Abstract
Background:
Hepatitis B virus (HBV) infection is a leading cause of chronic liver disease and cancer globally, with perinatal transmission representing the predominant route of new infections in high-burden countries such as Nigeria. Despite significant prevalence, screening, diagnosis, and timely intervention for HBV among pregnant women remain suboptimal in Nigeria, contributing to ongoing morbidity and mortality.
Objectives:
This analysis assessed the impact of interventions to reduce perinatal HBV transmission in five pilot health facilities in Delta State, Nigeria. It evaluated HBV and HCV prevalence among pregnant women, timing of antenatal care (ANC) booking, linkage to care for HBV-positive women, and HepB birth dose (HepB-BD) vaccination coverage and timeliness.
Design:
The project design was a prospective implementation research which involved a cohort of pregnant women registered for ANC in the five pilot healthcare facilities strategically located across the three senatorial districts of Delta State, Nigeria. These facilities were all high-volume public facilities located and were carefully chosen to ensure representation across different regions of Delta State, providing a diverse perspective on the project’s implementation and impact. The selected facilities provide secondary and tertiary care services.
Methods:
All pregnant women at five health facilities were screened at their first ANC visit for HBV and HCV using rapid testing. HBV-positive women were linked to care and initiated on tenofovir disoproxil fumarate (TDF) prophylaxis. The proportion of newborns receiving timely HepB-BD vaccination (within 24 h of birth) was measured pre- and post-intervention. Statistical analysis was performed using paired t-tests and Chi-squared.
Results:
A total of 2844 pregnant women were screened for HBV and HCV. The pooled prevalence of HBV infection among the study population was found to be 1.86% (95% CI: 1.35%–2.37%), six times higher than HCV (0.3%). A Chi-squared test for homogeneity was conducted to determine if the proportion of HBV-reactive individuals varied significantly across the five facilities. The test revealed a statistically significant difference in HBV prevalence across facilities (χ2 = 11.49, df = 4, p = 0.021). Most women (53.4%) booked ANC in the second trimester. Prior to intervention, the probability of administering the HepB vaccine to any child, whether newborn or older infants, on any day of the week, including weekends, was 40% because vaccination was not conducted daily in any of the pilot facilities. Post-intervention, this rose to 100% as all pilot facilities adopted daily vaccination practice. In comparison with the previous study of 5.8%, HepB-BD timely uptake within 24 h, post-intervention, was 100% across all pilot facilities.
Conclusion:
Project StopHepB interventions significantly improved early identification, linkage to care, and timely HepB-BD vaccination, effectively closing gaps in HBV prevention of perinatal transmission. These findings support the scale-up of similar interventions to advance HBV elimination goals across other facilities in the state, in Nigeria, and to prevent new HBV infections in the future.
Plain language summary
Hepatitis B is a serious liver infection that affects millions of people around the world and is one of the leading causes of liver cancer and related deaths. One of the most common ways it spreads is from mother to child during childbirth. Babies who become infected at birth are at high risk of developing chronic hepatitis B, which can cause lifelong health problems. Although there is a highly effective vaccine that can prevent this, it must be given within 24 hours of birth. Unfortunately, in many parts of Nigeria, including Delta State, not all babies receive the vaccine on time due to issues like vaccine access, staff shortages, and limited awareness among healthcare workers and families. To address this, a project called StopHepB: Break the Cycle, Protect the Next Generation was carried out in five hospitals across Delta State from February to May 2025. The project aimed to stop the transmission of hepatitis B from mothers to their babies by screening all pregnant women for the virus during their first antenatal visit, linking those who tested positive to free antiviral treatment, and ensuring that every baby born in the hospital received the hepatitis B vaccine within 24 hours. A key part of the project’s success was moving the vaccine supply directly into labor wards, allowing nurses to vaccinate newborns immediately after delivery. Before the intervention, only about 6% of babies received the vaccine on time. After the project was implemented, timely vaccination coverage jumped to 100% in the five pilot hospitals, with most babies receiving the vaccine within just two and a half hours of birth. All mothers who tested positive for hepatitis B were linked to care, and their babies received the vaccine on time. This represents a major step forward in preventing mother-to-child transmission of the virus.
Keywords
Introduction
Global HBV context
Hepatitis B virus (HBV) infection remains a major global public health challenge, with over 254 million people living with the virus. 1 Recent data highlight a troubling rise in viral hepatitis-related deaths, increasing from 1.1 million in 2019 to 1.3 million in 2022, with HBV accounting for 83% of these fatalities. 1 HBV is a leading cause of primary liver cancer (hepatocellular carcinoma) and cancer-related mortality.2,3 Transmission primarily occurs through direct contact with infected blood, unprotected sexual contact with an infected individual, use of contaminated or nonsterile medical or injection equipment, and, most commonly, from an infected mother to her newborn during childbirth (perinatal transmission). 4 The World Health Organization (WHO) recommends timely HepB-BD for every newborn within the first 24 h, followed by three subsequent doses of the hepatitis B vaccine as part of the pentavalent vaccination schedule. 5 Globally, hepatitis B birth dose coverage stands at 45% as of 2022, with significant regional disparities. The WHO African Region faces notable challenges, reporting the lowest coverage rate at 18%. This variability underscores the need for targeted interventions to improve access to timely hepatitis B vaccination, particularly in underserved regions. 6 While all countries in the African region, including Nigeria, have adopted the pentavalent infant vaccination series, only 15 out of 47 WHO AFRO member countries (32%) have introduced the HepB-BD. 7 Without timely administration of the birth dose, children will remain at risk for infection during the first 6 weeks of life, 8 with risk of transmission being particularly high among those born to mothers who are hepatitis B e-antigen positive. 9
Nigeria-specific burden
Nigeria contributes significantly to the burden in Africa, with an estimated 20 million people infected. 4 In Delta State, with a population of 6.7 million, HBV prevalence is estimated at 8%, translating to over 500,000 individuals living with the virus. 10 Despite this high prevalence, screening, diagnosis, and treatment rates remained suboptimal, creating a substantial gap in efforts to prevent liver cancer and HBV-related mortality. Children infected with HBV at birth have a greater than 90% risk of developing chronic hepatitis B.6,11 Since 2004, Nigeria has provided HepB-BD followed by the pentavalent vaccination series for every child. 12
Specific perinatal HBV barriers
All newborns delivered within public health facilities are eligible for these routine immunizations. However, despite the availability of the hepatitis B vaccine, uptake remains limited by factors such as cost, knowledge gaps among providers and communities, and access issues. Studies have documented significant barriers to birth dose uptake in Nigeria, including limited accessibility of the birth dose, staffing shortages, high prevalence of home births, inadequate knowledge among healthcare workers regarding HBV transmission, misconceptions about the timeliness of birth dose, and concerns about vaccination wastage.13–16 A recent study in Delta State identified significant discrepancies between reported and actual timely uptake of birth dose. It found a 5.8% timely uptake, 10 which was substantially lower than the 87.3% reported in the Nigeria Demographic and Health Survey (NDHS) 2023–2024, where the latter figure included birth doses administered up to 14 days after birth. 17
Project StopHepB: A novel approach
Significant gaps have existed in perinatal HBV prevention programming in Delta State, including inadequate, timely administration and reporting of HepB-BD vaccine coverage, as well as a lack of assessment of hepatitis B prevalence among pregnant women. Project StopHepB was a novel intervention that aimed to clearly establish HBV prevalence among pregnant women and to link and increase the timely HepB-BD vaccine uptake among newborns in the five healthcare facilities in Delta State, Nigeria, between February and May 2025. This project introduced innovative approaches to perinatal HBV prevention, including: Routine HBV testing for all pregnant women at antenatal booking, early linkage of HBV-positive women to antiviral treatment at 24 weeks of gestation, in line with WHO guidelines, and timely administration of the HepB-BD vaccine to all newborns within 24 h of birth through vaccine decentralization to delivery wards.
Methods
Design
The project design was a prospective implementation research which involved a cohort of pregnant women registered for antenatal care (ANC) in the five pilot healthcare facilities strategically located across the three senatorial districts of Delta State, Nigeria. These facilities were all high-volume public facilities located and were carefully chosen to ensure representation across different regions of Delta State, providing a diverse perspective on the project’s implementation and impact. The selected facilities provide secondary and tertiary care services (see Table 1).
Pilot sites and characteristics.

Inclusion criteria
Riding on the Delta State’s policy to screen all pregnant women attending ANC. Participants were enrolled if they were above the age of 18 and were able to provide verbal consent. Only pregnant women registered for ANC in the 5-pilot facilities were included in this project. Only pregnant women who screened positive for HBV were enrolled for antiviral prophylaxis, and only pregnant HBV-positive women at 24 weeks of gestation were qualified for initiation of TDF prophylaxis.
Exclusion criteria
All males, non-pregnant women, pregnant women under the age of 18, and pregnant women who declined consent were excluded from this intervention.
Time frame
This study was a 4-month intervention appraisal from February through May 2025.
Project implementation
Within the selected health facilities, a collaborative community-driven research protocol was developed to improve strategies across ANC delivery, immunization services, and labor and delivery for perinatal prevention. Prior to the project start, a one-day training took place with health facility leadership from each selected study site. Training included an overview of the project, the significance of hepatitis B early screening, and the importance of its prevention. Thirty (30) healthcare providers directly involved in project implementation participated in the workshop. Each facility was represented by a team of five healthcare providers, comprising one physician, one pharmacist, one nurse officer, one laboratory scientist, and one medical records officer.
Following the training, site visits were conducted by study staff to ensure key health facility partners were clear on study objectives, purpose, and implementation.
In partnership with the Ministry of Health in the state, all pregnant women were screened for HBV during the ANC first visit in line with the state’s policy on screening all pregnant women attending ANC for HBV and HCV, covered under the State’s health insurance equity plan. Following testing at ANC, all pregnant women were automatically referred in the second trimester to medication access through each health facility pharmacy. Daily Tenofovir was provided to all positive pregnant women during the second trimester for treatment of HBV. The cost of treatment was covered by the project and was free to all participants.
All infants born within the facility were directed to receive a timely HepB-BD, and health care workers were trained on the importance of the timely birth dose and data tracking throughout the project period. To facilitate vaccination administration, refrigerators were donated and kept in the labor wards of the selected study sites, and a point person for each health facility was selected to lead efforts within each setting. Collaboration between immunization and labor and delivery was essential to ensure accurate data reporting and accessibility of the vaccination in the labor wards. Pentavalent vaccination was also encouraged for all infants according to the country schedule.
Sample size calculation
The sample size for this study was calculated based on the average monthly attendance of pregnant women at the ANC clinic. With an average weekly attendance of 265 women from the 5 pilot sites, and ANC visits occurring once every week (approximately 4 days per month per facility), the estimated monthly attendance was calculated to be 1,060 women (265 women/week × 4 weeks/month). Over a 4-month period, the total sample size was estimated to be 4240 women (1060 women/month × 4 months). However, accounting for 6 ANC visits missed due to public holidays, which would have resulted in 1590 women being missed (6 days × 265 women/ANC day), the adjusted sample size was recalculated to be approximately 2650 women (4240 women – 1590 women). Therefore, the estimated sample size for this study was approximately 2650 pregnant women. However, a total of 2844 pregnant women were eventually screened and reported because some months were not exactly 4 weeks, accounting for the slightly higher number above the recalculated sample size.
Data collection and analysis
Data on screening, treatment, ANC attendance, and HepB-BD administration were collected from the laboratory, pharmacy, and nursing departments using a pre-tested, custom-designed online reporting survey using the KoBoCollect tool. Data were reported weekly into the KoBoCollect tool and then exported to Excel and SPSS for cleaning and analysis. Bi-weekly virtual data review meetings provided a platform for sharing insights, addressing challenges, and troubleshooting across the five pilot sites. Descriptive statistics were generated in Excel and presented using bar charts and tables. Inferential analysis- including paired-sample t-tests and Chi-squared tests for homogeneity- was conducted using SPSS version 29.
Performance characteristics of test strips
Hepatitis B surface antigen (HBsAg) tests were conducted with Bio-Check rapid diagnostic test kits, manufactured by Wellkang Limited, London, UK, using serum samples after centrifugation of whole blood collected from pregnant women at first ANC visits. The manufacturer claims the HBsAg rapid test strips have been evaluated with an EIA test using clinical specimens, and the results for sensitivity, specificity, and accuracy were 99.9% respectively, relative to the EIA test. Also, the quality control tests conducted by the state ministry of health gave sensitivity and specificity of 99.9% respectively, before deployment to pilot facilities for use.
Results
ANC booking and screening
A total of 2844 pregnant women were screened for HBV and HCV during the project implementation period (February–May 2025). Dis-aggregated data show that 666 women (23.4%) booked ANC in their first trimester, 1518 (53.4%) in the second trimester, and 660 (23.2%) in the third trimester (Figure 1). The majority of women initiated ANC in the second trimester, a critical finding given the importance of early identification, counseling, and initiation of antiviral prophylaxis for HBV-positive pregnant women (Figure 2). Early ANC booking is essential to maximize the effectiveness of interventions aimed at preventing mother-to-child transmission.
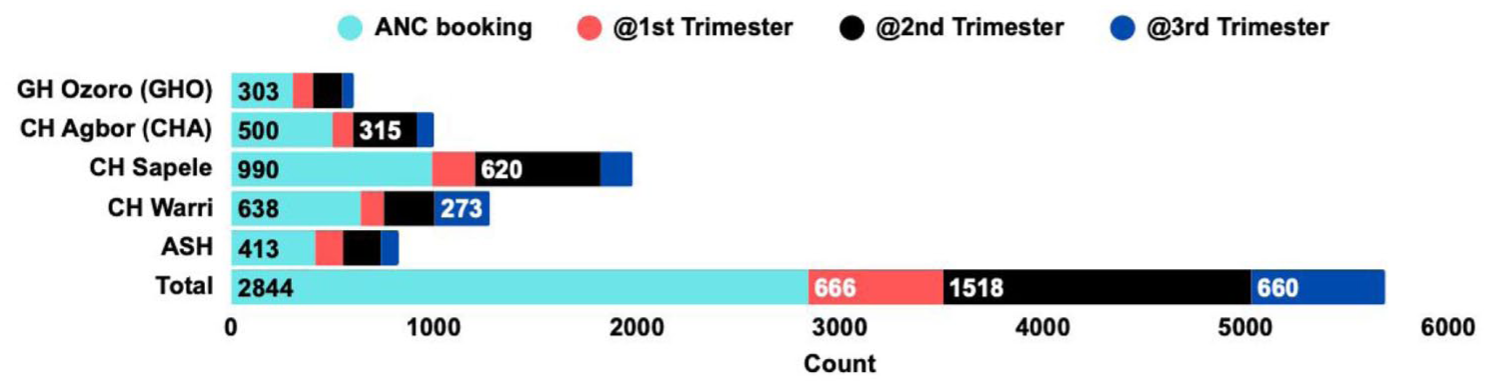
Summary of ANC booking across the five pilot facilities based on location and trimester.

Antiviral prophylaxis for pregnant positive women at 24 weeks of gestation across pilot and spoke sites.
Prevalence of HBV and HCV in pregnancy
The pooled prevalence of HBV infection among the study population was found to be 1.86% (95% CI: 1.35%–2.37%), approximately six times higher than the prevalence of HCV (0.3%) during the same period. A Chi-squared test for homogeneity was conducted to determine if the proportion of HBV-reactive pregnant women varied significantly across the five facilities. The test revealed a statistically significant difference in HBV prevalence across facilities (χ2 = 11.49, df = 4, p = 0.021), indicating that the null hypothesis of homogeneity can be rejected. These findings suggest that the proportion of HBV-reactive pregnant women is not uniform across the facilities, and that there may be underlying factors contributing to the observed heterogeneity. Table 2 shows the distribution of HBV and HCV screenings conducted across the pilot facilities for all pregnant women booked for ANC.
HBV/HCV screening data and positivity yield from 5-pilot facilities from February–May 2025.

This project was primarily piloted in five facilities. However, two patients were referred from off-pilot facilities we considered as spoke sites. Data from spoke sites were not factored into the positivity rate computation for HBV.
Linkage to care and antiviral prophylaxis
A total of 55 cases (including those from both hub and spoke sites) were reported and linked to care. As of the current reporting period, three out of four HBV-positive pregnant women who had TDF prophylaxis had delivered, and their infants received the birth dose of the HBV vaccine within 3 h of birth. See Figure 1.
Livelihood of timely HepB-BD vaccine administration
Prior to the intervention, the average likelihood of a newborn receiving the HepB-BD vaccine at all on any day of the week in the pilot facilities was 40% (range: 14%–71%), as vaccinations were not done daily. Notably, none of the facilities offered vaccination services on weekends, limiting access. Following the intervention, a paired Student’s t-test demonstrated a statistically significant increase in both daily (100%) and timely HepB-BD administration (t(4) = −7.43, p = 0.002), with a mean difference of 60.0% (95% CI: 43.1, 76.9). This indicates a substantial positive impact of the intervention.
Post-intervention, the likelihood of timely HepB-BD uptake increased to 100% across all pilot facilities, with an average administration time of 2 h and 30 min after birth, regardless of the day or time of delivery. This analysis demonstrates the substantial impact of the interventions in strengthening the prevention of perinatal transmission of hepatitis B in Delta State, Nigeria. See Figure 3.

Prior and Post-Intervention launch probability of newborns receiving HepB-BD vaccine on any day of the week after birth across the five pilot facilities.
Coverage and timeliness of birth dose administration
A total of 1,727 live births were recorded in the five pilot facilities during the study period. Most birth doses were administered within 3 h of birth (average time: 2 h, 30 min). Specifically, 85.2% of newborns received the birth dose within 3 h; 8.8% between 3 and <6 h; 3.7% between 6 and <12 h while 1.3% between 12 and <24 h. Importantly, no newborns received the birth dose after 24 h, a marked improvement from the 5.8% timely uptake reported at baseline in 2024. This achievement surpasses the WHO global target of at least 90% timely HepB-BD coverage within 24 h. The results underscore the effectiveness of decentralizing HepB-BD vaccine availability to labor wards in optimizing timely uptake. See Figure 4.

Distribution of HBV-BD administered to newborns across the 5-pilot facilities.
Discussion
HBV is a significant health threat in Nigeria, accounting for most liver cancer cases and ranking fourth among cancer-related deaths. However, the epidemic remains largely hidden, with most infected individuals undiagnosed and untreated, highlighting the need for increased awareness, screening, and access to care,18–20 especially for women of childbearing age and pregnant women.
Our study revealed a notably lower pooled HBV prevalence of 1.9% among pregnant women compared to the 5.9% reported in the 2018 NAIIS household survey 4 and the 6.5% reported in a separate systematic meta-analysis. 21 Although the HBV prevalence is lower, the risk of MTCT remains a concern due to gaps in ANC attendance, with only 60.5% of women attending at least four visits, as per the 2023-2024 NDHS report. This is juxtaposed with a higher rate of in-facility births (83.0%). 17 Furthermore, late ANC booking, predominantly in the second trimester, as observed in this study, poses a significant barrier to early diagnosis and timely initiation of antiviral prophylaxis for hepatitis B-positive pregnant women, thereby increasing the risk of perinatal HBV transmission. These findings underscore the need for targeted community education and engagement of young women and women of childbearing age to promote earlier ANC attendance, healthcare facility utilization, and improved maternal health outcomes. In addition, the significant variability in HBV prevalence across the five pilot facilities suggests possible underlying sociocultural or systemic factors that merit further investigation. Understanding these disparities is essential for designing context-specific and culturally sensitive interventions.
The WHO recommends a hepatitis B vaccination schedule that includes a timely birth dose (within 24 h of birth), followed by at least two additional doses. When administered within 24 h, the birth dose is 90% effective in preventing perinatal transmission.22,23 However, an earlier study in Delta State reported only a 5.8% timely uptake of the HepB-BD vaccine among children under five, lower than the 13% timely coverage reported in a 2018 study spanning select health facilities in Botswana, The Gambia, Namibia, Nigeria, and São Tomé and Príncipe (STP). 24 This underscores the urgent need for strategies to improve timely administration in line with the WHO recommendations. 1 Increased hepatitis B birth dose vaccination coverage could avert approximately 6.0 million new neonatal HBV infections from 2024 to 2030, with the greatest impact in the WHO African Region. 25
A key achievement of the intervention was the dramatic improvement in daily and timely HepB-BD vaccine administration. Prior to intervention, the probability of administering the HepB vaccine to any child, whether newborn or older infants, on any day of the week, including weekends, was on average 40%, because vaccinations were not conducted daily in any of the pilot facilities. Post-intervention, this rose to 100% as all pilot facilities adopted daily vaccination practice, including on weekends. In terms of timely administration, an earlier study showed a 5.8% timely uptake within 24 h, 10 hindered by factors such as poor providers’ knowledge, centralization of vaccine storage, limited vaccination days, and lack of services on weekends. These barriers are consistent with findings from earlier studies in Nigeria.15–17
Post-intervention, daily and timely HepB-BD administration reached 100% in all pilot facilities, including on weekends, with most doses administered within 3 h of birth—surpassing the WHO’s 90% target within 24 hours. All newborns born to HBV-positive mothers received the HepB-BD within 24 h. This achievement demonstrates the effectiveness of decentralizing vaccine storage and availability to labor and delivery wards, ensuring immediate access at the point of need. The elimination of delays beyond 24 h represents a key success in preventing perinatal HBV transmission.
Furthermore, all HBV-positive pregnant women were identified and only those up to 24 weeks of gestation were commenced on TDF prophylaxis to reducing the risk of MTCT. The 2024 WHO guidelines for management of HBV in pregnancy recommend a “test-and-prophylaxis” approach, which allows for treatment at 24 weeks based on HBsAg positivity in settings where HBV DNA or HBeAg testing is not available—making this approach highly feasible in resource-limited, high-burden settings. 1 The combined strategy of maternal antiviral therapy and universal, timely HepB-BD vaccination aligns with global best practices and presents a scalable model for other healthcare facilities in the state and beyond.
Despite these successes, they were not without challenges. The low rate of first-trimester ANC booking continues to limit opportunities for early diagnosis and intervention. While several factors may contribute to this, one observable barrier in some pilot facilities was the practice of “triage,” necessitated by limited facility capacity to accommodate high patient volumes. Further research is needed to identify and understand other barriers to early ANC attendance and to inform the development of targeted interventions. Finally, while the pilot facilities achieved excellent outcomes, scaling up these interventions across the state and nationally will require sustained political commitment, adequate resource allocation, and broader health system strengthening.
Also, during the project implementation, we encountered notable challenges that almost impacted timely vaccine administration. One major issue was the limited quantity of vaccine supplies due to logistical constraints, which threatened to impede the successes gained. However, this was successfully addressed by redistributing vaccine vials from neighboring primary healthcare centers (PHCs), facilitated by vaccine officers, to meet the demands. Another significant challenge was the knowledge gap among healthcare workers who were not part of the initial training but were involved in direct ANC management of pregnant women. A significant lag in this project from the facilities was the delay, and sometimes, the outright miss of initiation of antiviral at 24 weeks. To mitigate this, we conducted targeted virtual quasi-training sessions with these healthcare workers, supplemented by regular phone calls and follow-ups. While not exhaustive, these efforts helped bridge the knowledge gap to a reasonable extent and improved the overall PMTCT care for pregnant women in the affected facilities.
Conclusion
In conclusion, the Project StopHepB interventions have demonstrated that targeted, facility-based strategies can close critical gaps in PMTCT of hepatitis B. The Project StopHepB interventions in Delta State have demonstrated significant improvements in early ANC HBV/HCV screenings at booking, linkage to care, and, most notably, the timely administration of the HepB-BD vaccine to newborns within 24 h after birth in the five pilot facilities. These findings provide compelling evidence to support the scale-up of similar direct interventions across other healthcare facilities in the state, Nigeria, and other high-burden countries to enhance the prevention of mother-to-child transmission of HBV.
The benefits of scaling up PMTCT interventions are clear, as increased hepatitis B birth dose vaccination coverage could avert approximately 6.0 million new neonatal HBV infections from 2024 to 2030, with the greatest impact in the WHO African Region. The hepatitis B vaccine, described as the first cancer-preventing vaccine, is one very important tool in the fight to eliminate new infections, which has to be sufficiently and efficiently deployed. By prioritizing PMTCT interventions, Delta State can make significant contributions and strides toward eliminating hepatitis B new incidences and reducing the burden of future liver cancer and related deaths in its growing population. There is a need to further strengthen linkage to care and follow-up through a well-established referral system across the state.
Supplemental Material
sj-pdf-1-taj-10.1177_27558428251400452 – Supplemental material for Insights from strengthening perinatal hepatitis b prevention in healthcare facilities in Delta State, Nigeria: A prospective, pilot implementation intervention
Supplemental material, sj-pdf-1-taj-10.1177_27558428251400452 for Insights from strengthening perinatal hepatitis b prevention in healthcare facilities in Delta State, Nigeria: A prospective, pilot implementation intervention by Prince Ochuko Okinedo, Catherine Freeland, Ehimario U. Igumbor, Samuel Okwuchukwu Ilikannu, Funso Joshua Eniojukan, Ngozi Roy Maduka, Gregsonil Ogheneruemu Idibofu, Linda Peter-Enyi, Kevin Igweh, Dafe Akokotu, Sabastine Edokpayi, Anita Abifade, Sandra Igah, Ossai Abaniwa Theresa, Aki-Famous Betty, Victoria Idegbesor, Ubiri Obaherhekre, Idanesi O Stella, Kelechi Okorie, Kiyem Vivian Wamari and Christian Tetsola in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-pdf-2-taj-10.1177_27558428251400452 – Supplemental material for Insights from strengthening perinatal hepatitis b prevention in healthcare facilities in Delta State, Nigeria: A prospective, pilot implementation intervention
Supplemental material, sj-pdf-2-taj-10.1177_27558428251400452 for Insights from strengthening perinatal hepatitis b prevention in healthcare facilities in Delta State, Nigeria: A prospective, pilot implementation intervention by Prince Ochuko Okinedo, Catherine Freeland, Ehimario U. Igumbor, Samuel Okwuchukwu Ilikannu, Funso Joshua Eniojukan, Ngozi Roy Maduka, Gregsonil Ogheneruemu Idibofu, Linda Peter-Enyi, Kevin Igweh, Dafe Akokotu, Sabastine Edokpayi, Anita Abifade, Sandra Igah, Ossai Abaniwa Theresa, Aki-Famous Betty, Victoria Idegbesor, Ubiri Obaherhekre, Idanesi O Stella, Kelechi Okorie, Kiyem Vivian Wamari and Christian Tetsola in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors would like to first express profound gratitude to God Almighty for His divine benevolence and guidance from conception to planning, execution, and conclusion of this pilot project. Also, would like to thank the Wife of the Governor and Founder, You Matter Charity Foundation, Her Excellency, Deaconess Tobore Oborevwori, for her immense support, which culminated in the inclusion of viral hepatitis B/C screening policy for all pregnant women receiving ANC free-of-charge, covered in the Delta State Contributory Health Commission. Special thanks to the Hon. Commissioner of Health, Dr. Joseph Onojaeme, members of the Department of Public Health, Delta State Ministry of Health, and all healthcare providers who volunteered as focal persons at the pilot facilities. The authors would also like to thank the Medical Directors and their management team for providing the needed support for a smooth project implementation. Lastly, the authors thank the Hepatitis B Foundation, Doylestown, Pennsylvania, U.S.A., for providing funding for TDF, cooling systems, training, and bi-weekly calls and stipends for all healthcare workers who were directly involved in this project. Lastly, my great team at Hepatitis Advocacy Foundation for their invaluable support of this project.
Ethical considerations
Ethical approval for this study was obtained from the Delta State Ministry of Health Research Ethics Committee (MoHREC) (Approval Reference: HM/596/T2/24). This approval covered all pilot sites, as all participating healthcare facilities operate under the regulatory oversight of the Delta State Ministry of Health. The Committee reviewed and approved the study protocol, including the use of verbal informed consent, which was deemed appropriate given the minimal risk to participants and the need to preserve anonymity while addressing potential linguistic, literacy, and cultural barriers. The ethics approval letter has been uploaded as a ![]() . During antenatal care (ANC) visits at each of the five pilot facilities, designated ANC nurses explained the study objectives and the voluntary nature of participation to all eligible pregnant women. Verbal informed consent was obtained prior to hepatitis screening and subsequent linkage to care, and each consent was documented in participant logs by the attending nurses.
. During antenatal care (ANC) visits at each of the five pilot facilities, designated ANC nurses explained the study objectives and the voluntary nature of participation to all eligible pregnant women. Verbal informed consent was obtained prior to hepatitis screening and subsequent linkage to care, and each consent was documented in participant logs by the attending nurses.
Consent to participate
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki (2013 revision). No identifiable or sensitive personal information was collected. All data were fully anonymized and stored in secure, password-protected systems, with no identifiable information linked to any participant.
Consent for publication
Not applicable. No individual participant data, images, or other personal identifiers are included in this manuscript.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: However, the Hepatitis Advocacy Foundation received funding for the project implementation from the Hepatitis B Foundation in collaboration with the Delta State Ministry of Health and the Office of the First Lady of Delta State through her You Matter Charity Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The full dataset supporting this study is provided within the manuscript. Informed consent for available as a Supplemental Document.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
