Abstract
Background:
Chiropractors commonly manage cervicogenic headache (CGH) and tension-type headache (TTH) using nonpharmacological interventions such as spinal manipulative therapy. However, dedicated guidelines on chiropractic management of headaches are outdated.
Methods:
We first conducted an umbrella review. A search for systematic reviews and clinical practice guidelines on nonpharmacological interventions for adults with CGH or TTH published from 2017 to August 2023 was conducted. At least two authors independently performed article screening, risk of bias/quality assessment, certainty of evidence, and data extraction. A steering committee developed statements from the synthesized data and seed documents, which were refined by anonymous feedback from a 57-member Delphi panel until reaching at least 80% consensus. The statements were then subjected to public comment, prompting further revisions that were subsequently reviewed by the Delphi panel.
Results:
Thirty-two relevant articles were identified (31 systematic reviews and 1 clinical practice guideline). Statements included recommendations regarding history (e.g., red flags) and examination for CGH/TTH, recommendation to use of spinal manipulation for CGH, and for TTH only within multimodal care. Certainty of evidence and strength of recommendations for other nonpharmacological interventions (e.g., acupuncture and exercise) varied. Limitations in evidence precluded strong recommendations for acupuncture, education, meditation/mindfulness, and modalities used in isolation for CGH and electroacupuncture for TTH.
Conclusions:
This clinical practice guideline created evidence-based consensus recommendations for chiropractic management of adults with CGH and TTH. Chiropractors may appropriately care for individuals with CGH and TTH using a variety of nonpharmacological interventions, while considering best practices in diagnosis, referral, and other aspects of care management.
Background
Tension-type headache (TTH) is a common primary headache disorder with a prevalence of 26% in adults worldwide. 1 Manifesting as a pressing or tightening sensation with referred head pain, TTH affects the muscles around the head and neck. Cervicogenic headache (CGH) is a secondary headache arising from a disorder of the cervical spine, 2 with a variably reported prevalence ranging from 0.2% in individuals aged 30–44 3 to 4.1% in those aged 18–65. 4 CGH arises from cervical structures including facet joints, intervertebral discs, and/or soft tissues, 5 whereas TTH is multifactorial, with potential sources including genetic predisposition and peripheral and central sensitization.6–8
Chiropractors are conservative care clinicians who commonly treat patients with musculoskeletal disorders including spinal pain and headache.9,10 Headache is one of the most common reasons that patients present to chiropractors, occurring in about 6% of their patients.9,10 Furthermore, chiropractors are the most commonly sought first provider for the management of new-onset neck pain. 11 The most frequent treatment chiropractors use for headache is spinal manipulative therapy, defined herein as a high-velocity, low-amplitude thrust applied to the spinal joints. 12 While spinal manipulative therapy accounts for approximately 79% of chiropractor-delivered treatments, chiropractors may use a broad range of other nonpharmacological treatments. 9 These include soft tissue therapy, spinal mobilization, acupuncture, transcutaneous electrical nerve stimulation, therapeutic exercises, nutritional supplements, and patient education.9,13
Although TTH and CGH have different etiologies, neck pain and dysfunction are common in both.14,15 Neck pain affects 92% of those with CGH 16 and 88% of those with TTH. 17 Cervical spine-related symptoms including tenderness and limited range of motion are often present in both CGH and TTH.14,15,18 A key distinction is that neck symptoms in TTH are not intrinsic in causing the headache. 18 Despite being classified differently, with TTH as a primary headache and CGH as a secondary headache arising from cervical spine disorders, both conditions frequently involve neck pain and dysfunction.14,15 Patients often seek chiropractic spinal manipulative therapy to address neck pain and/or headache,9,10 possibly due to its potential to alleviate associated muscle tension or cervical dysfunction. Recent U.S. survey data further highlight this observation, indicating that chiropractors encounter neck pain and headache in 81% and 66% of their patients on a daily basis, respectively. 19 However, the biomechanical or neurophysiological mechanisms of spinal manipulative therapy remain unclear, 20 and the evidence supporting the use of this therapy for TTH and CGH requires an update and re-appraisal.
Nonopioid analgesics (e.g., acetaminophen) and nonsteroidal anti-inflammatory drugs (e.g., ibuprofen) are common first-line treatments for CGH and TTH,6,21 although there is little evidence supporting acetaminophen for pain relief.22,23 Other medications with a greater potential for dependency and medication overuse headache, for example, opioids and butalbital, are also commonly used.16,17,24 Some headache sufferers prefer to avoid pharmacological regimens altogether. 25 According to the 2012 United States National Health Interview Survey, spinal manipulative therapy was one of the most commonly used complementary and integrative health approaches for headaches, being used in 22% of those with headache or migraine. 26 Patients also were reported to use herbs and supplements and engage in movement therapies, among other nonpharmacological interventions. 26
The most recent clinical practice guidelines for chiropractic management of adults with headaches were published in 2011 and 2019,27,28 with the latter representing a broader guideline focusing on nonpharmacological care for headaches associated with neck pain. Both guidelines concluded that spinal manipulative therapy was beneficial for CGH, yet evidence was limited supporting its efficacy for TTH.27,28 In addition, these guidelines did not address the use of dry needling, another treatment used by chiropractors.27,28 Therefore, updated clinical practice guidelines are needed to capture potential growth in the literature while considering a broader range of therapies used by chiropractors for headache.
In addition to therapeutic interventions, effective management of CGH and TTH depends on accurate diagnosis, which is informed by thorough history taking and physical examination. Considering chiropractors are often first-contact providers for patients with neck pain and headache,9–11 they must be able to distinguish between headache types, identify red or orange flags suggestive of serious pathology, and recognize yellow flags that may influence treatment response.29–31 Accordingly, an updated, broad-scope review and guideline addressing these diagnostic and assessment elements may support appropriate decision-making regarding care planning and referral.
For this clinical practice guideline, we define nonpharmacological care as interventions that do not involve the use of medications, injections, or surgeries. This category includes, but is not limited to, manual therapies (such as spinal manipulative therapy, joint mobilization, and soft tissue manipulation), exercise, education, mind–body interventions (e.g., mindfulness, yoga), acupuncture, dry needling, and physical modalities (e.g., electrotherapy, heat, or cold therapy). This definition is informed by a previous review of nonpharmacological therapies for chronic pain 32 and interventions that may be used or considered in chiropractic practice.9,10,33
Although chiropractors are the most frequent providers of spinal manipulative therapy in the United States and use a range of nonpharmacological therapies sought by those with headache, there are no recent, comprehensive clinical practice guidelines addressing the use of these therapies by chiropractors.
The primary aim of this project was to evaluate the effectiveness of nonpharmacological interventions for adults with CGH or TTH and to use this evidence to inform the development of a clinical practice guideline for chiropractors. Specific objectives included: (1) identifying and appraising existing systematic reviews and clinical practice guidelines relevant to chiropractor-delivered care for CGH and TTH; (2) synthesizing evidence related to diagnosis, history taking, physical examination, imaging, care management, and nonpharmacological treatments related to care delivered by chiropractors, and (3) formulating evidence-based recommendations for both clinical decision-making and treatment through a Delphi process. The resulting guideline addresses nonpharmacological chiropractic management of adults with CGH or TTH, including examination, clinical decision-making, and treatment. The intended impact is to support improvements in pain, headache frequency, disability, and quality of life for adults affected by TTH and/or CGH.
Methods
This clinical practice guideline provides evidence-based recommendations for chiropractic management of adults with TTH or CGH using nonpharmacological interventions, covering aspects of diagnosis, examination, imaging, and care management. The target audience is chiropractors, with the goal of improving pain, frequency, disability, and quality of life for patients with TTH or CGH.
We followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses when reporting the review and the Appraisal of Guidelines for Research and Evaluation for the clinical practice guideline (Supplementary Data S2, Supplementary Table S1). 34 Checklists detailing our adherence to these frameworks appear in Supplementary Data S4 and Supplementary Data S5. We adhered to a registered protocol (International Prospective Register of Systematic Reviews #CRD42023461883).
The Institutional Review Board of Texas Chiropractic College approved the project prior to the Delphi panel formation. Delphi panelists signed a consent form affirming their participation was voluntary and without compensation.
Steering committee
The steering committee performed the main project functions of identifying, assessing, and synthesizing the evidence; developing evidence-based recommendations; and integrating the Delphi panel and public comments into the recommendations. The steering was composed of 12 members: 10 doctors of chiropractic, 1 medical physician, a neurologist specializing in headaches, and 1 health sciences librarian. Their mean years of professional experience were 20 (min–max: 5–38). Four were health professions faculty (three chiropractic and one medical), one was a postdoctoral trainee as well as practicing doctor of chiropractic, and the rest were in full-time practice, four of which were interdisciplinary practices. All have been involved in research as investigators, and nine had previously participated in developing clinical practice guidelines.
Seed documents
The steering committee identified documents providing background information regarding the current evidence for interventions, especially nonpharmacological interventions for CGH and TTH. These also aided in developing the literature search parameters and clinical practice guideline structure.27,28,32,35–38
Literature search
To provide a foundation for clinical practice guideline development, we performed a comprehensive literature search. Our research question was: Which nonpharmacological interventions for adults with CGH or TTH are effective? We focused on nonpharmacological interventions because the chiropractic scope of practice does not typically include medications or surgery. In addition, the steering committee was provided with lists of included and excluded articles and was asked to contribute additional references.
To answer the research question, we formulated elements defining the population, interventions, comparisons, and outcomes as follows:
Population: Adults (age ≥18) with CGH or TTH. Interventions: Nonpharmacological interventions (e.g., spinal manipulative therapy, manual therapy and soft tissue manipulation, education, exercise, mind–body interventions, acupuncture, dry needling, and modalities such as electrotherapy, heat, or cold). Comparisons: Any comparison, no restrictions. Outcomes: Improvement in headache outcomes, including pain intensity, frequency and duration, function, and quality of life.
Our search strategy was developed by a research librarian (SW) alongside several coauthors (CH, DT, KA, and RT). We conducted initial test searches in PubMed, refining our query by adding and removing terms. The librarian translated our research question into search concepts, using filters for date, language, and publication type reflecting the above-stated elements and eligibility criteria. The librarian then adapted the PubMed query for other databases, including the Cochrane Database of Systematic Reviews and Cumulative Index of Nursing and Allied Health Literature, with all searches conducted in August 2023. The complete search strategies are available in Supplementary Data S1. Additional articles were sought via reference tracking and consulting experts in the field.
In addition to the primary literature search focused on nonpharmacological treatments, the steering committee conducted targeted searches to support sections of the guideline related to history taking, physical examination, and clinical decision-making. These searches and corresponding article selection relied upon section-specific eligibility criteria without limitations by study type, corresponding to their subject focus in the guideline rather than requiring solely systematic reviews or clinical practice guidelines of randomized controlled trials. These searches included queries of PubMed and Google Scholar, citation tracking of seed documents,27,28,32,35–38 and the inclusion of key references suggested by the steering committee based on previous clinical, guideline, and research experience. Further targeted searches were conducted in response to the Delphi process and public comments.
Eligibility criteria
Inclusion criteria
Published in peer-reviewed journal from 1/01/2017 through 8/24/2023. This start date precedes the end dates two seed document searches; a systematic review covering nonpharmacological treatments for chronic TTH 32 and clinical practice guideline. 28
English language.
Study population comprised of adults treated for CGH or TTH.
Study intervention(s) included at least one nonpharmacological therapy.
Publication types including clinical practice guidelines and systematic reviews and meta-analyses of randomized controlled trials, or a combination of randomized and nonrandomized studies of interventions.
Exclusion criteria
Reviews, commentaries, abstracts from conference proceedings, theses, cross-sectional descriptive surveys, observational studies, and gray literature. These were excluded due to being considered lower quality evidence.
Articles addressing only migraine or other headache types and not CGH or TTH.
Articles only addressing acute or post-traumatic headache.
Articles that did not include nonpharmacological interventions.
Articles not providing separate analyses of CGH or TTH.
Screening
Search results were screened in duplicate (C.H. and R.J.T.), first by title/abstract, and then by full text. Disagreements regarding eligibility were resolved by discussion.
Evaluation of article quality
We evaluated the quality of articles meeting inclusion criteria. We used the Appraisal of Guidelines for Research and Evaluation instrument Global Rating Scale to assess clinical practice guidelines 34 and the Scottish Intercollegiate Guideline Network checklist for systematic reviews and meta-analyses (Supplementary Data S2, Tables S1 and S2, respectively). 39 At least two investigators rated each study and discussed differences in ratings until they reached agreement. Scottish Intercollegiate Guidelines Network scores were graded using threshold of high quality, low risk of bias (score of 10–12; majority of “yes” responses), acceptable quality, low risk of bias (7–9), low quality, high risk of bias (4–6), or unacceptable/reject (0–3). In this model, no items were assigned a higher weight than others. Appraisal of Guidelines for Research and Evaluation followed a similar threshold-driven approach with the maximum scores of 6 and 7 based on the main component items, with a determination that allowed raters to weigh the various factors.
Evaluation of evidence certainty
To evaluate the overall level of evidence for interventions, we adapted the Grading of Recommendations Assessment, Development, and Evaluation approach to account for the synthesis of systematic reviews rather than individual randomized controlled trials (Supplementary Data S2, Supplementary Table S3).40–42 Initial determinants of our initial certainty ratings were article quality ratings from the Scottish Intercollegiate Guideline Network ratings (which incorporate risk of bias and publication bias) and Appraisal of Guidelines for Research and Evaluation ratings, and the number and consistency of studies contributing to each rating for each intervention. We secondarily considered Grading of Recommendations Assessment, Development, and Evaluation domains of imprecision, inconsistency, and indirectness to determine if additional downgrading was necessary. Each rating was derived de novo, rather than relying on the cited study authors’ own ratings. Finally, we incorporated article quality/risk-of-bias assessments of randomized controlled trials as provided within our synthesized articles, rather than replicating these de novo, which would have been impractical for a large number of studies and cited trials. These quality scores were pragmatically categorized as high, acceptable, low, or very low. When available, pooled estimates from meta-analyses (versus narrative syntheses) and recent publications (versus older) were prioritized when deriving evidence certainty scores per condition and outcome, which enabled better assessments of precision and more updated data, respectively. At least two investigators independently performed evidence certainty assessments, with discrepancies resolved via discussion (performed by C.H., R.J.T., and C.J.D.).
After the quality assessment was completed, the steering committee was provided with all included articles and quality assessments.
Data extraction
Tables were constructed with the key characteristics of included study. Extracted items were determined a priori, including publication date, number of studies included, article quality, total number of patients, interventions, and summary of outcomes. To determine treatment effectiveness, data extractors considered effect estimates and measures of precision and statistical significance from synthesized studies, reviewing forest plots and subgroup analyses when available and appropriate. We avoided relying on authors’ broader conclusions that may have been oversimplified or not directly applicable to our research questions. One investigator (C.H.) extracted data and at least one other investigator (R.J.T., M.R.P., D.N.T., and C.J.D.) verified it, discussing disagreements until they were resolved. Completed data tables were provided to the steering committee to consider when making the clinical practice guideline recommendations.
Development of seed statements
The steering committee first agreed on domains to be covered in the clinical practice guideline, deciding the structure would follow the order of the clinical encounter. The initial proposed structure was adapted from a prior chiropractic clinical practice guidelines.35,43,44 Other seed documents played a role in helping structure material related to diagnosis, clinical decision-making, and red/orange flags including The International Classification of Headache Disorders, 3rd edition, 36 an overview of clinical practice guidelines for musculoskeletal pain, 37 and a primary care resource from the European Headache Federation and Global Campaign against Headache. 38 Steering committee members then developed statements addressing key aspects within each domain. Domains for both CGH and TTH included informed consent; history, physical examination and special tests; diagnosis; patient-reported outcome measures; frequency and duration of treatment; follow-up/re-evaluation; and concurrent care and co-management and/or referral. Interventions were addressed separately for each type of headache. Steering committee members drafted and revised content for specific domains iteratively, reaching consensus over several rounds of internal review. Each set of statements included supporting citations, with a reference list accessible to Delphi panelists during the consensus process to facilitate evidence-informed ratings.
Delphi consensus panel
We identified a list of possible candidates to form a panel of Doctors of Chiropractic and other health professionals with experience in managing headaches and who also valued scientific evidence. We estimated a panel of approximately 50–60 members would provide an appropriate range of viewpoints, based on our previous Delphi projects. We aimed for the majority to be doctors of chiropractic, given the goal of developing profession-specific recommendations. However, we also sought to include medical physicians, particularly headache specialists. We attempted to ensure geographical and ethnic diversity among the panelists. We identified candidates by (1) consulting lists of previous Delphi panelists for chiropractic clinical practice guidelines35,43–45 and (2) inviting nominees and volunteers among the Clinical Compass board, which includes representatives of ChiroCongress, the American Chiropractic Association, the American Black Chiropractic Association, and the Association of Chiropractic Colleges. We limited panelist selection to practitioners based in the United States, as clinical recommendations may vary due to differences in international regulatory environments. We asked volunteers and nominees to submit a form describing their practice characteristics and their curriculum vitae. These materials were reviewed by the steering committee to guide panelist selection. We aimed to include practitioners with a range of years in practice to capture a diversity of perspectives. Selection prioritized individuals with previous experience in guideline development, additional academic degrees or certifications (e.g., diplomate status), interdisciplinary practice experience, and/or involvement in research or professional education. Final panelist selection was based on group discussion and consensus among the steering committee.
Methodology of the modified Delphi process
In attempt to reduce bias, panelists were anonymous during the consensus process, being identified by a number only, which only the project director (C.H.) and project coordinator (CE) knew. This was done because all raters’ comments were shared among the SC and the Delphi panelists. We used an established systematic and quantitative methodology of incorporating evidence and expert opinion to reach group consensus. 46 Panelists used a 9-point ordinal Likert “appropriateness” rating scale for each seed statement. “Appropriate” means that the expected patient health benefits exceed expected negative effects by a large enough margin that the recommended action is worthwhile, without considering costs. 46 The scale is anchored by “highly inappropriate” (1) and “highly appropriate (9), with “uncertain” placed over the middle of the scale. Unlimited space was provided for comments after every statement. We asked that panelists provide supporting citations when disagreeing with recommendations. In addition to the reference list they received accompanying the rating statements, we offered to email panelists a bibliographic file of all citations to aid in their deliberations.
Data management and analysis
The project coordinator entered rating scores into a Statistical Product and Service Solutions (version 25, IBM Corp., Armonk, NY, US) database. Medians and percentages of agreement were calculated for each statement. To be congruent with the methodology, we required an a priori of at least 80% agreement with a median rating of at least 7 to be considered consensus.46,47 The project coordinator pasted panelists’ comments into a Microsoft Word® table, organized by panelist identification number, statement number, and rating. The steering committee examined the ratings and their associated deidentified comments. Balancing the comments with all supporting evidence, the steering committee revised statements that did not reach 80% agreement. Revised statements were returned to the Delphi panel, with their deidentified comments, for another round of rating.
External review: public comments
Multiple methods of dissemination are recommended by organizations such as Appraisal of Guidelines, Research and Evaluation for ensuring stakeholder involvement in clinical practice guideline development.
34
We included stakeholders from the inception of the project by representing chiropractic organizations on the steering committee and Delphi panel. These included representatives of US’ state chiropractic associations, faculty from health professions institutions, practitioners of other health professions such as medicine, nursing, physical therapy, or massage therapy, and interested laypeople such as vendors. After achieving consensus on the recommendations in the modified Delphi process, we solicited public comments on the draft clinical practice guideline, using several methods:
A MailChimp (Atlanta, GA, US) email blast to the Clinical Compass contact list, which included the Clinical Compass Board (including United States’ state and national chiropractic organizations, academic institutions, vendors, and interested laypersons [∼900 individuals]). Direct invitations through the chiropractic organization ChiroCongress to its member associations (∼35,000 chiropractors). Postings on Facebook (Meta Platforms, Inc., CA, US) and LinkedIn (Microsoft, WA, US) via the Clinical Compass page, which is available to health professionals and interested laypersons. Social media posts were publicly visible and actively shared to a broader audience. Notification to the Chiropractic Summit, a national organization of various chiropractic groups and interested individuals.
Using multiple methods with some overlap served to reinforce the message, as did a follow-up mailing 2 weeks later. The comment period was 30 days.
The Clinical Compass website (https://clinicalcompass.org/) provided links to several portable document files: a request for input; summary of methods; and the draft recommendations accompanied by the reference list. We also posted a fillable comment form to facilitate response. The project coordinator collected the comments and entered them into a Microsoft Word® table. The steering committee read all comments and considered revisions as appropriate. If a comment resulted in requiring a substantive change, the revised statements would be recirculated to the Delphi panel to reach consensus.
Results
Literature search
The search yielded 204 citations; 1 additional citation was added from expert consultation and reference tracking, for a total of 205 (see Fig. 1 for study selection flowchart). After removing duplicates, 179 citations remained. A total of 137 citations were excluded; reasons for exclusion are categorized in Figure 1 and detailed in Supplementary Data S3. This left 32 articles to be included in the evidence synthesis: 3132,48–77 were systematic reviews/meta-analyses and 1 was a clinical practice guideline (Table 1). 28
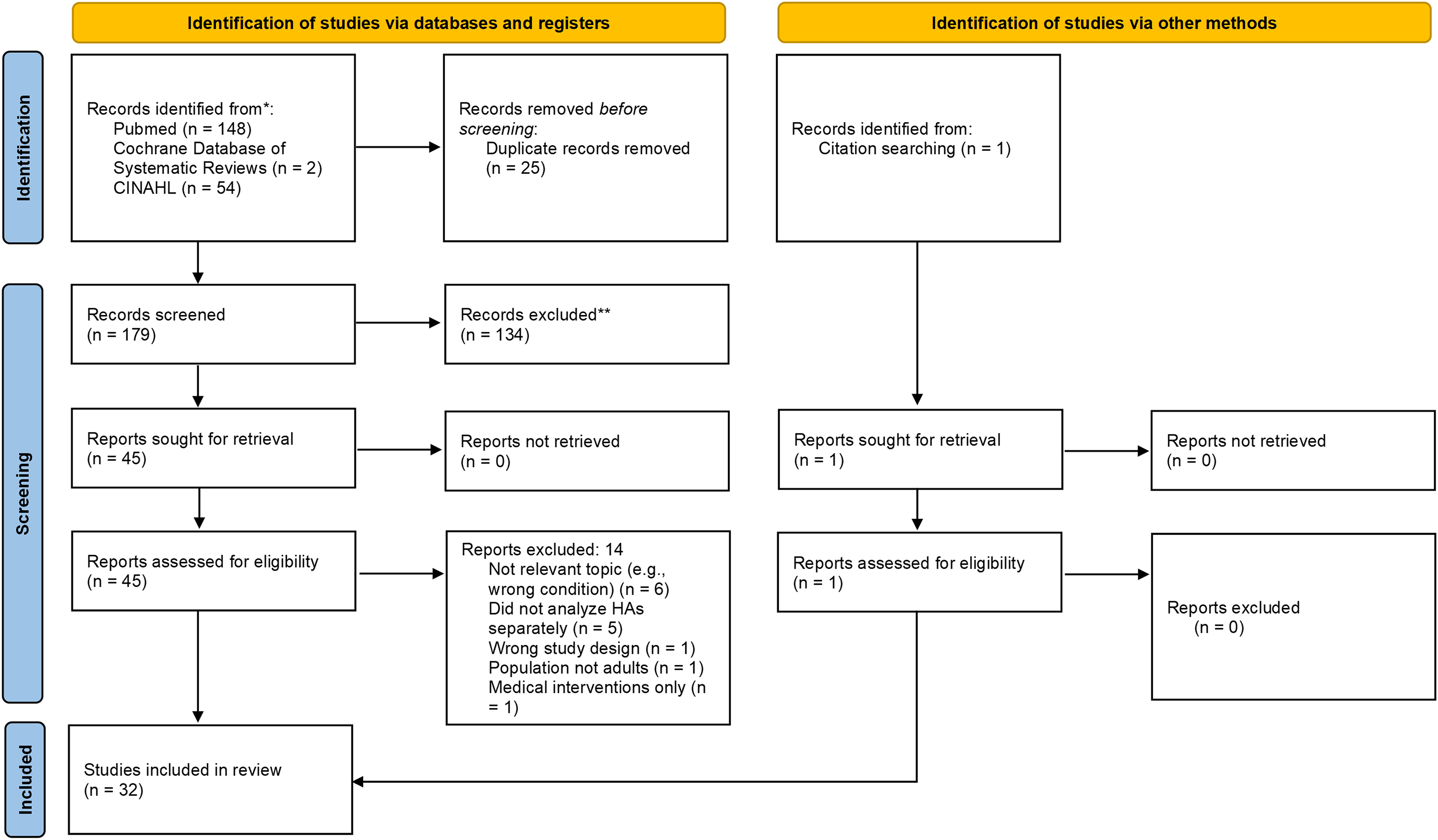
Article selection flow diagram (Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020). CINAHL, Cumulative Index to Nursing and Allied Health Literature; HA, headache.
Included Studies by Headache Type and Study Design
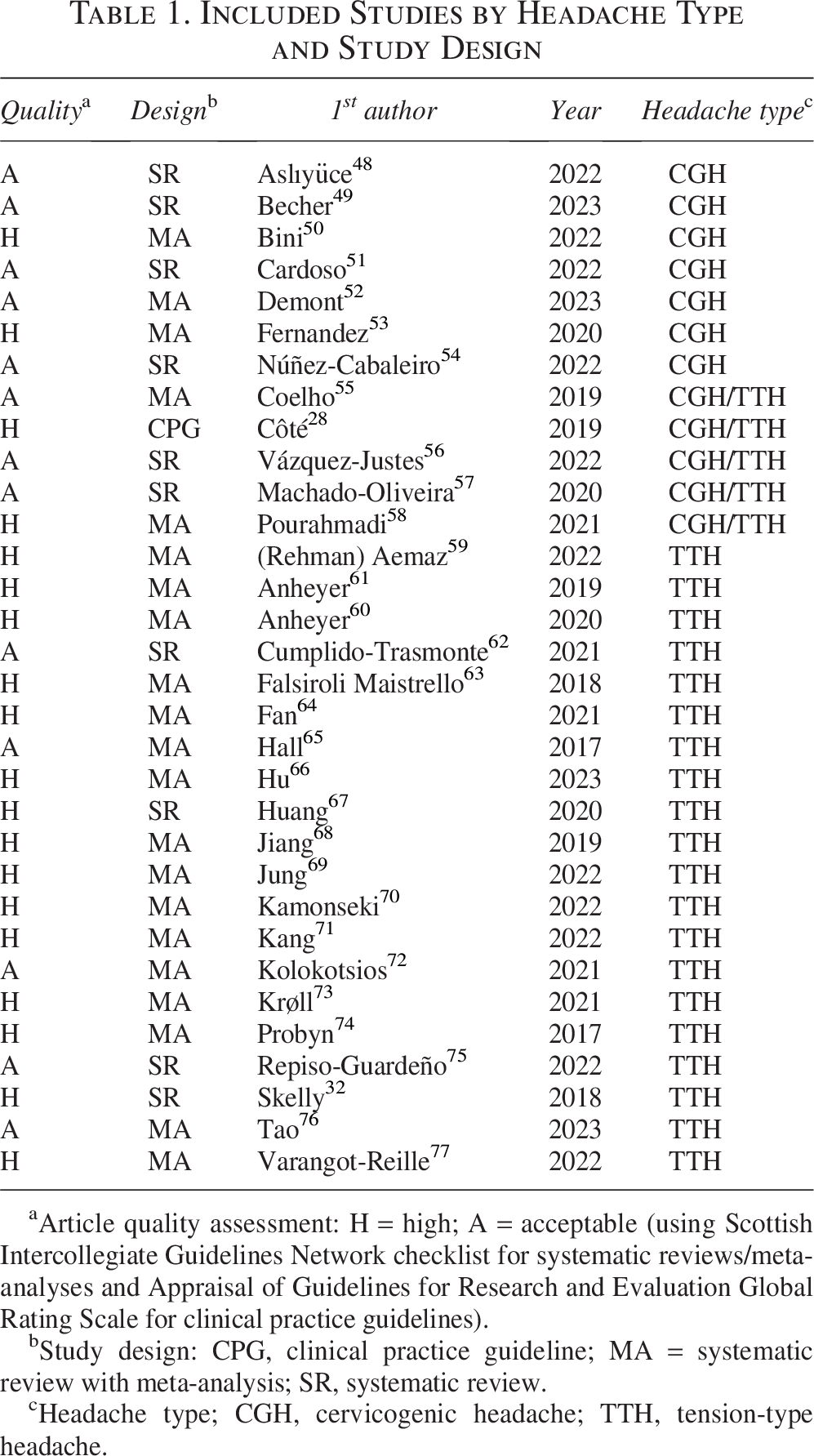
Article quality assessment: H = high; A = acceptable (using Scottish Intercollegiate Guidelines Network checklist for systematic reviews/meta-analyses and Appraisal of Guidelines for Research and Evaluation Global Rating Scale for clinical practice guidelines).
Study design: CPG, clinical practice guideline; MA = systematic review with meta-analysis; SR, systematic review.
Headache type; CGH, cervicogenic headache; TTH, tension-type headache.
Article quality assessment
All included studies were considered “high” or “acceptable” quality and thus were retained in the analysis (Table 1).
Data extraction
Tables 2 and 3 provide the results of the data abstraction for systematic reviews/meta-analyses of CGH and TTH, respectively. Table 4 summarizes the one included clinical practice guideline. Table 5 provides evidence certainty ratings. Data extracted in our umbrella review synthesis highlighted a greater quantity of literature regarding nonpharmacological interventions for TTH compared with CGH. A single clinical practice guideline provided pertinent recommendations, 28 which were considered alongside our updated evidence synthesis when drafting clinical practice guideline recommendations. The certainty of evidence supporting individual treatments varied from “high” to “very low.” We did not downgrade for indirectness, considering the study outcomes aligned with our eligibility framework. We did downgrade when article quality scores indicated risk of bias or publication bias, imprecision with respect to study effect estimates, and inconsistency when study findings varied. The information derived from this synthesis is best interpreted within the context of the guideline recommendations presented below, which are based on included articles’ quality, certainty of evidence, and expert and public feedback.
Systematic Reviews/Meta-Analyses on Cervicogenic Headache.a
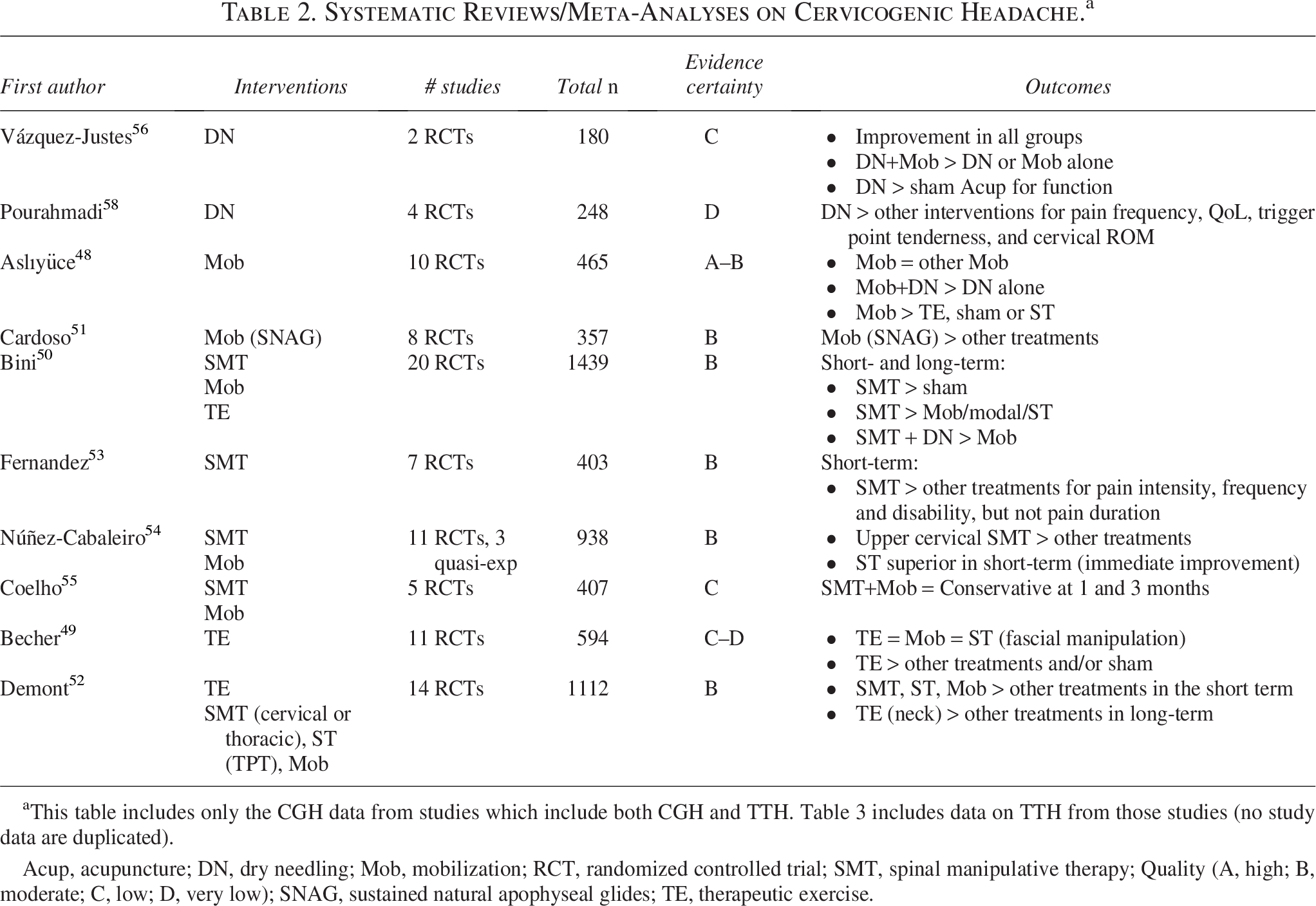
This table includes only the CGH data from studies which include both CGH and TTH. Table 3 includes data on TTH from those studies (no study data are duplicated).
Acup, acupuncture; DN, dry needling; Mob, mobilization; RCT, randomized controlled trial; SMT, spinal manipulative therapy; Quality (A, high; B, moderate; C, low; D, very low); SNAG, sustained natural apophyseal glides; TE, therapeutic exercise.
Systematic Reviews/Meta-Analyses on Tension-Type Headache
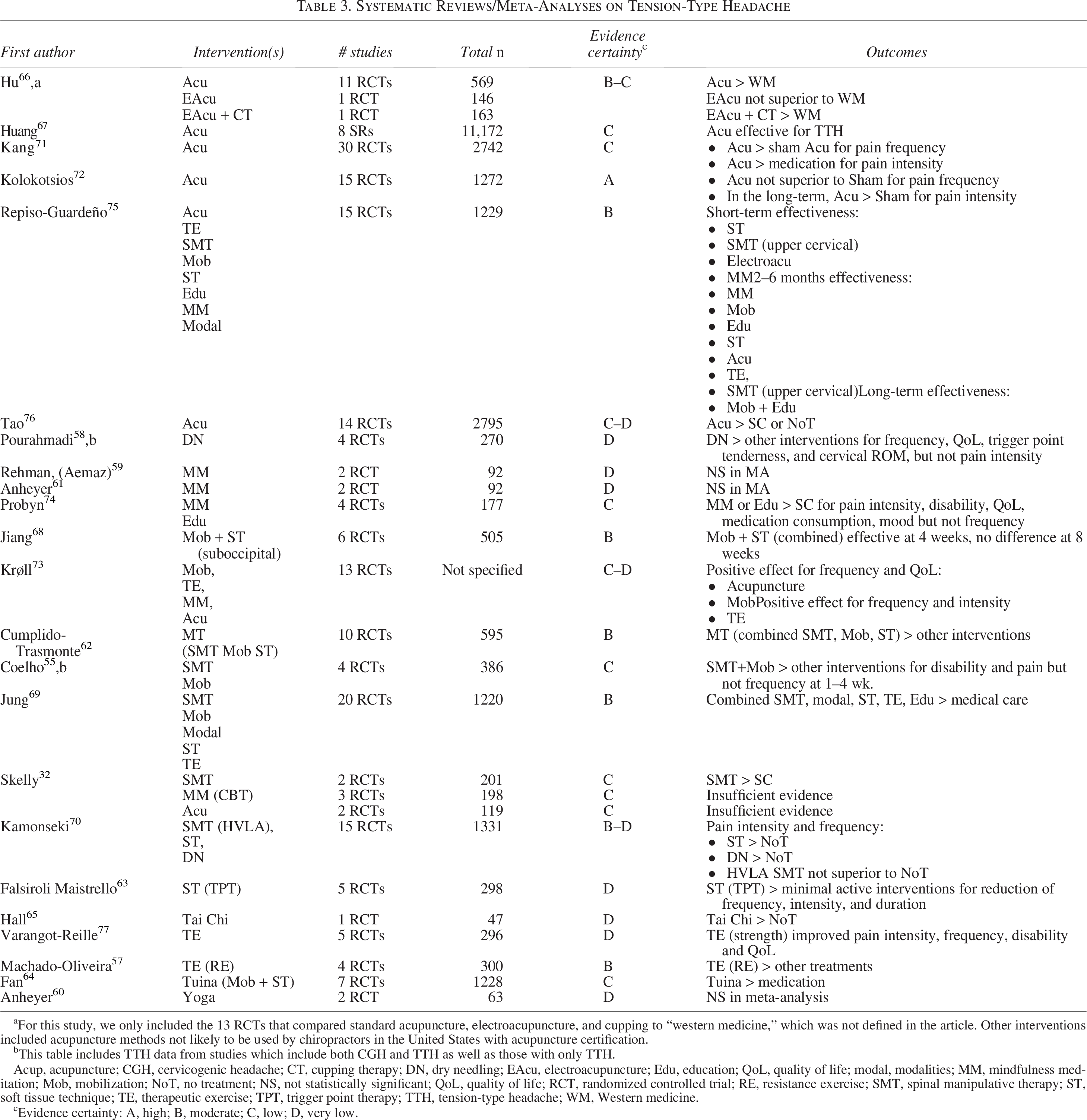
For this study, we only included the 13 RCTs that compared standard acupuncture, electroacupuncture, and cupping to “western medicine,” which was not defined in the article. Other interventions included acupuncture methods not likely to be used by chiropractors in the United States with acupuncture certification.
This table includes TTH data from studies which include both CGH and TTH as well as those with only TTH.
Acup, acupuncture; CGH, cervicogenic headache; CT, cupping therapy; DN, dry needling; EAcu, electroacupuncture; Edu, education; QoL, quality of life; modal, modalities; MM, mindfulness meditation; Mob, mobilization; NoT, no treatment; NS, not statistically significant; QoL, quality of life; RCT, randomized controlled trial; RE, resistance exercise; SMT, spinal manipulative therapy; ST, soft tissue technique; TE, therapeutic exercise; TPT, trigger point therapy; TTH, tension-type headache; WM, Western medicine.
cEvidence certainty: A, high; B, moderate; C, low; D, very low.
Prior Clinical Practice Guideline Recommendations for Treating Cervicogenic and Tension-Type Headache 28

Edu, education; Mob, mobilization; NA, not applicable; RCT, randomized controlled trial; RoB, risk of bias; SMT, spinal manipulative therapy; ST, soft tissue; TE, therapeutic exercise; TTH, tension-type headache.
Summary of Findings for Interventions for Cervicogenic and Tension-Type Headache

We were unable to derive an evidence certainty or recommendation for the following intervention-condition pairs as no available evidence was identified: acupuncture, electroacupuncture, education, mindfulness medication or psychological self-care, modalities, and Tuina for cervicogenic headache (CGH); education for tension-type headache (TTH). Multimodal therapies are defined as any combination of spinal manipulative therapy, joint mobilization, soft tissue manipulation, and modalities. The certainty of evidence ranges from A (highest) to D (lowest) as defined in Supplementary file 2 Supplementary Table S3: A, high; B, medium; C, low; D, very low. RCT, randomized controlled trial; SR, systematic review.
Modified Delphi consensus process
Of 73 individuals invited, 58 accepted (80%). One panelist did not respond after accepting, thereby yielding 57 final panelists. Of the 57, 43 (75%) identified as male and 49 (86%) as white. There were two who identified as Black/African American (5%); two as Hispanic (4%); and one each as Asian (2%), Native American (2%), and multiracial (2%). Fifty-five were doctors of chiropractic (96%); there was one medical physician (Family Medicine; 2%) and one pharmacist (2%). Seven doctors of chiropractic were cross-trained (12%): seven in acupuncture (11%) and one in nursing (Bachelor of Science in Nursing; 2%). Eight of the doctors of chiropractic had academic master’s degrees (Master of Arts, Master of Public Health, or Master of Science; 14%). Nineteen (33%) of the panelists are employed by a Veterans Administration facility. One panelist was a faculty member at a chiropractic college (2%), and two were faculty at nonchiropractic institutions (4%). Fifty-two were practitioners (91%) with a mean time in practice of 24 years (min–max: 1–45). The 52 practitioners reported a mean of 84 patient visits per week (min–max: 6–650). The mean estimated proportion of their patients with a chief complaint of headache was 17% (min–max: 2–80%). Panelists’ locations represented 28 US’ states as follows: five from New York (9%); four each from Arizona, Florida, and Wisconsin (7% each); three each from California, Kansas, New Hampshire, Oregon, Texas, and Washington (5% each); two each from Iowa, Missouri, Pennsylvania, and South Dakota (4% each); and one each from Colorado, Georgia, Hawaii, Illinois, Kentucky, Maryland, Michigan, Minnesota, Mississippi, Montana, New Jersey, Ohio, Rhode Island, and Tennessee (2% each).
On Delphi Round 1, the panel reached consensus (>80% agreement) on 21 of the 31 recommendations (68%). Comments indicated the primary reason for disagreement was a need for greater clarity of the recommendations for interventions, rather than disagreement with the statements’ evidence base. The steering committee clarified the statements after reviewing the panelists’ comments. Agreement was reached for all but one statement in Round 2. Round 3 consisted of a revision of that statement, clarifying the recommendation concerning the appropriateness of dry needling for CGH; the panelists then reached consensus.
Public comments
Five comments were submitted by four individuals (all doctors of chiropractic) during the 28-day comment period. The comments were deidentified and sent to the steering committee, who carefully considered each comment. One commenter requested a greater level of detail in the outcome measures recommendations to comply with Centers for Medicare and Medicaid Services regulations; the steering committee agreed this was not within the purview of making evidence-based recommendations. Another commenter requested consideration of additional conditions and medications as red flags; however, the steering committee agreed that these additions would be redundant with the history taking statements. The third and fourth commenters were concerned with contraindications and precautions regarding transcutaneous electrical nerve stimulation and high-velocity, low-amplitude spinal manipulative therapy for complicated or late pregnancy (Table 6) and provided several references. The steering committee considered their citations, conducting targeted literature searches on these topics. They concluded that the language used for contraindications/precautions was ambiguous, found more clearly delineated definitions, and changed five of the recommendations from “possible contraindication” to “precaution.” This necessitated an additional Delphi round to reach consensus. The Delphi panel reached at least 80% consensus on four of the statements, with a response rate of 95% (54 of 57 panelists). They did not reach agreement on the fifth revised statement (on transcutaneous electrical nerve stimulation), so it was retained in its original form.
Precautions and Contraindications for Nonpharmacological Interventions
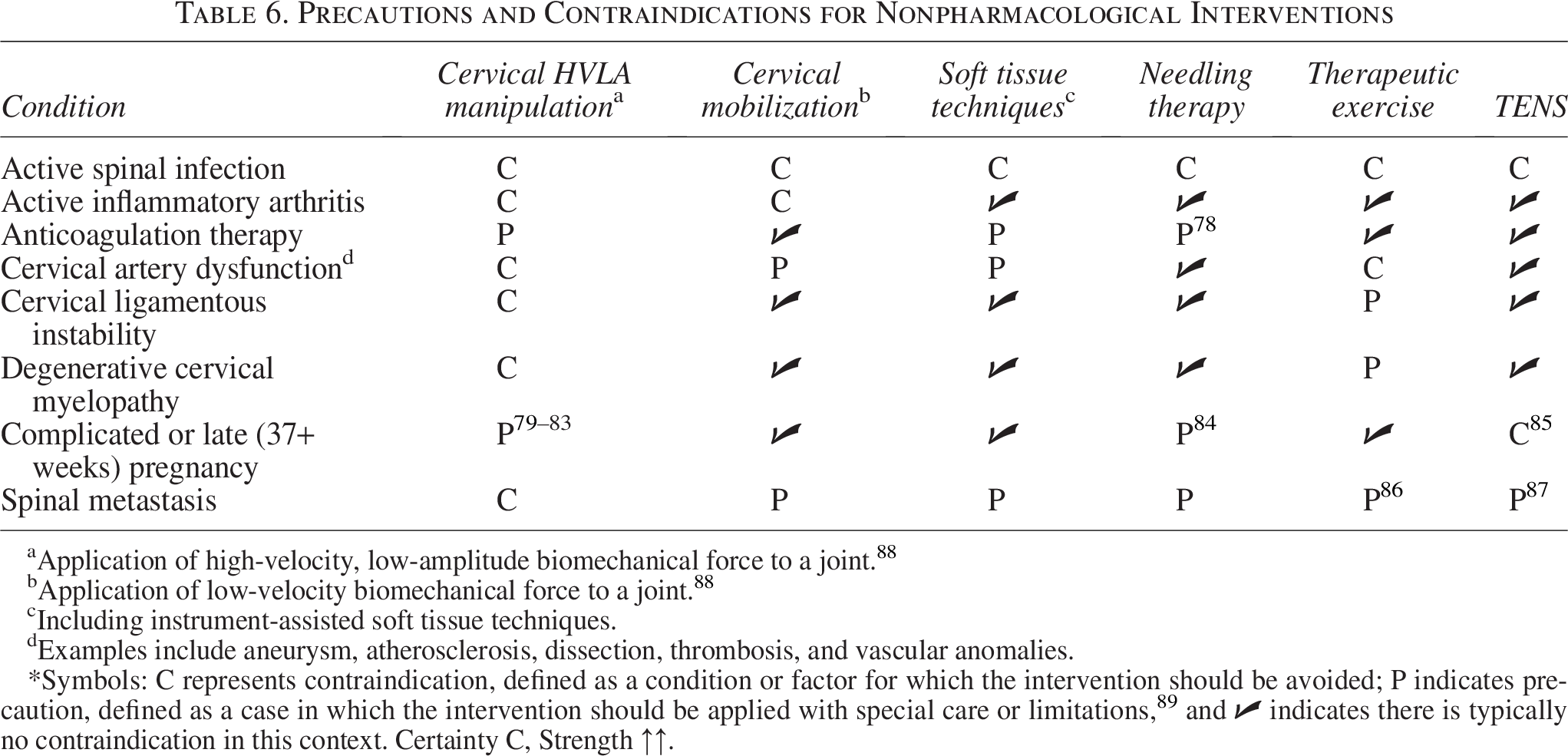
Application of high-velocity, low-amplitude biomechanical force to a joint. 88
Application of low-velocity biomechanical force to a joint. 88
Including instrument-assisted soft tissue techniques.
Examples include aneurysm, atherosclerosis, dissection, thrombosis, and vascular anomalies.
*Symbols: C represents contraindication, defined as a condition or factor for which the intervention should be avoided; P indicates precaution, defined as a case in which the intervention should be applied with special care or limitations,
89
and
Evidence-based recommendations reaching consensus
Following each statement is the rating for certainty of evidence, as described in Supplementary Data S2, Supplementary Table S3, and strength of recommendation (strength) as strong ↑↑ or weak ↑. 40 Below are the finalized recommendations in the clinical practice guideline.
Informed consent/risks and benefits
IC1
Informed consent is a process involving direct communication between the clinician and patient. Explain all procedures, including examination, diagnosis and treatment options (including no treatment) in terms the patient understands. Explain both benefits and risks. 44 Ask the patient if they have any questions; answer them and affirm that the patient understands, in order to inform shared decision-making. 44 Record the discussion and patient’s consent in the medical record. Certainty D, strength ↑↑.
IC2
Adhere to local/regional legal requirements. If necessary, seek advice on compliance from the state licensing board. Additional guidance is available from the American Chiropractic Association and the Association of Chiropractic Colleges. Certainty D, strength ↑↑.
History
H1
Record a history that includes current symptoms, health status, previous and concurrent treatment, and psychosocial factors. Components should include:29,38 Certainty B, strength ↑↑.
Assessment of red/orange flags29–31
(Supplementary Data S2, Supplementary Table S4) and possible contraindications for nonpharmacological interventions, with particular emphasis on high-velocity, low-amplitude spinal manipulation79–85,87,89 (Table 6) Assessment of yellow flags (i.e., psychosocial factors that may influence the treatment response)
31
History of significant and/or recent physical trauma Headache onset, characteristics, and precipitating factors Pain type, severity, location, frequency, and duration Provocative and relieving factors Previous interventions and response Current and previous self-care strategies Diagnostic tests and results Current medications, both prescription and nonprescription, particularly those for HA Current nutraceuticals and supplements Comorbidities, family history Lifestyle factors such as diet, sleep quality, physical activity, alcohol, tobacco, and recreational drug use.
Diagnostic considerations
D1
Establish a working diagnosis of CGH or TTH from the history and examination. 36 Certainty A, Strength ↑↑.
Definitions
The following material is based on diagnostic criteria from The International Classification of Headache Disorders, 3rd edition: 36
TTH is classified as a primary headache with palpable pericranial tenderness, typically bilateral. It is characterized as episodic (infrequent or frequent) or chronic. 36 Infrequent episodic TTH occurs less than once a month and is not addressed in this guideline. Frequent episodic TTH is characterized by at least 10 episodes occurring on 1–14 days/month on average for >3 months and does not exhibit the additional symptoms characteristic of migraine. Chronic TTH is similar to frequent episodic except that it occurs more than 15 days/month for at least 3 months. 36
CGH is classified as a secondary headache caused by a disorder of the cervical spine and its related structures (bone, disc, and/or soft tissue), usually but not always associated with neck pain. When cervical myofascial pain is the cause, the headache is more appropriately classified as TTH. 36
Diagnosis of CGH
D2
Diagnosis of CGH relies on clinical and/or imaging evidence of a cervical spine disorder, reasonable exclusion of other headache disorders, and any one of the following:
36
Certainty B, strength ↑↑
Temporal relation to cervical disorder Symptoms correlate with cervical disorder (e.g., symptoms improve/resolve in parallel with improvement or resolution of the cervical disorder) Reduced cervical range of motion or exacerbation of headache with provocative maneuvers Headache alleviated after diagnostic block.
Diagnosis of TTH
D3
TTH is diagnosed based on recurrent episodes of typically bilateral, pressing, or tightening headaches lasting from minutes to days, which are mild to moderate in intensity, and examination findings of pericranial tenderness (i.e., frontal, temporal, masseter, pterygoid, sternocleidomastoid, splenius, and trapezius muscles). 36 TTH is not aggravated by routine physical activities and never includes moderate or severe nausea or vomiting, yet it may include one of photophobia, phonophobia, or mild nausea. Other headaches should be ruled out although TTH may overlap with migraine features. Certainty B, strength ↑↑.
D4
There may be overlap or difficulty making distinctions between TTH and migraine. Certainty B, strength ↑↑.
Examination
E1
For suspected TTH or CGH, conduct a physical examination focusing on musculoskeletal and neurological components related to the neck and head, including an assessment of pain and/or tenderness upon palpation of the neck, pericranial region, and appropriate provocative testing for the cervical spine when indicated.28,90 Certainty A, strength ↑↑.
Diagnostic imaging
DI1
Routine diagnostic imaging is not recommended for patients with already-diagnosed cervicogenic or TTH.28,29,38,90,91 Certainty A, strength ↑↑.
Factors which indicate the need for imaging are:
Red flags on history or physical exam Severe and/or progressive neurological deficits No improvement after a reasonable course (4–6 weeks) of care.
General management considerations
See Figure 2 for general care pathway.
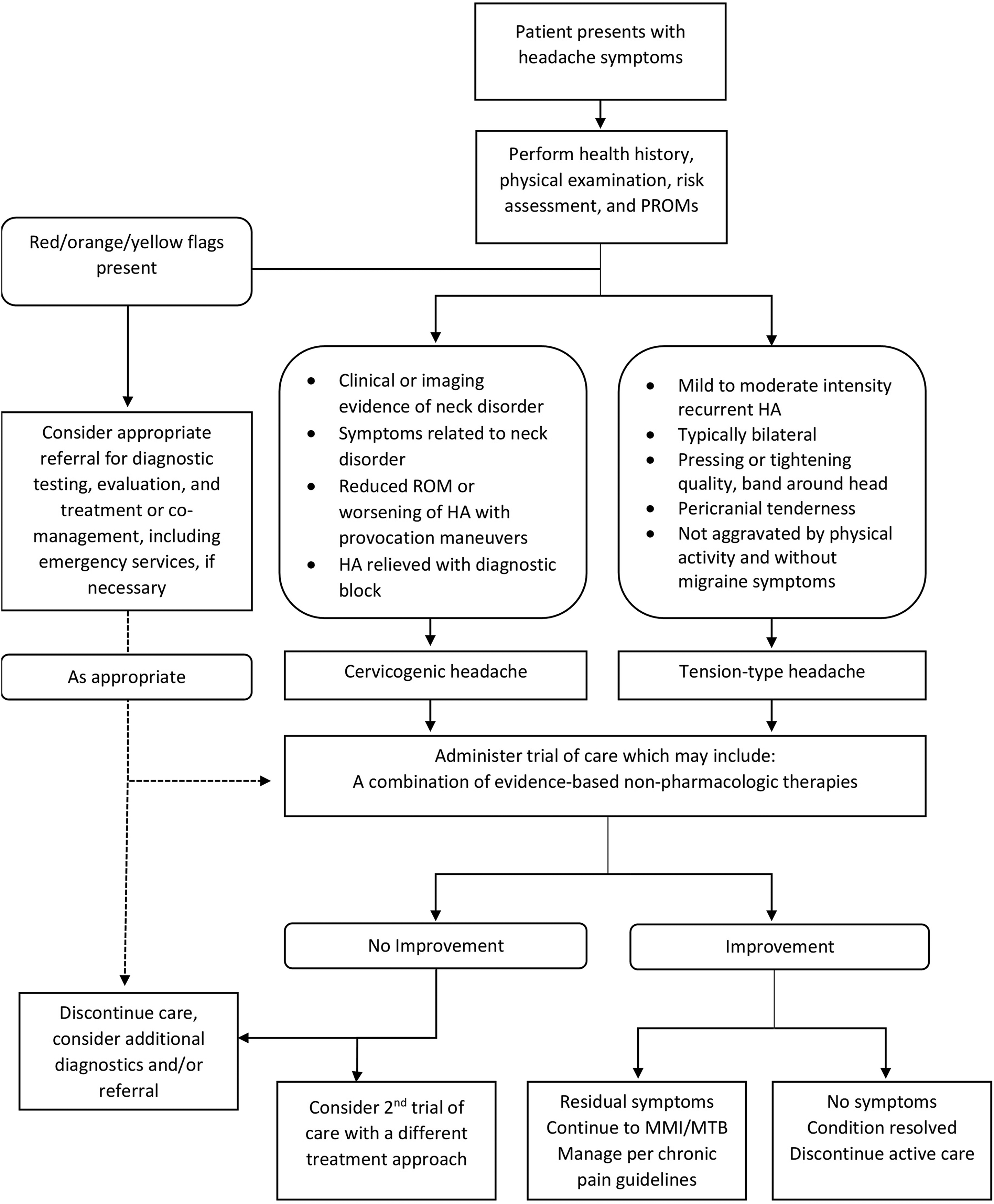
Care pathway for patients with cervicogenic or tension-type headache. HA, headache; MMI/MTB, maximum medical improvement or maximal treatment benefit; PROMs, passive ranges of motion; ROM, range of motion.
G1
For patients with contraindications, or who do not adequately respond to an initial course of approximately 4 weeks of conservative care, consider referral to and/or co-management with appropriate pharmacological providers or medical specialists for further evaluation and/or management of TTH/CGH. 38 Certainty B, strength ↑↑.
G2
Select intervention strategies that are congruent with patient preference, are without contraindications, and emphasize patient-centered care.28,37,90 Certainty B, strength ↑↑.
Outcome assessment
O1
Consider using patient-reported outcome measures to assess patient pain and function, and to assess progress over time. Certainty B, strength ↑↑.
Considerations for frequency and duration of treatment
F1
Administer any of the recommended interventions 1–2× per week for 4 weeks to 3 months, depending on clinical context (e.g., headache frequency/intensity), response to a trial of care, and patient preference (during or between headache episodes).27,28,54,69 Certainty C, strength ↑↑.
F2
Continue the course of care that best manages the patient’s headache pain intensity, frequency, and duration. Certainty C, strength ↑↑.
F3
Administer passive ranges of motion at endpoint of trial of care to assess response. Certainty B, strength ↑↑.
CGH interventions
CGH1
Offer spinal manipulation.28,50,52–55 Certainty A, strength ↑↑.
CGH2
Offer multimodal therapy that includes spinal manipulation and any combination of joint mobilization, soft tissue, and/or modalities. 28 Certainty B, strength ↑.
CGH3
Consider offering or referring for dry needling when appropriate (e.g., to address suspected trigger points of the cervical spinal musculature).56,58 Certainty B, strength ↑.
CGH4
Provide therapeutic exercises in a multimodal program (e.g., stretching, strengthening, endurance, and/or postural correction) and/or select individual exercises guided by patient preference.28,49,50,52 Certainty B, strength ↑.
TTH interventions
TTH1
Provide spinal manipulation combined with joint mobilization, soft tissue techniques, and/or modalities. 32 Certainty B, strength ↑.
TTH2
Offer joint mobilization and/or soft tissue techniques.32,68–70 Certainty B, strength ↑↑.
TTH3
Offer or refer for self-management strategies such as patient education, relaxation, and mindfulness-based stress reduction).37,74 Certainty C, strength ↑↑.
TTH4
Consider offering or referring for acupuncture, electroacupuncture, or dry needling as part of a multimodal approach.58,66–68,70–72,75,76 Certainty B strength ↑↑.
Discussion
The present umbrella review and clinical practice guideline builds upon prior recommendations regarding chiropractic and nonpharmacological management of CGH and TTH by encompassing a broader range of therapies27,28 and including a more comprehensive focus regarding patient evaluation and management. All recommendations were derived after synthesizing recent evidence from an umbrella review of the literature and attaining consensus from a large multidisciplinary panel. Several recommendations in this study differed from previous headache clinical practice guidelines, reflecting the higher certainty evidence due to the accumulation of more randomized controlled trials and systematic reviews regarding CGH and TTH. Considering the new and updated material focusing on broad aspects of care including diagnosis, history taking, treatment, and care management, the present guideline helps address gaps in prior guidance and aims to support chiropractors in their daily clinical decision-making.
Two statements on the topic of informed consent attained a strong recommendation despite being based on a very low certainty of evidence (i.e., D; ↑↑). Accordingly, this strongly encourages chiropractors to obtain informed consent despite limited evidence. The rationale for this apparent discrepancy stems from the source of evidence. Congruent with our approach to evidence certainty ratings, the supporting evidence for informed consent was based on consensus, expert opinion, and legal precedent, rather than arising from systematic reviews of randomized controlled trials. The informed consent example illustrates the necessity of separately evaluating certainty of evidence and strength of recommendation in guidelines, as these ratings may stem from distinct factors.94,95
The present clinical practice guideline makes a strong recommendation to offer spinal manipulative therapy for CGH based on high certainty evidence, yet it gives a weak recommendation to avoid spinal manipulative therapy as a stand-alone treatment for TTH based on low certainty evidence. This clinical practice guideline makes a weak recommendation to provide spinal manipulative therapy alongside other therapies for both CGH and TTH based on moderate certainty evidence. While prior clinical practice guidelines had already recommended spinal manipulative therapy for CGH,27,28 a clinical practice guideline in 2019 recommended avoiding spinal manipulative therapy for TTH. 28 In contrast to this prior recommendation, our findings were based on a larger number of studies and enabled a recommendation to use this therapy within a multimodal treatment strategy.
In a clinical practice guideline regarding the prescription of opioids published in 2022, the United States Centers for Disease Control and Prevention recommended the use of nonopioid therapies for individuals with subacute and chronic pain in the absence of serious pathology. 96 One of the recommendations was to use spinal manipulative therapy for TTH. In addition, the Centers for Disease Control and Prevention highlighted the relevance of patient preference when selecting therapies and that nonpharmaceutical treatments should be the initial approach to TTH and CGH. In the context of the current opioid epidemic, and considering patients’ potential preference to avoid opioids, our clinical practice guideline recommendation to use spinal manipulative therapy for TTH aligns with those of the Centers for Disease Control and Prevention.
New recommendations herein, which were not found in a prior similar clinical practice guideline for chiropractors, address specific elements of diagnostic criteria, history taking, red, yellow and orange flag screening, referral, and outcome assessment. 27 While our statements were based on moderate certainty evidence, each received a strong recommendation, highlighting the consensus around their necessity and importance in clinical decision-making for CGH and TTH. In addition, three of our statements regarding formulating a working diagnosis, conducting a physical examination, and avoiding routine diagnostic imaging received a strong recommendation based on high-certainty evidence.
The present clinical practice guideline also derived several statements that were based on a low certainty of evidence, suggesting additional research is needed to increase the certainty around these recommendations. One key example was the treatment frequency for headaches. While a prior clinical practice guideline arrived at more specific recommended frequencies and durations of care, our steering committee and Delphi panel were unable to derive similar statements due to the heterogeneity of the intervention characteristics included among relevant studies.27,28,54,69 Similarly, recommendations regarding self-management, soft-tissue therapies, dry needling, and mindfulness-based treatments were based only on moderate certainty evidence.
Other therapies had varying strengths of recommendation, based on a moderate certainty of evidence, including dry needling and therapeutic exercise for CGH (both having a weak recommendation), and acupuncture for TTH (strong recommendation). There was insufficient evidence to provide a recommendation either for or against several interventions that are potentially used for CGH such as acupuncture, education, meditation/mindfulness, and modalities used in isolation. Similarly, a weak recommendation was made to use electroacupuncture for TTH based on very low certainty evidence. Finally, we generally found a greater quantity and scope of available evidence for nonpharmacological treatments for TTH compared with CGH. These shortcomings reflect the gaps in the available literature and suggest randomized controlled trials should be conducted to continue examining a range of nonpharmacological interventions for CGH and TTH.
The Clinical Compass group, potentially including one or more of the current authors, expects to update the present guideline within 5–10 years using similar methods. Implementation of the present guidelines may be assessed through real-world longitudinal observational cohort studies, surveys, and cross-sectional analyses of large databases or registries. This may include an assessment of the clinical decision-making characteristics as well as treatments rendered for TTH and CGH.
Strengths and limitations
The present clinical practice guideline was strengthened by the review methods that were registered a priori, comprehensive search strategy, linkage between evidence and recommendations, incorporation of a large Delphi panel, and transparent team involvement and strategies.
Several limitations are noteworthy. Individualizing recommendations according to the frequency, duration, or chronicity of headaches (e.g., episodic vs. chronic TTH) was not feasible due to the lack of consistent definitions regarding these characteristics of the study populations among included studies. For TTH, the synthesized literature frequently did not analyze separate subgroups such as episodic versus chronic TTH, often due to primary randomized controlled trials including both types of TTH.55,60,62–64,66,69–72,76,77 As a result, there are instances where clinicians must rely upon the clinical expertise aspect of the evidence triad. 97 Future chiropractic clinical practice guidelines could also consider another common headache subtypes, migraine and concussion, which were beyond the scope of the present study. Meta-analysis based on raw data from randomized controlled trials was beyond our scope given the range of therapies assessed and corresponding strategy to focus on systematic reviews rather than primary studies. We did not consistently record individual justifications for downgrades in evidence certainty ratings, nor did we calculate kappa values to quantify agreement between assessors. While the data range was short (2017 onward), our approach obviated the need to include older studies, as these were inherently included within and/or superseded by more recent systematic reviews, which were included in this study. The Delphi consensus approach, while rigorous, is susceptible to bias due to the subjectivity and variability inherent among experts when interpreting evidence. Our restriction to English-language articles may have excluded relevant studies or guidelines, leading to language bias. However, two factors may have mitigated this potential bias. First, several of the systematic reviews we included had no language restrictions58,76,77 or incorporated studies published in non-English languages,50–52,56,60–64,66,68,69,75 and second, most systematic reviews and clinical practice guidelines are published in English. 98 Our use of predefined values for Scottish Intercollegiate Guidelines Network scores removes expert determination that may weigh certain items higher than others. Accordingly, article quality scores may have varied using different methods. This review and guideline did not assess the incidence of risks associated with chiropractor-delivered interventions for those with TTH/CGH, which was outside the scope of our primary objectives. Mild adverse events, such as transient muscle soreness or discomfort, are commonly reported with interventions such as spinal manipulative therapy, while serious adverse events are estimated at 1 per 2 million manipulations to 13 per 10,000 patients and may be better assessed through synthesis of large observational studies.99,100 Our guideline lacked a statement addressing marital status, work habits, family life, and leisure activities. These factors could be explored during history taking as they may influence headache frequency and/or severity. 101 Our guideline lacked specific recommendations on which components of the general physical or neurological examination should be conducted (e.g., blood pressure, cranial nerve assessment, and reflexes). Notably, blood pressure measurement has been encouraged by a previous guideline focusing on headache management in primary care settings. 38 Our recommendations did not differentiate between types of patient encounters (e.g., new vs. established patients or new vs. recurring headache presentations) or provide descriptive vignettes to guide interpretation. Accordingly, the utility of history taking and physical examination was left to the panelists’ judgment. Future guidelines may provide more explicit guidance regarding the frequency, timing, and clinical context of these recommendations. Our guideline did not evaluate the role of imaging in the initial diagnosis of CGH or TTH. While radiographs or magnetic resonance imaging may occasionally assist in differential diagnosis or help identify cervical spine pathology in suspected CGH, imaging findings are common among individuals without headache and are not required by The International Classification of Headache Disorders, 3rd edition for CGH diagnosis. 36 Recent evidence also indicates a lack of positive correlation between degenerative cervical magnetic resonance imaging findings and headache severity, suggesting limited diagnostic utility of imaging in CGH. 102 Future guideline updates could address appropriate indications and timing of imaging in greater detail. The present recommendations are not generalizable to pediatric populations and do not apply to those with serious pathology (e.g., cancer, infection, and spinal fracture).
Conclusions
This clinical practice guideline provides evidence-based consensus statements for nonpharmaceutical management of CGH and TTH in adults. Among other statements, recommendations encouraged a history and examination and use of spinal manipulative therapy for patients with CGH, whereas patients with TTH should receive spinal manipulative therapy as a component of multimodal care. In addition, our guideline highlights the lack of high certainty evidence, which precludes our ability to derive strong statements for several other nonpharmacological therapies such as acupuncture, education, meditation/mindfulness, and modalities used in isolation for CGH and electroacupuncture for TTH. This guideline provides evidence-informed direction to chiropractors regarding which nonpharmacological interventions are appropriate to consider for CGH and TTH in adults.
Ethics Approval and Consent to Participate
The Institutional Review Board of Texas Chiropractic College approved the project. All Delphi panelists consented to participate in the study.
Consent for Publication
All Delphi panelists consented to being acknowledged in the article.
Competing Interests
C.J.D., L.S.C., and S.A.M. are board members for Clinical Compass. C.J.D. is a member of the Washington State Health Technology Clinical Committee. K.R.A. is on the Board of Governors for the American Chiropractic Association. The authors have no additional conflicts to declare.
Authors’ Contributions
Study methodology was developed by R.J.T., C.J.D., C.H., D.N.T., S.A.W., M.R.P., K.R.A., L.S.C., S.A.M., C.A.S., and C.B.O. S.A.W. conducted the literature search. C.J.D. and C.H. secured funding. C.H. provided supervision and was responsible for the software and data analysis. Data curation was performed by R.J.T., C.J.D., C.H., D.N.T., and M.R.P. The investigation was carried out by R.J.T., C.J.D., C.H., D.N.T., S.A.W., M.R.P., K.R.A., L.S.C., S.A.M., and C.A.S. R.J.T. and C.H. prepared the first draft of the article, whereas R.J.T., C.J.D., C.H., D.N.T., S.A.W., M.R.P., K.R.A., L.S.C., S.A.M., C.A.S., and C.B.O. contributed to article revisions and editing.
Footnotes
Acknowledgments
The authors thank project coordinator, Cathy Evans, for her expert management of the consensus process and all project communications. The authors also thank the Delphi panelists for their generous contribution of their time and expertise to the consensus process: Logan Benjamin, DC, CSCS; Wayne Bennett, DC, DABCO; Craig R. Benton, DC; Derrell Blackburn, DC, MBA; Charles L. Blum, DC, CSCP; Gina M. Bonavito-Larragoite, DC, MBA; Amanda G. Brown, DC, PSP; Wayne H. Carr DC, DACRB, CCSP, AFMCP, CCA; Jeffrey R. Cates, DC, MS; Richard L. Cole, DC, DACNB, FICCN, DAAPM, FICC; Matthew Coté, DC, MS; Thomas Cotter, DC, DACRB; Diana Cowell, PharmD, MBA, BCPS; Zachary A. Cupler, DC, MS; Monica Curruchich, DC, RN, BSN; John Curtin, MSS, DC, DACO, CCIC; Mark D. Dehen, DC, FICC; Paul M. Ettlinger, DC; Jason T. Evans, DC, DIBCN, FIACN, ABIME, NASM; David Folweiler, DC, DACRB; Jonathan Free, DC; Margaret Freihaut DC; William P. Gallagher Jr., DC, CMVI; Justin Goehl, DC, MS; Kyle B. Hammes, DC; Jerrell Hardison, DC; Huyen “Lily” Ho, DC; Gary Alan Jacob DC, LAc, MPH, DipMDT; Jeffrey M. Johnson, DC; Valerie Johnson, DC, DABCI, DACBN; Gregory Katsaros, DC, DAAPM; Louis A. Kazal, Jr., MD, FAAFP; Robert E. Klein, DC, FACO; Lawrence J Larragoite DC, FIAMA, CFMP; Robert A. Leach, DC, MS, MCHES; Duane Lowe, DC; Eric Luke, DC, MS; Hans Mohrbeck, DC, CCN, CCSM; Mark Mulak, DC, MBA, MS, DACRB, DACBSP®; Marcus Nynas, DC; Juli Olson, DC, DACM, FAIHM; Colette Peabody, DC, MS; Mariangela Penna, DC; Lindsay Rae, DC; Shawn Reince, DC; Jeffrey W. Remsburg DC, MS, DACRB, Cert MDT; Christopher B. Roecker, DC, MS; Vern A. Saboe Jr., DC, DACAN, FICC, DABFP, DIANM; Scott Siegel, DC; Charles A. Simpson, DC, DABCO; Samantha Stolzel, DC, MS; Mignon “Mimi” Sweat, MS, DC; Susan Wenberg, DC, MA; John S. Weyand, DC, DABCO; Clint J. Williamson, DC; Morgan Young, DC.
Author Disclosure Statement
This material is the result of work supported with resources and the use of facilities at VA Puget Sound Health Care System. The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the United States Department of Veterans Affairs or the United States Government.
Funding Information
This study was funded in part by Clinical Compass and NCMIC Foundation. Clinical Compass funding partially covered the time and resources of C.J.D., S.A.M., and C.H., whereas NCMIC Foundation funding partially covered the time and resources of C.H. The study received no specific grant and NCMIC Foundation played no role in decision-making.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
