Abstract
Background:
Just in Time (JIT) and Lean manufacturing are concepts that originated in the automotive industry and were then adopted by pharmaceutical and biopharmaceutical companies during the 1990s. However, the Covid-19 pandemic and the urgent demand for pharmaceutical treatment challenged JIT and Lean manufacturing processes. Production of Covid-19-related medicines increased, putting pressure on global supply chains and operations. This also hindered the production of medicines using the same or similar materials. Thus, questions are raised concerning JIT and Lean supply chains in the pharmaceutical industry.
Objectives:
The present study aimed to explore (1) if material and supply constraints occurred due to the Covid-19 pandemic, (2) how companies were impacted and managed and (3) if changes are required to future proof the JIT supply chain approach for future global events.
Design:
A mixed-method cross-sectional survey design was used and focused on material supply, qualification and validation in Irish pharmaceutical manufacturing sites.
Methods:
Employees working in the Irish pharmaceutical manufacturing industry were recruited using convenience sampling through online advertisement using the social media platform ‘LinkedIn’. Quantitative data was analysed using percentages and qualitative data from free-text responses were used to add context to the quantitative survey questions.
Results:
A total of 41 participants were recruited. The results suggested that the pandemic had a negative effect on material availability according to 81% of participants. This translated to delays or stoppage of production activity and was mainly handled by sourcing new materials (70%). To cope with future global crises, 60% of participants recommended more flexibility in future validation processes while 78% of participants acknowledged the importance of validating additional suppliers. A hybrid model of manufacturing and supply chain management was also a preferred approach to exclusive Lean and JIT (42%).
Conclusions:
The production of non-Covid-19 medicines was adversely affected by the Covid-19 pandemic, but the pharmaceutical industry in Ireland demonstrated resilience and collaboration in response to these challenges. This study suggests that the JIT and Lean manufacturing model should be adjusted to ensure medicine supply chains are not disrupted during future global events.
Keywords
Introduction
JIT and Lean manufacturing
Early innovations of efficient manufacturing production operations have origins in the motor and textile industries. 1 At the start of the 20th century, the Toyota Motor Company pioneered the Toyota Production System (TPS), 2 a Lean method illustrated as the TPS house, which depicts the foundation, pillars and goals of the model (Figure 1). The two main pillars of the TPS system are Jidoka and Just in Time (JIT). Particularly, Jidoka is a process where an operation stops automatically upon defect detection, and JIT is a concept that offers efficiencies by minimising the raw material input and excess outputs of a process. JIT offers waste reduction, space saving in warehouses, low excess stock levels that can depreciate, and maximises the potential for reduced costs and increased profit if executed and maintained correctly. The efficiencies offered by the Lean model were obvious as Lean production focused on all aspects of waste, over-production, over-processing, errors, equipment reliability and inventory levels. This offered an insight into how agile working could benefit the western style model of mass production. Books like ‘The Machine that changed the World’ 3 brought these TPS production innovations to a mass audience that adopted its philosophy into their organisation. Lean manufacturing became a global phenomenon, transforming the process from a mass production mentality to a ‘consumer demand driven’ and ‘focus on efficiency’ model.
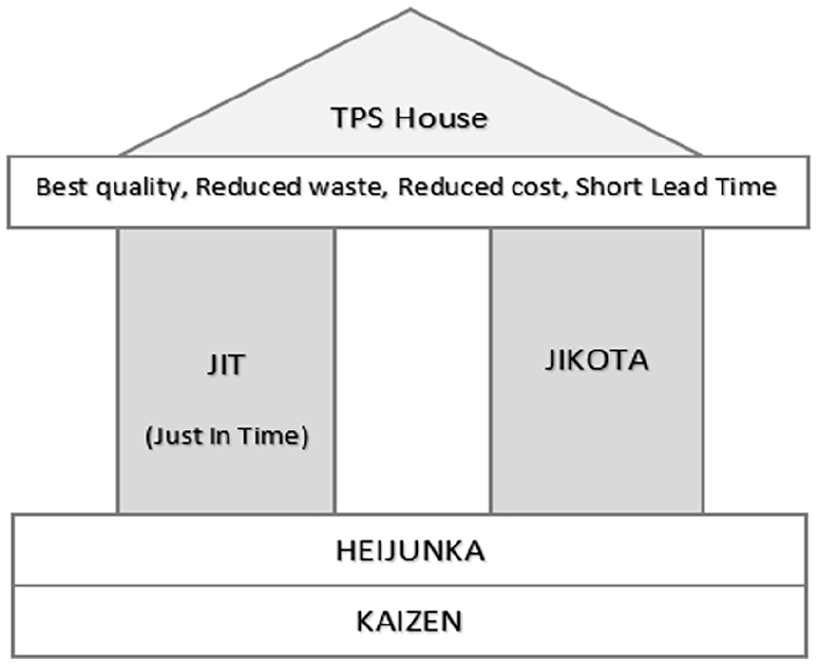
TPS house. 4
The global pharmaceutical industry, biopharmaceutical industry and MedTech industry are no different to most globalised industries. Supply chains are large, complicated, multi-layered and interdependent on each other. It is in essence an ecosystem of business partnerships, forged by a demand for materials which will ultimately support and produce medicines that are essential for both individuals and healthcare systems. 5 Although the pharmaceutical industry is not the first industry to have taken Lean manufacturing on board, the switch to the cost-saving principles of this philosophy has gathered momentum since the turn of the 21st century. 6 The emergence of a more competitive marketplace, the loss of patents on blockbuster sales products and the desire to maximise profit 6 increased created pressures on the pharmaceutical industries that have encouraged change. This environment highlighted the value of new innovation which in turn led to increased pressure for the pharmaceutical industry to adapt and become leaner and more innovative. 7 More and more drug companies are seeing the benefits of adapting to such a model. 6 More specifically, Lean Six Sigma methodologies that are in line with Operational Excellence have been found to be a beneficial and effective toolset for problem resolution and process enhancement in pharmaceutical manufacturing facilities. 7
Complexity
Research in the pharmaceutical industry shows a mixed picture of how successful adopting JIT manufacturing and Lean principles has been. It has shown that the benefits of Lean are several,7 –10 including its compliance with current Good Manufacturing Practices (cGMP). 8 Yet, there is also evidence of several pitfalls. 11 Geller 6 suggests that the value of JIT is observed in companies with low complexity and low variability, which aligns with its success in the automotive industry. Yet, pharmaceutical production processes, characterised by high complexity and high variability, mean it is harder to implement Lean. Particularly, large companies find easier paths to Lean manufacturing based on scale of operation, while smaller companies find it more challenging. 12
Regulation
There is added complexity to Lean implementation in highly regulated environments such as the pharmaceutical industry, as any changes to practice must also adhere to national or international regulation. 11 Change can often be considered cumbersome, as the rewards can be outweighed by the effort. 13 Indeed, regulation is demonstrated as a major factor in the decision to implement Lean with some researchers seeing an uneven fit between the two, and regulation as a barrier to implementation of Lean,11,14 while others see the two as a natural fit, because if a process can be simplified and made more efficient then it is easier to understand and thus regulate.9,15
Supply chain predictability
Managing market demand and supply chains is also challenging. It is noted that predicting market demand can present difficulty, especially since pharmaceutical manufacturers are known to sell to wholesalers and not directly to the customer, which places them one step away from accurately measuring the market demand. 14 Wang and Jie 16 and Bastani et al. 17 focus on supply chain uncertainty in the industry, acknowledging that a supply chain can also come under pressure from disasters like an earthquake, a tsunami or a hurricane. The ripple effects of such events can cause major supply chain disruption, due to the interlinking nature of the supply network. Delays at one supplier can lead to a knock-on delay at the next site and so on.
The COVID-19 pandemic and supply chains
Exposure to major global events like the Covid-19 pandemic provides a challenge to the successful operation of Lean and JIT manufacturing systems. 18 Evidence of this impact was made prominent over recent years of production (2021–2022) as global supply chains were squeezed to the point where some companies have had difficulty making product and others have had to stop or pause manufacturing. At the same time, the continuous monitoring of drug shortages as part of ongoing maintenance of healthcare systems during the Covid-19 pandemic became a critical task. 19 Regulatory agencies like the US Food and Drug administration (FDA) and the European Medicines Agency (EMA) expected and observed supply chain disruption, which caused lack of availability to consumers.
Early-stage development and manufacturing of Covid-19 vaccines and treatments for the global pandemic was initiated in early 2020. An initial unprecedented level of scientific sharing of data on the virus ensued its genetic makeup, characteristics and traits as a disease were shared. 20 These early studies suggested that medicines that were known to be effective against respiratory illnesses and infections could have an impact on Covid-19 disease progression. Thus, demand grew for non-Covid-specific anti-inflammatory treatments. 21 Moreover, as the race to develop a vaccine candidate for the virus intensified, access to the type of equipment needed in modern biopharmaceutical production processes grew. Single-use plastic products and bioreactors, filtration devices and raw materials needed in these processes grew scarce. 22
The issue of supply chain disruption has multiple root causes, and it is difficult to pinpoint a single definitive one. The logic of JIT manufacturing relies on prediction and planning, which in turn relies on a predictable system. When an event like the Covid-19 pandemic disrupts normal supply chain patterns, it is harder to predict and plan. Therefore, we propose the examination of the elements of the systems that create unpredictability. Taking a closer look into some of these causes, we can build a picture of how the pharmaceutical industry is set up to deal with pandemic-like crises and hopefully theoretically protect pharmaceutical supply chains against such events.
Research gaps and study motivation
The literature review shows conflicting views on the suitability of Lean and JIT approaches for the complex and highly regulated pharmaceutical manufacturing sector; yet, these approaches have been incorporated in pharmaceutical production. It would be interesting to review the potential benefits and/or challenges of such approaches in a time of global crisis (Covid-19 pandemic) that shook the status quo of pharmaceutical manufacturing and added further to existing complexity and regulations. The proposed supply chain predictability challenges in times of disaster make this attempt even more significant, in preparation for future turbulences.
Aims and objectives
The aim of the present study is to assess the impact of the COVID-19 pandemic on the Irish Pharmaceutical Manufacturing Sector. The study’s objectives are to explore the material availability during the pandemic and its effect on the non-Covid-19 medicine supply chain. Also, it aims to explore how companies managed and recovered from the crisis with a focus on the Lean and JIT framework. Finally, it aims to explore if current processes can be improved in preparation for future global events, focusing on the areas of material supply, qualification and validation.
Methods
Design
The present study adopted a cross-sectional design using an anonymous online survey administered to the pharmaceutical ecosystem, in Ireland. The survey was based on existing survey methodology theory.23,24 The questionnaire approach was used to increase reach to a small and difficult to access population of study participants.
Participants
Participants were highly skilled professional adults working in Irish pharmaceutical manufacturing sites.
Recruitment
The study used convenience sampling for participant recruitment and followed the social media recruitment approach used by Middleton et al. 25 and Stoke et al. 26 Social media platforms allow rapid recruitment with minimal financial resources and promote snowballing. Convenience sampling also benefits the exploration of emerging research areas where there is little or no existing literature, while promoting engagement.
Response rate
Due to the voluntary nature of the survey, and the online audience, a response rate cannot be calculated. In the case of the present study, the ease of access to a wide spectrum of hard-to-reach candidates from the pharmaceutical industry was argued to outweigh the inability to calculate a response rate.
Materials
Google forms were used to administer the online survey encompassing 16 questions to the target audience, with an option for further clarification or discussion after each of the core questions. The final two questions of the survey were free-text response options (see Appendix 1 for survey questions).
Common method bias
Participants were given a detailed information sheet with the aims of the study and how their data will be used to motivate them for more accurate responses. Also, the questionnaire was relatively short with no repeated measure to increase engagement with the survey. Scale items were phrased in a straightforward and clear way to avoid ambiguity and an option for free-text complementary answer was provided. Finally, the survey incorporated items with various scale properties (e.g. strongly agree–strongly disagree, strong positive effect–strong negative effect, multiple answer questions, free-text answer questions). These measures were taken to counteract common method bias that can be present in studies with survey designs. 27
Procedure
Data collection took place in autumn 2022. Participants received an information leaflet and invitation letter which outlined the study and the aim of the survey (see Appendix 3 for leaflet and invitation letter). Survey completion was estimated to take 15 min and was active for 2 months. The virtual data was stored in the Google Forms interface prior to data entry and then exported onto a password-protected Excel file on a secure network.
Analysis
Question by question analysis was performed using Microsoft Excel. Each quantitative question was analysed, and percentages were produced. Free-text responses were thematically analysed and reported alongside the trends from the quantitative questions to add further context to our findings.
Ethics
The study received full ethical approval from the School of Pharmacy and Pharmaceutical Sciences Research Ethics Committee, Trinity College Dublin (Application number 2022-06-01). Prior to the data collection process, a Participation Information Sheet and a consent form (Appendix 3) were built into the online survey as links and informed participants about their rights and the full oversight of the study. Participants were informed that their participation was voluntary and that answers would be anonymous. It was anticipated that no participant was at risk of physical or psychological damage as a result of the study, and no deception was present.
Results
Participant and organisational background
A total of 41 participants from mostly Irish Pharmaceutical Manufacturing sites responded to the survey. The initial four survey questions were aimed to establish the participant and organisational background (Figures 2–5).

Study participant and organisational characteristics: question 1.

Study participant and organisational characteristics: question 2.

Study participant and organisational characteristics: question 3.

Study participant and organisational characteristics: question 4.
Geographically, the Leinster region provided the largest sample set. This is expected as the Leinster region accounts for majority of the Irish population. This is additionally offset by the variation seen in the category of ‘product produced at the facilities’ where respondents originated and with good variation of the types of substances produced and evenly spread. The majority (66%) (Figure 5) of facilities were also confirmed as producing either non-Covid-19-related medicines or both Covid-19 and non-Covid-19 products. The quality area featured strongly with 63% (Figure 3) of respondents being from a quality background, but good representation from other areas of the pharmaceutical industry were also observed. The sample set provides a suitable sample for the purposes of this study, which aims to assess the impact of the Covid-19 pandemic on pharmaceutical supply chains and the production of non-Covid-19-specific medicines, with a focus on Ireland.
Manufacturing and Covid-19 impact
The next series of survey questions aimed to understand to what extent the pharmaceutical industry has been impacted by the pandemic. Questions were asked to explore how the pandemic has impacted production output, if the pandemic has led to a need to source new materials, or to a change in the way organisations source new materials or validate sourcing approaches (Figure 6).
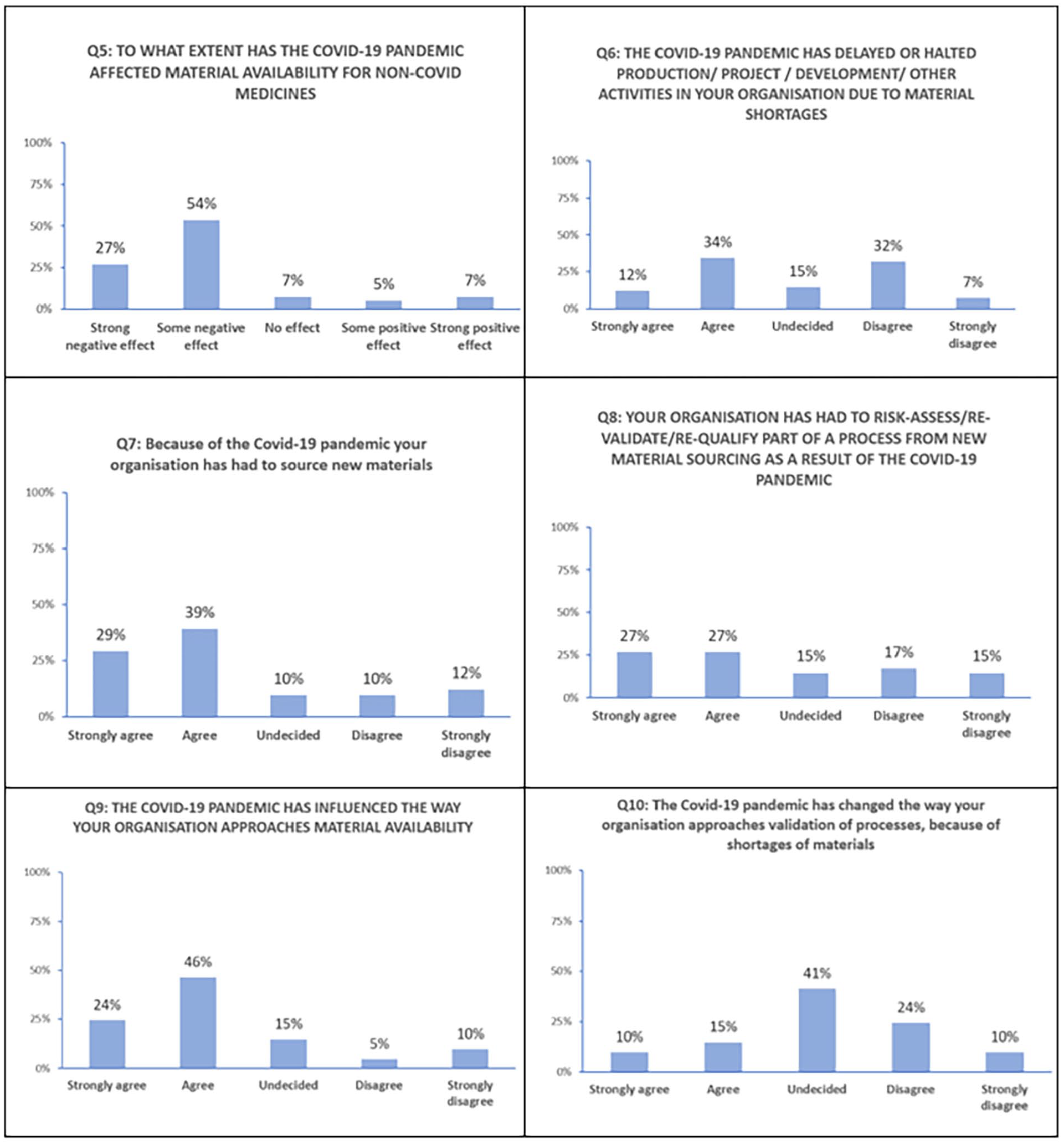
Survey questions concerning the impact of pandemic.
For questions Q5–Q10 in Figure 6, a clear trend emerges regarding the impact of the pandemic for the survey participants. There has been a negative effect in terms of material availability during the Covid-19 pandemic, with 81% (Figure 6, Q5) of the participants noting either a moderate or strong negative effect. This was supported by some qualitative quotes from participants also: ‘‘Caused a hard stop in facility for 6 months due to component shortage’’ (Participant 15)
This negative effect did not always translate into delays or stoppage of production activity, as approximately half of the respondents who acknowledged the negative effect did not report production delays. A great deal of effort and time was involved in sourcing new materials to offset the shortages as declared by almost 68% of respondents (Figure 6, Q7). There is a strong positive response (70%) (Figure 6, Q9) to the question related to whether the pandemic influenced how their organisation approached material availability but less of a conclusive trend regarding approaches to revalidation and future validation. This is supported by participant quotes (see Table 1 in Appendix 2), as below: ‘‘Due to no availability of critical reagents, material or components from qualified suppliers/vendors, production and shipping schedules had to be rearranged. In some cases, alternative material/components had to be sourced and qualified with additional quality/technical risk assessments thus an increase in change control activity’’ (Participant 22)
Organisational challenges, solutions and future direction
The qualitative questions in free-text boxes (see Table 3 in Appendix 2) gave the participants the opportunity to describe challenges experienced in relation to the manufacturing and supply of non-Covid-19 medicines during the pandemic, while reflecting on technical, political, social or emerging challenges. In one case, they were questioned regarding ideas to reduce the risk of shortages due to supply chain issues moving forward. These questions have provided similar themes seen in the previous question sets, such as the challenges of working with China, who is a major manufacturer of active pharmaceutical ingredients (APIs), due to their strict restrictions: ‘‘Hesitancy to work with China based CDMO’s due to travel restrictions. Unable to perform tech/quality visits’’ (Participant 9)
Other themes included the disruption caused to the supply chain and the theme of nationalised approaches by certain states created a challenge in terms of supply: ‘‘De-prioritisation due to Covid-19 vaccine supply. Also challenges with nationalization approach by certain states’’ (Participant 11)
From a social aspect, the stress of keeping processes going appears evident in the responses, along with an acknowledgement of the need to keep graduates coming into the industry. Q16 looked more at the solutions side, moving forward. Some interesting themes are mentioned ranging from better planning, better use of Lean tools, dual sourcing, increasing stock levels and increased bulk ordering. Also mentioned is the desire for flexible working arrangements to allow for continuity of work: ‘‘Adapting to working from home in relation to QP release of materials’’ (Participant 19) ‘‘Lack of staff due to sickness, social distancing affecting day to day meetings, group projects held off due to working from home or implemented shift within certain groups’’ (Participant 12)
The final set of questions (Figure 7) looked at how organisations considered protection against future shocks to the global industry like the Covid-19 pandemic, exploring how organisations would look to protect future material availability. This is also surveyed in the context of Lean practice and JIT manufacturing and the survey’s aim was to question whether this approach would be maintained or adapted in future years.

Survey questions concerning organisational future direction.
The responses indicate that the immediate trend seen in the previous data sets of sourcing new or replacement material will continue with most respondents (78%) (Figure 7, Q11) looking to source new material by validating additional suppliers. Further to this, almost 60% (Figure 7, Q12) of respondents agreed that their organisations will look to build more flexibility or robustness into future validation processes to cope with future events: ‘‘Organizations are more aware now and will take other options into account whereas they may not have previously’’ (Participant 8) ‘‘We always try to validate with flexibility’’ (Participant 20)
With reference to the previous data set, it is shown that there is more willingness to modify the ‘validation approach’ going forward. As seen in data set Q5–10, retrospective revalidation did not score as highly. Most organisations reported that they operate a Lean/JIT production/supply chain model and 42% (Figure 7, Q14) of respondents confirmed that they would look to adapt a hybrid model, JIT for some products but not others: ‘‘Continue the lean method with variability there in the case where lean cannot be adhered to, and bulk ordering is required’’ (Participant 12) ‘‘Hybrid work practices’’ (In relation to Lean and JIT) (Participant 30)
Whether this represents a change in how Lean/JIT is perceived by the pharmaceutical industry remains to be seen but what is evident is that the Covid-19 pandemic has provided enough of a shock to the supply chain network to encourage organisations to think about ways to strengthen the supply chain and ensure continuous production. An example of some of the commentary from respondents can be seen in Table 2 in Appendix 2.
Response rates
Response rate/non-response rate as described in the survey plan is not possible to determine, however, the extent of the sample audience and variety of responders can be determined from the data obtained in the initial survey questions and from the analytics on LinkedIn. A total of 937 impressions were made on the survey post on LinkedIn. Confirmation of the variety of potential responders was gained from the analytics on LinkedIn, illustrating that not all responders come from the same company, region or working background. The variety of responders can be confirmed by the answers to the initial four questions from the survey, which shows an adequate spread of data.
Discussion
Organisational impact: How manufacturers managed and recovered
The COVID-19 pandemic caused an obvious strain on the healthcare systems of most countries, 28 but supply chains were also put under severe strain. 29 The present study corroborates such claims with the majority of participants (81%) declaring that the pandemic had a negative impact on material availability which in some cases (almost 50%) delayed or haltered production. Fonseca and Azevedo, 30 also highlight how poorly insulated the global supply chain was against events like the pandemic. Shortages have always been common for a variety of reasons, but the pandemic amplified this problem and introduced several new factors that added further strain; for example, increased demand for certain components, lockdowns, difficulty in cross border travel and countries holding onto or stockpiling medicine supplies.29,31 Interestingly, the results showed that in the Irish pharmaceutical landscape, a lot of this impact was mitigated by companies sourcing alternative materials (68%). This enabled the full impact of the pandemic to be softened but did result in a significant increase in organisational workload and planning.
Strategic changes to practice
As seen in detail in the results section, the study participants provided ideas and solutions to the challenges faced during the Covid-19 pandemic. These solutions illustrate the need for strategic changes to practice insulating pharmaceutical manufacturing and supply chains from future crises.
Better planning and organisational management; use of operational excellence tools
Because of the added strain in organisational workload caused by the pandemic, participants highlighted the need for better planning and better use of the Lean tools. This is corroborated by previous literature. More specifically, better monitoring of supply chain weaknesses18,32,33 and better tracking of medicine stock levels 34 has been suggested from various scholars. Moreover, a study conducted by McDermott et al. 35 in 2021 highlighted the benefits of operational excellence methodologies and Lean and Six Sigma tools in the healthcare system. Flexible working was also highlighted by the study participants, which has been supported by the Information and Communication Technologies (ICT) as illustrated in an Indian study by Sharma et al. 36
A move away from Lean/JIT processes
The study provided some interesting suggestions for industry solutions, some of which suggest a move away from Lean principles and JIT manufacturing. The same idea is discussed in the study by Gereffi 31 focusing on personal protective equipment (PPE) products in the United States during the Covid-19 pandemic, in which it is suggested that the prevalence of Lean/JIT adaptation aiming to increase efficiency and reduce cost with reduced inventories has left the global supply chain in a fragile state. This notion is being highlighted among several studies.31,37 –39 However, Thakur-Weigold and Miroudot, in a study published in 2023, argue that the ‘too-lean’ concept is actually a myth that has been spread after the Covid-19 pandemic and the Russian invasion of Ukraine. They argue that there is no concrete scientific evidence to support such claims and that Lean/JIT processes aim to minimise waste by targeting redundant or faulty elements of production, transportation and supply. 40
Dual sourcing of materials
In addition, the concept of dual sourcing of materials arose as a solution to shortages by the study’s participants. A plus one approach, which is a way to reduce risk and diversify the supply chain network, was initially proposed to reduce the overexposure to the Chinese market and encourage investment in alternative supply chain options from other countries and markets. 41 Multisourcing of materials has been also suggested by previous literature, in an essay discussing the impact of the Covid-19 pandemic on the global supply chain and pharmaceutical manufacturing by Hausmann in 2020. 37 Yet, dual sourcing and validating a process for multiple input options of the same material add an obvious cost to the manufacturing profile of a medicine and this is likely to result in an increase in cost and complexity which goes against the principles of the Lean manufacturing model. 2 This approach does, however, reduce the risk of manufacturing shutdown and is being pursued by multiple organisations. Whether this is a shift in the Lean mindset or whether the Lean mindset will have to shift to catch up with the reality of global events remains to be seen. What is interesting though is that by its very nature Lean and continuous improvement are always adapting so events like the Covid-19 pandemic may speed up this evolution. These approaches could help to formalise some of the activities seen in the Irish pharmaceutical manufacturing setting and add further structural resilience to Irish and international pharmaceutical manufacturing systems.
Sustainable local production
The concept of sustainable local production featured in the survey, similar to the ‘One Roof’ 11 principle. The suggestion of a more regional supply chain was also highlighted by Hausmann in 2020. 37 This may be a logical reaction to global disruption but may not be practically possible. If we imagine the concept of the ‘One Roof’ principle of the Renault car company, which was based around a localised production unit, such a system might be more insulated from Covid-19-related supply problems. Indeed, there is a sustainability argument to be put forward about the nature of pharmaceutical production. In the future world, would more value be placed on a raw material that while more expensive, represents better value to the environment and assured supply? The causes of material and supply constraints may just be as simple as that. However, the world’s resources are not evenly distributed, and geographical and geopolitical factors will always play a role in the supply chain network.
Pivoting
Sourcing new or replacement materials and validating additional suppliers appeared as recommended future steps by the study participants. Socal et al. 42 proposed the introduction of a metric-based system for manufacturers, in which scores are assigned based on supply chain resilience and ability to meet demand. This could encourage companies to promote more safety stock into their manufacturing plans, as suggested by the study participants (i.e. increase stock levels and bulk ordering). Such solutions have also been suggested from various scholars.18,34 This will require robustness into future validation processes which can be facilitated by greater supply chain transparency. Literature has highlighted that greater supply chain transparency may be required to be able to monitor potential shortages more closely and put in place measures to combat supply chain stresses from events like the Covid-19 pandemic. China and its dominant market share in the manufacture of APIs and India’s large-scale manufacturing of generic medicines which predominantly use API from China is cited as an example of a supply chain that is not fully open, or transparent for analysis. 29 More cooperation in the area of mapping and creating a database of API manufacturers could also help speed up the approval of new sources of API. 42
Theoretical and methodological implications
There are several advantages of JIT and the system is overall successful but when global events or indeed significant local events happen, this can disrupt a supply chain. If the pandemic had not occurred, the weaknesses in the system may not have been exposed to the extent that they were. With every crisis, there is an opportunity to develop and grow new ideas. 18 The Covid-19 pandemic is undoubtedly a moment in time where the pharmaceutical industry and its Lean journey is under scrutiny.
The TPS ‘House’ 4 as a metaphor is rigid, secure and stable. We suggest that the challenges the pandemic imposed highlighted the need for flexibility and better use of the Lean tools rather than discrediting them. Perhaps this could be achieved by adding flexibility to the ‘house’ metaphor and diagram by making the foundations of this house more flexible in times of a crisis. The way Lean methodologies and tools are conceptualised and used in the pharmaceutical business model could be adjusted to align more with flexibility in the times of these significant contextual shifts. 43 As mentioned in the study by McDermott et al., 35 such events are so rare (0.001%) that do not justify the abandonment of a theoretical framework that has advanced business practice. Our theoretical contribution is simply underlying the importance of flexibility in the Lean/JIT model. We also propose that the framework should be re-examined to establish how this flexibility could be incorporated into the TPS House model.
Conclusion
It can be concluded that the pharmaceutical industry is a firm practitioner of the Lean and JIT principles and it experienced material and supply chain issues associated with non-covid medicines. During the pandemic, the industry pivoted and adapted its standard supply chain business model practices to manage the impact of the pandemic. The Covid-19 pandemic has shocked this sector; however, Irish pharmaceutical manufacturing demonstrated evidence of resilience throughout the pandemic. The pandemic has also made the industry evaluate its future business model, and a further evolution of the supply chain model may surface. In the spirit of continuous improvement, we encourage future research to explore how the modification of JIT/Lean model will be achieved, adding flexibility to the TPS house, to ensure that public health is not disrupted during future global events like the Covid-19 pandemic.
Limitations and future scope
This study has several limitations; the number of participants is limited and is representative only of the Irish pharmaceutical industry. We suggest that similar research studies with larger sample sizes in different geographical locations would be useful to identify if similar effects were present in larger countries. Moreover, the self-reported survey design has its own limitations and even though it is suitable for initial exploration of research topics, future studies could use more complex research designs that use multiple data sources to explore the proposed variables.
