Abstract
Background:
Evidence-based early intervention practices, such as screening, brief intervention, and referral to treatment (SBIRT), are recommended to identify unhealthy use and provide linkages to treatment to prevent substance use disorder. However, there is a lack of screening frequency recommendations. Pennsylvania (PA) SBIRT was a five-year initiative to implement SBIRT in primary care sites. This study evaluated the effects of screening policies in PA SBIRT on changes in substance use risk category over time.
Methods:
Quantitative data were obtained from seven primary care sites implementing SBIRT, with patients who were screened twice using the Drug Abuse Screening Test, US Alcohol Use Disorders Identification Test, or the Alcohol, Smoking, and Substance Involvement Screening Test and experienced any risk category change (n = 1,364). Patients were 49% male, 51% female, 81% White, and 14% Black. An intercept-only generalized estimating equation model tested whether category changes between screen one and screen two were statistically significant.
Results:
The average number of months between screenings for those experiencing a risk category change was 9.01 with a range of 0 to 46 months. There was a statistically significant change between screening one and screening two (P > 0.001), with 44% undergoing a decrease in risk category and 56% undergoing an increase in risk category. Of those undergoing an increase in risk category, 91% moved from a negative/low risk category to a positive/higher risk category.
Conclusions:
Results suggest that frequent screening policies may improve identification of substance use risk category changes. The results act as a catalyst to further evaluate recommended screening intervals for detecting unhealthy substance use to increase identification and patient connection.
Introduction
Substance use disorder (SUD) and overdose deaths are critical public health concerns nationally and in Pennsylvania (PA).1 -5 The 2022 National Survey on Drug Use and Health data show that 57.8%, or 161.8 million Americans aged 12 and older in 2021, used tobacco, alcohol, or an illicit drug in the past month. 6 The DSM-5 defines SUDs as “a maladaptive pattern of substance use leading to clinically significant impairment or distress.” 7 The US Preventive Services Task Force (USPSTF) defines unhealthy use of alcohol and other drugs as any use that “increases the risk of likelihood of health consequences or has already led to health consequences.” 8
Evidence-based early intervention practices, such as screening, brief intervention, and referral to treatment (SBIRT), are recommended to identify unhealthy use and provide linkages to treatment to prevent SUD. 9 Previous studies show SBIRT as a feasible option to reduce alcohol and other drug use in healthcare settings.10 -16 Research has demonstrated some benefits of SBIRT, especially related to alcohol use, including reductions in healthcare costs in emergency departments and improvements in the perceived attitudes of nursing staff toward patients who use alcohol.13,17 -22 The flexibility of screening and brief intervention (BI) can be a realistic option to integrate and expand services, as there is evidence of positive patient satisfaction using SBIRT via telehealth during emergency department visits.23,24 The USPSTF recommends screening adults, 18 years of age and older, in primary care settings for unhealthy use of alcohol and other drugs and engaging those using them at a harmful or hazardous risk level in brief counseling. 18
The Pennsylvania SBIRT (PA SBIRT) initiative provided SBIRT services to over 20,000 patients between 2015 and 2021. PA SBIRT included technical assistance (TA) from the University of Pittsburgh’s Program Evaluation and Research Unit (PERU) in partnership with nine healthcare sites in three counties in PA to integrate optimal screening into the workflow and monitor funder data requirements. PA SBIRT aimed to ensure that every patient received the appropriate SBIRT services from the appropriate provider as early as possible. To support this vision, seven of the nine partnering healthcare sites implemented screening policies during the grant period to screen every patient during every visit. During conversations on sustainability policies for screening integration, providers reported mixed responses around the screening of every patient at every visit screening policy, reporting positive feedback to universal and frequent screening to increase patient rapport and reduce stigma, but also challenges in workflow implementation or possible patient frustration. Given the mixed reviews of the screening policy, leaders at participating healthcare sites inquired about best practices in screening frequency and provide timely intervention.
The USPSTF lacks explicit guidance regarding screening frequency, stating that there is little or inadequate evidence around the optimal screening interval. 8 Studies reviewing screening implementation to evaluation are plentiful, but also lack guidance around screening frequency.25 -30 In lieu of strong evidence to guide policies, healthcare sites may generate policies based on how heavily they weigh convenience versus perceived risk about how quickly substance use can escalate to hazardous levels. 31 Providers who weighed convenience tended to prefer annual screenings bundled with general health screening visits; providers who were concerned about how quickly substance use can escalate were more likely to advocate for more frequent screenings, as annual screenings alone could miss opportunities for early intervention. 31 Additionally, universal screening integrated within the workflow may reduce biases in screening and identification of individuals using substances. 16 Given the inquiries from the healthcare sites, lack of explicit guidance from the USPSTF, and PERU’s positionality with the robust data set, PERU conducted a secondary analysis from the PA SBIRT screening data to examine patterns and direction of change in substance use risk category among patients being screened on multiple occasions. The results generated initial insights into the applicability of frequent screening policies.
Materials and Methods
Quantitative data were collected as part of the grant reporting requirements, focusing on the volume of screening, screening outcomes, collecting the Government Performance and Results Act (GPRA) data, and entering the data into the Substance Abuse and Mental Health Administration’s (SAMHSA) Performance Accountability and Reporting System (SPARS). Nine healthcare sites were recruited as part of the grant. Before implementing SBIRT, participating sites were asked to select and implement one of the three evidence-based screening tools and a screening frequency policy. Screening rates at each participating site were assessed using a combination of aggregated patient volume by site, number of screenings administered, and outcomes from patient risk screening categories. Tailored TA was provided to each site regarding screening implementation; sites implemented screening modes based on what worked best for their existing workflow (e.g., verbal, paper, or electronic screenings; via medical assistant, nurse, or provider). The TA team monitored the screenings administered, patient volume, and categorical outcomes from screenings by the site and engaged in continuous quality improvement activities to increase screening rates and optimize screening administration. Therefore, data used for the current study were initially collected as part of the reporting requirements during grant implementation and was not collected for the specific purpose of examining repeated screenings.
The present study was a secondary data analysis of the quantitative screening data collected as part of the grant reporting requirements to understand patient screening frequency and screening outcomes. Interventions based on screening outcome was not a main objective of the grant, and thus, was unable to be retroactively traced due to patient de-identification. To test whether more frequent screenings, rather than only annual screening policies, impacted the number of patients identified as having a change in risk category, only sites that adopted a policy to screen every patient during each visit were included in the study. Two of the 9 sites were removed because one was a pregnancy care center working with a special population with a trimester screening frequency policy and the other adopted a 90-day screening frequency policy. This resulted in a final sample of seven sites that adopted a policy to screen every patient during each visit. In total, sites screened 20,152 unique patients.
Measures
Sites used the Alcohol, Smoking, and Substance Involvement Screening Test (ASSIST), or both the US Alcohol Use Disorders Identification Test (US AUDIT) and the Drug Abuse Screening Test (DAST-10) as evidence-based screening tools. Scores on the respective screening tools were categorized into SBIRT risk categories: low, hazardous, harmful, and high, which corresponded to suggested interventions: none, BI, brief treatment (BT), and referral to treatment (RT). Though the specific items and numeric scoring systems are unique to each tool, SAMHSA recommends evidence-based screening tools and provides standard recommendations and interventions for outcomes across all three tools.17,32 -35 The screening scores and recommended interventions are as follows: DAST-10 (ten items) has scores ranging from 0 to ten. DAST-10 risk scores are: 1 to 2 = low, 3 to 5 = hazardous, 6 to 8 = harmful, and 9 to 10 = high; US AUDIT (ten items) has scores ranging from 0 to 40. US AUDIT risk scores are: 0 to 6/7 (women/men) = low, 7/8 to 15 (women/men) = hazardous, and 16 to 24 = harmful, 25+ high; and ASSIST (8 items) has scores ranging from 0 to 35. ASSIST risk scores are: 0-3*/10 = low, 4*/11 to 26 = hazardous, and 27+ = harmful.36,37 For the ASSIST, the higher number is for alcohol use only, and the cutoff score is for other substances. Thus, tools were compared using the categorical screening outcomes for standardization.
Data Analytic Plan
More than 80,000 patient screenings occurred throughout the grant cycle and were associated with 20,152 unique patients. Because of the de-identification procedures in data sharing adopted for the project and the SPARS reporting process rules, patients who may have been screened multiple times but did not change screening categories were unable to be differentiated from those patients only receiving one screening during the study period. Therefore, repeat screeners with no category change are included in the group with patients who only received one screening. Sixty-five patients from the two sites that did not screen every patient every visit were removed resulting in a final study sample size of 20,087. Patients were divided into two groups: those with only one screen or with multiple screens and no category change (n = 18,742; 93%), and the 1,345 patients (7%) with repeated screens that resulted in at least one SBIRT risk category change (Figure 1). This latter group was the sample of interest for the observational study.
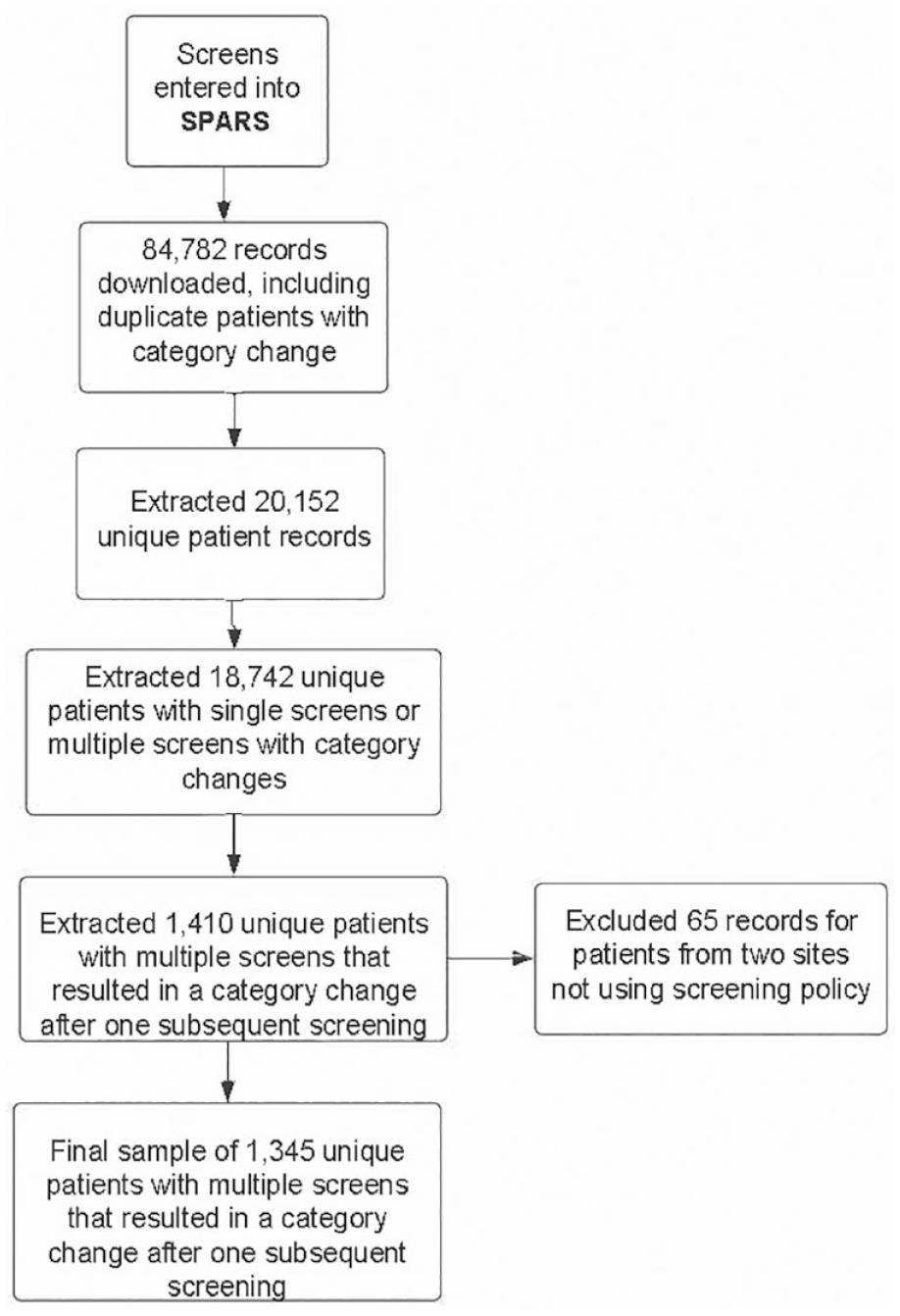
Shows the PA SBIRT data collection and extraction pathway, including exclusion criteria such as duplicate screening and sites not adhering to the screening policy. SPARS, Substance Abuse and Mental Health Administration’s (SAMHSA) Performance Accountability and Reporting System.
All 1,345 patients who experienced a risk category change between the first and second screens and were from a practice that adopted a screen every patient every visit policy were included in the sample. The screening category (e.g., screening, BI, BT, RT) was used as the outcome to account for the different screening tools used across sites. Descriptive statistics concerning demographic categories (age, gender, military service, race, ethnicity) between patients with category changes and patients with no category change or only one screening, frequency and proportion of patients in each risk category, number of patients changing categories and direction of change, numbers of months and days between screenings, and average number of days of self-reported use of drugs and alcohol between patients with category changes and patients with no category change or only one screening were explored. Traditional descriptive statistics were used to explore frequencies and proportions. To examine patterns of change, each risk category was assigned a value (1 = low, 2 = harmful, 3 = hazardous, 4 = high), a concatenated string variable was created from those values, and frequencies for each pattern were calculated, specifically the increase and decrease in risk category with each successive screen. To investigate the change patterns, a Sankey diagram was generated using the “ggalluvial” package in RStudio Version 0.12.3 (RStudio Team, http://www.rstudio.com/). In addition to descriptive statistics, an intercept-only generalized estimating equation (GEE) was performed using patients as subjects, screening number as the repeated measure (limited to two time points), and SBIRT risk category as the outcome. The GEE model was originally chosen because specific covariates (age, gender, military service, race, ethnicity) were to be added to the model. However, none of the covariates were significant in any of the models and were eventually dropped. This research was classified as exempt from the University of Pittsburgh Institutional Review Board because the data were collected only for program evaluation purposes and the project intended to inform adult primary care practitioners about the effectiveness of the SBIRT activities.
Results
The sample of patients used for the study reflected the overall sample of patients participating in the grant initiative (The demographic proportions were calculated using all available data resulting in variable n’s by characteristic. Missing data for age and gender were small; n = 7-16, while missing data for military service and ethnicity were larger; n = 283-738; Table 1). The study sample and population from which it was drawn were mostly White, roughly equally divided between males and females, had a similar distribution across age categories, and a similar proportion of the sample and population served in the military. No significant differences were found in demographics between the group of repeat screeners with a category change and the group screened only once or not experiencing a category change. Repeat screeners also reported similar average days of use for alcohol, use of both drugs and alcohol, and cannabis (analyses were performed on all available data). Only patients reporting use answered these questions. (N’s for the repeat screeners ranged from 110-654 while n’s for the overall group ranged from 581-2,704 depending on substance; Table 2). However, those with repeated screens had a statistically significant (F = 5.14, P = 0.02) lower self-reported average number of days of illegal drug use (mean = 7.80 days) compared to the overall participant group (mean = 8.96 days; A Brown Forsyth robust test of equality of means was performed in conjunction with the F-test. The pattern of results was the same, therefore, the F-test results were reported).
Demographics of Repeat Screeners With a Category Change Versus Non-Repeat Screeners and Repeat Screeners With No Category Change.

Select demographics available for all patients were compared for repeat screeners experiencing a category change versus non-repeat screeners and repeat screeners not experiencing a category change to determine if systematic differences between groups are present. No significant differences were found between the groups for any demographic category.
Low response categories included Alaskan Native, Native American, and Asian.
For Those Testing Positive, Comparison of Self-Reported Days of Use for Different Substances for Repeat Screeners with a Category Change Versus Non-Repeat Screeners and Repeat Screeners With No Category Change.
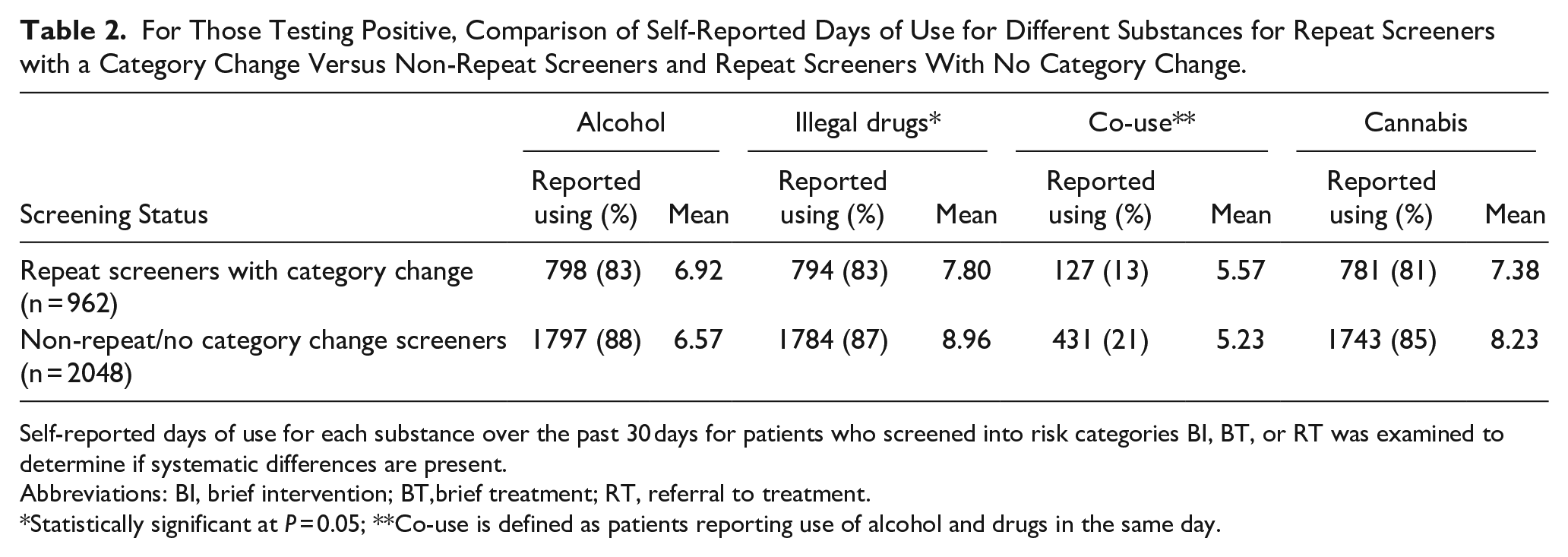
Self-reported days of use for each substance over the past 30 days for patients who screened into risk categories BI, BT, or RT was examined to determine if systematic differences are present.
Abbreviations: BI, brief intervention; BT,brief treatment; RT, referral to treatment.
Statistically significant at P = 0.05; **Co-use is defined as patients reporting use of alcohol and drugs in the same day.
The average number of months between screenings for those experiencing a risk category change was 9.01 with a range of 0 to 46 months. As shown in Figure 2, this distribution was negatively skewed with 50% of the second screenings happening between 0 and 6 months. The GEE model revealed a statistically significant intercept (P > 0.001) indicating a significant change between screening one and screening two. The model was also performed with demographic information used as predictors; however, age, race, and gender did not account for a statistically significant amount of variance in screening score changes (P > 0.05) and were dropped.
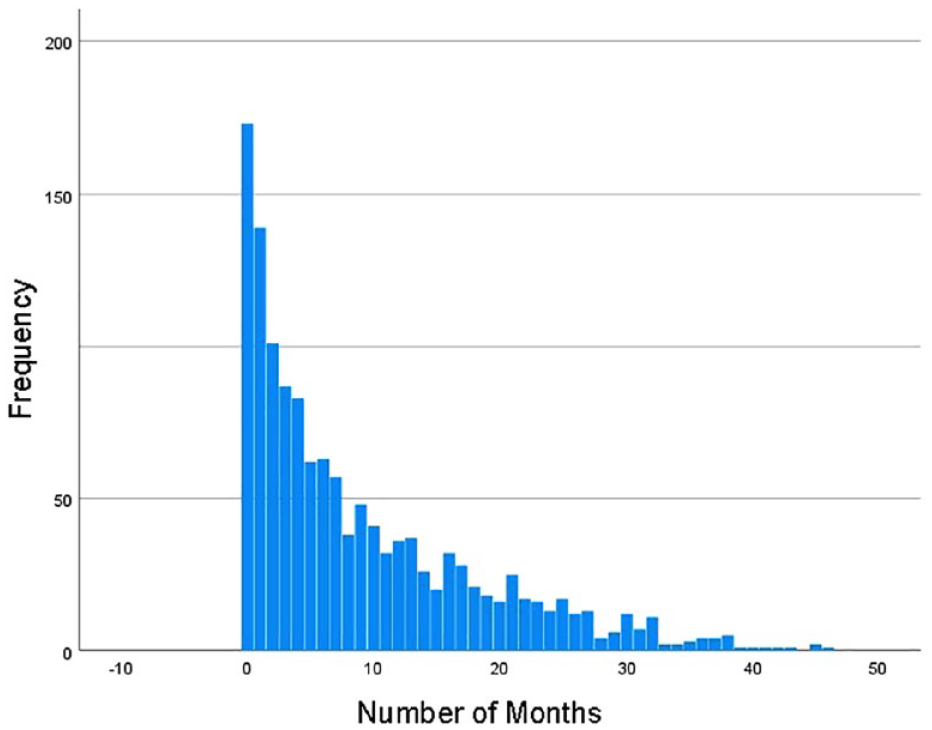
Distribution of the number of months between alcohol and substance use screenings.
The Sankey diagram (Figure 3) mapped the direction of change between screen one and screen two. The large majority (90%) of patients with initial negative screens increased in risk, moving from a negative screen to a positive screen (Figure 3). 37 Of those experiencing any risk category change (Figure 4), 581 (43%) had a decrease in risk while 758 (56%) had an increase in risk.
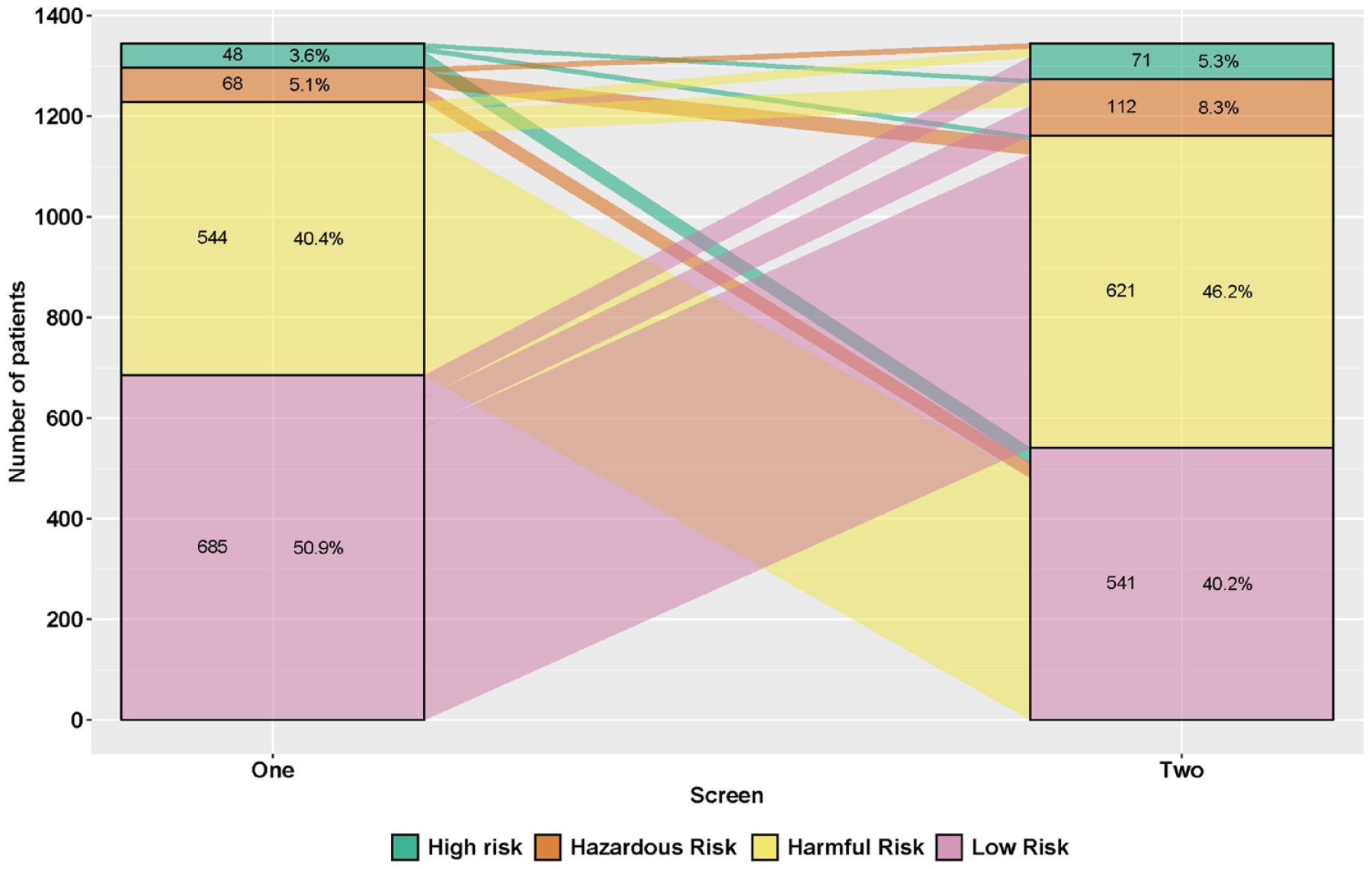
A Sankey diagram/alluvial plot depicting the change patterns in the frequency distributions of patients’ risk categories between their first and second alcohol/substance use screening assessment. Overall, 43% moved to a lower risk level between the first and second screen, 56% moved to a higher risk level. N = 1339 unique patients.
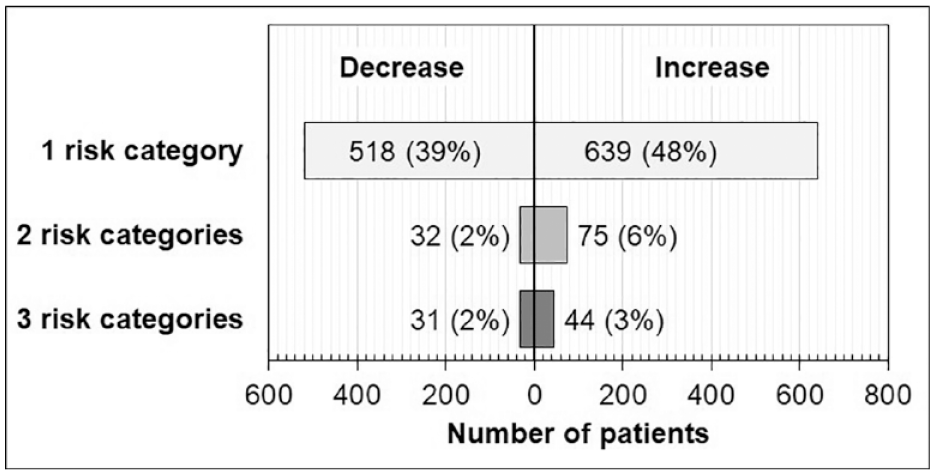
Shows the change in risk categories from a patient’s first screen to their second screen. Forty-eight percent of patients increased by one risk category between the first and second screen. Thirty-nine percent of patients decreased by one risk category between their first and second screen. N = 1,339 unique patients.
Discussion
Recommended screening frequency in adult primary care settings varies across conditions and guidelines to capture early detection of unhealthy conditions and gives providers the ability to provide appropriate early intervention.38,39 The USPSTF recommends alcohol and drug use screening in adult primary care settings, and explicitly states the need for continued research to understand optimal screening intervals. 8 The findings from this secondary analysis from the PA SBIRT initiative are an initial step to quantify and validate the perceived insufficiency in annual screening while working to understand the optimal screening rate. The statistically significant change between first and second patient screenings observed and depicted in Figure 2 as well as results showing half of the screenings which changed in category occurred between 0 and 6 months suggest that annual screenings alone may not quickly capture the incidence in change.
A change from a lower risk category to a higher risk category can have several implications, such as a change in patient’s behavior or a change in comfort level to disclose. Of the patients who reported an increased risk of substance use between screen one and screen two, most patients first reported no/low risk on the first screening, but then moved to a higher risk on the second screen. The change in risk category through more frequent screening can provide an opportunity for the provider to intervene early to reduce riskier use. In contrast, a change from higher to lower risk between screening one and screening two, as shown by 43% of the patients in this analysis, can indicate a positive change in health behaviors. Future studies should consider exploring the chance of changing categories based on the initial risk and impact of BIs and RTs.
Evidence points out that providers may support a more frequent screening policy by expressing concerns about infrequent substance use screening practices given perceptions about how quickly harmful or hazardous substance use can escalate without earlier intervention, explicitly calling out that annual screenings alone were insufficient for capturing critical windows for timely intervention. 31 Our findings support an increased screening frequency, capturing the average number of months between screening for those experiencing a risk category change was 9.01 months. While the range for the screenings was 0 to 46 months, about half of the second screenings occurred between zero and six months. Although research on screening frequency for SUDs in adult primary care settings is limited, these findings showing a direction in change with frequent screening policies offer some empirical support for existing guidelines and support for qualitative reports from providers.31,40
Adult primary care providers support universal and frequent screening, as it can open the door for a discussion with patients who may have been unlikely to disclose substance use, or are unaware of the consequences of unhealthy use as well as reduce biases in who is screened.16,40,41 However, some patients may refuse the offered screening, or worry about confidentiality, particularly if they do not have a positive relationship with their provider. 40 One study discussed the difference in demographics for those who consent compared to those who do not consent related to SBIRT via an electronic tablet. While there were no differences between age, gender, race, or education, those who did consent were more likely to be unemployed and uninsured, and more likely to live with someone who reported drug misuse, report a family history of drug use, and experience an episode of physical violence within the past year compared to those who did not consent. 42 The PA SBIRT initiative monitored healthcare site screening rates and implemented interventions for sites to increase screening rates and monitored consent for screening. Thus, it is important if conducting universal and frequent screening to understand the population consenting and not consenting to screen to identify at-risk use and provide timely intervention.
Healthcare professionals value the educational component of screening questions, but may feel unprepared to address substance use with a patient once identified or challenged with time constraints.41 -43 It is important to equip providers with evidence-based screening tools and clear rubrics for identification and next steps. In one study, of the providers who screen for alcohol and drug use, only 31.8% reported using a screening tool. 44 This is problematic, as one report shows as many as 94% of physicians can fail to accurately diagnose an alcohol disorder and intervene. 45 Results for the PA SBIRT initiative and previous literature reinforce that implementing and frequent use of evidence-based screening tools and processes can help providers accurately identify patients who could benefit from intervention.8,30,44
Additional barriers include time constraints, competing priorities, perceived patient frustration, and lack of time for relationship building between a provider and a new patient.41,42 Further, billing and low reimbursement remain challenges for SBIRT implementation. There are also challenges in training staff, modifications to workflows, and using approved procedure codes.45,46 Additionally, there are other complexities for screening patients under 18 years of age, with different guidelines for standardized screening tools and confidentiality concerns. There is an increased opportunity to change workflows and administer frequent screening, as screening and BIs can be delivered in office, telephone, web, mobile applications, or telemedicine.23,24 While initial findings from this analysis show promising results for optimal screening intervals, systemic changes in workflow, billing, and reimbursement should be considered for sustainable change. Screening every patient during each visit adds to the increasing demands on adult primary care physicians to provide high-quality care for individuals with an at-risk screen. One possible strategy is to implement team-based models, which may reduce up to 65% of a primary care physician’s service through having these services performed by healthcare team members or move away from a fee-for-service model and toward a value-based payment model. 43 Future research should consider exploring the support of the healthcare workforce.
The current study should be considered in light of several limitations inherent to all secondary data analytic designs.47,48 First, data collection and management prioritized clinical practice needs to provide for patients over systematic and comprehensive measurement to facilitate robust statistical analyses. Given patient de-identification and data sharing to protect confidentiality, the data analyzed only reflected one category change if an individual changed in risk level. Patients who received frequent screenings and remained in the same risk category, regardless of an increase or decrease in score within that category, are not captured in the analysis. Likewise, the use of different, albeit evidence-based, screening tools prevented the use of scores that would have provided a more robust statistical analysis of change. Further, because of the scope of the original project, other key variables were not measured, including whether interventions between screenings occurred or the extent to which training on stigma reduction skewed the results. Future research explicitly focused on evaluating the ideal screening frequency and stigma reduction training is needed. Second, findings related to perceived stigma reduction and improved patient care are based on the qualitative reports of providers and may not reflect the perspectives of the patients who were not interviewed. The extent to which patients perceived stigma reduction following implementation should be evaluated in future research. Finally, this study was conducted using a sample of patients from a specific geographic region of the nation with a rather ethnically homogeneous patient population. Given the historical underrepresentation and oppression of racial, gender, and sexual minorities within research and healthcare settings, future implementation research geared toward implementing and evaluating healthcare improvements for these populations is critically needed.49-51
Conclusion
Screening in adult primary care settings is recommended to reduce substance use. 9 The results from this analysis function as a catalyst to further evaluate recommended screening intervals for detecting unhealthy substance use to increase identification and patient connection to care. This analysis points to potential benefits of frequent screening policies. Evidence-based screening tools, along with policies that implement frequent screening, may further support providers to identify patients at risk of SUD and connect them to appropriate care. Healthcare professionals should consider these findings supporting frequent, universal screening when determining policies on screening frequency for substance use identification.
Footnotes
Acknowledgements
We wish to acknowledge the helpful comments and suggestions provided by members of the University of Pittsburgh, Program Evaluation and Research Unit, located in the School of Pharmacy, including Abigail Cotton, John Miller, and Rahul Amruthapuri. We wish to acknowledge the partnering primary care centers through the PA SBIRT initiative. The research presented in this article is that of the authors and does not reflect the official policy of the Pennsylvania Department of Drug and Alcohol Programs (DDAP) or the Substance Abuse and Mental Health Administration (SAMHSA). We wish to acknowledge the generous funding support from the DDAP and SAMHSA.
Author Contributions
GD: Conceptualization, Investigation, Writing—Original Draft, Project administration. DM: Conceptualization, Methodology, Validation, Formal Analysis, Data Curation, Writing—Original Draft, Visualization. GS: Investigation, Writing—Original Draft. RC: Writing—Review and Editing, Supervision. JP: Methodology, Resources, Writing—Review and Editing, Supervision, Funding acquisition. All authors participated in interpreting the results, contributed to the writing of the article, provided critical feedback to the article, and approved the final article draft for submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Substance Abuse and Mental Health Services Administration [Grant Number 1H79TI026666-01]. The financial support was not involved in the research and preparation of this article.
Compliance,Ethical Standards,and Ethical Approval
Institutional Review Board approval was not required.
