Abstract
The entry of falsified and substandard medicines into the legitimate pharmaceutical supply chain has negative impacts on healthcare systems, patient safety, and patient access to medicine. The COVID-19 pandemic has highlighted the importance of access to safe medicine through legitimate pharmaceutical supply chains and the willingness of criminals to target medical products such as PPE (personal protective equipment) and COVID-19 treatments. In this article, we analyse data from the United Kingdom (UK) national medicine alert and recall database to identify and understand recent cases of substandard and falsified medicine in the UK’s healthcare systems. Using the UK as a case study, we describe that national drug alert and recall data are useful in their current form to record and understand cases of substandard and falsified medicines in the supply chain. However, if regulatory agencies published further data, these drug recall databases may be useful to support longitudinal and international comparative medicine quality studies. We suggest that regulatory agencies publish the number of affected medicine packs in each recalled batch, as part of the recall process. This will help policy makers, practitioners, and researchers to better understand, monitor and compare the quality of medicines within legitimate supply chains.
Keywords
Summary Box:
National medicine alert and recall databases are a rich source of data concerning substandard and falsified medicines in the legitimate supply chain.
These databases exist across many countries and record much of the same drug recall variables.
If regulatory agencies published the exact number of recalled packs in the country, we could move closer to the longitudinal and international estimation of medicine quality.
By comparing international and longitudinal medicine quality, we can identify positively and negatively performing countries and manufacturers, and learn from their practices.
Introduction
The issue of Substandard and Falsified medicines (SF Medicines) affects high-income and low- and middle-income countries. 1 Substandard medicines are authorised medical products which are ‘out of specification’ and fail to meet either their quality standards or specifications, or both. Falsified medicines are ‘medical products that deliberately/fraudulently misrepresent their identity, composition or source’. 2 Within the medicine quality context, these terms are increasingly replacing the term counterfeit, which is more closely aligned with copyright and intellectual property infringements. 3 The term falsified medicine is preferred by the European Commission and the Medicines and Healthcare products Regulatory Agency (MHRA). One might expect that high-income countries with established medicine regulatory bodies have lower rates of SF Medicines in their legitimate supply chains, and higher standards of medicine quality compared to lower income countries. 4 However, we cannot be sure how medicine quality changes over time, nationally or internationally. In this article, we use the (United Kingdom) UK medicine alert and recall database as a case study to discuss the opportunities and challenges of comparative and longitudinal medicine quality studies in high-income countries.
Empirical examples of SF Medicine in high-income countries
SF Medicines have been linked to thousands of deaths internationally. 5 Furthermore, SF Medicines have an enormous economic impact, which is estimated at between US$10 billion and US$200 billion annually worldwide.6–8 Moreover, SF Medicines negatively impact global access to essential medicines 9 and adversely impact antimicrobial resistance. 10 In the high-income country context, a falsified version of Bevacizumab (Avastin®), an anticancer medicine, was found in the legitimate supply chain. This medicine, which is usually manufactured by Roche, and contained starch and salt instead of containing the active pharmaceutical ingredient (API). 11 Recently, there has been a case of falsified Iclusig® (Ponatinib) identified in Switzerland and circulated in Europe and the Americas which contained paracetamol instead of Ponatinib. This medicine was circulated through the legitimate supply chain, via pharmaceutical wholesalers. 12 Even more recently, due to the COVID-19 pandemic, there have been numerous cases of falsified COVID treating medical products circulating across the globe. 13 More broadly, the Pharmaceutical Security Institute (PSI) publishes annual figures concerning counterfeiting, illegal diversion and theft incidents; they too have identified that illegal medicine activity is an increasing problem. From 2015 to 2019, the PSI described an increase in incidence of 69% from 3002 cases in 2015 to 5081 incidents in 2019. 14 This contributes to a general international picture of increasing medicine falsification which has been described as a ‘wicked problem’.15,16 Furthermore, substandard medicines are regularly unintentionally released from legitimate pharmaceutical manufacturers. These known examples of SF Medicines in the legitimate supply chain are recalled by national regulatory authorities and published on open access databases in many high-income countries.17–22 These alerts and recalls are communicated to wholesalers, healthcare practitioners and, in some circumstances, the public, to alert and recall substandard medicines from the legitimate supply chain.
Operational impact of medicine recalls on healthcare provision
The UK medicine alert and recall process covers SF Medicines found within the legitimate supply chain. From a clinician’s perspective, when medicines are recalled for quality reasons, doctors can be required to prescribe, and nurses are required to administer, alternative treatments. SF Medicines impact pharmacists as they have an operational administrative burden to remove recalled medicines from the supply chain, and are responsible for sourcing alternative supplies or suggesting alternative treatments.
Lack of prevalence data to support stakeholder decisions
Despite the case study examples, and descriptive statistics mentioned above, there is a lack of accurate, understandable, and freely available data regarding medicine quality in the legitimate pharmaceutical supply chain in high-income countries. The prevalence of SF Medicine in high-income countries is largely unknown, with little available data to estimate and monitor the incidence of SF Medicines in the legal supply chain. This correlates with a lack of evidence to support balanced debate and decision making by practitioners and policy makers, regarding poor-quality medicine, issues relating to medicine recall, and its impact on practice. Systematic reviews of medicine quality studies in low-income contexts aim to amalgamate prevalence data to estimate the regional prevalence of SF medicines.6,8,23–30 However, studies examining medicine quality are rarely seen in high-income countries.This lack of data inhibits our understanding of the extent of medicine quality issues in the high income context and restricts our ability to identify and compare good manufacturing and distribution practice on an organisational and national level.
Existing policy and innovative solutions
In order to reduce the incidence of medicine falsification and streamline the recall of substandard medicine, international governments in high-income countries have introduced and implemented innovative medicine serialisation and traceability regulations.31–36 These regulations require pharmaceutical companies to serialise medicine packs and mandate that supply chain partners track and trace or authenticate medicines as they pass through the legitimate supply chain. The US approach is called the US Drug Supply Chain Security Act (DSCSA), and the European Union (EU) approach is called the EU Falsified Medicines Directive (EU FMD). 31 The EU FMD requires pharmacists to decommission medicines as they are dispensed to patients, and requires hospitals, community pharmacies and wholesalers to implement software and hardware systems to facilitate medicine authentication. 37 At the point of scanning, this software alerts the user via pop-up messages regarding the medicine’s status, for example, expired, recalled or potentially falsified, an approach which is already having an impact on pharmacy and general practice.38,39 However, these extensive policies and regulations are not the absolute solution to address poor-quality medicine. In high-income countries, regulations such as the EU FMD have been contested due to a lack of reliable prevalence data, which describe the extent of SF Medicine in high-income countries. There is a perception that falsified medicines are not a prevalent problem in high-income countries when compared to the operational and financial impact of these regulations.37–40 Many healthcare professionals in high-income countries have questioned whether an approach, such as the FMD, is proportionate to the problem. 41 To encourage the adoption of these regulations in high-income countries, we should first estimate and communicate the extent of the SF Medicine problem in the legitimate supply chain.
Illustrative case study
The aims of this case study are achieved by analysing data collected from an 8-year period (2012–2020) and discussing national medicine quality. A search of the UK MHRA website data 20 and MHRA archives was conducted. The first search assessed drug alerts and recalls for defective medicines issued by the MHRA, and the second search investigated company-led recalls. Data searches between 1 January 2012 and 1 January 2020 were performed. Company-led recalls occur when defective medicines with a ‘limited and known’ distribution, leave the manufacturing site and are recalled by the manufacturing company before they are completely distributed into the supply chain, and therefore do not require widespread communication. 42 As company-led recalls on the MHRA’s website do not backdate to January 2012, the MHRA archives were also searched. The date of the alert, name of the medicine, type of the defect, dosage form, number of affected batches, class of drug alert and recall level were recorded.
An exclusion criterion was applied in Figure 1, according to the Almuzaini et al. 42 methodology. Drug alerts and recalls issued by the MHRA are graded into four different classes (1–4) according to the level of risk they pose to public health. Class 1 relates to life-threatening defects and requires an urgent recall. Class 2 medicines relate to harmful defects, which require a recall within 48 h; although the defect can be harmful to the patient, the effect is not life-threatening. 42 Class 3 alerts are not necessarily issued for the safety of the patients and require attention within 5 days; these alerts are often concerning issues associated with marketing authorisation. 42 Class 4 alerts are ‘caution in use’ notices which do not pose a serious threat to the patients and are only there to provide advice. 42 Two types of defective medicines were identified in these drug alerts, recalls and risk communication documents issued by the MHRA; these were substandard (licensed and unlicensed/unregistered) and falsified medicines. The MHRA decided which products were falsified. The UK dataset was chosen as the example context as recalled data were freely available, and it is one of the top 10 biggest global pharmaceutical markets in the world. 43

Flow diagram of MHRA’s website and archive search strategy, and the number of drug alerts and company-led recalls between 1 January 2012 and 1 January 2020.
Using the UK as a case study, this article describes the number of falsified and legitimate but poorly manufactured or distributed medicines recalled in the UK from the legitimate supply chain in recent years. Between 2012 and 2020, there were 210 drug alerts and recalls (Figure 1; 136 MHRA alerts/recalls and 74 company-led alerts/recalls). The mean annual number of MHRA recalls was 17 during this same period. Between 2012 and 2020, there were 342 different medicines recalled as part of these 210 recalls. This includes 233 individual medicines recalled by the MHRA and 109 medicines recalled from pharmaceutical companies. The majority (n = 325) of the defective medicines identified in this period were substandard and 17 were falsified. The mean annual number of medicines recalled by the MHRA for the period 2012–2020 was 29 (Figure 1). 42
There were five cases of Class 1 drug recalls and alerts, which are the most serious cases according to the level of risk they pose to public health (Table 1). The other alerts and recalls were often due to contamination, and device or packaging defects (Table 2).
A description of Class 1 recalls reported by the MHRA from 2012 to 2020.

MHRA: Medicines and Healthcare products Regulatory Agency; NDMA: N-nitroso-dimethylamine.
A summary of defect details of all the company-led recalls and MHRA drug alerts (2012–2020; excluding falsified medicines).

MHRA: Medicines and Healthcare products Regulatory Agency; PIL: Patient Information Leaflet; GMP: Good Manufacturing Practice.
The most common reason for substandard medicine recall was contamination, which demonstrated an annual mean of 13 recalled products per year (2012–2020). Delivery defects also contributed significantly to medicine recalls during this period.
Between 2012 and 2020, 3 falsified medicine recall alerts and 17 individual falsified medicines (annual mean of 2 per year) were reported by the MHRA (Table 3)
Drug alerts relating to falsified medicine.
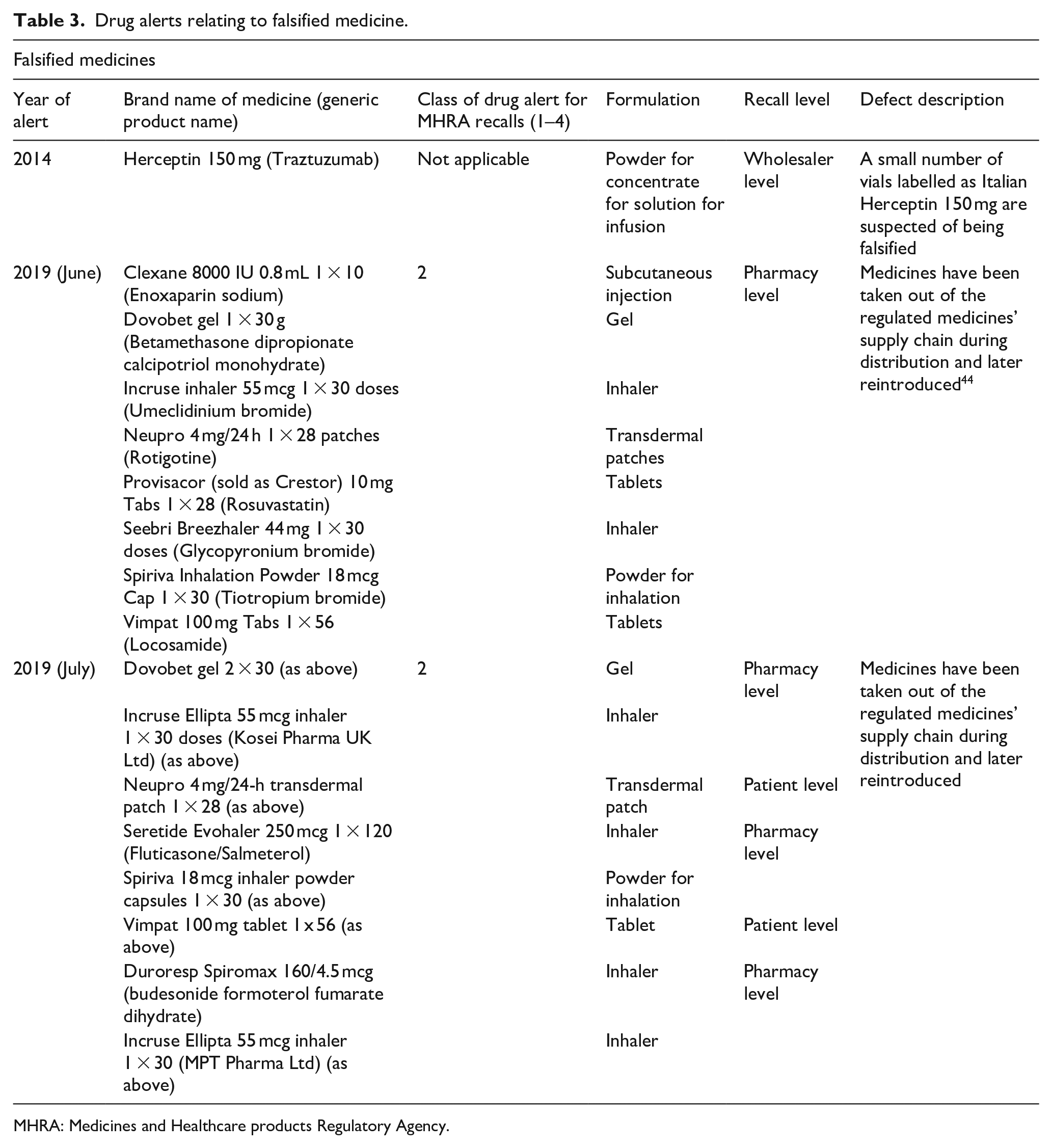
MHRA: Medicines and Healthcare products Regulatory Agency.
Medicine alerts, recalls and national medicine quality
When known SF Medicines make their way into the legitimate supply chain in high-income countries, these cases are reported to wholesalers, healthcare practitioners and the public in an effort to alert and sometimes remove these products from the supply chain. The data concerning these alerts and recalls are often stored on open access databases. This approach is adopted by many high-income countries, including Australia, Canada, Germany, New Zealand, Ireland, the UK and the United States. The data presented in this article describes the number of recalls from the UK drug recall database from 2012 to 2020. In an attempt to use these data as a proxy or surrogate marker for national medicine quality in the legitimate supply chain, some challenges were faced. This case study has identified that the mean annual number of MHRA recalls and alerts in the UK over the past 8 years is 17 (2012–2020), which equates to an annual mean of 29 individual products per year (2012–2020), including a total of five cases of Class 1 medicine recalls, and an average of two falsified medicines per year. It would be useful to compare these data with other periods of time in the United Kingdom to understand whether the problem is changing. It would also be useful to compare these data with other countries to understand how the United Kingdom’s medicine quality standard compares internationally. From here, we could establish whether other countries or manufacturers manage the issue of medicine quality in a more effective way and learn from their practice. If we could identify that there had been a change in medicine quality we could investigate through qualitative and quantitative research, why this was the case. From these data, we could perform further qualitative research to better understand the influence of changes in the regulatory authority strategy, or pharmaceutical company management and governance on medicine quality. It is clear, in the UK, that substandard medicines are a more common problem than falsified medicines 45 in the legitimate supply chain. What is not clear is whether the problem is changing or how the problem compares with other nations.
Ideally, the prevalence of poor-quality medicine (substandard and falsified) could be calculated by dividing the number of medicines recalled (the numerator) by the number of medicines used within the country (denominator). The number of medicine packs used in any given country is often available, for example, the national health service (NHS) business service authority provides prescription data which are freely available to the public. 46 However, freely available recall data typically include the product name, a description of the recall and the affected batch numbers, but does not contain data concerning how large the batch was or the number of affected packs. It also does not state what proportion of a batch went to the reporting country. Considering these points, we believe that there is currently no accurate available numerator to estimate the prevalence of poor quality medicine in a high-income country. Therefore, we call on medicine regulators to publish the total number of affected packs in each batch, delivered to each country, when recalling medicinal products from the market. This will help policy makers, practitioners and researchers to explore, estimate, monitor and compare the prevalence of poor-quality medicine in international legitimate supply chains. It may also help to inform and support future research, as well as policy and management work in the area of medicine quality in the legitimate pharmaceutical supply chain.
Limitations
Regarding substandard medicine, this study considers only legitimately manufactured medicines and defects at manufacture and distribution. This study does not consider medicines which have become substandard after manufacture and distribution due to inappropriate storage conditions within a healthcare facility. Concerning falsified medicine, this study considers falsified medicines identified in the legitimate supply chain and does not aim to estimate the total number of falsified medicines in circulation in the UK in illegitimate supply chains.
Conclusion
Substandard medicine recalls are a more common issue in the UK legitimate pharmaceutical supply chain than falsified medicine recalls. It is important to explore, estimate, monitor and compare drug recall data to understand whether the overall manufacturing and distribution of SF Medicines is changing in high-income countries. However, freely available drug alert and recall data in its current form are not a reliable estimate measure of medicine quality due to the lack of a suitable numerator. We propose data sharing to facilitate comparative medicine quality studies in legitimate pharmaceutical supply chains. We call on policy makers and regulators to work with pharmaceutical companies to publish the total number of affected medicine packs in each batch, entering each individual country when publishing drug alert and recall data.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Oxford University Hospitals NHS Foundation Trust has supported Dr Naughton’s time in preparing this manuscript.
Ethics and consent statement
This article conveys the opinions of the authors. Neither approval from an ethics committee nor informed consent is required.
