Abstract
Background:
Pharmaceutical drug recall is a relentless issue that is composed of multidimensional criteria. The distinct criteria that contributed to drug recalls have been identified in previous literature; however, there is limited information regarding the causal relationships between each criterion. Highlighting key influential aspects and criteria of pharmaceutical drug recall is critical in addressing this ongoing issue and promoting patient safety.
Objective:
The objective of this study is as follows: (1) identify critical criteria of pharmaceutical drug recalls for improvements, (2) determine the interrelationships among the criteria, and (3) define the causal relationships of pharmaceutical drug recall and provide theoretical insights and practice recommendations to minimize risks associated with pharmaceutical recalls and maximize patient safety.
Design:
This study proposes five aspects and 42 criteria to identify the impact of pharmaceutical drug recalls on patient safety by evaluating the interrelationships between the criteria by employing the fuzzy decision-making trial and evaluation laboratory method.
Methods:
A group of 11 professionals across the pharmaceutical industry, hospitals, ambulatory care, regulatory authority, and community care settings were selected for interviews.
Results:
Risk control is the influencing aspect of pharmaceutical drug recalls that has the most substantial impact on risk assessment and risk review; it generates medium effects on risk communication and technology. Risk assessment, risk communication, and risk review demonstrated comparative weak interrelationships, while risk communication exhibits a weak unidirectional effect on risk review. Finally, risk assessment exerts a weak influence on technology application and development. Product contamination, product subpotent or superpotent, injury to patients, product not sterile or impure, and system detectability of hazards have the strongest influence in the causal group of pharmaceutical drug recalls.
Conclusion:
The study shows that risk control drives risk assessment and risk review in the pharmaceutical industry manufacturing process. To achieve patient safety, this study suggests focusing on risk control strategies, as this aspect displays the most substantial effect on influencing other critical risk management aspects such as risk assessment and risk review.
Introduction
Pharmaceutical drug recalls are significant factors of therapy discontinuation, limited access to care, and adverse events that constitute persistent threats to patient safety. Pharmaceutical drug recalls often occur as a consequence of concerns over product defects and deficient quality that has posed harm to the safety of public health. Studies indicated approximately half of all pharmaceutical product market withdrawals appeared within 2 years of a product’s introduction to market. These numbers have created significant mistrust from the public and impacted on treatment continuity. 1 In the United States, clinically significant drug recalls occur nearly every month, affecting thousands of pharmaceutical supplies distributed across the country or internationally. 2 In October 2012, the United States Food and Drug Administration (USFDA) recalled thousands of vials of injectable corticosteroid medications that were caused by fungal contamination. 3 The contaminated products resulted in 753 reported cases of fungal meningitis and 64 deaths in multiple states.3,4 Risk mitigation actions were carried out by health regulatory agencies and public health authorities; however, safety events with various magnitudes of patient impact were continuously seen in different parts of the world. 5 The medication withdrawal rates due to safety events has not improved in the past 60 years, particularly in the African continent where the rate of drug recalls per country was very low (1.17%) in comparison with the western countries (6.18% in Europe and 5.83% in North America). 6 Most recently in 2020, the USFDA announced a substantial recall of ranitidine and metformin extended-release form that derived from contamination of N-nitrosodimethylamine (NDMA), a carcinogenic substance that increases the risks of cancer under high-level chronic exposure.5,7 This resulted in both short-term and long-term impact on patient safety, treatment access, and public trust around the world. While patient safety is being recognized globally as a top priority that requires a collaborative and coordinated response, it is imperative to address the issue of pharmaceutical drug recall promptly. 8
Pharmaceutical drug recall is a persistent issue that is composed of multidimensional criteria. Sandle 9 provided a comprehensive analysis for current state of pharmaceutical recalls across the globe, including recall trend, the economic impact, and predictive models for recall assessments. The distinct criteria contributed to recalls had been identified in previous literature; these include but are not limited to contamination, mislabeling, packaging issues, adverse reactions, incorrect potency, substandard and falsified components, and cross-contamination.3,10–12 These issues, specifically substandard and falsified medications, existed worldwide and impacted high-income and low- and middle-income countries.13,14 The traditional analysis approaches adopted in previous literature are limited to the consideration of direct effects or single directions of criteria; oftentimes, these approaches fail to identify the interrelations, interdependency, and causal relationships between each factor.15,16 Meanwhile, the International Conference on Harmonization (ICH) had issued pharmaceutical practice guidelines on the systematic approach for quality risk management to facilitate shared understanding of risk management strategies throughout the product lifecycle. 17 This guideline established critical infrastructure of the pharmaceutical quality risk management process to ensure patient safety and was issued as Guidance for Industry by the USFDA and adopted by the European Commission and became part of the European good manufacturing practice (GMP). 18 Nevertheless, highlighting the most critical aspects of this process is a key to addressing the ongoing issue of pharmaceutical drug recalls impact on patient safety.
In addition to a lack of well-defined causal relationships between recall criteria and a highlight of pivotal aspects for risk management strategies, there are significant perception gaps between consumers, health care professionals, and pharmaceutical industries’ apprehension of the causes and effects of drug recalls. 3 Understanding the causal relationships helps determine the positive and negative associations between each attributing factor. 19 Hence, despite the perception gaps among patients, clinical practitioners, pharmaceutical industries, and regulatory agencies, it is imperative to utilize an effective tool to establish the causal effects and interrelations between each contributing factor and identify best practices for policymakers and all stakeholders to ensure patient safety.
This study proposes fuzzy Decision-Making Trial and Evaluation Laboratory (Fuzzy DEMATEL) method to interpret the causal relationships and interrelationships between the key attributing criteria of pharmaceutical drug recalls 20 based on the experts’ linguistic preferences. Linguistic preferences can embrace fuzzy human assessment and represent more comprehensive evaluation. The objective of this study is threefold: (1) identify critical criteria of pharmaceutical drug recalls for improvements, (2) determine the interrelationships among the criteria, and (3) define the causal relationships of pharmaceutical drug recalls on patient safety. This study aims to provide theoretical insights and practice recommendations for clinical practitioners, pharmaceutical industries, and regulatory agencies to minimize risks associated with pharmaceutical drug recalls and maximize patient safety. The theoretical contribution is to identify and conceptualize a set of mediation recall criteria, present a hierarchical model that extends the current ICH guidelines, and determine strategies to address the ongoing issue of pharmaceutical drug recalls on patient safety. Practice recommendations are provided with important implications for clinical practice, pharmaceutical industries, and policymakers to maximize the safety of public health. This article is organized as follows: “Introduction” section introduces the significance of this research through literature background and identifies the research gap and the proposed research method. “Literature review” section reviews the literature on medical recalls and the decision criteria. “Methods” section elaborates the method and data analysis. “Results” section discusses the analytical results of criteria and figures. “Discussion” section depicts the theoretical and managerial implications, and finally, “Conclusion” section discusses the potential future extension of this study.
Literature review
Previous literature highlighted contamination, mislabeling, adverse reactions, incorrect potency, wrong dose or release mechanism, and cross-contamination as the major contributory causes of the overall pharmaceutical drug recalls.2,3,6,10 These criteria were identified via retrospective data analysis, the review of USFDA enforcement reports, and utilizing statistical tools; however, the interrelationships among the complex drug recall criteria have not been elucidated, and there were no existing studies that adopted analytical models to identify the causal interrelationships among recall criteria in the pharmaceutical field. This study proposed five recall aspects containing 42 recall criteria. The aspects include risk assessment (A1), risk control (A2), risk communication (A3), risk review (A4), and technology (A5), as indicated in Table 1.
Pharmaceutical drug recalls aspects and criteria.

IoT: Internet of Things; 3D: three-dimensional
Risk assessment
Risk assessment (A1) plays a vital role in the identification, analysis, and evaluation of risks associated with the exposure to hazards.17,21 The USFDA, European Medicines Agency (EMA), and many other health authorities strongly endorse integrating risk assessment in the decision-making process pertinent to product development, manufacturing, and distribution to ensure patient safety and product quality. 18 Twenty-one criteria have been identified in the risk assessment aspect (A1): nonsterility and failed impurities (C1) entail microbial contagion of products were described as the common causes of pharmaceutical drug recalls; 10 however, the USFDA enforcement reports were not analyzed directly in the studies to reach this conclusion. Wang et al., 2 on the contrary, classified the reasons for drug recalls in a 30-month analysis study. These results indicated product contamination (C3) accounted for 50.1% of drug recalls, 21.87% were due to product mislabeling (C4), 9.81% were attributed to injury to patients (C15) that caused harm or hazard to patients, including but not limited to adverse drug reactions, 7.06% due to defective products such as product unsealed or improperly sealed (C5) or product missing or unusable product (C6), and 6.21% were related to incorrect potency (C2) and ineffectiveness of the product (C7) that failed to exert pharmacological effects. 3 Abraham 17 and Azghandi et al. 22 argued that while product recalls are common for pharmaceutical products, the characteristics and patterns of recalls can vary in terms of a lack of product supply (C8) and the disruption of product supply (C16); therefore, outlining a supply chain that remains resilient to different types of interference remains a challenge of risk management.
Nagaich and Sadhna 10 also identified improving compliance with industry and government regulations and standards (C9) and increasing process efficiency (C11) as critical criteria to overcoming drug recalls. Meanwhile, product rejection (C10) in the context of failing to receive product approval after submissions and products marketed without an approved new drug application (NDA) or abbreviated new drug application (ANDA) (C10) are considered critical criteria for drug recalls. 10 Mollah et al. 18 described poor process yield (C13) as an indicator of the manufacturing process performance and reflect the overall proportion of products that pass through the compliance check. In addition, risk assessment is used to examine systematic hazard under various conditions such as the system's ability to evaluate the intended use (C17) or unintended use (C18) of a product and normal and fault conditions within the manufacturing processes (C19). 18 Finally, according to ICH risk assessment is used to determine the magnitude of the risk, which includes determining the risk is acceptable or unacceptable risks (C20, C21) perceived by the entity. 17
Risk control
The second aspect is risk control (A2), which is the process of mitigating and reducing risks to an acceptable level. 17 Mollah et al. 18 outlined the importance of implementing protective measures (C22) to reduce the probability and occurrence of a hazardous situation or the severity of the risk. This component resonates with the initial approach of the hazard analysis and critical control points (HACCP), suggested by the USFDA Guidance for Industry on Quality Risk Management. 18 Although MacKenzie 19 and Liberati et al. 24 considered administrative controls (C23) as weak interventions and only limited to controlling the exposure to hazards on a prevention level, the National Institute for Occupational Safety and Health (NIOSH) (2015) highlighted the role of administrative controls in risk reduction based on the hierarchy of controls. 25 The NIOSH described its effectiveness in minimizing risks and hazards by adjusting the technical difficulties during workflows to address the root causes of the system problem. 25 Validation and verification procedures (C24) were described as critical components in risk control to assure systems’ compatibility to mitigate risks pertinent to the product, process, or project throughout the manufacturing, drug preparation, and administration processes.18,26,27
FDA (2006) identified the system’s detectability of hazards (C25) as pivotal element in risk control; it reflects a system’s capacity in determining the existence of a hazard. 23 While ICH suggests processes in place to improve a system’s detectability of hazards, it also discussed the probability of introducing new risks into the system or increasing the significance of other existing risks. 17 Furthermore, risk control encompasses risk tolerance (C26) and risk mitigation capacity (C27) as determining criteria for an entity’s plan for risk acceptance to establish the level of risk that the organization is willing to allow in the process, product, or project. 18
Risk communication
The third aspect is risk communication (A3), which involves sharing information regarding the identified risks and risk management strategies among all interested parties, such as regulators, sponsors, health care professionals, and patients. 17 Mollah et al. 18 suggests organizations allow information available to assess the mitigation actions (C28) and include assessments of residual risks and recommendation for further action. 18 Lundgren and McMakin 23 described risk communication as a two-way communication, in which the organization manages the risk, and the audience carries on a dialogue. Meanwhile, outlining an effective risk communication plan (C29) that is audience focused and combining the elements of strategic planning and public engagement is essential in an organization’s risk communication process. The internal and external audiences in risk communication, thus, involve communication between regulators and industry (C30), communication between industry and the patient (C31), and internal communication (C32). 28
Risk review
The fourth aspect is risk review (A4), which is a continuous process of following up and reevaluating the system to determine the existence and significance of risks; it is an ongoing part of the quality risk management process for performing risk assessment and risk control.17,23 This process entails identifying new risks or changes to current risk profile during routine operations (C33), introducing new risk controls or changes made to existing risk control (C34), identifying the need for change in previous risk control decisions (C35), reconsidering risk acceptance decisions (C36), and establishing a system that allows the review and monitor of at-risk events (C37). 18
State-of-the-art technology
The fifth aspect is the applicability of the state-of-the-art technology (A5), which is key to performing risk mitigation and ensuring the intactness of the pharmaceutical supply chain, ranging from manufacturers, distributors, and patients. Liberati et al. 24 discussed the roles of artificial intelligence and machine learning (C38) in ensuring risk management and regulatory compliance, as well as its rapid growth in the pharmaceutical manufacturing industry. The application of artificial intelligence has been adopted throughout a product’s lifecycle, which includes drug discovery, manufacturing, quality control, and quality assurance. 29 Meanwhile, Liu et al. 25 demonstrated machine learning’s role in detecting risks associated with medications. 30 With the digitization of manufacturing processes, O’Donovan et al. 26 and Hernandez and Zhang 27 discussed the role of big data analysis (C39) in predictive analytics in identifying risks.31,32 Blockchain technology (C40) has been recognized as a critical intervention in providing manufacturing transparency, traceability, and maintaining the integrity of the pharmaceutical supply chain.33,34 This helps ensure the intact of supply chain and logistics blockchain-based supply chain management for pharmaceutical drugs. Liberati et al. 24 also identified the role of Internet of Things (IoT) (C41) in manufacturers to optimize factory operations, product quality, and the manufacturing logistics. 29 In addition, the application of three-dimensional (3D) printing (C43) in pharmaceutical manufacturing assists in risk management. Through the utilization of 3D printing, patient safety can be achieved via the production of personalized medicine. 35
Methods
This section explains the fuzzy DEMATEL method used in this study. This study examines the causal relationships between criteria using linguistic expressions to determine the relative degree of importance; the qualitative information is transformed into quantitative crisp values for visual analysis. 36 The DEMATEL method is then used to interpret the causal relationships and interrelationships between the attributing criteria of pharmaceutical drug recalls. 20 Visual analysis is then presented to facilitate the discussion. 37
Study background
In this study, a set of criteria is formulated based on the literature that follows a questionnaire for linguistic evaluation. A group of 11 experts were selected across the pharmaceutical industry, hospitals, ambulatory care, regulatory authority, and community care settings for interviews. These experts include seven pharmacists with more than 12 years of experience in hospital administration, inpatient pharmacy, ambulatory care, and academia, two representatives from community care practice with more than 10 years of experience managing pharmaceutical drug recalls, one government regulatory authority with 11 years of work experience in medication safety and risk mitigation, and one expert from the pharmaceutical industry with more than 30 years of experience in pharmaceutical drug recall and quality management.
Fuzzy DEMATEL
The uniqueness of fuzzy DEMATEL entails only requiring a small sample size from experts within the respective disciplinaries. This study included experts from five different disciplinaries in pharmaceutical practices as aforementioned. The detailed formula derivation of fuzzy DEMATEL can be found in Jaberidoost et al., 15 O’Donovan et al., 31 and Hernandez and Zhang. 32
The first important process of fuzzy DEMATEL is to convert the human/expert assessment into fuzzy linguistic variables 20 (see Table 2) following the application of defuzzification operation of fuzzy set theory to convert fuzzy number into an effective crisp value. The assessment scale of “relative influence” between any two criteria determined by expert respondent is categorized into five fuzzy linguistic scales: N (no influence), VL (very low influence), L (low influence), H (high influence), and VH (very high influence) to accumulate the fuzzy direct relation matrix of the criteria. 36 The defuzzification of fuzzy data into crisp values was calculated using the fuzzy minimum and maximum numbers to determine the left and right limit values.20,38 The resultant cause–effect diagram generated by fuzzy DEMATEL provides a comprehensive visual analysis for the criteria which are categorized into cause and effect groups to indicate their interrelationships and the influential effects between the groups. This study employs the following four analytical steps:
Converting linguistic assessment into fuzzy numerical data
Defuzzifying triangular fuzzy numbers into crisp values
Constructing interrelationship matrix for the crisp values and aspect-criteria grouping
Mapping of a cause–effect diagram.
Fuzzy linguistic preferences and scales.

Results
The results are presented in the analytical steps, as follows.
The assessment data from the expert respondents are available in linguistic forms. These linguistic data are then transformed into triangular fuzzy numbers, as Table 3 shows.
Converted triangular fuzzy numbers for aspects.

VH: very high influence; L: low influence; H: high influence.
The same data collection technique is implemented for 42 criteria as indicated in Table 1. The linguistic form of data ranging from “VL” to “VH” is using the same scale for evaluating the interrelationships among criteria.
Because these fuzzy numbers of the assessment of relative influence among aspects are not possible to compute, procedures need to be followed to resolve the vague meanings and obtain precise, crisp values. Table 4 presents the average crisp values of the aspects for all the expert respondents. Meanwhile, Tables 5–8, show the crisp values of the criteria for all the expert respondents.
Crisp values of relative influence assessment for the aspects.

Crisp values of relative influence assessment for the criteria ((C1 − C21) × (C1 − C21)).

Crisp values of relative influence assessment for the criteria ((C22 − C42) × (C1 − C21)).

Crisp values of relative influence assessment for the criteria ((C1 − C21) × (C22 − C42)).
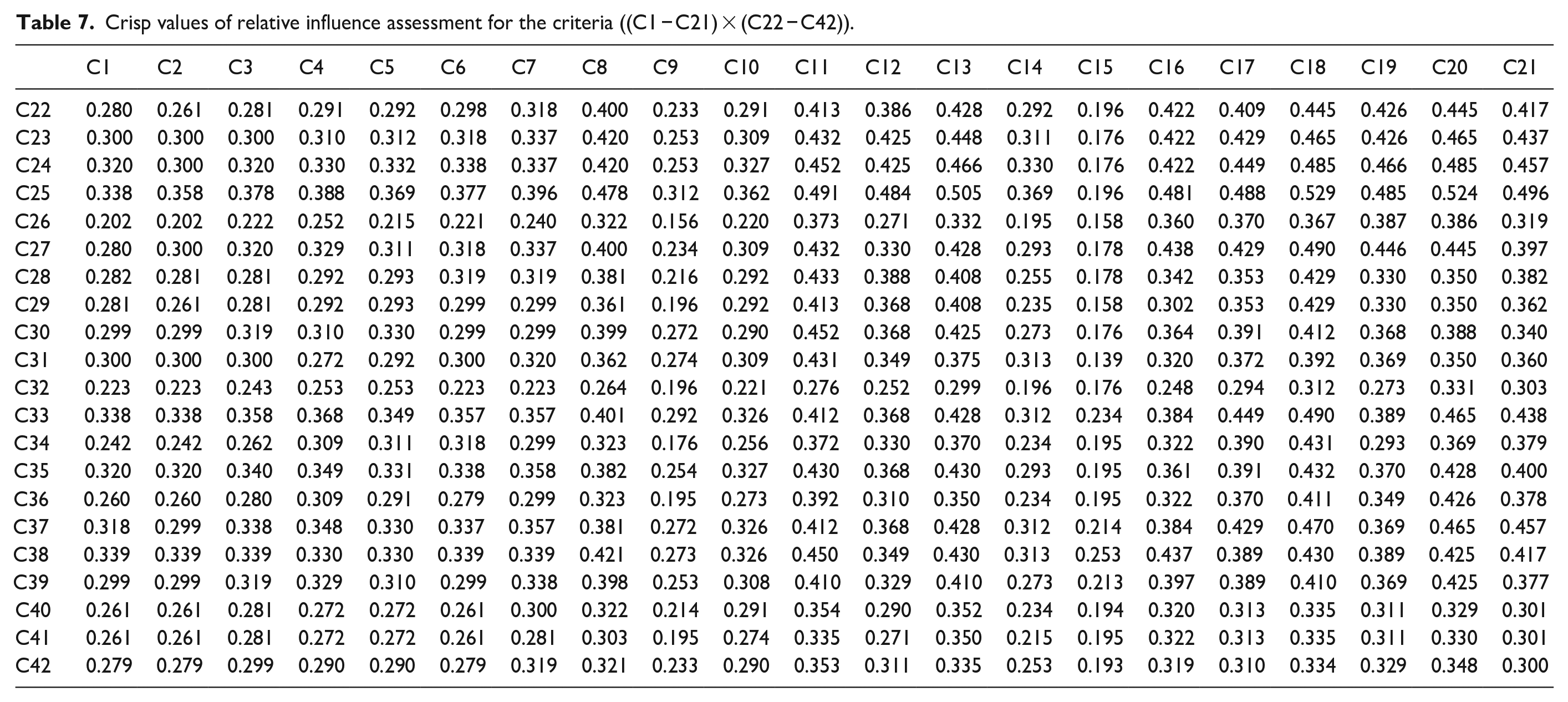
Crisp values of relative influence assessment for the criteria ((C22 − C42) × (C22 − C42)).

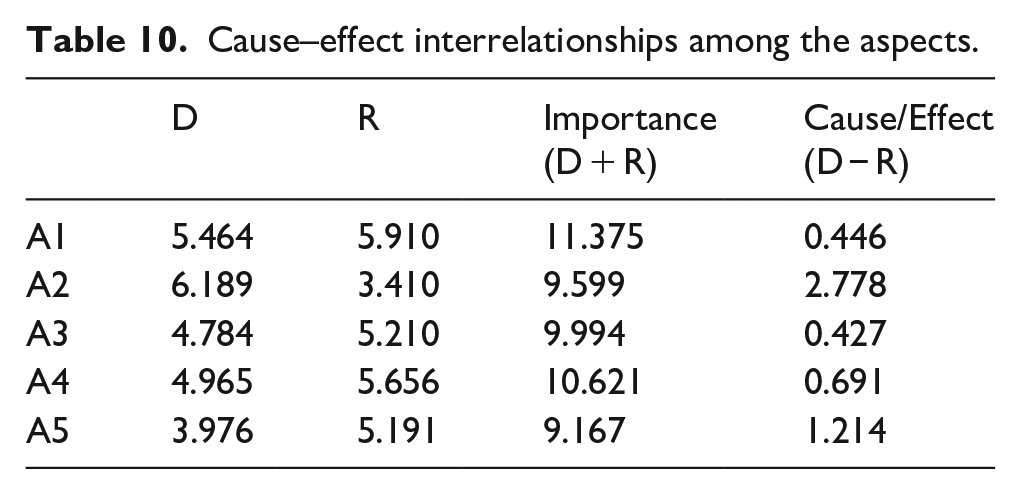
The aspects, including risk assessment (A1), risk control (A2), risk communication (A3), risk review (A4), and technology (A5), are set into an interrelationship matrix. Table 9 shows the total interrelationship matrix of the aspects. This matrix is then transformed into cause–effect interrelationships among the aspects, as seen in Table 10. The same method is employed in the analysis of criteria. There are 42 criteria that are involved and are set into an interrelationship matrix, as seen in Tables 11–14, showing the total interrelationship of the criteria. The matrix is then converted into cause–effect interrelationship matrix, as Tables 15 and 16.
Interrelationship matrix of the aspects A1–A5.

Cause–effect interrelationships among the aspects.
Interrelationship matrix for the criteria ((C1 − C21) × (C1 − C21)).

Interrelationship matrix for the criteria ((C22 − C42) × (C1 − C21)).

Interrelationship matrix for the criteria ((C1 − C21) × (C22 − C42)).

Interrelationship matrix for the criteria ((C22-C42) × (C22-C42)).

Cause–effect interrelationships among the criteria.

Cause–effect interrelationships among the criteria.

In either Table 9 or Tables 11–14, there are driving power (D) and dependence power (R); the values of D and R are obtained from the accumulation of rows and columns, respectively. (D + R) represents the importance of aspects/criteria. Also, (D − R) >0 represents the aspect/criteria belonging to the causal group while (D − R) <0 represents aspect/criteria belonging to effect group. The mapping of (D + R) and (D − R) is then transferred into the horizontal and vertical axes of the cause–effect diagram as shown in Figure 1.

Cause–effect diagram for aspects with interrelationship strength indicators.
Figure 1 concludes the interrelationships between each aspect, and as seen in the figure, there is one aspect that falls within the causal group: Risk control (A2), while the remaining aspects belong to the effect group. Risk control (A2) is the main influencing aspect of pharmaceutical drug recalls and has the strongest impact on risk assessment (A1) and risk review (A4); in the meantime, it generates medium effects on risk communication (A3) and technology (A5). On the contrary, risk assessment (A1), risk communication (A3), and risk review (4) demonstrated comparative weak interrelationships, while risk communication (A3) exhibits a weak unidirectional effect on risk review (A4). Finally, risk assessment (A4) exerts a weak influence on technology (A5) application and development.
Figure 2 illustrates the distribution and the cause–effect interrelationships among the criteria. The results show that C1, C2, C3, C4, C5, C6, C7, C9, C10, C14, C15, C22, C23, C24, C25, C27, C28, C33, C35, C37, C38, and C39 are the cause criteria and that C8, C11, C12, C13, C16, C17, C18, C19, C20, C21, C26, C29, C30, C31, C32, C34, C36, C40, C41, and C42 fall within the effect group. Among the causal group, product contamination (C3), product subpotent or superpotent (C2), injury to patients (C15), product not sterile or impure (C1), and system detectability of hazards (25) have the strongest influence in the causal group.

Cause–effect diagram for criteria.
Discussion
This section discusses the study’s theoretical and practical implications for minimizing pharmaceutical drug recalls and maximizing patient safety in the literature and industries.
Theoretical implications
This study presents the causal aspects of pharmaceutical drug recalls and discusses the theoretical implications to the literature. Based on the results, this article successfully identified that among the five important aspects, risk control (A2) is the main and dominating causal aspect of pharmaceutical drug recalls and has leading influence on risk assessment (A1) and risk review (A4); in the meantime, it yields medium effects on risk communication (A3) and technology (A5). The remaining aspects are within the effects group. This indicates risk control plays a significant role in reducing pharmaceutical drug recalls and maximizing patent safety. Prior studies also resonated with this finding in which risk control has a substantial impact on pharmaceutical manufacturing, drug administration, and patient safety.39,40
Risk control is a decision-making process intended for reducing and accepting risks. The primary goal of risk control is to minimize the risk to an acceptable level that prevents patients from harm. 39 Risk control activities typically involve identifying controls and measures that may minimize or manage the risks associated with a failure mode or negative event throughout the pharmaceutical manufacturing, delivering, and administration processes. Risk control activities are key for identifying pivotal process parameters for certain controls, how they will be oversaw, and the level of qualification and validation for such controls. 39 It helps determine optimal balance between benefits, risks, and possibilities of introducing new risks within the context of patient safety. Although risks should be eliminated in all circumstances and mitigated based upon severity, probability of occurrence, and detectability, the acceptability of a risk is critical for determining further actions for these measures. 40
The study findings also suggest that risk control displays a strong influence on risk assessment and risk review and demonstrated the interrelationship between all three aspects within the domain of quality risk management. Risk assessment is the routine of identifying risks and understanding the standard procedures. According to the ICH Q9 risk management, risk is composed of the probability of occurrence of harm and the severity of that harm. 17 Detectability of risks entails identifying and evaluating the probability of a risk occurrence; however, devoting resources and time to identifying risks without constructive strategies to minimize and reduce risks have minimal efficacy to prevent patient harm. Moreover, many systems highlighted for risk monitoring and detection (i.e. quality control) rely on small sample sizes, which could potentially introduce errors and inaccuracy. 41 Thus, the process of risk assessments should be centered around risk control to better ensure risk management. Risk review, on the contrary, represents the process of periodically reviewing and reassessing current procedures and information that may impact on the original quality risk management decisions.18,39 Content of the review should include changes to existing control systems, equipment and procedures, and organizational restructure. Monitoring is the scheduled measurement or observation of a specific risk control measure relative to its acceptance limits. 39 Hence, the process of risk review highly relies on risk control to carry out effective measures to monitor and reduce risks.
Risk assessment and risk review are conventional processes already integrated in all practices ranging from pharmaceutical industry manufacturing, supply chain, and health care services. Determining the risk was the objective of risk management; however, improving process through risk control and risk mitigation is a key to ensuring product quality and patient safety. The strategies for risk control ought to entail risk reduction to minimize any potential risks, as well as establishing the level of risk acceptance that helps determine an acceptable risk level or if additional mitigation strategies are needed. 18 Defining risk acceptance is key within the domain of risk control as it determines whether additional actions should be taken to mitigate the associated risks. Future studies on pharmaceutical drug recall’s impact on patient safety could place an emphasis on various risk control measures and the extent of effectiveness in mitigating different levels of risks.
In sum, to achieve patient safety, this study suggests focusing on risk control strategies, as this aspect exhibits the strongest effect on influencing other critical risk management aspects such as risk assessment and risk review. Improving risk control measures in all pharmaceutical practices, ranging from pharmaceutical industries, health care professionals, and regulators is critical for preventing patient harm and optimizing patient safety.
Practical implications
This section presents the criteria to provide practical insights to improve patient safety associated with pharmaceutical drug recalls. The important causative criteria, including product contamination (C3), product subpotent or superpotent (C2), injury to patients (C15), product not sterile or impure (C1), and system detectability of hazards (25), provide practical insights regarding pharmaceutical drug recalls effects on patient safety. The associated action plans that can help improve the sector are discovered.
Product contamination (C3) has the strongest influence on pharmaceutical drug recalls. It entails any substances other than the drugs that are present in the product, which includes bacterial, fungal, heavy metal, or other foreign particulate contaminants. This also entails any suspected or confirmed contamination, as well as product adulteration. 3 Based on previous retrospective reviews on regulatory alerts in the United States and Canada, product contamination was found to be the most common and concerning issue causing recalls for dietary supplements, prescription, and nonprescription products.3,42 To effectively reduce the risk of pharmaceutical drug recalls and ensure patient safety, pharmaceutical industries should emphasize risk control systems on product contamination to maintain product quality and timely mitigate any associated risks. Based on this finding, the prevention of product contamination should be the top priority of all key stakeholders as it is more likely to cause patient harm and leading to a pharmaceutical drug recall. Health care providers should remain vigilant on information regarding product contamination to ensure these products do not reach patients and inform patients about the risks. Meanwhile, if patients develop unexpected adverse events while taking these products, health care professionals should immediately report the events and advise patients to seek medical attention. This finding also suggests the urge for a more stringent regulatory framework and industry standards to protect the public from being exposed to suspected and confirmed contaminated products. Increased implementation of existing regulations improved self-regulation by manufacturers, and robust regulations should be incorporated to improve the overall medication quality and patient safety.
Product potency such as subpotency or superpotency (C2) is considered one of the core quality criteria (CQAs) that affect the safety and efficacy of a product.18,43 The change of product potency impacts the strength of a drug substance, which could lead to treatment failure or unexpected adverse events. Thus, it is imperative for the pharmaceutical industries to always include potency as one of the CQAs of all drug products during the pharmaceutical development process to ensure the consistency and better monitor the potency of the product. Patient safety is jeopardized when product quality deteriorates; hence, industries should strongly adhere to guidelines to ensure product potency during the pharmaceutical and manufacturing processes. Meanwhile, health care professionals should assist with the process for preventing patients from being exposed to these types of products and educate patients to avoid purchasing deviated products online. Health authorities should enforce regulations to prevent the public’s susceptibility to substandard and falsified medications.
Products causing harm or hazard to patients including but not limited to adverse drug reactions, hospitalizations, or fatal events fall under the category of patient injury (C15). Pharmaceutical and health care industries have the obligations to ensure patient safety; thus, all stakeholders should focus risk management approaches on patient safety and explore optimal risk management tools within standard practices to avoid patient injury to all extent. Consistent application of risk control measures is critical to ensure risks are mitigated and removed in the exposed populations. To achieve this, the pharmaceutical industries should enhance routine pharmacovigilance safety surveillance activities during premarketing and postmarketing drug development phases, as well as monitoring product quality throughout the manufacturing processes. Health care providers may conduct a root cause analysis to determine the patient, personnel, policy, environmental, and procedural factors that contribute to the issue and implement risk control strategies to prevent medication errors and human errors from reaching patients in all aspects of prescribing, dispensing, and administering medications. 44 In the meantime, health authorities must consider expanding the use of adverse reaction reporting systems to receive reports promptly and execute mitigation actions expeditiously. Health authorities may also educate the public the use of recall notification systems and how to stay informed on the status and updates on pharmaceutical drug recalls.
This study also recommends product not sterile or impure (C1) as one of the top five leading criteria of pharmaceutical drug recalls. Unsterilized or microbial contamination of purportedly sterile products is considered as hazards that may pose harm to patients. Achieving sterility for parenteral products has been the ultimate goal of both industry and regulators. While aseptic processing procedures, such as sterile sampling, filtration, filling, and lyophilization, are considered as higher risk processes, the primary source of nonsterility and microbial contamination in aseptic processing is recognized to be personnel. 18 To better minimize the risks associated with sterility, the manufacturing processes should tailor control strategy to include frequent microbial monitoring of air, surfaces, and personnel. Incorporating sterility assurance in validation processes would also help ensure the level of sterility. Health care professionals should carefully inspect sterile products before sterile compounding and drug administration for potential cloudiness, crystallization, or unusual colors prevent unsterile and impure products from reaching patients. In addition, regulators should perform frequent testing on sterile products to ensure the quality and safety of these marketed products.
The system’s detectability of hazards (C25) is critical for an entity or organization to identify and determine any new or existing hazards. This influences the decision-making process in manufacturing risk management, and specifically in risk control. Detection measures the capability of existing controls to detect the failure event and to prevent a substandard product from reaching patients. 18 Having a sensitive detection system allows expeditious action plans for risk mitigation and risk control; thus, stakeholders across pharmaceutical industries, health care settings, and regulatory authorities should strive to enhance the system’s detectability of hazards within each entity.
Limitations and future study directions
There are a few limitations to this study. The study did not differentiate the severity of risks and different types of recalls in the discussion. Future studies can consider examining the different classes of recalls and their impact on patient safety. This study is based on expert opinions, which could potentially introduce bias when limited number of participants are included. In future studies, the sample of experts can be enlarged to further develop the interrelationship DEMATEL model using linguistic preferences. The generalizability of this study excluded certain categories such as medical devices, combination products, and herbal and dietary products. Alternatively, future studies could overcome this limitation by including other subgroup products to examine their causal relationships of risks in these categories on patient safety from a different angle. There is also an imbalanced distribution among the expert respondents’ professional background and country representations; hence, future studies should involve a more diverse and balanced professional background to ensure experts’ representations from all fields.
Conclusion
Pharmaceutical drug recalls are driving factors that pose harm to patient safety and constitute threats to global public health. This study involves 42 criteria grouped under five aspects including risk assessment, risk control, risk communication, risk review, and technology. This study applied fuzzy DEMATEL to the structure of quality risk management to examine the pharmaceutical drug recall criteria’ causal interrelationships by clarifying the experts’ opinions using their linguistic preferences. Based on the adaption of the ICH Q9 framework, this study proposed a set of criteria to assess the impact of pharmaceutical drug recalls on patient safety using linguistic preferences. This assessment offers a decision-making tool that will be helpful for decision makers and policy-makers, such as the government (regulatory and health authorities), health care providers, and pharmaceutical industries.
The results show risk control exhibits the strongest effect toward the aspects in the effect group including risk assessment and risk review. Essentially, prioritization of risk control in all areas is needed to maximize patient safety. Overall, 42 criteria are categorized into cause and effect criteria groups. This study recommends top five causative criteria for pharmaceutical drug recalls as product contamination, product subpotent or superpotent, injury to patients, product not sterile or impure, and system detectability of hazards. To achieve the goal of reducing pharmaceutical drug recalls and improving patient safety, all key stakeholders should prioritize practical changes in these top five causative criteria.
This research suggests future studies examine different types of recalls and their impact on patient safety and ensure a proportionate distribution of the experts’ professional background and country representations. As an extension of this study’s results, future studies should consider investigating criteria in a wider range by including other categories of medications and medical device. More studies are needed to better understand risks associated with pharmaceutical drug recalls on patient safety, not only by identifying the causing and affected criteria, but also challenges in implementation.
Supplemental Material
sj-docx-1-map-10.1177_27550834231170075 – Supplemental material for Risk control drives risk assessment and risk review: A cause and effect model of pharmaceutical drug recall on patient safety
Supplemental material, sj-docx-1-map-10.1177_27550834231170075 for Risk control drives risk assessment and risk review: A cause and effect model of pharmaceutical drug recall on patient safety by Irene D Lin and John B Hertig in The Journal of Medicine Access
Footnotes
Acknowledgements
The authors would like to thank Dr. Ming-Lang Tseng for generously providing his fuzzy DEMATEL software for the data analysis. Also, the authors would like to thank the following expert respondents in their support of this research: Drs. Lubna Merchant, Erin Fox, James Stevenson, Scott Ciarkowski, Dan Degnan, Alex Varkey, Katie Hufft, Stephanie Arnett, and Ms. Grace Yep.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
