material, and 5 completed the module during the study period.
Table 1 details overall BUP/NX and NNS availability within the intervention and control groups. A nonsignificant decrease in BUP/NX availability between pre- and post-intervention audits was observed in both the intervention (35.3% vs 34.8%, −0.5%, P = .8415) and control group (38.1% vs 36.3%, −1.8%, P = .4772). Willingness to order BUP/NX increased nonsignificantly in the intervention group (+5.7%, P = .1967), and decreased nonsignificantly in the control group (−3.4%, P = .2579). Median BUP/NX ordering time did not differ between groups or across audits. Both intervention (+6.8%, P = .1266) and control (+8.7%, P = .0067) groups exhibited increased NNS availability between pre- and post-intervention audits. NNS was primarily available behind the pharmacy counter in both pre- and post-intervention audits (intervention: 49.6%-51.2%; control: 59.9%-58.9%). NNS stocking in both the counter and aisle sections increased over the study period in both intervention (20.5% vs 25.4%, +4.9%) and control (21.1% vs 24.6%, +3.5%) groups.
BUP/NX and NNS Availability Overall.
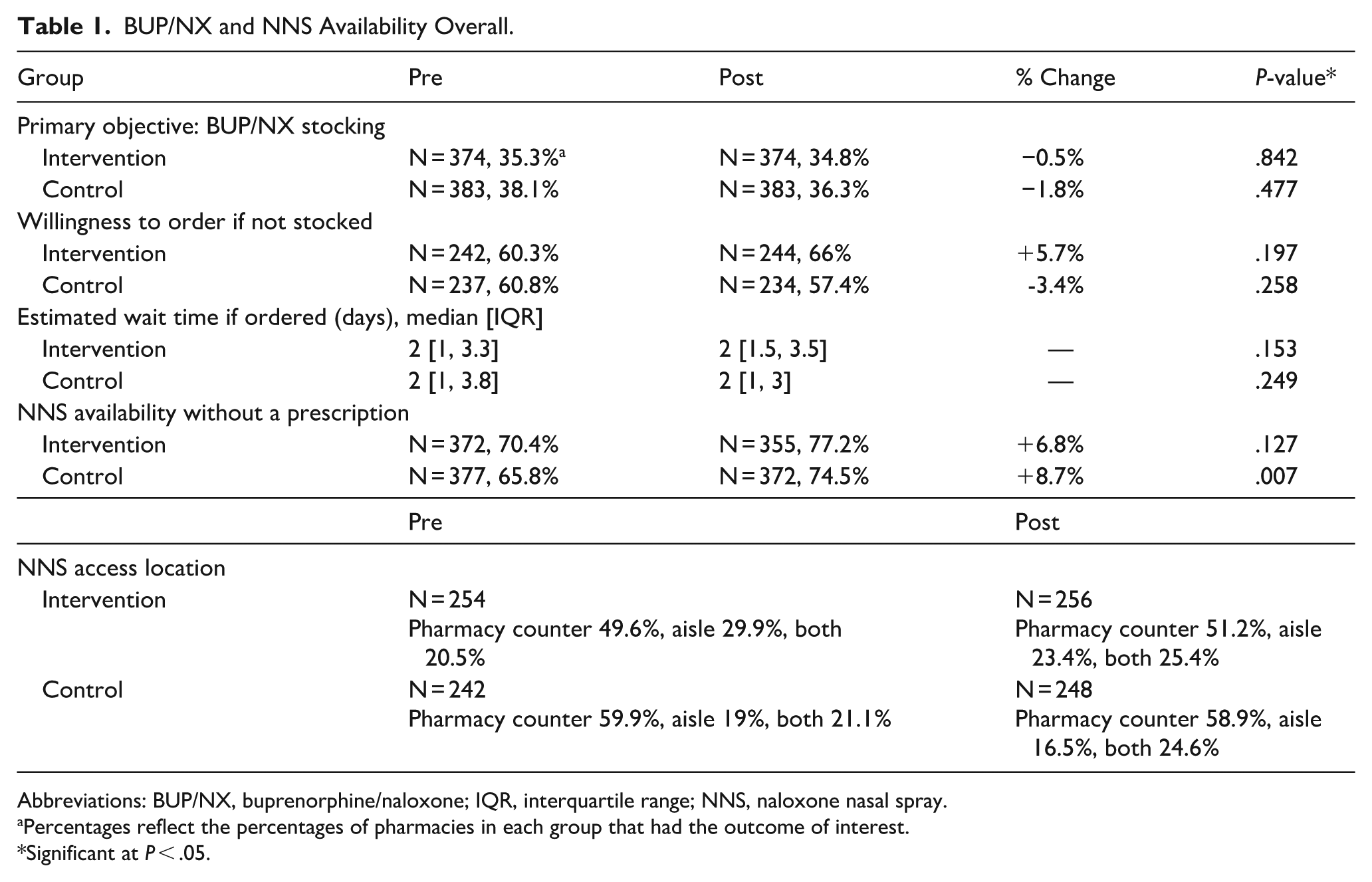
Abbreviations: BUP/NX, buprenorphine/naloxone; IQR, interquartile range; NNS, naloxone nasal spray.
Percentages reflect the percentages of pharmacies in each group that had the outcome of interest.
Significant at P < .05.
Table 2 details the subgroup analyses for primary and secondary outcomes by pharmacy type. In the intervention group, chain pharmacies were almost twice as likely to stock BUP/NX than independents in both the pre- and post-intervention audits (pre: 41% vs 21.3%; post: 40.2% vs 21.3%). Similar patterns were observed in control groups (pre: 47.5% vs 19.5%; post: 43.9% vs 21.1%). Neither pharmacy type exhibited significant changes in BUP/NX stocking after the intervention. Among chains, willingness to order BUP/NX increased significantly in the intervention group (+11.2%, P = .0196), while independents showed a nonsignificant decrease (−4.7%, P = .4142).
BUP/NX and NNS Availability by Pharmacy Type.
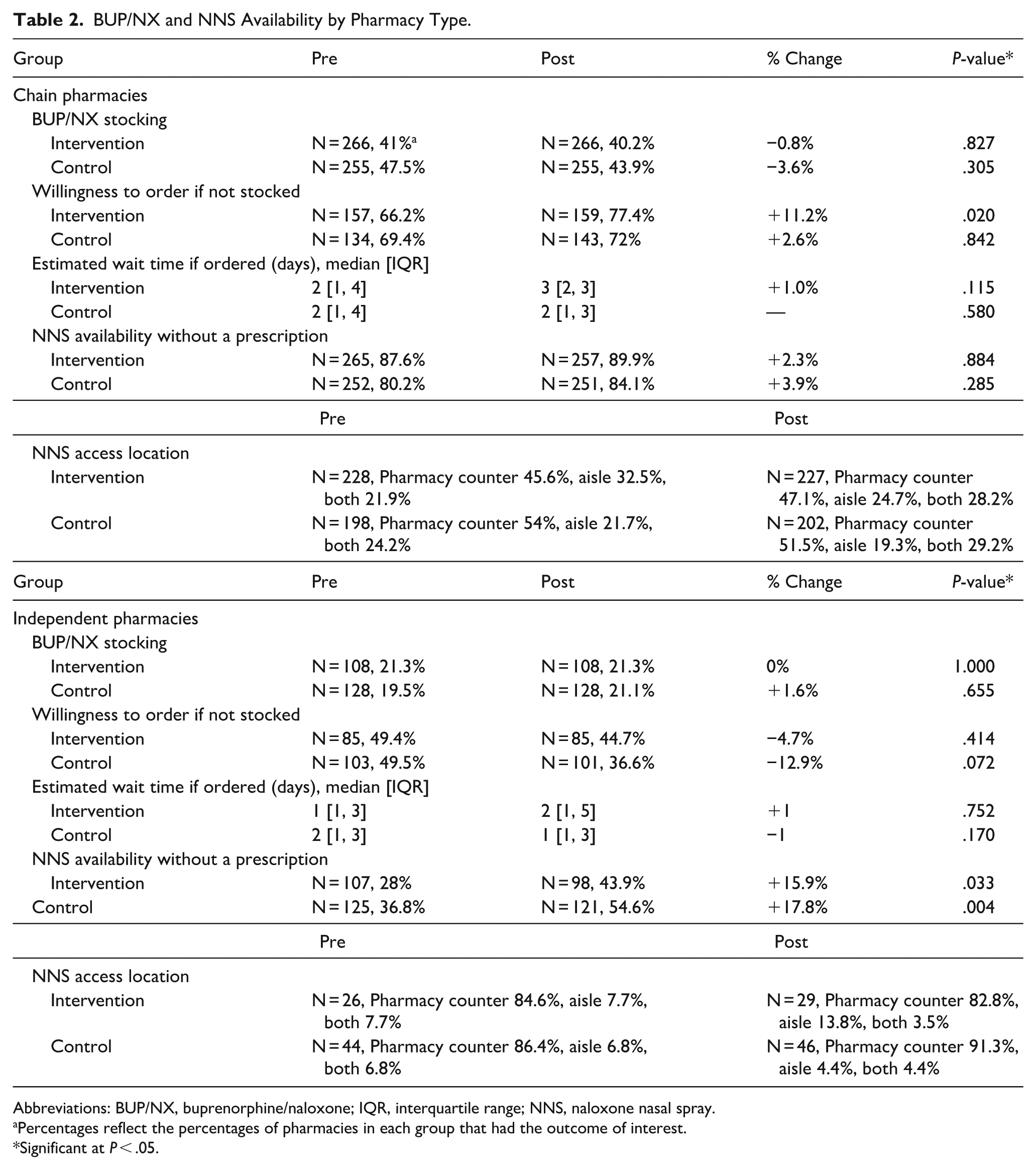
Abbreviations: BUP/NX, buprenorphine/naloxone; IQR, interquartile range; NNS, naloxone nasal spray.
Percentages reflect the percentages of pharmacies in each group that had the outcome of interest.
Significant at P < .05.
Table 3 details GEE analyses, which displayed no difference in post-intervention BUP/NX stocking between intervention vs. control pharmacies (aOR = 1.08, 95% CI = 0.69-1.70, P = .73), controlling for pre-intervention BUP/NX stocking and the interaction between pharmacy group (intervention/control) and pre-intervention BUP/NX stocking. However, significantly greater willingness to order BUP/NX (aOR = 2.25, 95% CI = 1.16-4.36, P = .02) and an improvement in the composite outcome of stocking or being willing to order (aOR = 2.17, 95% CI = 1.21-3.92, P = .01) were identified for intervention pharmacies controlling for pre-intervention BUP/NX stocking and the interaction between pharmacy group (intervention/control) and pre-intervention BUP/NX stocking. In contrast, no between-group difference was observed for NNS availability (aOR = 1.00, 95% CI = 0.58-1.71, P = .99).
Generalized Estimating Equations Comparing the Change in Outcomes Between Intervention and Control Groups.
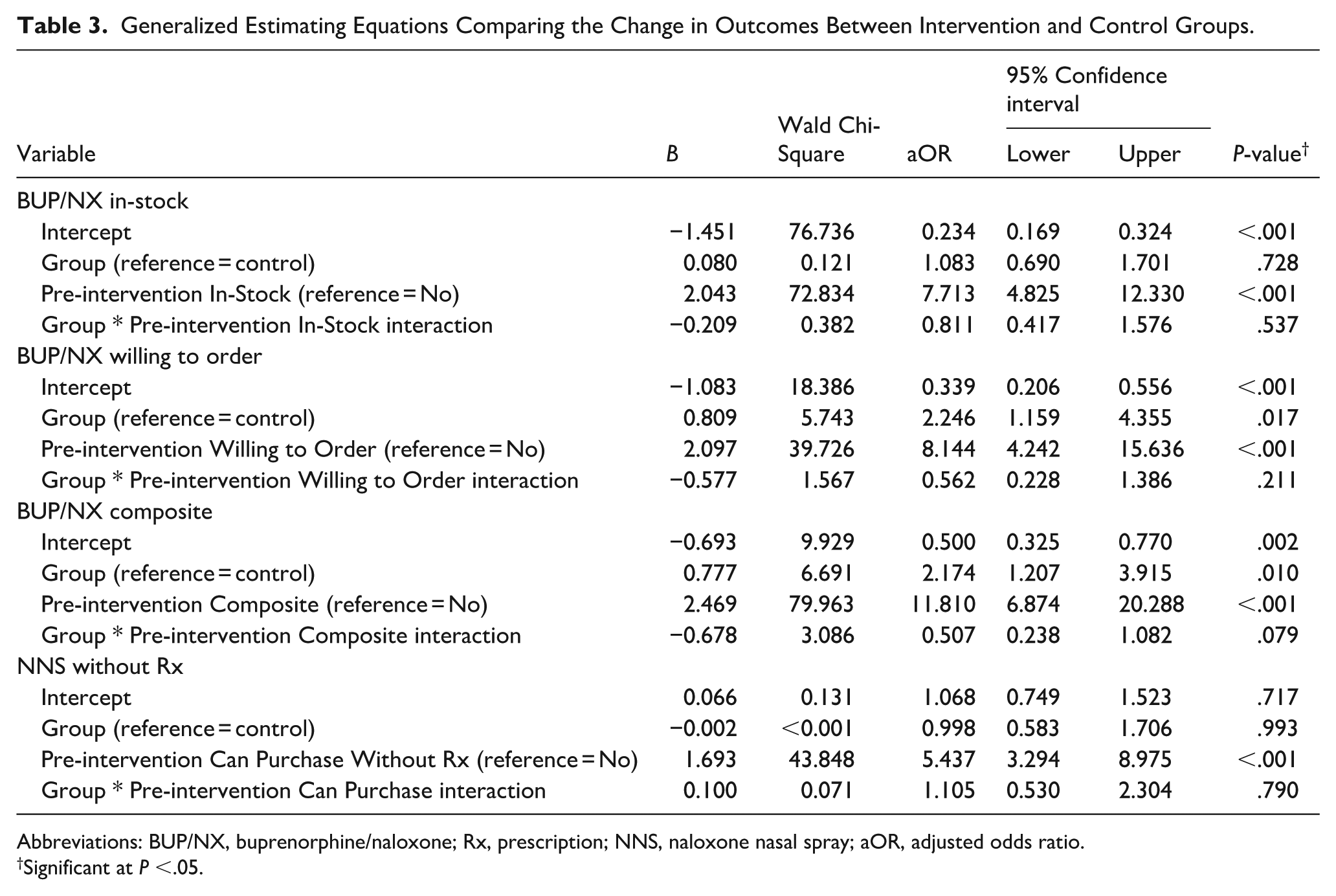
Abbreviations: BUP/NX, buprenorphine/naloxone; Rx, prescription; NNS, naloxone nasal spray; aOR, adjusted odds ratio.
Significant at P <.05.
Discussion
In this statewide audit of 757 Texas community pharmacies, a low-intensity, mailed educational intervention demonstrated limited efficacy in improving BUP/NX stocking. No significant change in the immediate availability of a common BUP/NX formulation or NNS for dispensing was observed. Overall, BUP/NX stocking remained low throughout the study, which is consistent with prior research and highlights an important concern for people living with OUD. 3 Maintaining BUP/NX stock is critical because delays in access may increase risk for recurrence of opioid use, overdose, and death, particularly among recently discharged or otherwise high-risk populations.14,15 In cases where pharmacies may not stock BUP/NX due to low demand, willingness to order this medication is also valuable. Alternative treatment options—such as long-acting injectable buprenorphine and oral methadone—are not typically available from community pharmacies. As a result, BUP/NX remains the predominant formulation of OUD medication treatment dispensed in community pharmacies and requires reliable and timely availability.
The DATA waiver, established under the Drug Addiction Treatment Act of 2000, was a prescriber-level barrier to buprenorphine access until its removal in December 2022.16,17 Buprenorphine prescribing via telemedicine is also evolving as policymakers and advocates address the opioid crisis. 18 These changes aim to increase BUP/NX prescribing but do not address limited availability in community pharmacies. Many pharmacy-level barriers further reduce access to buprenorphine and naloxone.3,4,8 From 2010 to 2019, gaps between substance use treatment records and outpatient pharmacy claims left approximately 87% of people with OUD who could benefit from medications such as buprenorphine untreated. 19 Recent studies quantifying buprenorphine stocking indicate this remains an ongoing issue.3,8
Several studies have targeted pharmacy-level barriers by conducting in-person academic detailing, 11 identifying pharmacies more likely to stock the medication, 20 and evaluating the feasibility of pharmacist-led buprenorphine induction through a collaborative practice agreement. 21 As policies are evolving to increase buprenorphine prescribing, pharmacies must be prepared to increase dispensing to avoid “buprenorphine bottlenecks.” 9 Notably, in March 2024, the Drug Enforcement Agency (DEA) stated its commitment to safe and readily accessible OUD treatment, recognized increased buprenorphine demand, and directed distributors to examine thresholds to ensure buprenorphine accessibility without delay. 22 The Pharmacy Access to Resources and Medication for Opioid Use Disorder (PhARM-OUD) Guideline, published in September 2024, further demonstrates the commitment of critical regulatory and professional organizations to addressing the pharmacy-level buprenorphine barriers. 23 This joint consensus practice guideline from the National Association of Boards of Pharmacy and the National Community Pharmacists Association has been endorsed by the American Society of Addiction Medicine, the American Pharmacists Association, and other professional pharmacy and addiction medicine organizations. It includes 9 recommendations to inform pharmacy service provision for people with OUD, primarily through addressing pharmacists’ concerns and questions related to dispensing buprenorphine in a rapidly evolving clinical and regulatory landscape. Wholesaler-controlled substance monitoring programs, “red flags” of misuse and diversion, and best practices for evaluating telehealth prescriptions are prominently featured.
While the primary outcome of BUP/NX stocking did not significantly improve post-intervention, post-hoc analyses of willingness to order and a composite of stocking and willingness to order were significantly more positive in the intervention versus control groups. These findings must be interpreted in the context of minor implementation deviations. For example, a single pharmacy initially assigned to the intervention group did not receive the materials and was analyzed with the control group. This minor deviation is unlikely to have affected the overall findings but demonstrates the practical challenges of delivering low-intensity, mailed interventions in real-world pharmacy settings. This suggests the intervention may have influenced pharmacy staff behavior, moving them along the stages-of-change continuum toward willingness to order, even if it did not produce rapid changes in stocking. Individual chain pharmacy staff may have limited control over inventory and shelf placement, which are often determined by district or corporate management. These structural constraints may have further limited the intervention’s measurable impact on stocking.
Increased NNS availability without a prescription was observed in both groups, which is likely explained by the changing landscape involving approval of OTC NNS in March 2023 and availability in September 2023.5,24 An interesting exploratory finding was that most pharmacies continued to carry NNS behind the pharmacy counter only. As this OTC transition period progresses, increased store aisle availability is expected. Future studies should evaluate this outcome, as the current practice of keeping OTC NNS behind the pharmacy counter is likely to attenuate the impact of OTC reclassification.
Many factors impact willingness to dispense, including concerns about thresholds on controlled substance ordering from manufacturers and related enforcement actions from the DEA and state boards of pharmacy. 9 Additional barriers to buprenorphine dispensing may include pharmacists’ mistrust of prescribers, stigma against people who use drugs, concerns about diversion or DEA investigation, and confusion regarding changing guidelines.25,26 This study corroborates previous research identifying that independent pharmacies are considerably less likely than chain pharmacies to stock BUP/NX and NNS.3,4,8 While evidence explaining this difference is limited, it is hypothesized that independent pharmacies may have greater financial stakes in stocked products and greater concern about exceeding controlled substance ordering thresholds. Education has been identified as a key facilitator of increased BUP/NX dispensing, along with trust, improved provider communication, payor reimbursement, and removing or clarifying ordering thresholds. 26 Some of these concerns could be reasonably addressed through written communication or CE similar to the study intervention.
This study’s academic detailing intervention highlighted the critical issue of poor BUP/NX and NNS availability in community pharmacies, its impact on OUD patient care, and provided online CE to pharmacy staff interested in learning more about these topics. Studies have shown that online buprenorphine training increases willingness to dispense; unfortunately, low engagement with the online content in the present study likely reduced the overall impact of the intervention. 26 While the physical handouts provided important education to pharmacists, this content was designed to be brief and not as all-encompassing as the linked online material. Furthermore, it is unclear how many individuals engaged with the physical handouts. Only 5 pharmacy staff from the 393 pharmacies that received handouts completed the online CE content despite the incentivization that CE met a state board requirement. It is possible that some pharmacies had already received education, while others may not have had the time, interest, or motivation to engage in a voluntary hour-long program. While in-person academic detailing interventions increase the likelihood of pharmacist engagement with educational content and have been proven effective at increasing accessibility of OUD medications, 11 this method has greater logistical restrictions, including geographical and time limitations for the investigator, and is likely not feasible on a large scale, particularly in more rural locations. While the present intervention is feasible and scalable, it would likely need to be modified to significantly improve engagement and outcomes. However, there are also pharmacy-related barriers that may not be addressable through educational interventions. For example, education alone cannot facilitate increased trust and communication between pharmacists, prescribers, and patients. While education can be a valuable tool for addressing BUP/NX and NNS stocking issues, it may not be a sufficient solution to address the problem.
Limitations
This study involved an indirect intervention that was highly scalable but also susceptible to low engagement. Researchers attempted to counteract this by calling each pharmacy in the intervention group, notifying them of the mailed handouts, and offering to email PDF versions. Almost 50% of pharmacies in the intervention group (186/393) received both the physical and electronic handouts, though engagement with the more extensive online CE was minimal. It is unclear to what extent pharmacists engaged with the handouts, and these handouts alone may not have been sufficient to effect change. It is unclear if a significant improvement would have been observed with more engagement with the online CE. Pre- and post-intervention audits collected cross-sectional data by speaking to one pharmacist. These audits may have been impacted by confounding factors independent of the intervention (eg, the stocked product could have been dispensed earlier in the day). Pharmacy staff also had limited time (4-8 weeks) to engage with the handouts and online CE before the post-intervention audit; it is possible that greater engagement may have been observed if the post-audit had been conducted at a later time. A secret shopper approach was conducted, which decreases the likelihood of social desirability bias and mimics the patient experience, but this method may not provide a true estimate of medication stocking, and it is possible that different information would be provided to an actual patient presenting in person with a buprenorphine prescription. Inter-auditor variability could impact study results, though this was minimized by providing consistent training and a standardized script and data collection sheet. Audits specifically inquired about the availability of a 1-month (quantity #60) supply of BUP/NX 8/2 mg sublingual films. It is possible that pharmacies had an alternate buprenorphine formulation on hand, but this specific dose and formulation were selected based on expert opinion of what is commonly used in practice and informed by previous research.3,8 While a quantity of 60 films was requested, pharmacies were determined to have the medication stocked if they indicated they had the full or partial quantity on hand. Given that no similar studies had been previously reported, the initial sample size calculations were based on the investigator’s conjecture as to the intervention impact. However, the observed effect size of this intervention was much lower than anticipated, so it is possible that this study was underpowered. Furthermore, given the limited prior research into similar interventions, a novel, written academic detailing intervention was developed for this study. It is possible that different written tools could impact these results differently. Auditors sought to speak exclusively with pharmacists; pharmacy technicians were not intended to participate in the audits. However, this CE was available to both pharmacists and pharmacy technicians, and the study did not differentiate CE participation by profession, which could be a future area of research, as it is possible that results between these groups may differ. Finally, it is unclear if results are generalizable outside of Texas, or even across each region of Texas, given the large, diverse geography and demography of the state, though the large sample size and statewide distribution of the intervention should improve generalizability compared to many academic detailing studies that are limited to a small geographic area within driving distance of the research center.
Conclusions
In this study, crucial gaps were observed in BUP/NX and NNS availability from Texas community pharmacies. A mailed academic detailing intervention with links to online CE was found to be feasible and scalable, but was not sufficient to significantly increase BUP/NX and NNS availability, possibly due to low engagement or a need to address alternate pharmacy barriers. Higher-intensity interventions or strategies to increase engagement with similar low-intensity interventions, in conjunction with policy and regulatory interventions, are likely needed to address this critical issue. Future research incorporating qualitative follow-up could help clarify barriers and facilitators to engagement among both chain and independent pharmacy staff.
Supplemental Material
sj-docx-3-saj-10.1177_29767342261422061 – Supplemental material for Increasing Buprenorphine and Naloxone Access in Texas Community Pharmacies Through an Academic Detailing Intervention
Supplemental material, sj-docx-3-saj-10.1177_29767342261422061 for Increasing Buprenorphine and Naloxone Access in Texas Community Pharmacies Through an Academic Detailing Intervention by Sorina B. Torrez, Kirk E. Evoy, Kelly R. Reveles, Lucas G. Hill, Kenneth A. Lawson and Lindsey J. Loera in Substance Use & Addiction Journal
Supplemental Material
sj-pdf-1-saj-10.1177_29767342261422061 – Supplemental material for Increasing Buprenorphine and Naloxone Access in Texas Community Pharmacies Through an Academic Detailing Intervention
Supplemental material, sj-pdf-1-saj-10.1177_29767342261422061 for Increasing Buprenorphine and Naloxone Access in Texas Community Pharmacies Through an Academic Detailing Intervention by Sorina B. Torrez, Kirk E. Evoy, Kelly R. Reveles, Lucas G. Hill, Kenneth A. Lawson and Lindsey J. Loera in Substance Use & Addiction Journal
Supplemental Material
sj-pdf-2-saj-10.1177_29767342261422061 – Supplemental material for Increasing Buprenorphine and Naloxone Access in Texas Community Pharmacies Through an Academic Detailing Intervention
Supplemental material, sj-pdf-2-saj-10.1177_29767342261422061 for Increasing Buprenorphine and Naloxone Access in Texas Community Pharmacies Through an Academic Detailing Intervention by Sorina B. Torrez, Kirk E. Evoy, Kelly R. Reveles, Lucas G. Hill, Kenneth A. Lawson and Lindsey J. Loera in Substance Use & Addiction Journal
Footnotes
Acknowledgements
The authors thank Sofia Garcia, Emily Johnson, Kristopher Rodriguez, Daniel Sledge, Victoria Altenberg, Hannah Angeles, Lauren Burnett, Casey Carswell, Jamie Chan, Chad Dittmar, Chanelle Duncan, Maria Gugliano, Nahal Iranpour, Berkin Kutluk, Esther Lin, Kim Nguyen, Irene Raju, Maya Ramirez, Delandra Robinson, Niraja Surendran, Fre’Daiza Tucker, and Kelly Yu for contributing to data collection on a volunteer basis.
Author Note
The views expressed do not necessarily reflect the official policies of the Department of Health and Human Services or Texas Health and Human Services; nor does mention of trade names, commercial practices, or organizations imply endorsement by the US or Texas Government. The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation of the manuscript; or the decision to submit the article for publication.
Ethical Considerations
The University of Texas at Austin Institutional Review Board determined this study was not regulated human subjects research.
Author Contributions
SBT: project administration, conceptualization, investigation, writing—original draft; KEE: conceptualization, methodology, supervision, writing—review & editing; KRR: conceptualization, methodology, supervision, formal analysis, writing—review & editing; LGH: funding acquisition, methodology, supervision, writing—review & editing; KAL: methodology, formal analysis, writing—review & editing; LJL: project administration, methodology, writing—review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Texas Targeted Opioid Response, a public health initiative operated by the Texas Health and Human Services Commission through federal funding from the Substance Abuse and Mental Health Services Administration grant award number 1H79TI087739.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Hill has been paid to serve as an expert witness on behalf of Kroger, Albertsons, Publix, and Meijer in civil litigation related to opioid dispensing. The other authors report no conflicts of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
