Abstract
Background:
Injectable opioid agonist treatment with hydromorphone (iOAT-H) is effective for persons who inject drugs (PWID) with opioid use disorder (OUD) but remains unavailable in the United States. Our objective was to determine interest in iOAT-H among syringe services program (SSP) participants.
Methods:
We recruited PWID with OUD from SSPs in New York City. Interest in iOAT-H was assessed on a 4-point scale. We compared participants who were and were not interested in iOAT-H regarding sociodemographic characteristics and self-reported variables (past 30 days): heroin use, public injection practices, and participation in illegal activity other than drug possession. Participants reported their preferred OUD treatment and reasons for these preferences.
Results:
Of 108 participants, most were male (69%), Hispanic (68%), and median age was 42 years. The median number of prior OUD treatment episodes was 6 (interquartile range: 2-12). Most (65%) were interested in iOAT-H. Interested participants (vs not interested) reported, over the prior 30 days, greater heroin use days (mean, 26.4 vs 22.3), injecting in public more times (median, 15 vs 6), and a higher percentage having participated in illegal activity (40% vs 16%). Preferences for OUD treatment were: iOAT-H (43%), methadone (39%), and buprenorphine (9%). Participants who preferred iOAT-H to conventional OUD treatments reported preferring injection as a route of administration and that available OUD treatments helped them insufficiently.
Conclusions:
SSP participants with OUD reported high interest in iOAT-H. Participants had attempted conventional treatments but still used heroin almost daily. We identified PWID at risk for opioid-related harms who potentially could benefit from iOAT-H.
Keywords
Introduction
Injectable opioid agonist treatment (iOAT) is a safe and effective opioid use disorder (OUD) treatment for persons who inject drugs (PWID).1 -3 Diacetylmorphine (DAM), also known as heroin, and hydromorphone are strong opioids that can be provided for self-injection under medical supervision, typically up to 3 times per day. 4 Oral methadone or another long-acting opioid may be prescribed with iOAT. Like other agonist medications for opioid use disorder (MOUD), the rationale for prescribing injectable opioids is to minimize opioid withdrawal, reduce opioid cravings, and avoid adulterants in non-prescribed opioids, thereby mitigating risks of overdose, acute and chronic infections, and other harms. 5 At least one form of iOAT is available in Canada, Denmark, Germany, Switzerland, the Netherlands, and England; however, iOAT remains unavailable in the United States. 2
In the early 20th century, US physicians commonly prescribed injectable opioids at low doses to manage morphine addiction. 6 The Harrison Narcotic Act of 1914 and subsequent US Supreme Court decisions, positioned opioid prescribing to treat OUD as outside of legitimate medical practice, forcing most opioid maintenance or detoxification clinics to close by the 1930s. 7 However, in 1965, with new research demonstrating that methadone treatment was highly effective, US federal law created an exception for licensed opioid treatment programs to administer oral methadone under strict regulations. 8 Methadone remained the sole approved MOUD option in the United States until the Drug Addiction Treatment Act of 2000, which allowed some physicians to prescribe buprenorphine. Thus, US laws strictly regulate what opioids may be prescribed to manage OUD, but there is a growing need for a range of effective OUD treatments.
Nine international randomized controlled trials (RCTs) demonstrate the safety and efficacy of iOAT with DAM or “heroin-assisted treatment.”2,3,9 When compared to enhanced methadone treatment, iOAT with DAM reduces non-prescribed opioid use, participation in illegal activity (including drug offenses and property crimes), and improves health (including physical and mental health) and well-being. 2 For example, in a Canadian RCT, participants receiving iOAT with DAM (vs oral methadone) had greater 12-month retention in treatment (88% vs 54%), reductions in days per month using non-prescribed heroin (from 26.6 to 5.3 vs 27.4 to 12.0), and more participants had reduced illegal activities (67% vs 48%). 10 Fewer studies have examined iOAT with hydromorphone (iOAT-H), but the Study to Assess Longer-term Opioid Medication Effectiveness (SALOME) was a phase 3, double-blind Canadian RCT that found iOAT-H to be non-inferior to iOAT with DAM.11 -13 Participants in both arms (hydromorphone vs DAM) greatly reduced the average number of days per month that they used non-prescribed opioids (from 27.3 to 5.8 vs 28.6 to 4.9) and participated in illegal activity (from 12.8 to 3.8 vs 15.5 to 2.8), and most reported health improvements. The group receiving hydromorphone had significantly fewer serious adverse events, including seizures, somnolence, and life-threatening opioid overdoses, than those receiving DAM. 12 Treatment retention for iOAT-H over 180-day periods during the trial and in an open-label extension was 87% and 78%, respectively. 14 Trial participants could not determine whether they received hydromorphone or DAM.11,15 In 2019, based on SALOME findings, iOAT-H was approved in Canada to treat severe OUD. 16 These data are compelling; however, iOAT efficacy may differ in the current era defined by synthetic opioids in unregulated drug supplies, and additional clinical trials will likely be necessary to confirm iOAT-H feasibility, acceptability, and safety.
In planning for an iOAT-H pilot in the United States, we sought to determine whether PWID were likely to use iOAT. We focused on iOAT-H because hydromorphone’s status as an approved (Drug Enforcement Agency [DEA] Schedule II) drug in the United States that is commonly used to treat severe pain may enhance its feasibility in comparison to DAM, which is a Schedule I drug and therefore prohibited for medical use. 2 Our main objective was to determine whether PWID with severe OUD would be interested in iOAT-H. We hypothesized that participants interested in iOAT-H would have more OUD-related problems than those who were not interested, including more frequent heroin use, public injection, and greater participation in illegal activity. These data should inform potential benefits that could stem from making iOAT-H available in the United States. We also explored iOAT-H acceptability by inquiring about participants’ preferences for OUD treatment and perceptions of potential benefits that could result from iOAT-H.
Methods
We conducted a cross-sectional survey recruiting PWID with OUD from 3 syringe services programs (SSPs) in New York City. This study was approved by the Albert Einstein College of Medicine institutional review board.
Setting
Combined, the 3 SSPs annually provide services to approximately 14 500 unduplicated persons, including provision of sterile syringes and injecting equipment; human immunodeficiency virus (HIV), hepatitis C virus (HCV), and sexually transmitted infection screening and medical treatment; case management; hot meals and food pantry; and other social services. The SSPs serve clients who use drugs regardless of whether they inject. All 3 SSPs offer onsite buprenorphine treatment (sublingual but not extended-release injectable buprenorphine), but there is no iOAT available.17,18
Recruitment
We recruited a convenience sample of PWID between November 2020 and August 2021. We posted and distributed study flyers at SSPs, staff members referred interested candidates to the research team, and a study coordinator recruited at SSPs when allowed by COVID-19 health and safety protocols. We intended to administer all surveys in-person, but in compliance with social distancing guidelines, informed consent and surveys were conducted by telephone when necessary. Research staff described the study, elicited informed consent (oral or written), and screened for eligibility. Recruitment was conducted in English or Spanish by bilingual staff members. Each participant was compensated with $20 cash and a roundtrip transit pass worth $5.50. The sample size was limited by available funds; therefore, recruitment continued until funds were expended.
Participants
Eligibility criteria were: (1) age ≥18 years; (2) lifetime history of OUD (self-reported); (3) lifetime history of injection opioid use (self-reported); (4) ability to provide informed consent; and (5) fluency in English or Spanish.
Questionnaire
Items were adapted from studies that examined SSP participants’ attitudes toward MOUD and harm reduction services.19 -22 We piloted questions with SSP staff members who had lived experience of injecting opioids and adapted content based on their feedback. For example, staff members recommended including synthetic cannabinoids among potential substances being used, because of their knowledge of participants’ substance use patterns. Because iOAT-H would be a new modality, the questionnaire included a scripted overview of iOAT-H that described hydromorphone, side effects, and iOAT treatment goals based on published iOAT guidelines (see online Appendix 1). 4 The instrument was programmed in English and Spanish in the online survey platform Qualtrics (Provo, UT, USA) and administered by research staff.
Measures
Substance Use and Treatment
We used the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) criteria to categorize participants as having mild (2-3), moderate (4-5), or severe (≥6) OUD. 23 We assessed current substance use with an adapted version of the Addiction Severity Index. 24 Participants self-reported past 30-day use of several substances, their most common route of administration, and whether the substances were prescribed to them. We also asked about prior OUD treatment history, including the number of past treatment episodes for outpatient treatment with group or one-on-one counseling, residential treatment programs, or MOUD (methadone, buprenorphine, or naltrexone). Participation in medically managed withdrawal (“detox”) and mutual aid organizations (eg, Narcotics Anonymous) were reported separately but were not considered OUD treatment episodes. As per the definition used in iOAT clinical trials, we categorized participants as treatment experienced if they reported at least 2 OUD treatment episodes in the prior 5 years. 11 We also inquired whether participants had ever experienced a nonfatal opioid-involved overdose, and if so, the number of overdoses in their lifetime. 25
iOAT Knowledge
Participants reported whether they had ever heard about clinics where people with OUD are prescribed DAM (yes/no) or injectable hydromorphone (yes/no), whether they had ever been treated at an iOAT clinic (yes/no), and whether they had ever taken hydromorphone tablets (Dilaudid) in the past without a prescription (yes/no).
Interest in iOAT
Our primary outcome was interest in iOAT-H. Participants reported their interest in starting iOAT-H treatment if it were made available (very interested; somewhat interested; undecided/unsure; not interested). We considered those very or somewhat interested as endorsing interest in iOAT-H.
Preferences Concerning OUD Treatment
Participants also reported preferences for OUD treatment by selecting one option from the following: buprenorphine (Suboxone or Subutex); methadone; naltrexone (Vivitrol); injectable hydromorphone (Dilaudid); a program without medications; or not interested in treatment. In a prior study, SSP participants reported preferring sublingual to injectable buprenorphine, so we chose not to include the injectable formulation as an option. 26
Participants who reported iOAT-H as their preferred MOUD, were asked to report their agreement on a 5-point scale (1 = strongly disagree, 5 = strongly agree) with several statements regarding potential reasons for choosing iOAT-H over other MOUDs. These statements were developed with SSP staff members and phrasing was adjusted to use common terms. Results were dichotomized with scores of 4 and 5 representing agreement with proposed statements. For participants who reported preferring other MOUDs, we also assessed potential reasons for these preferences (data presented elsewhere 27 ).
Potential Personal Benefits of iOAT-H
Participants who indicated interest or were unsure about starting treatment with iOAT-H were presented with several statements that described potential benefits of starting treatment. These statements were developed with SSP staff members and phrasing was adjusted to use common terms. Statements presented potential perceived personal benefits: I would be less likely to inject in public; less likely to drop syringes on the street; less likely to put myself or others at risk; community members would be less likely to report me; it could reduce my chances of overdosing; it could lower my chances of getting abscesses or skin infections; and I could get care for other medical problems like HIV or HCV. Participants reported their level of agreement on a 5-point scale (1 = strongly agree, 5 = strongly disagree). Results were dichotomized with scores of 4 and 5 representing agreement with statements.
Covariates
Covariates included sociodemographic data: age, race, ethnicity (Hispanic, yes/no), primary language (English; Spanish; other), assigned sex at birth (male; female), gender identity (male; female; transgender; gender nonconforming), employment status, health insurance status, and housing status (rent/own; unstable housing). Participants also reported criminal-legal involvement: if they were currently on probation or parole (yes/no); whether they are mandated to treatment (yes/no); and how many months they had spent incarcerated in their lives. We also inquired if, in the past 30 days, they were involved in illegal activities that they could be arrested for (excluding substance use or possession offenses; yes/no), and if so, the number of times they have been arrested. Lastly, participants reported if they had ever injected in public (yes/no), and if so, the number of times injected in a public setting in the past 30 days (full data presented elsewhere). 28
Data Analysis
We used descriptive statistics to summarize participants’ characteristics. Specifically, we report means and standard deviations for normally distributed continuous variables, medians and interquartile ranges (IQRs) for skewed continuous variables, and frequencies and percentages for categorical variables. Next, we determined the number of participants who were interested in and expressed preference for iOAT-H. We then compared sociodemographic and clinical characteristics among participants who were and were not interested in iOAT-H. Chi-square testing was used for categorical variables, and T-test or Mann–Whitney U tests were used for continuous variables. All analyses were performed using Stata software (version16.1, College Station, TX, USA).
Results
Sample Description
We screened 134 SSP participants and 108 enrolled and completed the questionnaire. Most questionnaires were administered in English (87%) and conducted remotely via telephone (70%). Participants were predominantly male (69%), identified as Hispanic (68%), with a median age of 42 years (IQR: 36-51). Most participants were unstably housed (78%) and reported having health insurance coverage (89%), including 86% with Medicaid and 11% with Medicare.
Substance Use
All participants had injected opioids in their lifetime and 94% reported injecting heroin or fentanyl in the past 30 days. Nearly all participants (98%) met the DSM-5 criteria for severe OUD. Half (48%) were currently receiving MOUD, including 44% who were prescribed methadone. Participants reported using heroin or fentanyl on 25.0 (SD ±8.9) of the past 30 days, with most (90%) reporting intravenous administration as their typical route of administration. Participants who reported receiving MOUD (N = 52) also used heroin or fentanyl on 22.4 (SD ±10.6) of the past 30 days. Other substance use was also very common (see Table 1). Most participants had experienced an opioid-involved overdose during their lifetime (69%), and the median number of lifetime opioid-involved overdoses was 2 (IQR = 0-3). Based on past OUD treatment, most participants (79%) were considered treatment experienced. The median number of lifetime OUD treatment episodes was 6 (IQR = 2-12), including 1 (IQR = 0-1) methadone treatment episode.
Sociodemographic and Clinical Characteristics by Interest in iOAT-H.
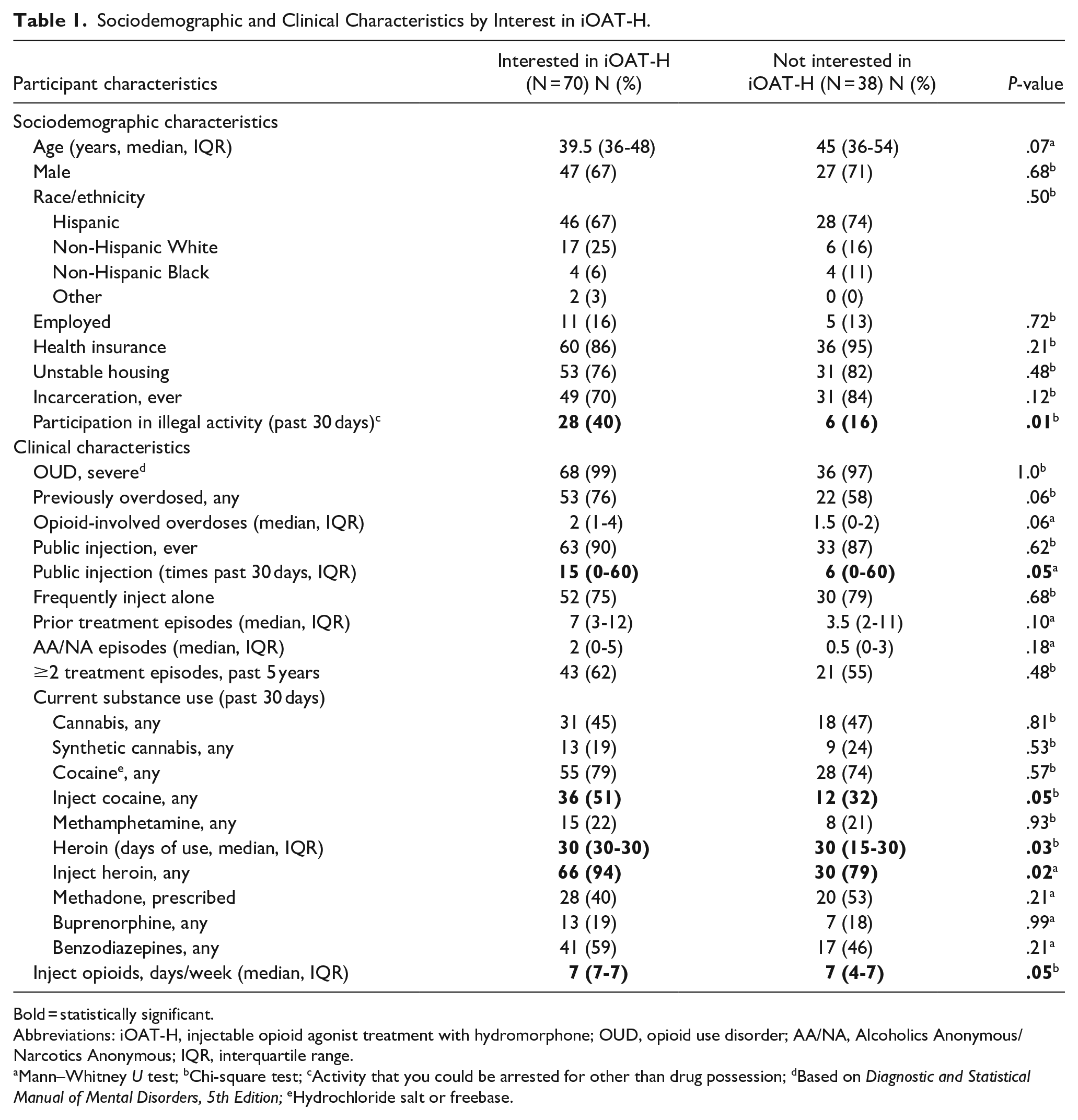
Bold = statistically significant.
Abbreviations: iOAT-H, injectable opioid agonist treatment with hydromorphone; OUD, opioid use disorder; AA/NA, Alcoholics Anonymous/Narcotics Anonymous; IQR, interquartile range.
Mann–Whitney U test; bChi-square test; cActivity that you could be arrested for other than drug possession; dBased on Diagnostic and Statistical Manual of Mental Disorders, 5th Edition; eHydrochloride salt or freebase.
iOAT-H Knowledge and Interest
Few participants (26%) had heard of iOAT-H clinics, while just over half (52%) had heard about clinics where people are prescribed DAM. Two participants reported having been treated with iOAT-H, and 30% reported previously taking hydromorphone tablets.
Most participants (65%) were interested in iOAT-H. In bivariate analyses (Table 1), participants who were interested in iOAT-H (vs not interested) were significantly more likely to have engaged in an illegal activity (other than drug possession) in the past 30 days (40% vs 16%), and they reported, within the past 30 days, greater number of days using heroin or fentanyl (mean, 26.4 vs 22.3) and injecting in public more times (median, 15 vs 6). Excluding participants (N = 9) who had not injected opioids in the past 30 days did not significantly change the proportion reporting interest in iOAT
Personal Preferences for OUD Treatment and Perceived Benefits
iOAT-H was the most commonly preferred OUD treatment reported by participants (43%), closely followed by methadone treatment (39%; see Figure 1). Among participants taking prescribed methadone (N = 48), more reported methadone (48%) than iOAT-H (42%) as their preferred OUD treatment. Participants who preferred iOAT-H endorsed the following reasons: other MOUDs had not worked for them (83%), the potential euphoria or high (70%), and preferring injection over other routes of administration (94%; see Figure 2a). Most participants agreed that if available, iOAT-H could result in many benefits to themselves: avoiding fentanyl (79%), saving money (92%), avoiding the police (96%), and getting other medical treatment (95%; see Figure 2b).
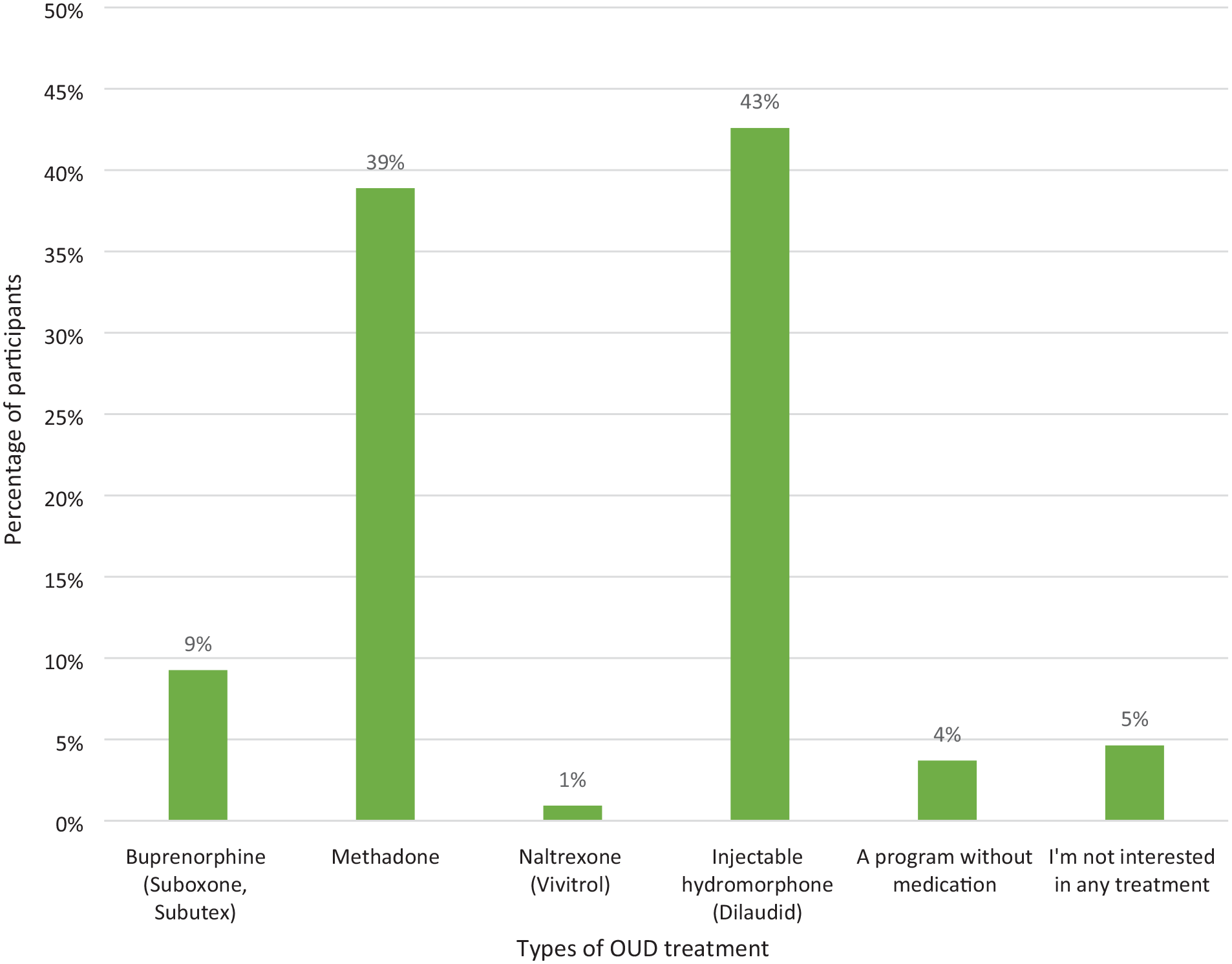
Preferences for OUD treatment among syringe service program participants (N = 108).
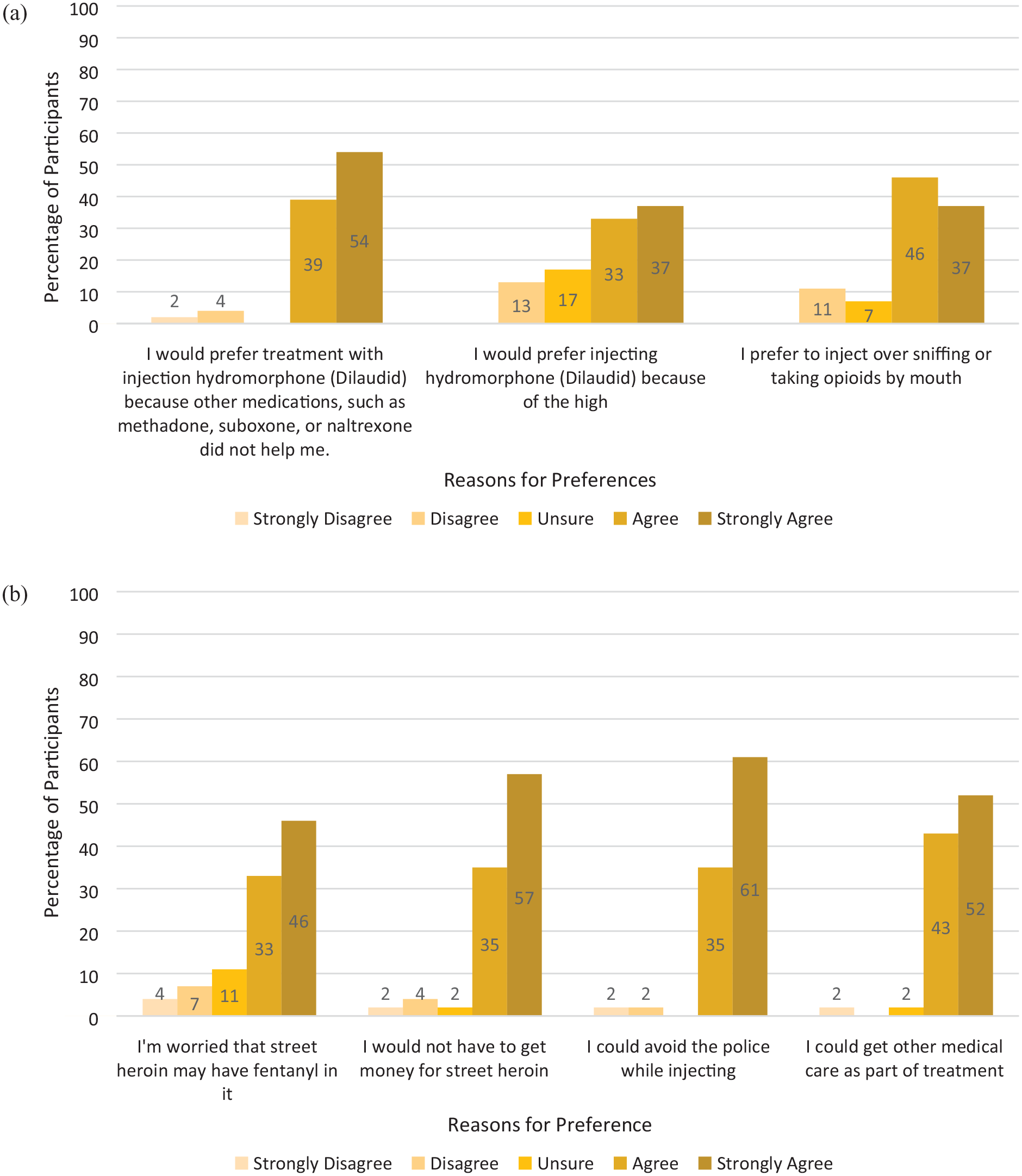
Among syringe services program participants who prefer iOAT-H (N = 46): (a) Reasons for preferring iOAT-H to other OUD treatment options. (b) Potential personal benefits of iOAT-H.
Discussion
In a sample of 108 SSP participants with OUD and a history of injecting opioids, we found that most were interested in receiving iOAT-H. More than 40% of participants reported that iOAT-H would be preferable to other forms of OUD treatment. Participants who were interested in iOAT-H injected heroin on more days per month, injected in public more frequently, and were more likely to participate in an illegal activity (other than drug possession) than participants who were not interested, suggesting that those who could most benefit from iOAT were also those who were most interested in the treatment. Though interest in iOAT-H was not universal, we identified a highly interested group of treatment-experienced PWID with severe OUD who were at high risk for OUD-related harms.
This is the first US study that explores the views of persons with OUD regarding iOAT-H as a MOUD. We described goals and procedures for iOAT-H, and 65% of participants expressed interest in potentially pursuing treatment. When given the option of asking questions about iOAT-H, few participants had specific concerns but 2 questioned whether hydromorphone could be addictive (data not shown). When prompted to choose a preferred OUD treatment, some participants still reported preference for methadone, which demonstrates that treatment preferences can be nuanced. Recent work examining iOAT-H interest in Australia similarly identified a population of PWID with considerable treatment experience and frequent opioid injection and 53% of participants perceived iOAT-H to be a good treatment option for them. 29 Based on our survey, we have no way of knowing whether participants expressing interest in iOAT-H would start treatment if it were available, but we identified treatment-experienced PWID to recruit for a future clinical trial.
Most participants had used conventional OUD treatments but remained at risk for OUD-related harms, which highlights a role for new treatment options. Nearly all the met criteria for severe OUD, approximately half reported that they were receiving MOUD when surveyed, yet most participants continued to use heroin or fentanyl frequently—on average 25 days/month—and most still injected non-prescribed opioids. The median number of prior opioid-involved overdoses was 2, participants reported participation in illegal activity (other than drug possession), and they injected in public a median of 10 times per month. The association between interest in iOAT and OUD-related problems may reflect that participants who are unable to stabilize their opioid use would look to new OUD treatment options to help mitigate risks. In the context of the current US overdose crisis, where opioids being sold as heroin frequently contain high-potency synthetic opioids (eg, fentanyl) in unknown concentrations and possibly xylazine or other sedatives, 30 maintaining the current OUD treatment status quo leaves participants like ours, and other PWID, at high risk for overdose, arrest, and other opioid-related complications. Other measures will be necessary to reduce these harms, but iOAT could be included in a comprehensive response to the US overdose crisis.
Participants’ reasons for preferring iOAT-H to other OUD treatments, considered with the finding that participants receiving conventional MOUD were still frequently injecting other opioids, reinforces that available options are not meeting the needs of many treatment-experienced individuals. Many participants reported dissatisfaction with available MOUD. Participants who were receiving methadone or buprenorphine reported using heroin or fentanyl on average 22 days in the prior 30. Many participants also acknowledged that they preferred to inject opioids and that the pleasure or euphoria from iOAT-H would make it a more acceptable treatment for them. In the United States, research on new MOUDs often prioritizes agents with low “abuse potential” or absence of reinforcing effects; however, if iOAT-H retains people with OUD in treatment, improves quality of life, and lowers opioid-involved overdose risk, then the pleasurable aspect of treatment may not conflict with patients’ own treatment goals.31,32 Treating OUD with methadone has remained controversial in the United States despite many decades of supportive research and clinical experience 33 ; therefore, there would likely be ideological opposition to iOAT-H as well. 34 Nonetheless, for many treatment-experienced persons with OUD, iOAT may be an acceptable and desirable option.
One potential downside to offering iOAT is that it could “crowd out” other OUD treatments, meaning that PWID would preferentially choose iOAT instead of other options that are less expensive and resource intensive. 3 Experience from Switzerland, where there is a range of MOUD options including DAM, demonstrates that over time, methadone treatment remains the mainstay and only a small fraction of people with severe OUD choose iOAT with DAM. 35 This is consistent with our finding that methadone was a preferred option for nearly 2 in 5 participants. Instead of “crowding out” other treatments, iOAT may engage underserved PWID who do not want conventional treatments, emphasizing the need for a spectrum of evidence-based MOUD options.
Future Directions
The next step in this work will be planning for an iOAT-H clinical trial in the United States. To our knowledge, hydromorphone is currently only used for iOAT in Canada, but comprehensive guidelines are available that provide recommendations for clinical management and program implementation. 4 However, the SALOME trial was conducted in 2011 to 2013, before fentanyl had replaced heroin as the predominant opioid in British Columbia.11,36 Fentanyl penetration into US drug supplies may be creating challenges with other MOUDs 37 ; thus, confirming that iOAT-H efficacy in the fentanyl era will be important before making wide-spread investments. We have chosen to focus on hydromorphone for pragmatic reasons due to its established domestic supply chain and DEA scheduling; however, there is also strong rationale for testing iOAT with DAM due to the more robust evidence base. 2 In either case, injectable therapies could provide benefits in comparison to conventional MOUDs for PWID with severe OUD.
Limitations
Our study has several limitations. Few participants had heard of iOAT-H as an OUD treatment, so their perceptions were based on a brief description provided by study staff and not direct knowledge of the treatment modality. The description of iOAT provided by our team may not have given sufficient detail about the pharmacologic effects of hydromorphone or the structure of potential treatment programs for some participants to accurately estimate their interest in the treatment modality. We recruited a convenience sample of participants from SSPs in New York City, and findings may not generalize to PWID with different racial/ethnic backgrounds, those who do not use SSPs, or those living in other geographic areas where injection drug use is common (eg, non-urban areas) but access to conventional MOUD is more limited. The sample size was limited by available funds, and we cannot be confident that the convenience sample here reflected the typical characteristics of the SSPs’ clients. The proportion of male to female participants was 2:1, but this is consistent with the demographics of SSP participants nationally. 38 Data were all self-reported, therefore treatment history and other measures are subject to recall bias. We did not ask whether participants preferred fentanyl or other synthetic opioids to heroin, or whether they were using xylazine, which may influence attitudes toward iOAT-H. Finally, expressing interest in iOAT-H treatment does not guarantee future treatment engagement; however, we previously published on interest in buprenorphine treatment at SSPs and have since initiated clinical services at SSPs with good treatment uptake. 17
Conclusion
First-line MOUDs, such as methadone and buprenorphine, work well for many persons with OUD; however, even when these conventional treatments are available, important groups of PWID remain out of treatment and at high risk for opioid-related harms. iOAT-H provides a promising option to engage treatment-experienced individuals who continue to inject opioids after trying first-line OUD treatments. Our study demonstrates interest in iOAT-H, especially among persons at high risk for overdose—those with severe OUD who frequently use non-prescribed opioids and inject in public places. Broadening the spectrum of MOUDs available in the United States could help address the unacceptably high rates of opioid-involved overdose death.
Supplemental Material
sj-docx-1-saj-10.1177_29767342231210552 – Supplemental material for High Interest in Injectable Opioid Agonist Treatment With Hydromorphone Among Urban Syringe Service Program Participants
Supplemental material, sj-docx-1-saj-10.1177_29767342231210552 for High Interest in Injectable Opioid Agonist Treatment With Hydromorphone Among Urban Syringe Service Program Participants by Aaron D. Fox, MD MS, Lindsey Riback, MPH, Andres Perez-Correa, MD, Eric Ohlendorf, BS, Megan Ghiroli, BA, Czarina Navos Behrends, PhD and Teresa López-Castro, PhD in Substance Abuse
Footnotes
Acknowledgements
We thank the staff and participants from New York Harm Reduction Educators, Boom! Health and Washington Heights Corner Project, without their assistance and participation this study could not have been completed. Bilal Abbas contributed greatly to the project, including conception and data collection.
Author Contributions
ADF (PI) designed the study, extracted and managed the data, performed the analyses, interpreted the results, and drafted the article. AP-C, LR, and EO led initial data collection and project coordination. CNB, AP-C, LR, EO, MG, and TL-C revised the article and provided critical comments. All authors approved of the final article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ADF receives royalties for an online chapter on primary care for patients with opioid use disorder from Wolters Kluwer. Other authors report no financial conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was awarded by the New York Academy of Medicine to explore attitudes toward novel OUD therapies among SSP participants.
Compliance,Ethical Standards,and Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki. The Institutional Review Board (IRB) at the Albert Einstein College of Medicine reviewed the study protocol (2020-12002) and determined this to be exempt research.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
